Impact Factor : 0.548
- NLM ID: 101723284
- OCoLC: 999826537
- LCCN: 2017202541
Xingyu Wang1*, Zhengchao Wang1, Xin Zhang1, Yuxuan Shao2, Leyang Ma1, Lvhao Cao1, Yanjing Zhu1 and Tongyuan Zhang1
Received: February 23, 2026; Published: March 17, 2026
*Corresponding author: Xingyu Wang, Department of Emergency Surgery, The First Affiliated Hospital of Anhui Medical University, Hefei, Anhui, People’s Republic of China
DOI: 10.26717/BJSTR.2026.65.010135
Intestinal ischemia/reperfusion injury (II/RI) is a critical complication in gastrointestinal surgery, critical care medicine, and organ transplantation, characterized by high incidence and mortality rates. The pathophysiological process is highly complex, involving oxidative stress surges, uncontrolled inflammatory responses, activation of multiple forms of programmed cell death (such as ferroptosis, pyroptosis, and necrotic apoptosis), severe disruption of intestinal barrier function, and significant dysbiosis of the gut microbiota. In recent years, research has not only profoundly elucidated the core regulatory roles of key signaling pathways such as nuclear factor E2-related factor 2 (Nrf2), the NOD-like receptor protein 3 (NLRP3) inflammasome, and silent information regulator proteins (Sirtuins), but has also highlighted the pivotal position of the gut microbiota and its metabolites in disease onset and progression. In therapeutic strategies, approaches ranging from traditional antioxidant and anti-inflammatory drugs (such as dexmedetomidine and resveratrol) to gut microbiota-based interventions (such as probiotics and fecal microbiota transplantation), and extending to cutting-edge stem cell/exosome therapies and smart nanomedicine delivery systems, have all demonstrated significant potential. This review aims to systematically summarize the latest research advances in II/RI, integrating the evidence chain from molecular mechanisms to therapeutic interventions, to provide a comprehensive theoretical framework for the clinical precision treatment and future research directions of this disease.
Keywords: Intestinal Ischemia-Reperfusion Injury; Oxidative Stress; Cell Death; Intestinal Barrier; Gut Microbiota; Therapeutic Targets
Abbreviations: II/RI: Intestinal Ischemia/Reperfusion Injury; Nrf2: Nuclear Factor E2-Related Factor 2; NLRP3: NOD-Like Receptor Protein 3; MODS: Multiple Organ Dysfunction Syndrome; SIRS: Systemic Inflammatory Response Syndrome; ROS: Reactive Oxygen Species; mtDNA: Mitochondrial DNA; DAMPs: Damage-Associated Molecular Patterns; GPX4: Glutathione Peroxidase 4; MLKL: Mixed Family Kinase-Like Domain; TFEB: Translocation of Transcription Factor EB; ILA: Indole-3-Lactic Acid; CAT: Capsaicinate; MAMs: Mitochondrial-Associated Endoplasmic Reticulum Membranes; MSCs: Mesenchymal Stem Cells; cfDNA: Can Efficiently Clear Cell-Free DNA
Ischemia-reperfusion injury of the intestine commonly occurs in various acute and critical conditions, including acute mesenteric artery embolism, intestinal volvulus, traumatic shock, cardiac extracorporeal circulation procedures, and major abdominal vascular surgery [1,2]. The damage is not confined to the local intestinal tract. The “cascading” inflammatory response triggered by reperfusion and the collapse of the intestinal barrier can lead to bacterial/endotoxin translocation, subsequently inducing systemic inflammatory response syndrome (SIRS) and multiple organ dysfunction syndrome (MODS), which are the primary causes of patient mortality [3,4]. Despite advances in supportive therapies, there remains a lack of specific effective drugs for II/RI, reflecting an insufficient understanding of its complex mechanisms. In recent years, with the advancement of molecular biology, metabolomics, and high-throughput sequencing technologies, our understanding of II/RI has deepened from macroscopic pathology to microscopic molecular networks and ecological levels. This paper systematically reviews the core pathological mechanisms, key regulatory signaling networks, and the resulting diverse novel therapeutic strategies for II/RI based on extensive recent research.
Oxidative Stress and Inflammatory Storm: Initiation and Amplification of Injury
The essence of II/RI lies in the excessive production of reactive oxygen species (ROS) due to mitochondrial dysfunction following the restoration of blood flow to hypoxic tissues, thereby triggering oxidative stress [4,5]. ROS not only directly damages cellular macromolecules but also acts as a key signaling molecule, activating pathways such as nuclear factor-κB (NF-κB) to promote the expression of proinflammatory factors, including tumor necrosis factor-α (TNF-α), interleukin-1β (IL-1β), and interleukin-6 (IL-6) [1,6]. Simultaneously, damage-associated molecular patterns (DAMPs) released by damaged cells, such as mitochondrial DNA (mtDNA), can further exacerbate the inflammatory response by activating the cGAS-STING pathway [7,8]. The assembly and activation of NLRP3 inflammasomes serve as a pivotal link between oxidative stress and pyroptotic inflammation. Downstream, Caspase-1 cleaves Gasdermin D (GSDMD) to form pores in the plasma membrane, leading to the maturation and release of IL-1β and IL-18, ultimately triggering cell pyroptosis [9,10]. On the other hand, the body’s endogenous antioxidant system, particularly the Nrf2/HO-1 pathway, has been demonstrated to be a core protective mechanism against II/RI. Multiple drugs (such as isoquercitrin and dimethyl fumarate) and metabolites (such as NADPH) exert antioxidant and anti-inflammatory effects by activating this pathway [11-14].
Diversity of Programmed Cell Death: Key Pathways for Damage Execution
Cell death directly reflects damage to the II/RI intestinal epithelium. Beyond classical apoptosis, the role of novel regulatory cell death mechanisms is increasingly prominent. The mechanisms, key molecules, and intervention strategies are summarized in Table 1.
Table 1: Key types of programmed cell death and their roles in intestinal ischemia/reperfusion injury.
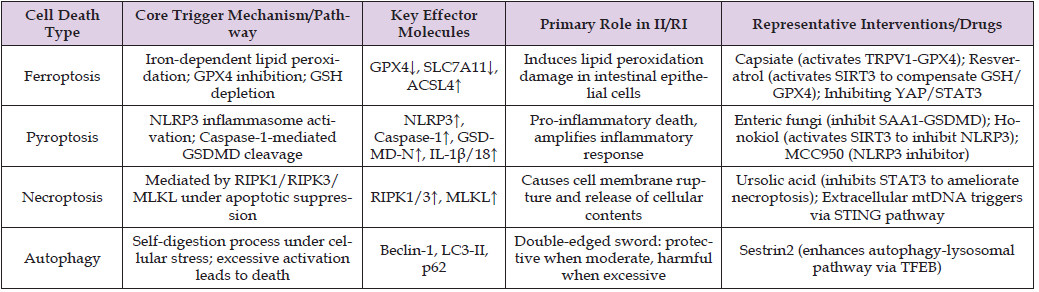
• Ferroptosis: A form of cell death that is iron-dependent and driven by the accumulation of lipid peroxides. In II/RI, reduced glutathione peroxidase 4 (GPX4) activity and suppressed expression of solute carrier family 7member 11 (SLC7A11) lead to weakened antioxidant capacity, accumulation of lipid peroxidation products, and ultimately trigger ferroptosis [3,5]. Research confirms that capsaicin esters suppress ferroptosis by activating TRPV1 to promote GPX4 expression [15]; whereas ferroptosis inhibitors (such as Ferrostatin- 1) or activation of the YAP/Nrf2 pathway can effectively mitigate injury [16-18].
• Pyroptosis: As previously described, it is primarily mediated by the NLRP3 inflammasome-GSDMD axis [9,19]. Gut fungi exert a protective effect by reducing GSDMD-mediated pyroptosis through the suppression of serum amyloid A1 (SAA1) expression [10]. The small-molecule compounds MCC950 and magnolol have also been shown to alleviate pyroptosis by inhibiting NLRP3 activation [9,19,20].
• Necroptosis: Mediated by receptor-interacting protein kinase 1/3 (RIPK1/RIPK3) and the mixed family kinase-like domain (MLKL) when apoptosis is suppressed. Extracellular mtDNA can synergistically trigger necrotic apoptosis in intestinal epithelial cells through the STING pathway in conjunction with IFN and TNF-α signaling [7,8]. Ursolic acid can improve necrotic apoptosis by inhibiting STAT3 activation [21].
• Autophagy: Acts as a double-edged sword. Moderately activated autophagy clears damaged mitochondria (mitophagy) and endoplasmic reticulum stress products, maintaining cellular homeostasis. For example, Sestrin2 exerts a protective effect by enhancing the function of the autophagy-lysosomal pathway through promoting the nuclear translocation of transcription factor EB (TFEB) [22,23]. However, it should be noted that autophagy is a double-edged sword; excessive or dysregulated autophagy may instead promote cell death.
Complete Collapse of the Intestinal Barrier Function
The intestinal barrier consists of the intestinal epithelial barrier and the intestinal vascular barrier [24]. II/RI leads to significant downregulation or disrupted distribution of tight junction proteins (such as ZO-1, Occludin, and Claudins), resulting in a dramatic increase in intestinal epithelial permeability [24-26]. This not only allows for the translocation of intra-cavity bacteria and endotoxins, but also serves as a potent source of inflammatory stimulation. Notably, disruption of the intestinal vascular barrier is equally critical, as the opening of intercellular junctions in its endothelial cells facilitates the entry of bacteria and antigens into the systemic circulation [24,27]. Dexmedetomidine has been demonstrated to protect the intestinal vascular barrier by activating β-catenin signaling, thereby mitigating distal liver injury [24].
Core Regulatory Role of the Gut Microbiome and its Metabolic Products
The gut microbiota is referred to as the “forgotten organ,” playing a dynamic regulatory role in II/RI. Damage leads to reduced microbial diversity, decreased levels of beneficial bacteria (such as Lactobacillus and Bifidobacterium), and increased levels of potential pathogens [28,29]. This imbalance is not only a consequence of the injury but also a significant factor driving the progression of the condition. More importantly, microbial metabolites serve as key messengers mediating their functions, with their mechanisms summarized in Table 2.
Table 2: Protective roles of key gut microbiota metabolites in intestinal ischemia/reperfusion injury.
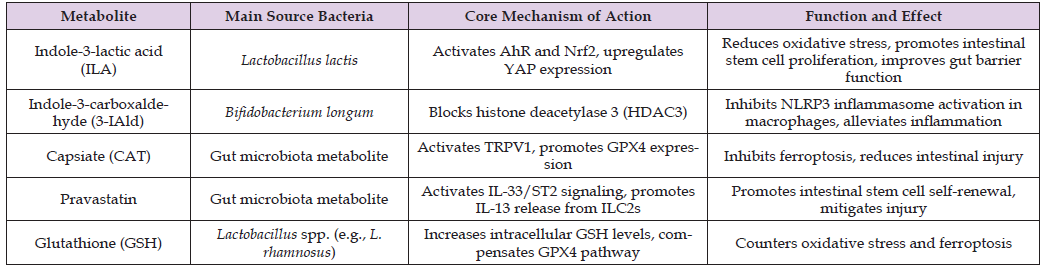
• Tryptophan metabolites: The protective effect of indole- 3-lactic acid (ILA) derived from Lactobacillus lactis is closely associated with its upregulation of YAP expression. Mechanistic studies indicate that ILA positively regulates YAP by activating AhR and Nrf2 signaling pathways, thereby alleviating oxidative stress and promoting intestinal stem cell proliferation [16,30]. Indole-3-carboxaldehyde, derived from bifidobacteria, inhibits NLRP3 inflammasome activation in macrophages by blocking histone deacetylase 3 (HDAC3) [31].
• Other protective microbial metabolites: Additionally, studies have identified multiple structurally diverse microbial metabolites with direct protective effects. For example, capsaicinate (CAT), defined as a gut microbiota metabolite, promotes GPX4 expression by activating the TRPV1 receptor, thereby inhibiting ferroptosis15. Pravastatin, another microbiota-associated metabolite, promotes intestinal stem cell renewal by activating the IL-33/ST2 signaling pathway to stimulate ILC2s to release IL-13, which in turn activates the Notch and Wnt signaling pathways [32].
• Microbe-Host Co-metabolism: Disruption of the gut microbiota also directly impacts the host’s metabolic pool. For example, the protective effect of metformin partially depends on its regulation of the gut microbiota, increasing the abundance of Lactobacillus species, thereby enhancing the capacity of microorganisms to synthesize glutathione (GSH) to compensate for the GPX4 pathway against ferroptosis [33].
Based on the aforementioned mechanisms, intervention strategies are evolving toward diversification and targeted approaches. A summary of representative studies for various treatment strategies is presented in Table 3.
Table 3: Major therapeutic strategies and representative agents/methods for intestinal ischemia/reperfusion injury.
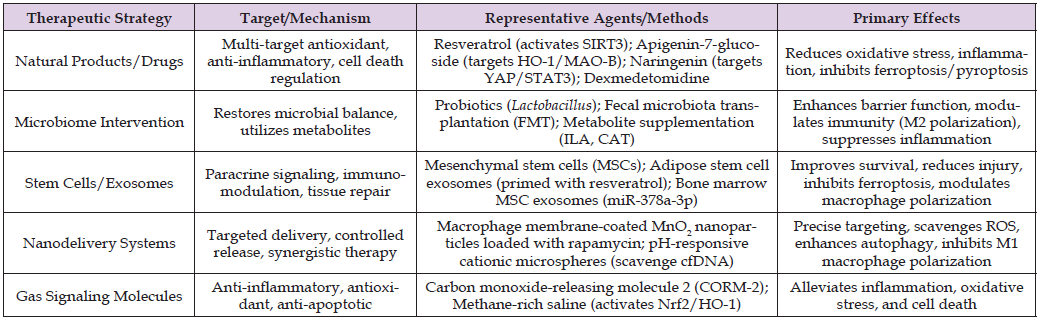
Drug Intervention: From Natural Products to Repurposing Existing Drugs for Clinical Use
• Natural bioactive compounds: Resveratrol (activates SIRT3, inhibits ferroptosis) [3], apigenin-7-glucoside (targets HO-1 and MAO-B, inhibits endothelial ferroptosis) [34], naringenin (inhibits ferroptosis) [5,17], and magnolol (activates SIRT3, inhibits mitochondrial ROS and pyroptosis) [19], among others, all demonstrate promising effects through multi-target mechanisms.
• Clinical Drug: Dexmedetomidine, as a highly selective α2-adrenergic receptor agonist, exerts protective effects through multiple mechanisms, including anti-inflammatory, antioxidant, anti-cell death (apoptosis, pyroptosis, ferroptosis), and enhanced mitochondrial autophagy. Meta-analyses have confirmed its efficacy in animal models [1,2]. Metformin not only modulates the microbiota but also inhibits ferroptosis by reducing the formation of mitochondrial-associated endoplasmic reticulum membranes (MAMs) [33].
Microbiome-Targeted Therapy
• Probiotics/ Prebiotics: Direct supplementation with specific probiotics, such as Lactobacillus lactis (which promotes IL-10 release from M2 macrophages via TLR2 signaling [35]) or Bifidobacterium longum (whose metabolites exhibit anti- inflammatory effects [31]), or administration of prebiotics, can mitigate II/RI injury by enhancing intestinal barrier function and modulating local immunity (e.g., promoting macrophage M2 polarization). A systematic review and meta- analysis confirmed that probiotic treatment significantly reduced intestinal injury scores (Chiu’s score), inflammatory factors, and oxidative stress levels in animal models [36].
• Fecal Microbial Transplantation (FMT): Compared to supplementing with a single or limited number of probiotics, transplanting the entire microbiota from a healthy donor to a recipient theoretically enables a more comprehensive restoration of the host’s microbial diversity and interaction networks. Animal studies indicate that FMT can effectively mitigate II/RI injury by reshaping the intestinal microenvironment28, demonstrating its unique potential as an “ecosystem- level” therapeutic strategy.
• Postbiotic therapy: Directly delivering microbial metabolites with well-defined protective effects (e.g., indole-3-lactic acid [16], capsaicin esters [15]) represents a more precise therapeutic strategy than live probiotic supplementation. Such formulations based on non-living microbial components are termed “postbiotics.” They circumvent the uncertainty of live bacteria surviving in damaged environments, can directly target specific pathways, and offer greater stability and improved safety.
Stem Cell and Extracellular Vesicle Therapy
Mesenchymal stem cells (MSCs) have been extensively studied for their potent immunomodulatory, anti-apoptotic, and tissue-repairing capabilities. Meta-analysis indicates that MSC therapy significantly improves survival rates in II/RI animals while reducing tissue damage, inflammation, and oxidative stress [37]. Its effects are largely mediated through paracrine mechanisms, particularly through the release of extracellular vesicles such as exosomes. Exosomes derived from adipose-derived stem cells, particularly after resveratrol “pretreatment,” can more effectively regulate macrophage polarization toward the M2 phenotype and alleviate inflammation [25]. Exosomes derived from bone marrow MSCs can counteract ferroptosis by delivering miR-378a-3p to inhibit the SREBF2/HMGB1 axis [38].
Novel Delivery Systems and Nanomedicine
To enhance drug targeting and reduce side effects, smart nanocarriers have been developed. For example, the manganese dioxide nanosphere-loaded rapamycin system encapsulated by macrophage membranes achieves targeted delivery through its membrane coating while utilizing MnO₂ to catalyze ROS clearance. It also enables controlled release of rapamycin under ROS conditions to activate autophagy, thereby synergistically mitigating damage [39]. Additionally, pH-responsive cationic polymer-functionalized microspheres can efficiently clear cell-free DNA (cfDNA) enriched at injury sites, exerting anti-inflammatory effects by inhibiting M1 macrophage polarization [12].
Gas Molecular Therapy and Other Approaches
Molecular signals such as carbon monoxide releasers (e.g., CORM-2) [40], hydrogen, and methane (e.g., methane saline) exhibit anti-inflammatory, antioxidant, and anti-apoptotic properties. Methane- containing saline has been demonstrated to inhibit ferroptosis by activating the Nrf2/HO-1 pathway [41].
In summary, the comprehension of II/RI has progressed from a basic ischemic-hypoxic model to a multifaceted network that includes oxidative stress, inflammation, cell death, barrier disruption, and dysbiosis. Intervention strategies aimed at critical nodes such as Nrf2, NLRP3, SIRT3, GPX4, and the gut microbiota have expanded and shown considerable efficacy in preclinical studies.
However, challenges persist:
1. The high complexity of the mechanism necessitates that future research prioritize network integration and dynamic analysis over single pathways;
2. The translational gap demands the development of models that more closely reflect human pathophysiology (e.g., human- derived organoids [42]);
3. Individual differences: The distinctive characteristics of gut microbiota require treatment strategies that are both precise and personalized;
4. Therapeutic time window: Most interventions demonstrate effectiveness when administered prior to or immediately following reperfusion. The feasibility of clinical identification and timely administration warrants further investigation. Future research should prioritize the following areas:
1. Employing multi-omics technologies to delineate a comprehensive molecular landscape of II/RI, thereby identifying novel key targets;
2. Developing combination therapies or multifunctional nanomedicines that exhibit multi-target synergistic effects;
3. Investigating the potential of gut microbiota metabolites as diagnostic biomarkers and therapeutic agents;
4. Facilitating the translation of advanced therapies, such as stem cell products and engineered exosomes, into clinical trials. Through robust interdisciplinary collaboration, it is expected that advancements in basic research related to II/ RI will ultimately translate into clinical applications that enhance patient outcomes.
The authors declare no competing interests.
Data sharing is not applicable to this article as no new data were created or analysed in this study.

