Impact Factor : 0.548
- NLM ID: 101723284
- OCoLC: 999826537
- LCCN: 2017202541
Ivan Malčić1*, Dražen Belina2, Jasna Stioć Brezak3, Teodora Zaninović Jurjević4 and Željko Čolak5
Received: February 02, 2026; Published: March 06, 2026
*Corresponding author: Ivan Malčić, Zagreb Medical School, Medical Faculty Zagreb, Department of Pediatric Cardiology, Zagreb, Croatia
DOI: 10.26717/BJSTR.2026.65.010126
We present the report of disease in a child with severe restrictive cardiomyopathy (RCM) resulting in the development of veno-occlusive pulmonary arterial hypertension (PAH) and cardiopulmonary insufficiency with NYHA II-III. Due to borderline criteria for heart transplantation (HT), it was difficult to find justification for the procedure, although it was the only way to save the child’s life. The boy was monitored for hepatomegaly one year before admission. At the age of 11, significant dilatation in both atria, accompanied by a reduced cavity of both ventricles led to the recognition that the clinical symptoms were attributed to restrictive cardiomyopathy (RCM) with preserved left ventricular ejection fraction (LVpEF – LV with preserved EF). Because of the evidence suggesting that it is a secondary development of veno-occlusive PAH, before the HT decision it was required to clarify distinguish from reactive PH, in accordance with the latest 6WSPH criteria (6th World Symposium on pulmonary hypertension) [1]. Pulmonary hypertension is considered when the mean pulmonary artery pressure (mPA) is >20 mmHg, transcapillary gradient (PAWP) > 15 mmHg and pulmonary vascular resistance (PVR) ≥ 3 iWJ. Since the patient did not meet the criteria for passive (isolated) precapillary PAH (mPA>20 mmHg, PAWP >15 mmHg and PVR < 3iWJ) with preserved LVEF /IpcPH/LVpEF=Isolated precapillary PH with LVpEF/, but according to the same criteria, he had reactive, or combined veno-occlusive pulmonary hypertension (PVR ≥ 3iWJ) /CpcPH/LVpEF= Combined precapillary PH with LV preserved EF)/.
Diagnosis indicated that it was a contraindication for HT. This position is valid only if the discussion about possible reversible changes in PAS (Pulmonary artery small – the former name of arteriole) are ignored. Aware of the fact that HT is risky, but also of the probability that changes caused by the Kitajew phenomenon are reversible [2] contrary to precapillary PH caused by CHD (Euler Ljiljestrand fenomen) [3], we decided to proceed with HT. We are predicting a difficult postoperative course but which can be overcome with adequate measures (strict supervision of the Frank-Starling law) [4] with mechanical and pharmacology support. Encouraged by reports from the literature indicating a significantly higher incidence of passive postcapillary than reactive pulmonary hypertension as well as rapid regression of pulmonary hypertension after elimination of the cause of PAS self perpetuation [5-7], we decided on HT as the only perspective for the survival of a severely ill child. Our arguments also included a discussion on the irreversibility of PAH before the development of Eisenman’s syndrome (ES) (“gray area”, 6-8 iWJ) [8,9]. HT decision despite the inconsistency of the criteria for defining pulmonary hypertension and the type of pulmonary vascular resistance according to the previous international criteria’s [10-13], including the 6WSPH criteria [1,8,14,15]. Our arguments were accepted by the EUROTRANSPLANT committee. A HT was successfully performed and boy was discharged home with good follow up more than > 10 years in good condition (NYHA I).
Keywords: Restrictive Cardiomyopathy; Isolated and/or Reactive Veno-Occlusive Pulmonary Hypertension; Heart Transplantation; COR/LOE Discussion
Abbreviations: RCM: Restrictive Cardiomyopathy; PAH: Pulmonary Arterial Hypertension; HT: Heart Transplantation; PVR: Pulmonary Vascular Resistance; PAS: Pulmonary Artery Small; CHD: Congenital Heart Disease; ES: Eisenmenger Syndrome; RAAS: Renin-Angiotensin-Aldosterone System; NP: Natriuretic Peptide; WSPH: World Symposia on Pulmonary Hypertension
Unlike in the past when in paediatric cardiology the main cause of pulmonary hypertension was L-D shunt at the level of the ventricles and large blood vessels, today the most common cause of PH is considered to be left ventricular diseases. In CHD (congenital heart disease) with L-D shunt, precapillary pulmonary hypertension always develops with gradual transformation of small pulmonary blood vessels until the development of Eisenmenger syndrome (ES). In these circumstances, the degree of precapillary pulmonary hypertension is always discussed, while postcapillary pulmonary hypertension is rarely or not discussed at all. According to the 6WSPH guidelines [1,15,16], pulmonary hypertension is considered as to be condition with mean pressure in PH > 20 mmHg, TPG > 15 mmHg and PVR ≥ 3 iWU. The automatic logic is that not all those who have such or even more significantly advanced PH can be considered candidates for HT, although according to the available literature this is not precisely stated anywhere. Here we would like to present a child who, due to severe RCM with LVpEF, had a high degree of pulmonary hypertension with clear criteria for reactive (precapillary) PH. This condition is designated as CpcPH/LHDpEF, and in our patient it can be read as reactive due CpcPH/LVpEF.
If we look at the definition according to the table recommended by the 6WSPH studies, our patient is classified in the reactive pulmonary hypertension group (therefore, with contraindications for TS). However, it seemed to us that no distinction was made between reversible (isolated) and irreversible (combined-reactive) PH, so we sought the possibility of determining the degree of histological changes on PAS, without lung biopsy (the method is not recommended) [16] and without liquid biopsy. Therefore, we decided to investigate the possibilities of determining the level of these changes based on the findings of cath. study and the definition of PH, according to the Heat-Edwards (HE) classification, as well as the exact relationship of the recognised to the “gray area” which is clearly defined in the knowledge of PH in CHD with L-D shunt. An additional question was to define CpcPH/ LHD also as combined PH (C) (which means isolated postcapillary + reactive precapillary). Where in that definition are the histological changes that mark the border between reversible and irreversible?
An 11-year-old boy was observed for hepatomegaly one year before the primary diagnosis was established. With the gradual development of heart failure, he had NYHA II at admission, and while waiting for the heart transplant, he progressed to the NYHA III. At admission, the diagnosis of RCM was established by echocardiography with extremely enlarged atria, mostly the right one, and reduced ventricular cavities, especially the right. Very severe tricuspid regurgitation with a velocity corresponding to a gradient of 90 mmHg with dilatation of the hepatic veins and inferior vena cava (Figures 1A & 1B). The diagnosis was also confirmed by magnetic resonance imaging (Figure 1C) showed thickening of the anterior wall of the right ventricle and flattening of the interventricular septum (Detailed description of the findings in Figure 1C). Given the possible etiology of RCM, it was necessary to perform extensive laboratory workup (idiopatic RCM, Sy. Löffler, infiltrative metabolic diseases, amiloidosis, genetic testing etc.), but the etiology was not clear. We decided to perform cath. study and myocardial biopsy the left ventricle. RCM was confirmed with previous myocarditis as a potential cause. According to this finding, it was RCM, probably after a previous myocarditis, with a high percentage of connective tissue in the cardiac musculature (20-40%). Bundles of connective tissue separated the musculature and thus reduced the distensibility of both ventricles (see Figure 2C).
Based on these findings, it was clear that this was RCM with severe PH/LHDpEF. Following the 6WSPH guidelines from 2018-2019 [1,8,14,15], the first diagnosis was determined as reactive PAH also CpcPH /LHDpEF) Hemodynamic features, oximetry analysis and hemodynamic features recorded by MRI are shown in Table 1. (Table 1) The exact MRI results showed differences in the hemodynamic parameters of the right and left ventricles that can be interpreted as a disturbance of the Frank-Starling law balance [4], which is manifested by significant preload due to pulmonary hypertension, but is compensated by still well-maintained left ventricular function. Although the latest 6WSPH guidelines present the definition of reactive pulmonary hypertension as shown in Table 2., we could not ignore the possibility that the PVR is not higher than the criteria for the “gray area”, which is by definition the ratio of systemic and pulmonary vascular resistance, actually an indicator of the beginning the correction of CHD with L-D shunt in which PVR > 6 iWU is found. With the calculation shown in Table 2, we searched for where the actual PVR in our patient is actualy located according to the mathematical principle. We did this with the belief that all previous definitions did not clearly show at what level thFe PAS is converted into the so-called “gray area” (> 6 iWU). We started from the knowledge that the calculation of the quotient Rp/Rs can be an orientation point, as well as from the definition of ES (Rp/ Rs = 1 or > 1) [6,8].
Note: PAP-pulmonary artery pressure, RV-right ventricle, TPG-transpulmonary gradient, Qp–pulmonary flow(q=quantity), Qs – systemic flow, Rs:Rs – the ratio of systemic (s) and pulmonary resistance (R), iWJ – Wood units (indexed (i) to the BSA), MR – magnetic resonance, EDV-end-diastolic volume, ESV- end-systolic volume, SV- (stroke volume, EF – ejection fraction
According to this logic, in our patient, PVR: SVR = 0.39:1, which is below the values mentioned as a state with advanced proliferation in CHD with L-D shunt with conversion degree > III (HE) [8,16]. So based on these considerations we decided to proceed with HT. Table 2 shows a calculation how to determine the place according to the “grey area”. We were guided by the belief that histological changes in numerous publications can be very quickly reversible. In one observation, it was found that in mitral insufficiency, the systolic pressure in the pulmonary artery was > 70 mmHg, and that a small number of patients (17%) had by definition CpcPH, and later it was proved that reactive capillary hypertension was shown in a relatively small number of patients [17-19]. We have also had similar experiences from our practice [20], as well as the knowledge that PH has various forms of histological changes on the different parts of the lung [21] (Table 3). Our basic question was how the PVR ≥3iWJ criterion alone can be sufficient to sugest irreversible changes in the precapillary system, and the same value is far from the “gray area” that manifests itself as a value of 6-8 iWJ in CHD with L-D shunt [6-8,15]. It is also known that in veno-occlusive PAH, the pressure in the lungs increases for a long time due to pericapillary edema [21], and not due to intracapillary conversion). We supported the pro/con dilemma with the following reasoning: according to the 6WCPH definition, the criteria for HT are not met due to increased PVR calculation (5.8 = ≥3 iWJ).
Table 2: Which group does our patient belong to? Should we performe
a HT?
(Calculation based on the Rs:Rp quotient according to the ES definition).
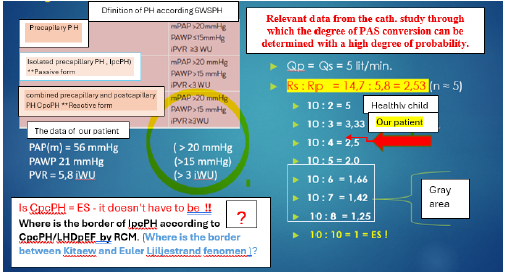
Table 3: A graph of the PVR calculation in a healthy child and in our
patient.
It is clear that the PVR in our patient is below the “gray area”.
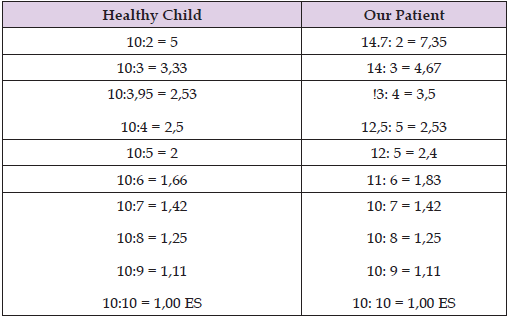
However, if we take the experience in children with long-term L-D overflow with consequent increase in pulmonary, according to the surgical protocol, the “gray area” for surgery is the value of PVR between 6-8 iWJ (provided that Eisemenger syndrome is defined as the quotient PVR: SVR = 1:1 or > 1). According to the described values, the PVR in the child with RCM was far below the values mentioned as a state with advanced proliferation in CHD with L-R flow with a conversion degree > of III (HE). Based on this reasoning, EUROTRANSPLANT accepted our arguments and we decided on HT. We were aware that we would face a great struggle for the post-transplant period, especially in the first days, which turned out to be true. The HT transplant was performed on August 3, 2014. using the Shumway biatrial technique [22]. Spontaneous establishment of sinus rhythm was achieved, with cardiac ischemia of 118/min. Aware of the borderline values according to the recommended criteria, inotropic support with milrinone and isoprotenerol (sympathomimetic) was first introduced. The first complications started immediately after being transferred to the ICU. Ultrasound diagnosed of PAH on the basis of pulmonary insufficiency and severe over the tricuspid orifice with a speed of 4.5m/ sec, along with pathological dilatation of the VCI and hepatic veins. Was consistent with severe imbalance of the Frank-Starling law (F-S l) (high preload and high afterload caused by a high dose of inotropic drugs (Isoprotenerol), especially in combination with Milrinone.
Consecutively, the cardiac output was significantly reduced, so heart failure developed rapidly. Peripheral vasoconstriction was treated with dobutamin, hidralasine and nitropruside. After vasodilation drugs we saw a gradual drop in lung pressure. Subsequent use of VA-ECMO support lead to the stabilization of F-S-l.
Along with medical support with peripheral vasodilators and iNO, a central V-A ECMO system is installed with a support of 3.5 lit/ min. After 18 days we changed VA-ECMO to right ventricular support device with an oxygenator (PLS Quadrox Maquet). Three weeks after surgery support with this device is discontinued and V-V ECMO was used for a week. This difficult treatment with mechanical support lasts a total of one month after the initial heart transplantation, with significant medication modification. Due to renal insufficiency and hyperkalemia, hemodialysis was introduced. Oral ACEi (angiotensin- converting enzyme inhibitors) such as Captopril or Enalapril are gradually introduced. In this way, the effects of the renin-angiotensin- aldosterone system (RAAS) are also reduced. These vasodilating drugs enhanced the beneficial effects of natriuretic peptide (NP) on fluid and sodium excretion. Due to sinus bradycardia, an electrostimulator was implanted, which further improves the child’s general condition, and after several hemodialysis procedures, kidney and liver function normalize.
Steroid therapy (prednisolone) and immunosuppressants (Tacrolimus) were used as usual, are introduced to stop rejection. One month after transplantation, an early myocardial biopsy was performed and the rejectin grade was IR(IA), and 3 months after transplantation, grade 1R. All other myocardial biopsyes findings at 3-month, 6-month, and 5x intervals over a period of one year were normal and there were no signs for rejection. The child gradually recovered with a normal mental status, and the pressure in the lungs gradually decreased to normal values two months after HT according to the ultrasound findings; minimal tricuspid insufficiency, physiological pulmonary valve insufficiency, one-dimensional representation of the pulmonary valve motion speaks against pulmonary hypertension, the right ventricular cavity was significantly reduced compared to the left ventricle. Due to steroids, Cushing’s syndrome developed with consequent iatrogenic arterial hypertension. Next comes maintenance of the condition according to the protocol and therapy with ARNi group of medication. The boy was discharged to home care with long-term good condition (NYHA I) (Tables 4 & 5).
Pulmonary hypertension was almost know two centuries ago [23-25]. Interest increased sharply from the mid of the 20th century, when the first definitions of ES [6] were adopted, and then, in its own way, a mathematical definition of ES (equalization of pressures in the pulmonary and systemic circulation) (Qp = Qs, Rp: Rs = 1 or > 1) [7]. The first classification of pathological changes in the small blood vessels of the lungs (which were then called arterioles) was adopted, and Wood directed the first research initiatives towards vasoconstriction factors [7,21]. The HE classification [25] includes six possible conversions of small blood vessels of the lungs, the first three being considered reversible, and the others irreversible [25]. Discussions about the possible reversibility of the process of conversion to the various histological description that represents the third degree of HE (progressive intimal fibrosis and intimal occlusion) provides important conclusions and serve significantly in the practice of CHD with L-D shunt at the level of large blood vessels and ventricles [25]. Between complete irreversibility (PVR > 8 iWU) and possible reversibility (PVR < 6 iWU) is also included the level of PVR of 6-8 Wood units which is still considered the so-called “gray area”. The possibility of calculating the flow through the pulmonary and systemic circulation can be attributed to Fick’s principle which was realized on the basis of Hüfner’s number [8,26] (who lived in the same time period at the end of the 19th century).
There is no doubt that until recently all attention was focused on precapillary PH, both diagnostically and as a guideline for therapy, until the majority of CHD with flow at the level of the ventricles and large blood vessels was eliminated by timely surgery in the majority of such patients thanks to the simultaneous rapid development of paediatric cardiac surgery. In precapillary conversion the term “arteriole” was used longer represented, which gradually receiving the name “small pulmonary vessel” (PAS- pulmonary artery small) and in some authors we also find the name “distal vessels. This change in the nomenclature comes together with a change in the paradigm in paediatric cardiology, because there are references that prove that the main reason for PAH is no longer L-R shunt in CHD, because the main reason for the development of PAH in childhood is the left heart disease (PH/LHD). In this way, veno-occlusive PH, which first manifests as isolated precapillary pulmonary hypertension (IpcPh/LHD), due to prolonged duration, turns into combined, or reactive pulmonary hypertension (CpcPH/LHD. Such a condition is too often associated with severe histological transformation at the postcapillary level (PAS - self perpetuation) is designated as combined PAH (precapillary + postcapillary). If we accept this definition as irreversible, we should abandon further consideration [14,15,21].
PH marked as CpcPH/LHD, more precisely in our case reactive pulmonary hypertension due to restrictive cardiomyopathy of the left ventricle with preserved EF (CpcPH/RCMLVpEF). Following by World Symposia on Pulmonary Hypertension (WSPH) which dealt with the problem of veno-occlusive pulmonary hypertension, even at 4WSPH both the criteria and the definition were confirmed according to which (Dana Point, 2009) [10,27] hypertension was again defined as any condition with a mean pressure in PA > 25 mmHg. However, if we refer to the latest criteria (6 WSPH 2019), when PVR > 20 mmHg it is considered a PH. With numerous examples and discussions about the relative benign course of PH due to obstructive lesions of the left ventricle, late correction of TAPVR, as well as literature references that find rapid recovery and regression of PH after surgical correction of the defect, they led us to consider whether the definition of reactive PH according to 6WSPH should also be understood as a contraindication for HT in our patient. All the aforementioned doubts had some sway in the discussion whether only a strict definition is important or whether it is important to consider the possibility of HT in the belief that in our patient the degree of small vessel obstruction does not reach the “grey area”, i.e. that the histological changes in PAS are still reversible.
In support of such doubts is the recognition that the early increase in PA pressure in veno-occlusive defects is caused by pericapillary oedema [21]. Based on the calculation of systemic and pulmonary resistance and their quotient (5,8 iWU) we realised that of PVR in our patient was below the “grey area” level. The calculation and explanation in Table 2 are mathematically correct, but a fatal outcome of HTTS could have occurred if we had not thoroughly prepared for postoperative care that includes not only strict adherence to F-S law [4] but also mechanical support and team work with anesthesiologists and cardiac surgeons. Although we appreciate the extensive and intensive scientific reading of all participants in the world symposia, as well as 6WSPH, we believe that the definition of CpcPH/RCM-LVpEF is only conditionally correct and allows for a more like approach presented in this paper. An additional question was to define CpcPH/LHD also as combined PH (C) (isolated postcapillary + reactive precapillary). Where in that definition are the histological changes that mark the border between reversible and irreversible? Here we also want to emphasize that an atmosphere of teamwork should be created according to the principles of COR/LOE (common of recommendation on the level of evidence). It is always easier to bear defeat together than when it falls on only one side, and human life is at stake.
With this review, we would like to stimulate a discussion on a possible theoretical assumption that the histological transformation of small blood vessels in the lungs in veno-occlusive pulmonary hypertension (which is currently the main cause of PAH) needs to be subjected to a more thorough analysis. The purpose of this analysis would be to determine the irreversibility or possible reversibility of the histological changes and thus open up a safer treatment option for most patients with such a condition. Today’s diagnostic and thresholding options must also include the so-called liquid biopsy, because lung biopsy has become an obsolete method. Such an approach would also help in the development of a medical approach as well as in the prevention of conditions that, due to a preformed heart, eventually collapse (failing Fontan) due to the lack of a pulse wave. Borderline and incompletely clear cases are often left in the care of only one doctor, who then bears too much responsibility. Therefore, the presentation of the course of the disease in our patient could also be an example of the proper application of the COR/LOE principle [28,29].

