Impact Factor : 0.548
- NLM ID: 101723284
- OCoLC: 999826537
- LCCN: 2017202541
Meirong Gu1#, Wenyi Chen1#, Xiaowen Wu1, Tianjun Wu1, Chengcheng Song2, Meilin Weng3, Wankun Chen3,4, Xin Zhang1,5* and Yiling Qian1,6*
Received: February 16, 2026; Published: February 26, 2026
*Corresponding author: Yiling Qian, Department of Anesthesiology, The Affiliated Wuxi People’s Hospital of Nanjing Medical University,
Wuxi People’s Hospital, Wuxi Medical Center, Nanjing Medical University. #299 Qingyang Road, Wuxi, Jiangsu, China
Xin Zhang, Department of Anesthesiology, The Affiliated Wuxi People’s Hospital of Nanjing Medical University, Wuxi People’s Hospital, Wuxi
Medical Center, Nanjing Medical University. #299 Qingyang Road, Wuxi, Jiangsu, China
DOI: 10.26717/BJSTR.2026.64.010115
Background: Effective postoperative pain management is crucial for patients undergoing endoscopic submucosal
dissection (ESD) for early esophageal carcinoma. Traditional analgesia methods involve oxycodone or
sufentanil administration. However, the efficacy of oxycodone in this context remains uncertain. This study aims
to explore the transitional analgesia with oxycodone and sufentanil in ESD patients under sufentanil general
anesthesia.
Methods: 86 patients scheduled for ESD under sufentanil general anesthesia were randomly assigned to oxycodone
(O group, n = 29), sufentanil (S group, n = 30), or normal saline (N group, n = 26). Before surgery completion,
O group received oxycodone (0.1 mg/kg), S group received sufentanil (0.1 μg/kg), and N group received
saline. Patient conditions, anesthetic medication, vital signs, recovery duration, decannulation time, adverse reactions,
Visual Analog Scale (VAS) scores, and pain relief incidence were recorded.
Results: O group showed reduced VAS scores post-surgery compared to S and N groups. S group had reduced
VAS scores postoperative 0 h and 12 h at rest compared to N group. No significant differences were observed
in patient conditions or intraoperative anesthetic dosages. O group had prolonged recovery and decannulation
times but similar overall recovery. No significant differences in severe vomiting were noted.
Conclusion: Transitional analgesia with oxycodone and sufentanil provided more prolonged and comprehensive
postsurgical analgesia compared to sufentanil in ESD patients, with no significant side effects. These findings
inform transitional analgesia strategies in this patient population.
Keywords: Postoperative pain; Esophageal ESD; Oxycodone; Sufentanil
Abbreviations: American Society of Anesthesiologists; ANOVA: One-Way Analysis of Variance; BIS: Bispectral Index; BMI: Body Mass Index; BP: Blood Pressure; CONSORT: Consolidated Standards of Reporting Trials; ECGs: Electrocardiograms; ESD: Endoscopic Submucosal Dissection; HR: Heart Rate; MAP: Mean Arterial Pressure; NBP: Noninvasive Blood Pressure; N group: Normal Saline Group; O group: Oxycodone Group; S group: Sufentanil Group; PONV: Postoperative Nausea and Vomiting; SPO2: Pulse Oxygen Saturation; VAS: Visual Analog Scale
Esophageal cancer, characterized by its malignant nature and prevalence, arises from the esophageal epithelium, posing a significant clinical challenge due to its insidious symptoms [1]. The diagnosis of early esophageal carcinoma remains intricate, but advancements in digestive endoscopy, particularly endoscopic submucosal dissection (ESD), have substantially improved diagnostic rates and therapeutic efficacy, particularly in cases of early esophageal cancer. ESD offers the advantages of low trauma, high resection rates, and enhanced safety [2]. Despite these advancements, postoperative pain following ESD is a frequently overlooked clinical complication [3]. The potential for moderate to severe pain [4] not only extends hospitalization but also adversely impacts the quality of life for affected individuals [5]. Timely intervention during the perioperative period is crucial to mitigate the risk of significant post-ESD pain in patients with early esophageal cancer. Sufentanil [6], a primary drug for general anesthesia and postoperative analgesia, primarily targets μ receptors, exhibiting well-established analgesic efficacy and safety [7]. However, escalating drug dosages may lead to undesirable side effects such as respiratory depression [8], nausea, vomiting, and urinary retention [9]. Oxycodone, a semi-synthetic opioid derived from tiabaine, activates μ receptors, effectively alleviating acute incision pain [10]. Moreover, its activation of κ receptors, widely distributed in visceral smooth muscles, raises the threshold of visceral pain [11]. Despite debates on oxycodone’s efficacy in clinical practice, animal experiments and clinical studies support its superiority [12], leading to its increased perioperative use. In comparison to sufentanil, oxycodone boasts a quicker onset and prolonged action duration [13]. Combining oxycodone with sufentanil may mitigate adverse reactions while enhancing overall benefits [14]. Although ESD is minimally invasive, acute incision pain and visceral pain remain common in clinical scenarios [15], demanding effective resolution. A noticeable gap exists in the literature concerning oxycodone’s application for transitional analgesia in patients with early esophageal carcinoma undergoing ESD under general anesthesia.
This study aims to establish the hypothesis that sufentanil can effectively alleviate postoperative pain in ESD patients with early esophageal carcinoma. Additionally, we seek to compare the analgesic effects of oxycodone with those of sufentanil in patients undergoing ESD under general anesthesia. Through this investigation, we aim to contribute valuable insights to the optimal perioperative management of pain in this specific patient population.
The present study received approval from the Institutional Research Ethics Committee of Wuxi People’s Hospital, Nanjing Medical University, and was registered in the Chinese Clinical Trial Registry (ChiCTR2400083623). Informed written consent was obtained from all eligible patients. The study adhered to the principles outlined in the Helsinki Declaration and followed the Consolidated Standards of Reporting Trials (CONSORT) guidelines. A total of 96 adult patients with American Society of Anesthesiologists (ASA) physical status I or II, scheduled for elective endoscopic submucosal dissection (ESD) under general anesthesia at the Affiliated Wuxi People’s Hospital of Nanjing Medical University between June 2022 and May 2023, were recruited. Exclusion criteria included severe systemic diseases, heart disease, hepatopathy or nephropathy, coagulation dysfunction, submucosal infiltration, acute or chronic pain, alcohol or opioid dependence, and allergy to trial agents. Patients were randomly assigned into the oxycodone group (Group O), sufentanil group (Group S), and saline group (Group N) using a computer-generated randomization list, ensuring no premedication. Routine monitoring in the operating theater included noninvasive blood pressure (NBP), pulse oxygen saturation (SPO2), electrocardiograms (ECGs), and bispectral index (BIS). Cannulation via the median cubital vein of the forearm with a 20G venous trocar needle was performed, followed by induction of general anesthesia with midazolam 2 mg, sufentanil (0.4 μg/kg), propofol (2.5 mg/kg), and cis-atracurium (0.25 mg/kg).
Endotracheal intubation was carried out five minutes later using a Glide Scope. Maintenance of general anesthesia involved propofol (4–6 mg/kg/h), remifentanil (0.1–0.3 μg/kg/min), and cis-atracurium as needed, with anesthesia targeted to maintain BIS between 40 and 60, ensuring no body movements throughout the operation. Blood pressure (BP) and heart rate (HR) were stabilized within ± 20% of baseline. Thirty minutes before the operation’s conclusion, oxycodone 0.1 mg/kg (diluted to 1 mg/ml with normal saline) was administered in Group O, sufentanil 0.1 μg/kg (diluted to 0.1 μg/ml with normal saline) in Group S, and an identical volume of normal saline in Group N. Mean arterial pressure (MAP), heart rate (HR), and pulse oxygen saturation (SPO2) were recorded at various time points: T1 (baseline, prior to general anesthesia), T2 (30 min before the operation’s termination, at the time of analgesic administration), T3 (end of operation), and T4 (5 min after intubation before returning to the inpatient department). Anesthetic dosages and postoperative reactions were documented, including depressed respiration, vertigo, lethargy, delayed emergence, and restlessness. Postoperative incision pain was assessed using the Visual Analog Scale (VAS) at T1, T2, and T3. Additionally, the incidence of postoperative nausea and vomiting (PONV) and remedial analgesia within 24 hours postoperatively were recorded.
GraphPad software (Boston, USA) was utilized for the statistical analysis of the data obtained in this study. Normal distribution of the presented data was assessed using the Kolmogorov–Smirnov test. Measurement data are expressed as the mean ± standard deviation, and intergroup differences were assessed using One-way Analysis of Variance (ANOVA). Additionally, the data were characterized using interquartile range. Categorical data, presented as absolute or relative effect sizes, were analyzed using the chi-square test or Fisher’s exact test. A significance level of P < 0.05 was considered statistically significant.
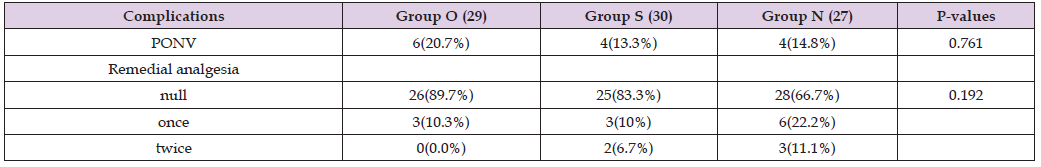
A total of 96 adult patients with ASA physical status I or II, aged between 55 and 78 years, weighing 47.5–93 kg, and with BMIs ranging from 15.3 to 29 kg/m2, were initially enrolled in the study. Three patients did not meet the inclusion criteria, and an additional three patients were excluded for other reasons, as illustrated in Figure 1. Consequently, 90 patients were enrolled, but one failed to follow up due to the inability to describe postoperative pain. In the oxycodone group, 29 patients were included, while 30 patients were recruited in the sufentanil group. In the control (normal saline) group, one patient failed to follow up due to intraoperative esophageal perforation, one patient presented with postoperative bleeding, and one was unable to describe postoperative pain, resulting in 27 cases ultimately included (Figure 1). The characteristics of the patients, as well as surgery and anesthesia profiles, were compared among the groups, revealing no statistically significant differences (p > 0.05) (Table 1). Medication quantities and perioperative complications are detailed in Table 2, with no observed statistical significance among the groups. No significant differences were noted in mean arterial pressure (MAP), heart rate (HR), pulse oxygen saturation (SPO2), and bispectral index (BIS) at time points T1, T2, T3, and T4 (Table 3).
Table 1: Demographic characterization of the patients, data were expressed as mean±SD, Group O Group Oxycodone, Group S Group Sufentanil, Group N normal saline group; BMI body mass index, ASA American Society of Anesthesiologists physical status.
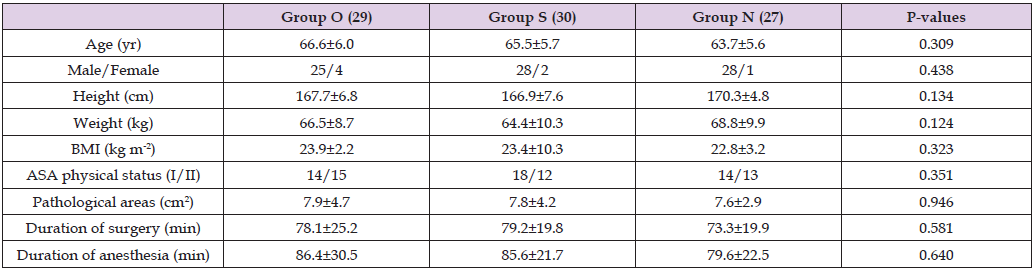
Table 2: Medication Quantities and complications during the treatment, data were expressed as mean±SD. Y yes, N no
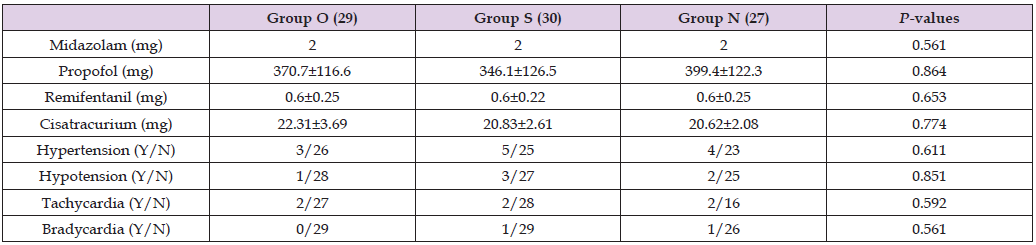
Table 3: Comparison of MAP, HR, SpO2 and BIS values at different time points, data were expressed as mean±SD. P-value < 0.05 statistically significant, HR heart rate, MAP Mean arterial pressure, SpO2 pulse oxygen saturation, BIS bispectral index.
No significant differences were noted in mean arterial pressure (MAP), heart rate (HR), pulse oxygen saturation (SpO2), and bispectral index (BIS) at time points T1, T2, T3, and T4 (Table 3). Evaluation of visceral pain at rest or in movement at 0h, 12h, and 24h post-operation revealed notable differences. In the rest state, the sufentanil group did not exhibit satisfactory analgesic effects at each time point, whereas the VAS score of the oxycodone group demonstrated significant statistical differences compared to the saline group. In the movement state, the sufentanil group showed slightly modest analgesic effects at 0h and 12h compared to the saline group, with no statistically significant difference at 24h. Conversely, the oxycodone group consistently demonstrated potent analgesic effects at each time point, markedly surpassing the sufentanil group (Figure 2). Figure 3 illustrates the quartile of data in each group.
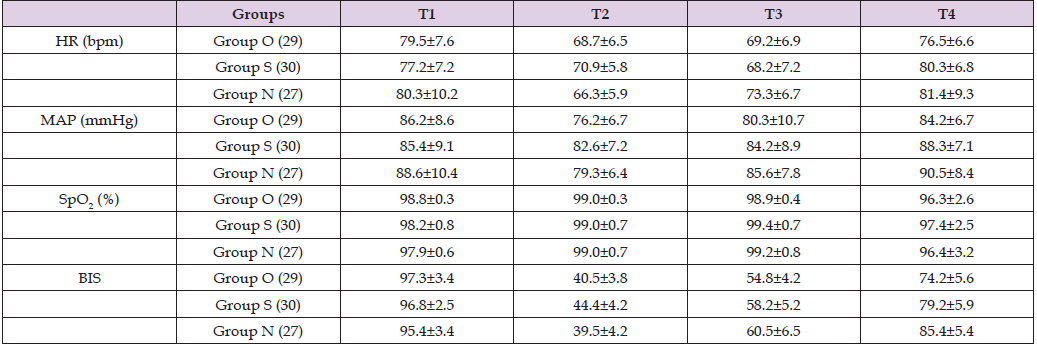
Evaluation of visceral pain at rest or in movement at 0h, 12h, and 24h post-operation revealed notable differences. In the rest state, the sufentanil group did not exhibit satisfactory analgesic effects at each time point, whereas the VAS score of the oxycodone group demonstrated significant statistical differences compared to the saline group. In the movement state, the sufentanil group showed slightly modest analgesic effects at 0h and 12h compared to the saline group, with no statistically significant difference at 24h. Conversely, the oxycodone group consistently demonstrated potent analgesic effects at each time point, markedly surpassing the sufentanil group (Figure 2). Figure 3 illustrates the quartile of data in each group. The comparison of reactions and complications during the recovery period in the three groups is presented in Table 4. The awakening and decannulation duration of the oxycodone group were slightly longer compared to the other two groups, showing statistically significant differences (Figure 4). However, no statistically significant differences were observed Postoperative malignant vomiting (PONV), a serious side effect of opioid medications. Despite the evident difference in postoperative VAS scores, the utilization rate of postoperative salvage analgesia showed no statistical significance in each group (Table 5).
Table 4: Comparison of reactions and complications during the recovery period, P-value < 0.05 statistically significant.
Table 5: Comparison of PONV and Remedial analgesia within postoperative 24 hours, P-value < 0.05 statistically significant, PONV postoperative nausea and vomiting.
Patients undergoing endoscopic submucosal dissection (ESD) for early esophageal cancer commonly experience substantial postoperative pain [16], necessitating effective pain management strategies [17]. In this study, we explored the analgesic efficacy and side effects of opioid analgesics, specifically oxycodone and sufentanil, during ESD surgery. Postoperative pain was assessed at 0h, 12h, and 24h using Visual Analog Scale (VAS) scores [18]. The results revealed that the oxycodone group consistently exhibited superior analgesic effects compared to the sufentanil and saline groups at all time points, regardless of rest or movement status (Figure 2). Quartile descriptive statistics further indicated that, compared with the sufentanil group, the oxycodone group demonstrated more significant VAS scores in the movement state at 0h and 12h postoperatively (Figure 3). Notably, both oxycodone and sufentanil, as opioid agonists, have been recognized as classic perioperative analgesics, with distinct receptor activation profiles—oxycodone primarily activating the κ receptor [19] and sufentanil predominantly activating the μ receptor [20]. Our findings align with existing research, emphasizing oxycodone’s superior analgesic effects [21].
The study focused on ESD patients experiencing inherent visceral pain [22], where oxycodone’s κ receptor activation demonstrated efficacy. Administering low doses of both oxycodone (0.1 mg/kg) and sufentanil (0.1 μg/kg) at 30 minutes post-operation demonstrated comparable efficacy. Oxycodone exhibited advantages within the first 24 hours after surgery, showcasing a longer analgesic duration [23] than sufentanil [24], encompassing the entire 24-hour period, while sufentanil’s effect lasted only up to 12 hours. Oxycodone also displayed efficacy in both rest and movement states [25], suggesting improved sleep and sedation at rest, potentially contributing to better recovery [26]. Additionally, oxycodone demonstrated superior analgesic effects in the movement state within 12 hours after surgery, facilitating early patient mobility and accelerating Enhanced Recovery After Surgery (ERAS) recovery [27]. Concerning side effects, decannulation time and recovery time were statistically significant in the oxycodone group compared to other groups, without causing delayed recovery. This could be attributed to oxycodone’s sedative effect through κ receptor activation [28]. Postoperative malignant vomiting (PONV) [29], a serious side effect in this patient population [30], did not exhibit statistical differences in the oxycodone group compared to the other groups. Despite consistent high VAS scores in the saline group, there were no statistical differences in the utilization rate of postoperative salvage analgesia. This aspect may warrant further investigation into the pain relief process in the ward. Sufentanil, while exhibiting analgesic effects, appeared suboptimal compared to oxycodone in this study. Limitations include a predominantly male participant population, necessitating further investigation into potential gender differences in oxycodone metabolism in female patients [31]. Moreover, surgical factors such as lesion location and circumference ratio, which may influence pain [32], were not comprehensively explored and warrant future investigation.
In conclusion, our study demonstrates that, when compared with sufentanil, oxycodone offers more prolonged and comprehensive analgesic effects in patients undergoing endoscopic submucosal dissection (ESD) for early esophageal carcinoma. Importantly, this enhanced analgesic efficacy does not come at the cost of increased side effects. The intravenous injection of oxycodone administered 30 minutes prior to the conclusion of the operation consistently provided superior analgesic effects for patients undergoing ESD under general anesthesia. This transition from anesthesia with sufentanil to analgesia with oxycodone represents a promising clinical approach. However, to establish its widespread applicability, further validation through multi-centered large-sample clinical trials is imperative. These findings contribute valuable insights into optimizing pain management strategies for patients with early esophageal carcinoma undergoing ESD, highlighting the role of oxycodone transitionally combined with sufentanil therapy in enhanced recovery in digestive endoscopic surgery.
We thank all the participants of this study. Special thanks to Changhong Miao from Fudan University for his contribution of the project design, Pan Li and Ying Zhuang for their contributions to the edition of this manuscript.
Y.Q and X.Z: Designed the study, wrote the protocol, and were the guarantors of this work and had full access to the data. M.G and W.C: Perform the clival trial and data collection. X.W, T.W, M.Land W.C: data collection and analyses. All authors contributed to the article and approved the submitted version.
The study was funded by National Natural Science Foundation of China, (82201378 Y.Q) and Outstanding Young Medical and Health Talents of Wuxi (HB2023005 Y.Q). It was also funded by National Natural Science Foundation of China (82271251 X.Z), Jiangsu Distinguished Medical Expert Project (X.Z) and Jiangsu Health Innovation Team Project (X.Z).
All data applied or analyzed in the present study are available from the corresponding author on reasonable request.
This randomized double-blind controlled study was conducted at the Affiliated Wuxi People’s Hospital of Nanjing Medical University between June 2022 and May 2023, with the approval from the Institutional Research Ethics Committee of Wuxi People’s Hospital, Nanjing Medical University, and the registration of the Chinese Clinical Trial Registry (ChiCTR2400083623). The study adhered to the principles outlined in the Helsinki Declaration and followed the Consolidated Standards of Reporting Trials (CONSORT) guidelines. The written informed consents were obtained from all patients before enrolled.
Not applicable.
No conflict of interest exists in the submission of this manuscript.

