Impact Factor : 0.548
- NLM ID: 101723284
- OCoLC: 999826537
- LCCN: 2017202541
Amar P Garg1*, Nilam Thakur2, Ajit k Saxena3 and Mehrukh Iffat4
Received: January 16, 2026; Published: February 18, 2026
*Corresponding author: Amar P Garg, Centre for Multidisciplinary Studies & Research, Swami Vivekanand Subharti University, NH-58, Subhartipuram, Meerut-250005, India
DOI: 10.26717/BJSTR.2026.64.010098
Male infertility is one of the major problems in human couples. It has been attributed to various factors like genetical, physiological, environmental and life style. The qualitative and quantitative problems in the production of sperms mainly accounts for male infertility. Sexual reproduction in humans promotes genetic variability through meiosis, recombination, and fertilization, thereby enhancing evolutionary adaptability to changing environment. The process of sex differentiation begins during embryonic development and is governed by the presence of the SRY (Sex-determining Region Y) gene on the Y chromosome that triggers the differentiation of bipotential gonads into testes, leading to the secretion of testosterone and anti-Müllerian hormone, which direct male reproductive tract development. In the absence of the SRY gene, the gonads develop into ovaries, resulting in the formation of the female reproductive system. This review deals with the biology of male infertility and the factors that govern it. The speculations of elimination of male populations have no sound scientific foundation and the male population reduction due to Y chromosome gene loss is unexpected. However, the fertility rates will decline due to lifestyle, environment, and socioeconomics rather than chromosome shrinkage.
Keywords: Biology, Male Infertility, Sex Chromosomes, Sex Hormones, Spermatogenesis, Y Chromosome Genes
Infertility has been defined as “conception failure after twelve months of unprotected intercourse”. Major global population surveys reveal that 5 to 20% of couples experience infertility, affecting approximately 72.4 million people [1]. World Health Organization has estimated approximately 9% of couples globally having primary fertility issues. These issues are due to male factors in 50% of cases where couples are struggling with sub-fertility, with men being solely responsible in 30% of instances and women co-contributing in 20% of cases [2]. Infertility encompasses a broad range of manifestations, including congenital or acquired genitourinary issues, hormonal imbalances, and immune system factors, as well as quantitative and qualitative problems with sperm production, impacting approximately 7% of the global male population [3]. Impotence in conception in a woman of normal reproductive health by a male partner is referred to as male infertility. Approximately 30 million males are affected by infertility in the region of Central Eastern Europe and Australia exhibiting the highest male infertility rates, respectively ranging from 8 to 12 % and 8 to 9 % [4].
Reproduction is a fundamental biological process that ensures the continuity of life by producing new individuals from parental organisms. In higher organisms, reproduction is primarily sexual, involving the fusion of male and female gametes to form a zygote, which subsequently develops into a new individual. Sexual reproduction promotes genetic variability through meiosis, recombination, and fertilization, thereby enhancing evolutionary adaptability [5]. The process of sex formation, also known as sexual differentiation, begins during embryonic development and is governed by genetic, hormonal, and environmental factors. In mammals, the presence of the SRY (Sex-determining Region Y) gene on the Y chromosome triggers the differentiation of bipotential gonads into testes, leading to the secretion of testosterone and anti-Müllerian hormone, which direct male reproductive tract development. In the absence of the SRY gene, the gonads develop into ovaries, resulting in the formation of the female reproductive system [6]. At the cellular level, reproduction involves gametogenesis, where spermatogenesis produces motile spermatozoa in males and oogenesis generates oocytes in females. These gametes unite during fertilization to form a diploid zygote, restoring chromosomal number across generations [7]. The interplay of sex chromosomes (XX in females, XY in males), gonadal development, and endocrine regulation determines phenotypic sex and sexual characteristics.
Aberrations in these processes can result in disorders of sex development (DSDs), which highlight the delicate balance of genetic and hormonal pathways in reproduction and sex formation. Sex formation in living organisms is the biological process that determines whether an individual develops as male, female, or intersex. It is regulated by genetic, chromosomal, and hormonal mechanisms that differ among species. In humans and most mammals, sex is determined by the sex chromosomes, XX for female, and XY for male. The Y chromosome carries the SRY (Sex-determining Region Y) gene, which triggers male development. Initially, embryos have bipotential gonads that can develop into testes or ovaries. Presence of SRY gene leads to testes formation. In absence of SRY development of ovaries will take place. Testes produce testosterone and anti-Müllerian hormone (AMH). Testosterone causes development of male reproductive ducts and external genitalia while AMH causes regression of female reproductive ducts. In the absence of these hormones, the embryo follows the default female pathway. At puberty, sex hormones (estrogen, progesterone, and testosterone) regulate the appearance of secondary sexual traits like breast development in females or facial hair in males. Sometimes, mutations, chromosomal abnormalities (e.g., Turner’s syndrome XO, Klinefelter’s XXY), or hormonal imbalances lead to differences in sex development (DSDs).
The male reproductive system can be broadly classified into internal and external structures. The internal structures include the testis, vas deferens, epididymis and prostate gland whereas external structures comprise externally visible structures like penis and scrotum. They are highly vascularized and help to promote production of male specific hormones and sperms and their transport during ejaculation [8]. They can also be divided into primary and secondary organs. The primary organs consist of gonad i.e., the testis responsible for gamete and hormone production whereas the secondary organs comprising ducts and glands having important role in maturation, growth and transmission of gametes [9]. The primary reproductive organ, the testis consists of three tunics i.e., tunica vasculosa, tunica albuginea and tunica vaginalis. The thick tunica albuginea provides the structural support to the testis and sends several septa inside to form many lobules. Each lobule has two morphologically and functionally separated components. Tubular components include seminiferous tubules and the other intercellular portions are sandwiched between seminiferous tubules. The intertubular components provide support and immune responses and comprise mainly Leydig cells and other supporting cells like immune cells, fibroblasts, blood vessels and nerves [10]. Likewise, seminiferous tubules comprise 60 to 80 % of testicular volume [11].
Seminiferous tubules consist of two types of cells, namely, Sertoli cells and spermatogenic cells. The Sertoli cells provide mechanical support as well as nourish the various stages of developing cells during spermatogenesis [12-14]. These cells produce inhibin and activin hormones that have negative and positive feedback to Follicle Stimulating Hormone. The Sertoli cells are meiotically inactive in adulthood and control the various stages of release of sperms and phagocytose the degraded germ cells [15-17]). The epithelial cells of seminiferous tubules are divided into two functionally different regions by the blood testis barrier (BTB) [18,19]. The two obvious functions of BTB are
i) To separate the germ cells physically providing protection against immune system and
ii) To provide optical environment for meiosis process and sperm development [20,21].
Development of Male Reproductive System
The sexual differentiation of human embryos is automatically female. In contrast, the Y chromosome specifies phenotypic and reproductive system differentiation in males. The Y chromosome contains the sex-determining region (SRY) gene, which codes for testicular development. At maturity, the testes lay in the scrotum after descending from the posterior abdominal wall throughout development. The testes develop Sertoli cells, which produce MIS to induce regression of the Mullerian ducts, which form the female reproductive system. The testes also develop another important type of cells called interstitial cells of Leydig cells, are found in between the seminiferous tubules. They produce testosterone and are the major driver of male reproductive development. Testosterone in male is crucial for promoting the growth of the Wolffian ducts, which ultimately give rise to the testes, epididymis, seminal vesicles and vas deferens. Testosterone is also responsible for various other important functions in male including pubertal growth spurt, epiphyseal plate closure, muscle growth, erythropoiesis, bone density, deepening of the voice, male body development, and desire. The growth of the testes symbols the start of puberty in males, which occur between 11 to 13 years of age. The anterior pituitary releases FSH and LH in response to a sharp increase in GnRH from the hypothalamus. The growth and coloration of the penis and scrotum are caused by the stimulation of Leydig cells to produce more testosterone by LH.
The following stage is secondary sexual features, including growth of pubic, chest, axillary, and facial hair, voice deepening, and growth spurt [22].
Hormonal Regulation and Postnatal Development
After birth, a transient but significant activation of the hypothalamic– pituitary–gonadal (HPG) axis occurs during the first six months of life, known as mini-puberty. During this period, increased secretion of gonadotropins luteinizing hormone (LH) and follicle-stimulating hormone (FSH) stimulates Leydig cells to produce testosterone and Sertoli cells to promote germ cell maturation, laying the foundation for future fertility. This early hormonal surge is essential for penile growth, testicular enlargement, and the initiation of spermatogonial differentiation. The reproductive system then enters a quiescent phase throughout childhood until the onset of puberty, when reactivation of the HPG axis drives progressive maturation. At puberty, pulsatile release of gonadotropin-releasing hormone (GnRH) from the hypothalamus stimulates the anterior pituitary to secrete LH and FSH. LH promotes testosterone production by Leydig cells, while FSH acts on Sertoli cells to support spermatogenesis by regulating the seminiferous tubule microenvironment. Testosterone at this stage not only supports spermatogenesis but also drives growth of the penis, scrotum, prostate, and seminal vesicles, completing maturation of the male reproductive system. [23].
Spermatogenesis
Spermatogenesis is the most important stage in male fertility [24,25]. Any deviation in the natural course of spermatogenesis can lead to infertility. Spermatogenesis is the process by which diploid spermatogonia give rise to differentiated haploid germ cells into the seminiferous tubules. Spermatogenesis cycle takes around 16 days in human and four to five cycles for the further development and differentiation of spermatogenic cells into adult spermatozoa altogether taking 74 days in humans [26,27]. The spermatogenesis event comprises four phases where,
1) Mitotic cell division leading to proliferation and differentiation of spermatogonial cells into preleptotene spermatocytes (spermatogoniogenesis;
2) Meiotic cell division of spermatocytes giving rise to spermatids;
3) Transformation of round spermatid into adult spermatids (spermogenesis) and finally
4) elongated spermatids are released into lumen of seminiferous tubules (spermatogenesis) [28] (Figures 1 & 2).
Spermatogoniogenesis
The germ cell lines that are found in seminiferous tubules are all derived from primary germ cells (PGC) [29]. PGCs begin to form during 3rd week of intra-utertine life from endodermal cells and by the time of 5th week, they migrate and reach to the gonadal ridge. Due to the influence of Y chromosome, the gonadal ridge proliferates and forms the primary male sexual organs [30-32]. PGCs are called as gonocytes during the first trimester of mitosis. Thereafter, they halt at G3 phase of cell cycle and remain silent until birth and subsequently they transform into spermatogonia that remain in dormant stage until puberty. Spermatogenesis process begins after birth by the proliferation of spermatogonia [33]. The difference between sexual and non-sexual reproduction is meiosis. Eventually, meiosis causes diploid cells to produce haploid gametes. Nuclear division happens twice in a DNA replication cycle during mammalian meiosis. Classically, meiosis divisions occur in two stages. Two types of meiosis are I and II [34-36]. Through the kinetochore, the microtubules are joined to sister chromatids and moved to opposing poles during meiosis or subtractive division. The number of diploid to haploid chromosomes decreases as a result of this shift. In the equational division known as meiosis II, four daughter haploid cells are formed when the microtubules bind to the centromere’s kinetochore and split the sister chromatids [37,38].
Spermiogenesis
The maturation of spermatids from round to elongated spermatids and finally to mature spermatozoa is the last stage of spermatogenesis. This process is called spermiogenesis, and it is followed by the release of spermatozoa into the lumen of seminiferous tubules (spermiation). The main alterations that take place during spermiogenesis are the following: the acrosomal cap, which contains proteolytic enzymes required for sperm penetration of the ovum; the sperm tail, or flagellum, which points into the lumen and allows motility; the phagocytic removal of excess spermatid cytoplasm (referred to as the residual body) by Sertoli cells; and the release of mature spermatozoa into the lumen. Progressive maturation of spermatids is accompanied by progressive movement of more mature spermatids toward the lumen of the seminiferous tubule. Spermiogenesis is directed by the Sertoli cells that sustain and support developing spermatids and by the major endocrine regulators of Sertoli cells, FSH and testosterone [39].
Disruptions at any stage of male reproductive development can lead to disorders of sex development (DSDs), infertility, or reproductive pathologies. Mutations in SRY, SOX9, NR5A1, or androgen receptor (AR) genes can result in gonadal dysgenesis, ambiguous genitalia, or complete androgen insensitivity syndrome [40]. Defects in AMH or its receptor may lead to persistent Müllerian duct syndrome, whereas 5α-reductase deficiency results in under virilization of external genitalia despite normal internal reproductive structures. Environmental factors, including prenatal exposure to endocrine-disrupting chemicals such as phthalates and pesticides, have also been implicated in testicular dysgenesis and reduced spermatogenic potential in adulthood [41]. Moreover, failure of testicular descent (cryptorchidism) not only compromises fertility but also increases the risk of testicular cancer later in life. A clear understanding of the normal developmental processes of the male reproductive system is therefore essential for elucidating the mechanisms underlying these clinical conditions and developing strategies for diagnosis, management, and prevention.
Diagnosis of Male Infertility
The male infertility can be exclusively diagnosed by semen analysis where three important parameters are observed; sperm concentration, motility and appearance. As per WHO 2010 male infertility can be classified based on seminogram under following categories: Azoospermia - Absence of sperm in the ejaculate. It can be classified as obstructive azoospermia [OA] where absence of sperm in the ejaculate is detected as a result of problems in sperm delivery or non-obstructive azoospermia [NOA] where there is absence of sperm in the semen due to abnormal sperm production. NOA constitutes 60% of all cases of azoospermia.
Oligozoospermia- Less than 15–20 × 106 mLspermatozoa in the ejaculate
Severe oligozoospermia- Less than 5 × 106 mL spermatozoa in the ejaculate
Normozoospermia- Normal values of sperms in the ejaculate
Asthenozoospermia- Low levels of motility observed in less than 50% of sperms
Teratozoospermia- Less than 30% of sperms have normal morphology Aspermia- Failure of sperm in ejaculating semen [42].
Genetics of Azoospermia
About 1% of males experience azoospermia, or the absence of spermatozoa in their ejaculate, and there are 3 main types of etiology for this condition: malfunction of the hypothalamic-pituitary axis, primary quantitative spermatogenic abnormalities, and blockage of the urogenital duct. Congenital and acquired variables can be linked to each of these groupings. The spermatogenic process remains unaffected when there is bilateral distal or proximal occlusion of the ejaculatory ducts; this pathologic situation is known as Obstructive Azoospermia (OA). However, Non-Obstructive Azoospermia (NOA) is the result of either primary or secondary testicular failure. At least three distinct forms of testis histology may be present in NOA, a phenotypic manifestation:
(i) Maturation Arrest (MA) at various phases of Sertoli-Cell-Only Syndrome (SCOS),
(ii) Maturation Arrest (MA) at different stages of germ cell maturation (such as Spermatogonial and Spermatocyte Arrest [SGA, SCA]),
(iii) Hypospermatogenesis. A number of acquired conditions (such as orchitis, cytotoxic treatment, ejaculatory duct obstruction, CNS tumors, systemic diseases etc.) may lead to azoospermia and may account for approximately 35–40% of cases [43].
Next Generation Sequencing (NGS)-based Whole Exome Sequencing (WES) or gene panel sequencing have allowed the identification of a growing number of novel monogenic causes. The detection of monogenic defects of quantitative alterations of spermatogenesis due to primary testicular failure is increasing constantly, but their screening has not been introduced into the investigative workup of NOA men, so far. In about 70% of NOA without known acquired causes, the etiology remains unknown, and we refer to it as “idiopathic” NOA (iNOA) (Figures 3-5).
Male infertility can be caused by a number of factors, including cryptorchidism (the absence of one or both testes in the scrotum), varicocele (the unusual enlargement of the veins; pampiniform plexus within the scrotum), hormonal imbalances, excessive alcohol consumption, blockages or partly or complete absence of seminal pathways, chemotherapy effects or infections [44]. In general, the male infertility is divided into four primary etiological groups:
(a) Quantitative defects in spermatogenesis
(b) Ductal obstruction or dysfunction
(c) Disturbances in the hypothalamic-pituitary axis
(d) Qualitative defects in spermatogenesis [45]. Some of the common reasons of male infertility according to mechanism are listed in Table 1.
Hormonal Factor
The hypothalamic-pituitary-gonadal axis is identified as the male reproductive hormone axis. The pituitary, testicular, and hypothalamus glands are its three main constituents. The hypothalamic-pituitary- gonadal axis has a vital role in promoting sexual maturity, sperm production, and the development of secondary sex characteristics. Throughout the male’s life, it maintains spermatogenesis and sexual function. To stimulate the anterior pituitary, the hypothalamus secretes GnRH into the hypothalamo-hypophyseal portal system. GnRH is a peptide hormone that is pulsatilely released by hypothalamic neurons. By attaching itself to and activating a G protein receptor, it influences the gonadotrophs in the anterior pituitary. By increasing intracellular calcium and activating inositol 1, 4, 5-triphosphate (IP3), this causes the anterior pituitary to release FSH and LH. Prolactin, testosterone, Estradiol and estrogen all suppress GnRH [48]. Any aberration in the system can cause infertility. If the brain is unable to produce gonadotropic releasing hormone (GnRH), this disorder results in an absence of testosterone and stopping sperm production [49]. Absence of GnRH causes a group of disorders known as hypogonadotropic hypogonadism [50]. One of them is known as Kallmann syndrome, which is associated with a change in sense of smell and immaturity [43].
Similarly, the pituitary’s incapacity to produce sufficient amounts of luteinizing hormone and follicular stimulating hormone leads to a failure in stimulating the testes and to produce testosterone and sperm [51]. Reduced sperm production, libido and impotence can be caused. By increased prolactin. Hyperprolactemia leads to infertility in 11% of men with oligospermia [52].
Physical and Sexual Factor
Physical conditions may hamper the ejaculatory pathway and interrupt sperm production. Varicocele, a condition with enlarged spermatic veins, is among the most common causes of male infertility, impacting approximately 40% of men [53,54]. Testicular torsion can result to testicular damage by exerting pressure on the spermatic vessels and compromising testicular blood flow. Additionally, both acute and chronic infections of the genital tract are prevalent contributors to male infertility. The mumps virus can cause testicular atrophy, ensuing in infertility. Furthermore, sexually transmitted infections such as chlamydia and gonorrhea can lead to male infertility by causing blockages in the epididymis [55]. Numerous sexual dysfunctions have both psychological and physical origins. Examples of sexual activity- related issues include premature ejaculation, inability to ejaculate, and erectile dysfunction, commonly known as impotence [56,57].
Environmental Factor
Infertility in men can be attributed to exposure to hazardous chemicals in the workplace, including radiation, solvents, pesticides, adhesives, silicones, and related substances [58]. Exposure to radiation can lead to diminished sperm production, and in cases of high radiation levels, it may lead to complete sterility. Additionally, temporary lower sperm count has been associated to excessive sun exposure [59]. The recurrent use of substances such as cocaine and cannabinoids has been linked to a substantial decrease in sperm concentration and urinary testosterone levels in men [60].
Genetical Factor
Genetic variations have emerged one of the principal factors contributing to male infertility. Common genetic disorders found in infertile males are karyotypic abnormalities, single gene mutations or polymorphisms, gene copy number variations (CNVs) and deletions on long arm of the Y chromosome (Yq micro deletions). These genetic disorders can hamper the development of male urogenital tract during fetal life which disrupt the production and maturation of germ cells, or may result to the formation of non-functional spermatozoa. Male infertility can be caused by variety of factors, among them karyotypic chromosomal abnormalities and Yq micro deletions are the most prevalent genetic causes [61]. The next-generation sequencing (NGS) technique, specifically whole exome sequencing (WES), serves as a robust and unbiased technique for finding the genetic variations by focusing on the coding regions of the genome. WES encompasses approximately 1%-1.5% of the human genome, where around 85% of causative mutations are found [62]. Infertility remains a prominent public health issue among Nepalese population. Infertility is projected to be 4% in Bangladesh, 6% in Nepal, 5% in Pakistan, and 4% in Sri Lanka according to the World Fertility Survey [63]. Though it is a major health problem, it has never been studied at molecular level. Especially, genetic causes of male infertility have never been studied. Therefore, the present study will not only help to understand the genetic variations of male infertility but also opens a pathway to design a protocol of genetic testing in male infertility.
Genetic Abnormalities of Male Infertility
A genetic abnormality can cause of infertility in 15% of men suffering this condition [45]. However, a recent investigation involving a large, unselected cohort of patients revealed that only 4% of infertile males received a definitive genetic diagnosis, despite the comprehensive application of karyotyping, screening for azoospermia factor (AZF) deletions, and examination of cystic fibrosis transmembrane conductance regulator (CFTR) mutations [64]. The identification and characterization of critical genes linked with numerous forms of male infertility have been facilitated by advancements in technology and the introduction of innovative molecular techniques (Tables 2-4). This ongoing research builds upon the initial discoveries made in the mid-twentieth century. In the 1950s, the identification of an additional X chromosome in individuals with Klinefelter Syndrome marked the first indication of a genetic basis for male infertility. The pursuit of additional genetic factors intensified in the late 1990s, concentrating initially on specific deletions and mutations in the androgen receptor and CFTR gene [65]. The first identification of Y chromosome abnormalities occurred in 1995 [66].
Table 2: Timeline of the discovery of key genes involved in male infertility identified using technological innovations.
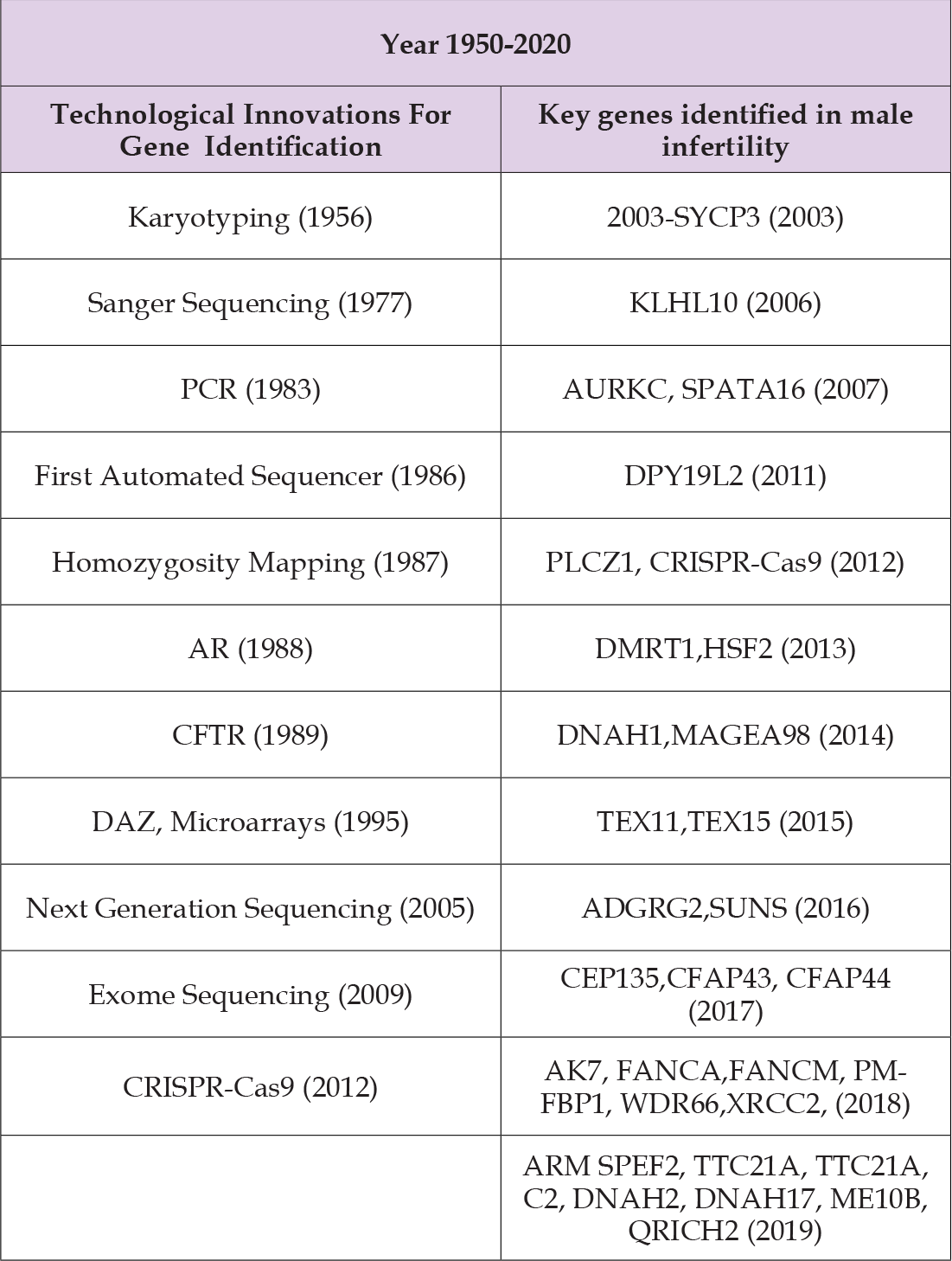
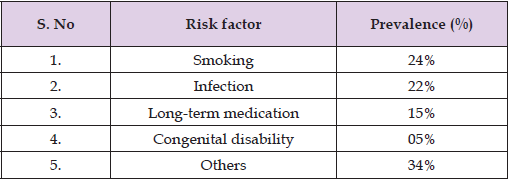
Table 3: Prevalence of risk factors contributing to male infertility in the studied population [123].
Cytogenetic Abnormalities
Chromosomal abnormalities encompass both numerical and structural chromosomal alterations that are sufficiently large (>4–5 Mb) to be observed microscopically. These aberrations are accountable for 5% to 10% of oligozoospermia [66] cases and 15% to 25% of cases involving nonobstructive azoospermia [67,68]. Karyotyping was the initial method employed to identify genetic abnormalities in infertile males and remains the predominant diagnostic test for male infertility in current practice.
Numerical Aberrations
Numerical chromosome abnormalities are a prevalent factor contributing to male infertility. The frequency of these abnormalities is inversely related to the sperm count in the ejaculate. Klinefelter syndrome (KFS) represents the most common numerical chromosomal anomaly and is the leading cause of azoospermia, occurring in 11% of azoospermic cases. Males with a 47, XXY chromosomal configuration are typically azoospermic due to dysgenesis of the seminiferous tubules, while those with mosaicism featuring a normal 46, XY cell line may present with oligozoospermia. Investigations into the additional chromosome in the sperm of Klinefelter men have indicated that it is eliminated during the process of spermatogenesis [69]. Although most offspring of these men possess normal chromosomal complements, there remains a considerable risk of chromosomal defects in the fetus [70]. The frequency of sex chromosome aneuploidy in Klinefelter mosaic sperm ranges from 1.5% to 7%, while in sperm from men with a nonmosaic 47, XXY karyotype, it varies from 2% to 45%. Males with 46, XX chromosomes are evaluated at 1 in 20,000 men. This condition arises from an abnormal translocation of Y chromosome material, including the sex-determining region (SRY), to the X chromosome during paternal meiosis. The presence of the SRY gene facilitates testicular differentiation; however, the absence of spermatogenesis is attributed to the lack of the long arm of the Y chromosome [71].
Structural Aberrations
Structural chromosomal aberrations are generally less harmful, occurring in less than 1% of cases, due to their propensity for repair in the subsequent phase of the cell cycle, and are often referred to as unstable genetic molecules. Robertsonian translocations (RT) are observed in approximately 1 in 1,000 individuals [72] and represent the most prevalent structural rearrangements among infertile men. These translocations involve the fusion of the long arms of two acrocentric chromosomes, specifically from Group D (chromosomes 13, 14, 15) and Group G (chromosomes 21, 22, and Y). The short arms of these chromosomes are typically lost, resulting in a chromosomal makeup of 45 chromosomes for the carrier. During meiosis, these chromosomes pair as a trivalent, leading to gametes that may possess an extra or missing long arm of DNA or may be chromosomally normal [71]. Reciprocal translocations happen when there is an exchange of genetic material between nonhomologous chromosomes, affecting approximately 1.17% of infertile men [73]. In this case, four chromosomes pair as quadravalents during meiosis, and their segregation results in a higher incidence of imbalanced chromosomes compared to RT carriers. The risk of meiotic imbalance is primarily influenced by the characteristics of the involved chromosomes and the locations of the breakpoints.
In prenatal diagnoses, the typical frequency of paternally derived translocation imbalances is around 12%, with many of these imbalances leading to fetal mortality [74]. Inversions, which are balanced structural rearrangements, are found in 10% to 15% of prenatal diagnoses and in 0.1% of infertile men. When two breaks occur on the same chromosome with 180° rotations before joining, there is occurrence of inversion. Inversions can be classified as paracentric, where both breakpoints are located on one arm of the chromosome and do not involve the centromere, or pericentric, where the breaks encompass the centromere and involve both the short and long arms of the chromosome [75].
Germline Mosaics
Germline mosaics refer to males who possess a normal somatic karyotype yet exhibit an abnormal cell line within their germ cells. Such cases are typically identified through testicular biopsy. Research indicates that between 1% and 17% of infertile men exhibit germline mosaicism [76]. The heightened incidence of chromosomal abnormalities observed in pregnancies resulting from intracytoplasmic sperm injection, as well as in newborns, correlates with the increased prevalence of these abnormalities in the sperm of infertile males. Studies have confirmed that these chromosomal defects are of paternal origin, highlighting the potential for chromosomally compromised sperm in these individuals to lead to offspring with chromosomal abnormalities [77]. Furthermore, it has been suggested that sperm obtained through surgical extraction may present centrosome irregularities, which could contribute to an elevated risk of mitotic nondisjunction and mosaicism in the resulting embryos [78]. Karyotype analysis played a crucial role in pinpointing the location of genetic elements responsible for normal spermatogenesis. This technique led to the discovery in 1976 of a deletion at the distal end of band q11 on the Y chromosome in six men diagnosed with azoospermia, establishing this region as vital for spermatogenesis [79].
Y-Chromosome Genes
The Y chromosome is one of the smallest chromosomes in the human genome, measuring 57 Mb, and exhibits significant length polymorphism. It comprises approximately 60 million nucleotides. The distal ends of both the short (p) and long (q) arms contain pseudo autosomal regions (PARs) that undergo recombination with the X chromosome during meiosis. The chromosomal segments outside these PARs do not participate in recombination, thus being classified as the non-recombining region of the Y (NRY), which constitutes 95% of the Y chromosome [78]. This chromosome is male-specific and represents the only haploid component of the human genome. To date, 156 transcription units, 78 protein-coding genes, and 27 distinct proteins (9 located on Yp and 18 on Yq) have been identified on the Y chromosome. The Y chromosome is categorized into seven deletion intervals, with intervals 5 and 6 being critical for spermatogenesis. The euchromatic DNA sequences on the Y chromosome cover approximately 23 Mb, including 8 Mb on the short arm and 14.5 Mb on the long arm. There are three classes of euchromatic sequences: those transposed from the X chromosome during Y chromosome evolution (X-transposed), those sharing sequence similarities with the X chromosome (X-degenerate), and those consisting of repeated units found across the proximal short arm of Yp and throughout most of Yq (amplicons).
Only two protein-coding genes have been identified: TGIF2LY and PCDH11Y in X-transposed segments. The 8.5 Mb, X-degenerate regions are spread with single-copy genes or pseudogenes that are largely expressed across various organs rather than being restricted to specific tissues. The sex-determining gene (SRY) is situated within this region, where it functions as a transcription factor that switches on the genes for male sexual differentiation. The X-degenerate region contains the AZFa genes USP9Y and DBY. The unique amplonic regions spanning 10.5Mb within the euchromatin signify the most complex areas of the Y chromosome. Amplicons, which are families of identical nucleotide sequences, are distributed across the euchromatin on the Yp and Yq arms in seven segments. The amplicons are particularly rich in Y chromosome genes that are exclusively expressed in the testes and enhance sexual fitness. Additionally, the ampliconic regions include genes associated with the AZFb and AZFc regions. An array of amplicons forms eight palindromic structures (P1–P8) [80]. The sequencing and assembly of the human Y chromosome have posed significant challenges due to its intricate repeat architecture, which features long palindromes, tandem repeats, and segmental duplications. Consequently, over half of the Y chromosome is absent from the GRCh38 reference sequence, making it the last human chromosome to be fully characterized.
The Telomere-to-Telomere (T2T) consortium has successfully delivered the comprehensive sequence of the human Y chromosome derived from the HG002 genome, which consists of 62,460,029 base pairs (designated as T2T-Y). This newly established sequence corrects multiple errors found in GRCh38-Y and adds over 30 million base pairs, thereby elucidating the complete ampliconic structures of gene families including TSPY, DAZ, and RBMY. Furthermore, it illustrates an alternating pattern of human satellite 1 and 3 blocks within the heterochromatic Yq12 region. The T2T-Y sequence has been incorporated with an earlier assembly of the CHM13 genome, facilitating the mapping of existing population variations, clinical variants, and functional genomics data, thus establishing a comprehensive reference sequence for all 24 human chromosomes [81].
Microdeletion of Y- Chromosome
Microdeletions on the long arm of the Y chromosome (Yq) represent a significant pathogenic factor linked to male infertility. Tiepolo and Zuffardi showed a connection between deletions of the Y chromosome and male infertility in 1976 [82]. These Yq microdeletions are observed in approximately 13% of azoospermic men and in 1% to 7% of men with severe oligozoospermia, defined as having a sperm count of less than 0.5 million/mL. The deletions are primarily concentrated in intervals 5 and 6 of the Y chromosome and are classified under the Azoospermia Factor (AZF), as the most severe phenotype associated with these deletions is azoospermia. The AZF region is further divided into three distinct, non-overlapping areas known as AZFa, AZFb, and AZFc. Six genes located within the AZF regions are exclusively expressed in the testes and are referred to as “AZF candidate genes.” The AZFa region includes about 400–600 kb of DNA. It is located in the proximal section of deletion interval 5. It contains two protein-coding genes, USP9Y and DBY (recently renamed DDX3Y). Deletions in the AZFa loci are associated with Sertoli-cell-only syndrome, type I. The AZFb region spans about 1–3 Mb of DNA and is located from the distal part of deletion interval 5 to the proximal end of deletion interval 6 (subintervals 5O–6B). The protein-coding genes within the AZFb region include EIF1AY, RPS4Y2, and SMCY, which are found in X-degenerate euchromatin, as well as HSFY, XKRY, PRY, and RBMY, located in the ampliconic region.
The AZFb gene is expressed in primary spermatocytes, and its deletion results in meiotic arrest, leading to the accumulation of primary spermatocytes [83]. The AZFc region spans 3.5 Mb of euchromatin and is positioned at the distal end of deletion interval 6 (subintervals 6C–6E) on the Y chromosome [82]. Deletions within the AZFc region are predominantly observed in men diagnosed with idiopathic oligozoospermia or azoospermia. This region encompasses eight gene families that play a crucial role in spermatogenesis, namely BPY2, CDY, DAZ, CSPG4LY, GOLGAZLY, TTY3.1, TTY4.1, and TTY7. There are four copies of the DAZ gene having autosomal homolog, DAZL, located on chromosome 3p24. The first gene identified in the AZFc region was the Deletion in Azoospermia factor (DAZ), which encodes RNA-binding proteins that are specifically expressed in germ cells [84]. A subset of infertile men presents with partial deletions in the AZFb and AZFc loci. Given the structural complexity of the Y chromosome and the mechanisms underlying deletions, it is plausible that additional deletions may also contribute to the effects observed with partial deletions of AZFb and AZFc. The frequency and clinical implications of these partial deletions remain unclear. A specific 1.6 Mb deletion on the Y chromosome, referred to as the “gr/gr” deletion, is particularly noted in infertile men experiencing varying levels of spermatogenic failure [85]. AZF deletions are directly associated with spermatogenic failure, where deletions of the AZFa or AZFb loci, as well as extensive deletions involving two or more loci, result in azoospermia and are linked to a poor prognosis for couples considering assisted reproductive technology (ART).
In cases involving AZFc deletions, the phenotypic expression varies, and while the likelihood of sperm retrieval is relatively high, these cases often exhibit a progressive decline in sperm count over time, transitioning from oligozoospermia to azoospermia [86].
Y chromosome Shrinkage and Gene Loss
The Y chromosome is the smallest human chromosome, carrying ~55–60 protein-coding genes, compared with ~1,000 plus on the X chromosome. Over evolutionary time (millions of years), the Y has lost most of its ancestral genes. For example, from an original ~1,000 genes, it has retained less than 10%. However, current research [87,88] shows that the remaining genes are highly conserved and functionally stable especially those controlling spermatogenesis and sex determination (SRY, AZF regions). Meaning that large-scale gene loss is not ongoing at a pace that would threaten male survival in the near future (Figure 6). Therefore, the idea that the Y will disappear soon is unlikely in the foreseeable evolutionary timeframe [87,88].
The first gene associated with male infertility outside the Y chromosome was discovered in 1988 on the X chromosome. In 1989, researchers identified mutations in the CFTR gene (Cystic Fibrosis Transmembrane Conductance Regulator) located on chromosome 7, which were found to be the cause of Cystic Fibrosis [89]. Subsequent studies have pinpointed specific mutations within this gene that lead to isolated infertility. Notably, mutations in CFTR have been linked to obstructive azoospermia due to Congenital Bilateral Absence of the Vas Deferens, as demonstrated through DGGE and SSCP techniques [90]. The high frequency of CFTR mutations in the global population, especially among individuals of European ancestry where approximately 1 in 25 are carriers of pathogenic variants—accounts for 60–70% of cases of congenital absence of the vas deferens (CAVD) [91]. Building on this knowledge, Brown et al. in 1988 employed a PCR-based method combined with southern blotting to demonstrate that the deletion of the human Androgen Receptor (AR, also referred to as NR3C4) was linked to infertility in patients with mild or partial androgen insensitivity syndrome [92], as well as sex reversal in those with complete androgen insensitivity syndrome. In 1989, the CFTR gene mutations were further explored using restriction fragment length polymorphism (RFLP) analysis to identify the relevant genomic locus, followed by PCR-based sequencing to detect mutations [89].
In the early 2000s, advancements in Sanger sequencing and the introduction of automated DNA sequencers led to the widespread use of DNA sequencing in research and diagnostics related to human diseases [93]. This approach proved particularly effective in testing for mutations in candidate disease genes identified through positional cloning or evidence from orthologous genes studied in model organisms [94]. Advances in sequencing technology and the introduction of single nucleotide polymorphism (SNP) microarrays in the 1990s facilitated a renewed shift in research methodologies aimed at exploring the genomes of infertile men and uncovering new genes linked to male infertility. In 2007, this innovative positional cloning strategy led to the identification of two new male infertility genes, AURKC and SPATA16, which are responsible for various morphological sperm abnormalities [95]. Likewise, homozygosity mapping was employed to detect a homozygous variant in the DNAH1 gene within a small group of infertile men exhibiting morphological defects in their sperm flagella [96]. The intricate interplay of gene products located on both sex chromosomes and autosomes is crucial for an individual’s fertility. Numerous autosomal genes, including acrosin, BAX, BCL16, c-kit, ATM, HSP70.2, RAD6B, MDHC7, CREM, and DNA11 and 12, are vital for germ cell development and spermiogenesis. Additionally, the methylenetetrahydrofolate reductase (MTHFR) gene, situated on chromosome 1, is essential for folate metabolism, DNA methylation, and spermatogenesis.
Any mutation in the MTHFR gene may interfere with the methylation of nucleotides in germ cells, a critical regulatory process for the precise transmission of genetic information to offspring [97].
The advancement of high-throughput next-generation sequencing (NGS) technologies over the last ten years has led to a significant reduction in sequencing costs, accompanied by a substantial increase in sequencing capacity. Instead of focusing on the selection and sequencing of individual candidate genes or assessing specific polymorphisms and large structural anomalies in a limited number of patients and controls, NGS facilitates the unbiased sequencing of extensive gene sets, encompassing all coding exons through whole exome sequencing, or even the complete human genome. This capability is increasingly being applied to large cohorts of patients and controls. NGS technology serves as a robust instrument for uncovering the genetic underpinnings of human phenotypes. Whole Exome Sequencing (WES), as a prominent molecular technique in reproductive medicine, has significantly enhanced our comprehension of the genetic factors contributing to male infertility. While WES produces a vast amount of genetic information, the analysis of this data can be intricate [98]. NGS has introduced a cost-effective and swift genetic screening method for the identification of novel disease-associated genes [99]. In recent years, exome sequencing has emerged as the primary technology for investigating disease genes related to male infertility. Male factor infertility presents a critical clinical challenge, with its most severe manifestations, such as severe oligospermia or azoospermia, arising from various genetic origins.
Some conditions, including Klinefelter syndrome and cystic fibrosis, are well characterized, yet many remain elusive, with Y chromosome microdeletions accounting for only a small portion of the unexplained cases. In both consanguineous and non-consanguineous families, whole exome sequencing has effectively identified likely causal mutations in instances of severe oligospermia [100]. Furthermore, exome sequencing has been successfully utilized to discover new disease genes in patients exhibiting acephalic spermatozoa. Notably, homozygous and compound heterozygous mutations in testis- specific genes such as BRDT, SUN5, and PMFBP1 have been identified as disruptors of the head-flagella junction in spermatozoa [101]. Next Generation Sequencing (NGS) has played a crucial role in the investigation of congenital isolated hypogonadotropic hypogonadism, a condition marked by incomplete or absent puberty and infertility. Numerous pathogenic mutations have been discovered in various genes and genetic loci, which lead to neurodevelopmental issues affecting the migration of gonadotropic hormone-releasing hormone (GnRH) neurons or interfere with the neuroendocrine functions of GnRH secretion and action [102]. The process of human spermatogenesis spans approximately 72 days, during which the activation of around 2000 protein-coding genes is essential for the development and maturation of millions of male gametes [103].
This complexity underscores the intricate genetic landscape associated with male infertility, necessitating specialized research methodologies [104]. The field of androgenetics has evolved through the traditional phases of molecular genetics, beginning with Sanger sequencing for identifying de novo mutations and progressing to the utilization of NGS. The past decade has witnessed a rapid acceleration in the identification of novel genetic variants linked to male infertility, propelled by advancements in genome-wide discovery tools. While early genome-wide association studies (GWAS) relied on array-based methods, NGS has recently emerged as the primary technique for exploring genetic diversity. The genes linked to male infertility encompass those involved in various biological processes, such as spermatogenesis (for instance, USP9Y, DBY, RBMY, TEX11, DAZ), the development of the male reproductive system (including AR, FSHR, CFTR, ADGR2) [105], and steroid hormone signaling (e.g., SHBG). Deficiencies in these genes can result in either qualitative or quantitative sperm abnormalities, ultimately leading to infertility [106]. The advent of high-throughput technologies, such as next-generation sequencing and genome-wide association studies, has facilitated the discovery of numerous novel candidate genes that may contribute to male infertility. The Male Infertility Knowledgebase (MIK) serves as a comprehensive resource, aggregating information on all documented genetic factors associated with male infertility sourced from the PubMed database.
This knowledgebase contains a detailed compilation of 17,754 genes (validated dataset: 1,564 genes; predicted (high-throughput) dataset: 16,190 genes), along with 738 single nucleotide polymorphisms (SNPs), 338 distinct pathways, 8,152 unique disease conditions, and 17,870 gene ontology terms related to these genes. Additionally, the knowledgebase provides insights into literature supporting congenital causes of male infertility and data derived from high-throughput technologies. The genes cataloged within this resource are annotated with information regarding known genetic abnormalities associated with male infertility, as well as functional and pathway-related details. Furthermore, the database includes disease conditions that share a common genetic basis [107]. The male infertility subtypes most extensively examined through next-generation sequencing (NGS) are the quantitative abnormalities, specifically non-obstructive azoospermia (NOA) and oligospermia. The earliest study in this area was conducted in 2013, where the researchers utilized NGS to enhance a previously identified genome-wide association study (GWAS) signal. This investigation focused on five genes located near peak association signals on chromosomes 12 [peroxisomal biogenesis factor 10 (PEX10), protein arginine methyl transferase 6 (PRMT6), and SRY-box 5 (SOX5)] and 20 [signal regulatory protein alpha (SIRPA) and signal regulatory protein gamma (SIRPG)] [108].
In a separate analysis, Xu et al. 2013, [109] identified a significant association with a single nucleotide variant (SNV) in SIRPA (rs3197744) through targeted panel sequencing of both cases and controls, thereby reinforcing the potential involvement of this gene in male infertility. Numerous other studies have employed NGS to evaluate individuals diagnosed with NOA. For instance, scientist examined two unrelated consanguineous families experiencing spermatogenic failure, one comprising three azoospermic brothers and one oligospermic brother, while the other included three azoospermic siblings. Their research utilized a hybrid methodology that combined whole-exome sequencing with SNV genotyping, enabling a focused analysis on runs of homozygosity to pinpoint the causative variant. This approach resulted in the identification of distinct genes for each family: TATA-box binding protein associated factor 4b (TAF4B) and zinc finger MYND-type containing 15 (ZMYND15), both of which carried recessive deleterious truncating mutations shared among all affected brothers in their respective families [110]. Additionally, Okutman et al. 2015, reported a recessive mutation in testis expressed 15, meiosis and synapsis associated (TEX15) linked to NOA in three affected siblings from a Turkish family, which was absent in their fertile brother and parents [111].
Ramasamy and coworkers discovered mutations in neuronal PAS domain protein 2 (NPAS2) in three azoospermic siblings from another consanguineous family in Turkey [62]. Gershoni with his colleagues employed a combination of whole-exome sequencing and whole-genome sequencing across various families to identify mutations in the following genes: meiosis specific with OB domains (MEIOB), testis expressed 14 (TEX14), and dynein axonemal heavy chain 6 (DNAH6) [112]. In a more recent development, five studies published in 2017 utilized next-generation sequencing (NGS) in patients diagnosed with non-obstructive azoospermia (NOA) or oligospermia to identify additional genes associated with quantitative sperm defects and male infertility. Four of these studies concentrated on multiplex consanguineous families, demonstrating the segregation of recessive mutations in serine peptidase inhibitor, Kazal type 2 (SPINK2), MAGE family member B4 (MAGEB4), Tudor domain containing 9 (TDRD9), and adhesion G protein-coupled receptor G2 (ADGRG2) in siblings with NOA, while no such mutations were found in healthy males within the same families [113-115]. Morphological abnormalities such as teratozoospermia, macrozoospermia, globozoospermia, and acephalic spermatozoa syndrome, which negatively impact fertility, manifest in various forms affecting the sperm’s head, neck, and tail.
Alazami and coworkers in 2014, employed whole-exome sequencing on a family exhibiting asthenozoospermia, revealing a nonsense mutation in nephrocystin4 (NPHP4) [116]. In a separate investigation, Sha and colleagues in 2017, sequenced a patient with flagellar defects and identified a recessive deleterious mutation in centrosomal protein135 (CEP135), which is essential for centriole biogenesis [117]. This mutation led to infertility by causing protein aggregates within the centrosome and flagella. Additionally, in 2017 Li found a mutation in bromodomain testis associated (BRDT) in a consanguineous patient suffering from acephalic spermatozoa [118]. In a study involving 21 patients with dysmotile spermatozoa (DFS), Sha and coworkers discovered 17 distinct DNAH1 mutations across 12 cases, which included one homozygous and 16 compound heterozygous patients. These mutations were present in the affected individuals but absent in unaffected family members and a cohort of 50 ethnically matched fertile men. Functional analyses conducted on a subset of these patients demonstrated reduced levels of DNAH1 and disorganized 9+2 microtubule structures [117]. Amiri-Yekta with his coworkers initiated their research on motility anomalies by examining ten men from six highly consanguineous families exhibiting flagellar abnormalities through whole-exome sequencing.
They discovered mutations in the DNAH1 gene in two of these families, which were subsequently confirmed in an additional sibling from each affected family via Sanger sequencing. Following this, the authors expanded their study to include an additional 38 men to screen for the same founder mutation, successfully identifying one more individual with the identical mutation [119]. More recently, Wang and coworkers employed whole-exome sequencing to uncover four more consanguineous Chinese men with frameshift truncating mutations in DNAH1, thereby reinforcing the gene’s significance in flagellar development and motility during spermatogenesis [120]. Additionally, in 2017 Xu and coworkers reported homozygous mutations in two siblings from consanguineous parents, which affected a highly conserved residue in sperm-associated antigen 17 (SPAG17), leading to asthenospermia. Functional studies indicated that this mutation resulted in a marked reduction of SPAG17 expression in the spermatozoa of the affected individuals, aligning with its functional role in motility [117]. Subsequently, Tang and coworkers investigated 30 independent cases of motility defects attributed to flagellar abnormalities and identified further recessive mutations (both homozygous and compound heterozygous) in three cilia- and flagella-associated protein (CFAP) genes: CFAP43, CFAP44, and CFAP65, in five men [121].
1. Irreversible diseases that can be treated by assisted reproductive technologies (ART) using the male partner’s sperm
2. Life-threatening conditions that may underlie infertility or related medical comorbidities that require medical attention
3. Genetic abnormalities or lifestyle and age factors that may affect the health of the male patient or offspring, particularly if ART is to be used
4. Irreversible conditions that are not amenable to the above and for which donor insemination or adoption are possible options.
Current sex ratio: India already faces a skewed sex ratio due to sociocultural factors (female feticide, son preference), not due to Y chromosome instability. According to Census 2011, India had ~943 females per 1000 males; NFHS-5 (2019–21) reported ~1020 females per 1000 males at birth. Government of India has launched several schemes for promotion and encouragement of birth and up-bringing of female child with strict punitive actions against female feticide and sex determination before birth. It has created great awareness in the society and it is expected that it will produce positive results in maintenance of sex ratio in the society [122-124].
Genetic Future
There is no evidence that Y chromosome reduction will biologically reduce the male population in the coming centuries. In fact, studies suggest the Y-chromosome has reached a stable evolutionary state. However, fertility challenges may rise, requiring stronger reproductive healthcare and awareness. Male fertility rate will get reduced, as Y chromosome microdeletions (especially AZF regions) contribute to 10–15% of male infertility cases in India. With lifestyle stressors (pollution, obesity, endocrine disruptors), this infertility burden may rise in future but the speculations of elimination of male populations have no sound scientific foundation.
Global Comparison
In western countries like Europe, USA, and Japan, similar infertility issues exist, but advanced reproductive technologies (IVF, ICSI, sperm banking) mitigate long-term impacts. Fertility decline is mostly sociological (delayed marriage, lower birth rates), not genetic. Like India similar situation exists in China, historically skewed sex ratios due to sociocultural preferences have created demographic imbalances (more men than women). Globally, male population reduction due to Y chromosome gene loss is unexpected. What’s more realistic is that fertility rates will decline due to lifestyle, environment, and socioeconomics rather than chromosome shrinkage. Birth data is usually reported as total live births per year and often broken down by sex ratio at birth (SRB), i.e., number of male births per 100 female births. Global biological norm is approximately 105 male births per 100 female births. Sex ratio at birth is around 1.05. Some countries deviate from this biological norm due to cultural, medical, or demographic factors [125].
Births by Sex (Male/Female) – Approximate Numbers
1. India & China: Higher Sex ratio at birth (~1.07–1.08), reflecting mild sex selection practices in past decades, though narrowing now.
2. USA, Japan, Russia, Africa: Closer to natural SRB (~1.05).
3. Africa: Highest absolute births, with growth continuing.
4. Japan & Russia: Facing severe fertility decline, <2M total births combined in 2023.
5. China: Massive drop in births (almost halved since 2016).
Sex Ratio at Birth (Male births per 100 Female births).
The process of Y chromosome degeneration has prompted the suggestion that continuing gene loss will lead to the eventual disappearance of the human Y80-83. These predictions are based on a naive model of a constant rate of gene loss from the Y chromosome. However, recent theoretical and experimental studies have clearly demonstrated that Y chromosome’s shrinkage is not an immediate threat and Y degeneration does not proceed in the simple linear fashion and refute these sensational claims of human Y extinction. Research confirmed that 45% - 50% of its genes are highly conserved and stable and guaranteed the continuity of male population in the future generation. In India and developing countries, male population trends are shaped by social and environmental factors, not chromosome loss. Compared with foreign countries, India may face higher infertility burdens unless public health, pollution control, and reproductive care are not improved. Technology-assisted reproduction will likely neutralize genetic concerns, but population structure will be determined more by culture, policy, and lifestyle than by Y chromosome evolution. The Y chromosome has lost many genes long ago, but modern genomic data show important protective mechanisms (palindromic gene conversion, selection) that stabilize remaining essential genes. Clinically important Y deletions (AZF) cause infertility for affected men, but these are relatively rare and do not translate into major shifts in the number of males worldwide.
Population numbers and sex ratios are dominated by demographic, social and health factors (fertility trends, life expectancy, migration), not by ongoing rapid Y-gene loss. Long-term evolutionary turnover of sex chromosomes is possible (seen in other mammals) but occurs on very long timescales (hundreds of thousands to millions of years), not within human historical timeframes. Human MSY (male-specific Y) is small compared with X but contains essential genes for spermatogenesis and male health; most dramatic gene loss in deep evolutionary past; present human Y chromosome shows structural features (palindromes, ampliconic genes) that help preserve key genes. Certain Y microdeletions (AZF regions) cause male infertility in affected men; but such deletions are relatively rare and usually familial/individual.
Acknowledgement: The authors are thankful to Dr. Atul Krishna, a medical surgeon and the President of Mahayana Theravada Vajrayan Buddhisht Religious and Charitable Trust, Meerut and Major Gen. Professor (Dr.) G.K. Thapaliyal, Director-General, Swami Vivekanand Subharti University, Meerut for their valuable suggestions during the finalization of the manuscript.
The authors received no funding.
The authors declare that they have no conflict of interest.

