Impact Factor : 0.548
- NLM ID: 101723284
- OCoLC: 999826537
- LCCN: 2017202541
González Javier1*, Torres Patricia2 and Olazaran Javier3
Received: February 06, 2026; Published: February 18, 2026
*Corresponding author: González Javier, Medical Department, ITF Research Pharma, S.L.U., Madrid, Spain.
E-mail: j.gonzalez@italfarmacogroup.com
DOI: 10.26717/BJSTR.2026.64.010096
Cognitive decline comprises a range from subjective memory complaints (SMC) to mild cognitive impairment (MCI) and major neurocognitive disorders such as Alzheimer’s disease (AD). Early identification and intervention are essential, yet many individuals with initial symptoms neither seek medical advice nor implement preventive lifestyle changes. Nutraceuticals have gained increasing attention as complementary strategies in the early phases of cognitive decline due to their potential neuroprotective effects, favourable safety profile, and accessibility. This narrative review summarizes current evidence on six nutraceutical compounds: docosahexaenoic acid (DHA), folic acid, selenium, vitamin D, lutein and zeaxanthin, and choline, in the context of cognitive impairment. These compounds, especially in adequate and studied doses, have been proposed to influence cognitive function through antioxidant, anti-inflammatory or methylation-related mechanisms, and in some cases by supporting neurogenesis or synaptic plasticity.
Clinical studies suggest potential benefits in specific cognitive domains, particularly memory, attention, and executive function, especially among individuals with SMC or MCI. The review also highlights the high prevalence of suboptimal intake of neuroprotective nutrients among older adults and the rationale for nutritional intervention in early stages of decline. Notably, nutrient combinations appear to yield greater benefits than single-nutrient approaches, reflecting the multifactorial nature of cognitive deterioration. Although emerging evidence is promising, further well-designed studies are needed to confirm efficacy, determine optimal formulations, and define which populations may benefit most. In any case, nutraceuticals may represent a valuable, low-risk option to support cognitive health and delay progression across the cognitive impairment spectrum.
Keywords: Subjective Memory Complaints; Mild Cognitive Impairment; Alzheimer’s Disease; Nutraceuticals
Abbreviations: ADAS-Cog: Alzheimer’s Disease Assessment Scale–Cognitive Subscale; AD: Alzheimer’s Disease; AMD: Age-Related Macular Degeneration; ANIBES: Anthropometry, Intake, and Energy Balance in Spain study; APP: Amyloid Precursor Protein; AREDS: Age-Related Eye Disease Study; BDNF: Brain-Derived Neurotrophic Factor; BCTs: Brief Cognitive Tests; CANTAB: Cambridge Neuropsychological Test Automated Battery; DSM: Diagnostic and Statistical Manual of Mental Disorders; DHA: Docosahexaenoic Acid; EPA: Eicosapentaenoic Acid; GPC: Glycerophosphocholine; IQ: Intelligence Quotient; IL: Interleukin; LZ: Lutein/Zeaxanthin; MCI: Mild Cognitive Impairment; MCS: Memory Complaint Scale; MIDAS: Memory Improvement with Docosahexaenoic Acid Study; MMSE: Mini-Mental State Examination; MoCA: Montreal Cognitive Assessment; MPOD: Macular Pigment Optical Density; NHANES: National Health and Nutrition Examination Survey; NPD1: Neuroprotectin D1; NTs: Neurotransmitters; PUFA: Polyunsaturated Fatty Acids; Se: Selenium; SMC: Subjective Memory Complaints; TNF: Tumor Necrosis Factor; VDR: Vitamin D Receptor; WAIS-RC: Wechsler Adult Intelligence Scale–Revised Chinese version; VITAL: Vitamin D and OmegA-3 TriaL; VITACOG: Vitamin B Intervention Trial in Cognitive Ageing
Cognition comprises a wide range of mental processes that allow individuals to perceive, process, store, and retrieve information. These functions are traditionally grouped into six core domains: attention, executive function, learning and memory, language, visuoperceptual abilities, and social cognition. Impairments in these areas may lead to cognitive decline, ranging from mild cognitive impairment (MCI) to major neurocognitive disorders [1]. At a molecular level, cognitive decline is driven by multiple, interrelated biological mechanisms that often precede clinical symptoms. These include synaptic dysfunction, oxidative stress, neuroinflammation, mitochondrial impairment, and epigenetic alterations, all of which contribute to impaired neuronal communication, reduced plasticity, and increased vulnerability to degeneration [2-4]. Until the Diagnostic and Statistical manual of Mental disorders, version 5 (DSM-5), cognitive impairment was clinically divided into three main categories based on severity and functional impact. Age-associated memory impairment referred to subjective complaints without functional decline, considered part of normal aging [5]. MCI, although not formally recognized as a diagnosis in the Diagnostic and Statistical manual of Mental disorders, version 4 (DSM-IV), was acknowledged as a condition of interest. It was commonly defined by Petersen’s criteria: subjective memory complaints, objective cognitive decline, preserved daily function, and no dementia [5-7]. Dementia involved a marked decline in cognitive abilities with significant interference in daily life and functional disability [5].
The DSM-5 restructured cognitive impairment into two categories: “Mild Neurocognitive Disorder” and “Major Neurocognitive Disorder” [8]. The mild form comprises impairments that do not notably interfere with daily life, including what was formerly defined as MCI, while the major form reflects more severe decline affecting daily functioning [8]. This new classification also expanded the scope of cognitive disorders to include impairments resulting from any etiology and across all age groups, such as dementia associated to neurodegenerative diseases, traumatic brain injury or epilepsy, among others [1]. A recent global meta-analysis estimated that approximately one in five adults over the age of 50 (19.7%) is affected by MCI, and studies estimate a 10–15% annual conversion rate from MCI to dementia [9]. However, rates vary widely depending on the diagnostic criteria and assessment methods used [10]. Subjective Memory Complaints (SMC) are part of the diagnostic criteria for MCI. While their association with current cognitive performance is inconsistent, longitudinal studies indicate they may predict future decline, especially when recent or progressively worsening [11]. In a 2019 Madrid-based study with 1,775 participants over 55, SMC prevalence was 16.3%, increasing to 19.6% in those over 65 [12]. A subsequent Spanish national study conducted in 2021 with 2,300 individuals over 50 reported higher rates: 31% overall and 44% among those over 75 [13].
To identify and monitor SMC and early cognitive decline, several Cognitive Tests (CTs) have been developed and validated. The Mini-Mental State Examination (MMSE) is the most established, evaluating mainly orientation, memory, attention, and language domains [14]. The 7-Minute Screen, including cued recall, verbal fluency, clock drawing, and orientation, offers high sensitivity and specificity for early Alzheimer’s disease, even in mild cases [15]. Other tools like the Fototest, AD8, and Memory Complaint Scale (MCS) are suitable for routine use (specially useful in a Primary Care scenario) due to their brevity, simplicity, and low educational bias; and, therefore, are considered as Brief Cognitive Tests (BCTs) [9,12]. Combined with functional scales such as the Lawton–Brody or Barthel Index [16,17], these tests support the distinction between SMC or MCI and major neurocognitive disorders. Despite the prevalence of SMC, most individuals take no specific action. A Spanish survey found that 78% had not sought professional advice and 90% had not made nutritional changes [13]. Additionally, the Anthropometry, Intake, and Energy Balance in Spain study (ANIBES) study revealed widespread inadequate intake of key neuroprotective nutrients, particularly omega-3 fatty acids and folic acid [18].
Both preventive and therapeutic strategies should be personalized and multidimensional, particularly in the early stages of cognitive decline [9]. Core strategies include physical activity, social engagement, smoking cessation, and vascular risk control, along with cognitive stimulation and psychological support. Dietary changes and supplements are proposed as complementary interventions in cases of SMC or MCI, and pharmacological treatment may be considered in selected cases based on etiology and severity [9]. Nutraceuticals have attracted growing interest as a complementary approach for cognitive support. These compounds include a range of micronutrients and bioactive substances with antioxidant, anti-inflammatory, or methylation- related properties; and, in adequate doses, may help preserve cognitive function during the initial phases of decline. Their favorable biological plausibility, safety profile, and accessibility make them particularly attractive for preventive use in individuals with SMC or MCI [9]. Nutraceuticals are naturally occurring bioactive compounds, such as vitamins, minerals, polyphenols, fatty acids, and plant extracts, derived from foods and administered in concentrated forms, with the goal of promoting health and preventing or mitigating disease.
They differ from conventional foods in that they are consumed in non-dietary formats (e.g., capsules or powders), and unlike pharmaceuticals, they are not intended to treat diseases directly but to support physiological functions [19,20]. In the field of cognitive health, various nutraceuticals have demonstrated potential to modulate neuroinflammation, oxidative stress, mitochondrial dysfunction, and amyloid or tau-related pathology. These mechanisms may contribute to the preservation of cognitive function, slow disease progression, or alleviate symptoms in both early and advanced stages of cognitive impairment [21-25]. The aim of this review is to summarize current evidence on the neuroprotective potential of nutraceuticals across the spectrum of cognitive impairment, from subjective memory complaints and mild cognitive impairment to more advanced stages such as Alzheimer’s disease. It outlines their proposed mechanisms of action, reviews experimental and clinical data, and explores their possible application in both preventive and complementary therapeutic strategies.
A comprehensive literature search was conducted in PubMed, ScienceDirect, Scopus, and Google Scholar using combinations of keywords such as “nutraceuticals”, “cognitive impairment”, “cognitive enhancement”, “neuroprotection”, or “memory”. Eligible sources included original research articles, systematic reviews, and meta-analyses published up to the date of manuscript preparation. Studies were selected based on their relevance and methodological quality, with particular emphasis on those describing mechanisms of action involving antioxidant, anti-inflammatory, or neuroplastic processes. Only articles published in English or Spanish were considered.
Key Nutraceuticals for Brain Health
DHA (Docosahexaenoic Acid): Docosahexaenoic acid (DHA) is a vital omega-3 polyunsaturated fatty acid (PUFA) and the predominant long-chain PUFA found in the human brain. It constitutes a major component of phospholipids in neuronal membranes, particularly in grey matter and synaptic regions. The incorporation of DHA into membrane phospholipids, such as phosphatidylethanolamine and phosphatidylserine, is crucial for maintaining membrane fluidity, receptor functionality, and synaptic signaling. Furthermore, DHA facilitates neurotransmitter release, synaptic connectivity, and myelination, a fundamental process for neuronal communication and cognitive function [26,27]. In addition to its structural roles, DHA modulates neuroinflammation and oxidative stress pathways, both of which contribute to cognitive decline. It influences the processing of β-amyloid precursor protein (APP), potentially reducing the accumulation of beta-amyloid peptides, a hallmark of AD pathology [28]. Moreover, DHA serves as a precursor to specialized pro-resolving lipid mediators, including neuroprotectin D1 (NPD1), which promotes neuronal survival and mitigates inflammatory responses in the brain. Evidence also indicates it may enhance synthesis of brain-derived neurotrophic factor (BDNF), a neurotrophin that stimulates neurogenesis, supports synaptic plasticity, and facilitates neuronal repair mechanisms [28].
Reduced DHA plasma and brain tissue levels have been linked with cognitive impairment, decreased brain volume, and an increased risk of dementia, even in individuals without overt clinical symptoms, emphasizing DHA’s importance as a modifiable nutritional factor in cognitive aging [28]. Numerous clinical trials have examined the effects of DHA supplementation on cognitive function in aging populations, including individuals with SMC, MCI, and early AD. Although the findings are heterogeneous, likely due to variations in study design, participant characteristics, dosage, and intervention duration, a substantial number of studies have reported beneficial effects on cognitive domains such as memory, attention, and overall performance. These effects appear more consistently when DHA is administered at daily doses of 800–900 mg or higher and maintained for at least six to twelve months [27]. The OmegAD study, a randomized, double-blind, placebo-controlled trial involving 174 patients with mild to moderate Alzheimer’s disease, investigated the effects of supplementation with 1.7 g/day DHA and 0.6 g/day EPA over six months. The primary cognitive outcomes measured were the MMSE and the Alzheimer’s Disease Assessment Scale–Cognitive subscale (ADAS-Cog).
Although no significant differences were observed between the treatment and placebo groups in the primary endpoints across the entire study population, in a subgroup of 32 patients with mild AD (MMSE ≥27 and Clinical Dementia Rating Scale scores 0.5–1), a significant treatment effect on MMSE scores was observed over time (p = 0.02), with the rate of cognitive decline being significantly slower in the treatment group at the 6-month follow-up (p = 0.01) [29]. The Memory Improvement with Docosahexaenoic Acid Study (MIDAS) was a randomized, double-blind, placebo-controlled, multicenter trial conducted at 19 sites across the United States, assessing the cognitive effects of daily supplementation with 900 mg of DHA (n=242) or placebo (n=243) over 24 weeks (or 6 months) in healthy older adults aged 55 years or above with age-related cognitive decline characterized by SMC [30]. The primary outcome measure was the Cambridge Neuropsychological Test Automated Battery (CANTAB) Paired Associate Learning (PAL) test, a computerized neuropsychological assessment designed to evaluate visuospatial associative learning and episodic memory. Participants were required to remember the locations of visual patterns displayed sequentially on the screen, with increasing task difficulty. PAL performance depends largely on the medial temporal lobe and hippocampus, areas involved in memory formation and sensitive to early cognitive changes.
The test’s computerized format and use of parallel versions minimize practice effects, providing reliable longitudinal cognitive data [30,31]. Secondary outcomes included verbal recognition memory (immediate and delayed recall), pattern recognition memory, spatial working memory, and executive function [30]. After 24 weeks, although both groups exhibited a decrease in errors over time, the reduction was notably greater in the DHA group, with a mean difference of 1.63 fewer errors (p = 0.032). This improvement corresponds to an estimated cognitive age enhancement of approximately 3.4 years based on normative data, suggesting a beneficial effect on early memory and learning impairments. Additionally, DHA supplementation resulted in significant improvements in verbal recognition memory (immediate recall p = 0.018; delayed recall p = 0.012), and in an increase in DHA plasma levels doubled after supplementation and positively correlated with cognitive improvement; no significant adverse events related to DHA were reported [30]. A subsequent meta-analysis systematically combined data from 15 randomized controlled trials that assessed the impact of DHA supplementation, either alone or in combination with EPA, on cognitive function in adults experiencing mild memory complaints. The pooled analysis revealed a statistically significant improvement in episodic memory performance in individuals receiving DHA/EPA supplementation compared to controls (p = 0.004).
Although most data come from studies providing DHA doses between 501 and 999 mg daily, doses around 900 mg could tend to show greater cognitive benefits, suggesting a dose-response relationship. In addition to episodic memory, the meta-analysis demonstrated significant gains in immediate recall (p = 0.012), a fundamental component of episodic memory, crucial for everyday functioning. The findings further indicated that participants with lower baseline omega-3 fatty acid levels experienced greater cognitive benefits, highlighting the potential moderating role of nutritional status on treatment efficacy. Furthermore, there was a trend (p < 0.058) for episodic memory improvement in response to supplementation in subjects aged 45 years or older [32]. Complementing these findings, a 24-month randomized controlled trial conducted in 2017 included 240 older adults diagnosed with MCI. Participants were randomized to receive either 2 grams per day of DHA or placebo.
The DHA group exhibited statistically significant improvements in cognitive domains measured by full-scale Intelligence Quotient (IQ; p = 0.037) and verbal IQ (p = 0.019). Improvements were also observed in memory-specific subtests, such as digit span, which showed a significant increase (p = 0.007). Importantly, these cognitive enhancements coincided with favorable neurobiological changes: elevated expression of autophagy-related proteins LC3-II and Beclin-1, and reduced levels of amyloid-beta 42 (Aβ-42) and APP mRNA, suggesting that DHA may contribute to cognitive improvement via mechanisms involving enhanced autophagy and modulation of amyloid metabolism [33].
Folic Acid (Vitamin B9): Folic acid (folate, vitamin B9) is an essential water-soluble vitamin involved in one-carbon metabolism, particularly in the methylation of homocysteine to methionine, a reaction that also requires vitamins B6 and B12 as cofactors [34-36]. The human body contains approximately 15 to 30 milligrams of folate, with roughly half stored in the liver and the remaining amount distributed between blood and various tissues, highlighting the liver’s role as a major folate reservoir and the systemic importance of folate homeostasis [34]. Deficiency in any of these B vitamins components leads to homocysteine accumulation, a neurotoxic metabolite that has been associated with vascular dysfunction, oxidative stress, β-amyloid toxicity, and neuronal apoptosis, all of which are mechanisms implicated in the pathogenesis of cognitive decline and brain atrophy [34,37]. The current dietary reference intake for adults aged ≥19 years is 400 micrograms per day, based on levels sufficient to maintain normal homocysteine metabolism and to prevent deficiency- related neurological and hematological manifestations [34]. Several clinical studies have demonstrated that supplementation with folate improves cognitive function, primarily through the reduction of homocysteine levels. The FACIT trial, a 3-year randomized controlled study, showed that daily supplementation with 800 micrograms of folic acid significantly improved memory performance and processing speed in older adults with elevated homocysteine [38]. Another 6-month randomized controlled trial conducted in older adults with mild cognitive impairment also assessed the impact of daily supplementation with 400 micrograms of folic acid. The study demonstrated that participants receiving folate supplementation showed significant improvements in cognitive performance compared with those receiving conventional care without supplementation. Biochemically, participants in the folate group exhibited significant increases in serum B9 and vitamin B12 concentrations, accompanied by notable reductions in plasma homocysteine levels. Cognitively, the most pronounced benefits were observed in working memory and short-term memory, as assessed by the Digit Span test (p = 0.009). Additional improvements were also reported in other cognitive domains evaluated by subtests of the Wechsler Adult Intelligence Scale-Revised Chinese version (WAIS-RC), such as the Block Design task, which reflect enhanced visuospatial processing and broader cognitive engagement.
These results were especially relevant as the study population had no exposure to mandatory folate food fortification and presented with baseline folate intake below recommended levels [39]. Additional studies also explored the combined effects of folate with other supplements. Thus, Homocysteine and B Vitamins in Cognitive Impairment (VITACOG) trial investigated combined B vitamin supplementation, including folate, B6, and B12, and found reductions in brain atrophy and cognitive decline in patients with mild cognitive impairment, especially in those with higher baseline homocysteine levels [40,41]. A 6-month, double-blind, placebo-controlled trial in elderly individuals with MCI (n=240), tested the cognitive effects of 800 micrograms/day of vitamin B9 combined with 800 mg/day of DHA, compared to either nutrient alone or placebo. The combination of folic acid and DHA resulted in significantly greater improvements in full-scale IQ scores, as well as in specific cognitive domains, including arithmetic, digit span, picture completion, and block design tests.
Importantly, these cognitive gains were accompanied by a more pronounced reduction in circulating inflammatory cytokines such as interleukin-6 (IL-6), interleukin-1β (IL-1β), and tumor necrosis factor- alpha (TNF-α) [42]. However, despite these benefits, data from the Spanish ANIBES study revealed that approximately 50% of individuals aged 65 to 75 years do not meet recommended folate dietary intakes, and this deficiency frequently coexists with inadequate omega-3 fatty acid consumption [18]. Additionally, results from the cross-sectional National Health and Nutrition Examination Survey (NHANES) in United States have recently suggested an association between higher serum levels of folic acid and lower levels of several epigenetic aging biomarkers [43].
Selenium (Se): Selenium (Se) is an essential trace element crucial for various physiological processes, including immune function, reproduction, and antioxidant defense. Its deficiency has been associated with multiple adverse health outcomes in animals and humans, including white muscle disease, reduced fertility, thyroid dysfunction, increased susceptibility to infections, cognitive impairment, and severe and chronic deficiency can lead to endemic disorders such as Keshan disease and Kashin–Beck disease [44]. Plasma Se levels decline with age and are associated with greater risk of cognitive deterioration [45]. Given Se’s narrow therapeutic window, appropriate intake is critical: the World Health Organization recommends 55 micrograms/ day for adults, while the tolerable upper intake level is 400 micrograms/day [44,46]. Notably, several studies suggest that Se supplementation within the physiological range may contribute to improved antioxidant capacity and cognitive function, reinforcing its potential role in supporting neurological health.
Recent preclinical research has demonstrated that Se supplementation may restore hippocampal function and improve learning and memory performance. In a murine model, dietary administration of Se was sufficient to stimulate the generation of new neurons in the hippocampus, and to reverse cognitive deficits associated with both aging and brain injury [47]. Complementary evidence arises from a systematic review and meta-analysis, which evaluated the effects of Se supplementation in patients with MCI or AD. The meta-analysis included 11 clinical trials, of which 6 were eligible for pooled analysis. The review found that Se supplementation significantly increased Se levels in plasma, serum, erythrocytes, and cerebrospinal fluid, as well as glutathione peroxidase activity in erythrocytes, an indicator of antioxidant defense. Cognitive outcomes such as MMSE and ADAS-Cog scores showed modest yet consistent improvement in both MCI and AD populations, particularly when Se was administered alongside other nutrients such as B-vitamins or DHA. However, the long-term effects of Se supplementation on neurodegeneration could not be determined within the scope of the included studies [48].
Cholecalciferol (Vitamin D3): Vitamin D has emerged as a biologically plausible factor in cognitive health due to the presence of its receptors and activating enzymes in key brain regions involved in memory and executive functioning. Specifically, vitamin D receptors (VDR) and the enzyme 1α-hydroxylase have been identified in the hippocampus, amygdala, prefrontal cortex, and cerebellum. These regions are functionally involved in memory consolidation, attentional control, emotional regulation, and higher-order executive processes. In addition to its neuroanatomical localization, vitamin D exerts modulatory effects on calcium signaling, oxidative stress, and neuroinflammation, and may enhance neurotrophin expression, supporting its potential contribution to cognitive function [49]. Current dietary recommendations established by the NIH focus on bone health and suggest an intake of 600 IU/day (15 μg) for adults up to 70 years of age and 800 IU/day (20 μg) for individuals over 70 years. The tolerable upper intake level is set at 4,000 IU/day (100 μg) [50].
Epidemiological data have consistently shown associations between low circulating levels of 25-hydroxyvitamin D (25(OH)D), the primary biomarker of vitamin D status, and cognitive impairment. This relationship has been documented across multiple publications, including observational studies, systematic reviews and meta-analyses, with similar trends reported in both cross-sectional and longitudinal research designs. In particular, low 25(OH)D levels have been repeatedly linked to an increased risk of cognitive dysfunction and dementia in older adults [51,52]. When examining specific cognitive domains, executive functions such as processing speed and cognitive flexibility appear to be more consistently affected by vitamin D status than episodic memory. Nonetheless, not all studies confirm these associations. Some prospective analyses adjusting for confounding variables such as physical activity or comorbidities have yielded inconclusive results, and several cross-sectional studies have failed to demonstrate robust correlations [49]. Randomized controlled trials assessing the impact of vitamin D supplementation on cognitive outcomes have produced heterogeneous findings. Supplementation has been associated with improvements in memory and global cognition in individuals with MCI, particularly when administered over 12 months [53,54].
Beneficial effects on executive performance have also been reported with high-dose monthly regimens [55]. In contrast, trials assessing both conservative and intensive vitamin D supplementation approaches in cognitively healthy individuals have generally failed to show meaningful cognitive benefits compared to placebo [56,57]. Additionally, recent studies have explored the potential role of vitamin D in cellular aging and neuroprotection through its effects on telomere biology. The Vitamin D and OmegA-3 TriaL (VITAL) study, a largescale four-year trial that evaluated combined supplementation with vitamin D and omega-3 fatty acids, reported that the group receiving 2,000 IU/day of cholecalciferol exhibited significantly less leukocyte telomere shortening compared to placebo. No significant effects were observed for omega-3s or the combined treatment, suggesting that the observed benefit was primarily attributable to vitamin D [58]. These results are consistent with those of a previous randomized controlled trial that observed improvements in cognitive performance in older adults with MCI after daily supplementation with 800 IU of vitamin D for 12 months, a significantly lower dose, accompanied by a reduced rate of telomere shortening and reductions in oxidative stress biomarkers [59].
Lutein: Lutein and zeaxanthin (LZ) are fat-soluble carotenoids from the xanthophyll family, with potent antioxidant and anti-inflammatory properties. They are found primarily in green leafy vegetables (lutein) and yellow-orange fruits and vegetables (zeaxanthin). Although they are chemically distinct, they are structural isomers and share similar biological functions. Both accumulate selectively in the macula of the retina, where they form the macular pigment and play a key role in protecting the retinal tissue of the eye from oxidative stress caused by blue light. A meta-analysis from nine randomized, controlled clinical trials involving 855 patients diagnosed with age-related macular degeneration (AMD) suggested that lutein supplementation (10-20 mg/day) was associated with relevant clinical benefits such as increase in macular pigment optical density (MPOD), and improvement in visual acuity and contrast sensitivity [60]. In relation to the above, and taking into account the close relationship between visual function and cognition, lutein is present in brain regions involved in visual and cognitive processing, suggesting their potential contribution to maintaining cognitive function in aging populations [61,62].
Several observational studies have reported associations between lower levels of lutein and zeaxanthin and cognitive decline. Plasma concentrations of these carotenoids have been linked to lower performance in various cognitive domains in older adults [63]. Similarly, a lower macular pigment optical density, which reflects retinal carotenoid content, has been associated with lower scores on general cognitive tests such as the MMSE and Montreal Cognitive Assessment (MoCA), as well as with deficits in memory and processing speed [64]. In parallel, large cohort AREDS (Age-Related Eye Disease Study) 2 concluded that higher dietary intake of lutein was associated with a lower risk of cognitive impairment, reinforcing their potential as indirect markers of cognitive vulnerability, as well as positive effects on AMD of lutein supplementation (10 mg/day) [65]. Intervention studies evaluating supplementation with lutein and zeaxanthin have shown promising results in specific cognitive domains. From both carotenoids, lutein has been suggested as the main responsible of the potential positive effects.
In community-dwelling older adults, 12 months of supplementation with 10 mg/day of lutein and 2 mg/day of zeaxanthin led to significant improvements in complex attention and cognitive flexibility [66]. In individuals with SMC, a 6-month supplementation protocol (10 mg lutein/day) resulted in improvements in visual episodic memory, although no changes were observed in mood or executive function [67]. Based on this body of evidence, a daily intake of 10 mg of lutein as main carotenoid component is considered both safe and effective, and is the most widely studied and recommended dose in clinical trials and expert reviews [68]. Further evidence suggests that the combination of lutein with other nutraceuticals may enhance their visual/cognitive positive effects. In this sense, and noteworthy, the omega-3 PUFA DHA is widely present in the macula and has a relevant role for regulating oxidative stress in retinal pigment epithelial cells. Based in this rationale, supplementation of high dose omega-3 PUFA including DHA has been shown to reduce AMD progression in some clinical studies, and its use in combination with lutein could produce synergistic effects in terms of protection against oxidative and photochemical damage to the macula [69].
Consequently, in a 4-month randomized controlled trial, combined supplementation with lutein and DHA improved verbal fluency, memory performance, and learning efficiency in older women, while the individual administration of each compound improved verbal fluency alone [62]. A more recent set of randomized placebo-controlled trials in older Japanese adults showed that combined supplementation with lutein, zeaxanthin, arachidonic acid, DHA, and EPA led to significant improvements in composite, verbal, and visual memory scores in individuals with mild cognitive decline. These effects were observed in healthy, non-demented, older individuals with cognitive decline, highlighting the potential benefit of targeted interventions in at-risk populations [70].
Choline: Choline is an essential nutrient involved in numerous physiological and neurochemical processes that are critical for brain function. It serves as a precursor of acetylcholine, a neurotransmitter implicated in attention and memory, and as a structural component of phosphatidylcholine and sphingomyelin, which are vital for membrane integrity and synaptic plasticity [71,72]. Furthermore, choline contributes to methyl-group metabolism through its oxidation to betaine, influencing epigenetic regulation [73]. Its activity is particularly relevant in the hippocampus and prefrontal cortex, where it modulates memory, attention and emotional regulation via cholinergic neurotransmission [73]. While endogenous hepatic synthesis occurs, it is insufficient to meet physiological demands, making dietary intake essential [71]. Population studies indicate that over 90% of individuals fall below these levels [68,72]. In an APP/PS1 transgenic model lifelong administration of a choline-enriched diet resulted in improved spatial memory performance, reduced β-amyloid plaque burden, and decreased expression of alpha-7 nicotinic acetylcholine and Sigma-1 receptors in microglia.
These receptors are involved in regulating the neuroinflammatory response, and their downregulation is associated with reduced microglial activation. In parallel, lower concentrations of both soluble and insoluble forms of β-amyloid peptides Aβ40 and Aβ42 were detected, indicating a reduction in amyloidogenic processing [74]. Additionally, a systematic review of 44 rodent studies reported consistent improvements in learning and memory tasks following choline supplementation at different life stages, particularly in recognition memory and hippocampal-dependent functions [73]. Consistent with these findings, population-based data have shown similar associations. In a prospective cohort study including 125,594 participants from the UK Biobank, with a median follow-up of 11.8 years, moderate dietary choline intake was associated with a lower risk of developing dementia and AD. The analysis revealed a U-shaped relationship between total choline intake and incident dementia, with the lowest risk observed in the second quartile (from 332.89 to 353.93 mg/day) [75].
Additionally, moderate consumption of choline-related compounds such as free choline, phosphatidylcholine, sphingomyelin and alpha glycerophosphocholine (GPC) was associated with a 17 to 23% reduction in dementia risk. In terms of cognitive performance, participants in the moderate intake range showed better results in visual attention, fluid intelligence and processing speed, although no association was observed for episodic memory [75]. The GPC compound is a natural source of choline found prevalently in milk and dairy products [76] and naturally resides in the brain, playing an important role in the synthesis of acetylcholine [75]. When absorbed into the bloodstream, GPC freely crosses the blood-brain barrier to deliver free forms of choline, which are then converted to acetylcholine. GPC has shown to spike acetylcholine levels after 1 - 3 hours post-ingestion [77,78]. The rationale above suggests GPC could be the preferent choline compound for choline supplementation.
This narrative review examines the potential role of nutraceuticals across different stages of cognitive decline, including SMC, MCI, and more advanced conditions such as AD. The analysis highlighted six nutraceuticals with recurrent and clinically relevant mechanisms of action in relation to cognitive decline: DHA, folic acid, Se, vitamin D, lutein and choline. Based on the findings reported in the literature, these compounds appear to exert their effects at different levels, including the reduction of oxidative stress, modulation of neuroinflammation, stimulation of neurogenesis and synaptic plasticity, and overall neuroprotection. According to the available evidence, each nutraceutical may influence cognitive function through distinct but sometimes overlapping biological pathways. DHA and choline support membrane fluidity and synaptic transmission [28,74], folate and B vitamins (B6, B9, and B12) regulate methylation and reduce homocysteine [35,36], and Se and lutein enhance antioxidant capacity and neuroprotection [48,61]. Vitamin D has been associated with the modulation of inflammation, the expression of neurotrophic factors [49,53,54], and the decrease of oxidative stress markers in the blood, which can affect neuroinflammation and contribute to cognitive decline [79,80].
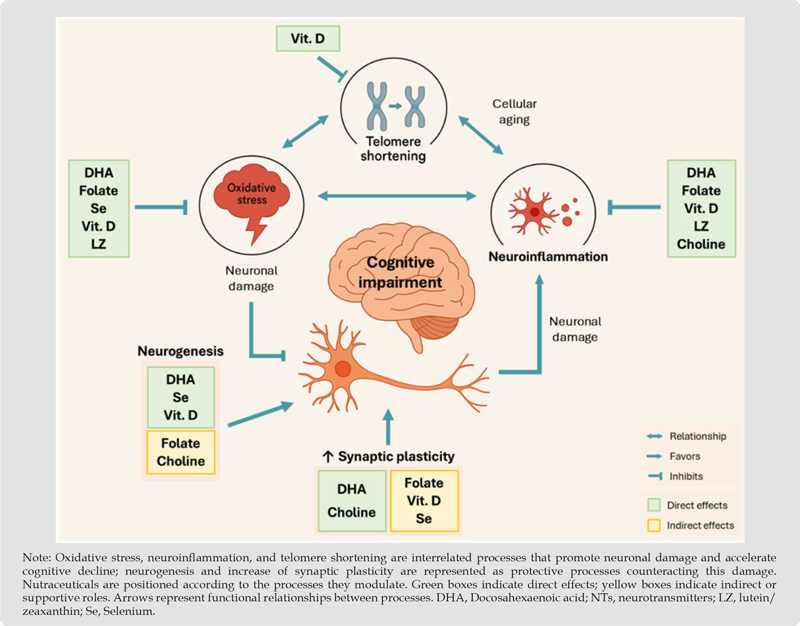
More recently, vitamin D3 supplementation has also been linked to the preservation of telomere length, suggesting potential systemic effects relevant to cognitive aging [58,59]. In addition to these effects, DHA, Se, and vitamin D have shown the ability to promote adult hippocampal neurogenesis [33,47,53], while folate and choline contribute indirectly through epigenetic regulation and structural membrane synthesis [36,73]. Regarding synaptic plasticity, both DHA and choline play a direct role in maintaining membrane fluidity and neurotransmitter synthesis [28,71,74], whereas vitamin D, folate, and Se support this process through neurotrophic modulation, methylation, and antioxidant protection, respectively [35,48,49]. The mechanisms and associated clinical benefits of each nutraceutical are summarized in Table 1. Additionally, Figure 1 illustrates the interrelation of the processes involved in cognitive impairments, and the effect nutraceuticals at multiple levels. Importantly, several studies suggest that the combination of nutraceuticals yields greater cognitive benefits than isolated compounds. As an example, VITACOG trial and a study carried out by Li et al. [40-42], demonstrated that folate combined with DHA or other B vitamins results in superior outcomes compared to monotherapy.
Table 1: Summary of the mechanisms of action and cognitive benefits associated with selected nutraceuticals.
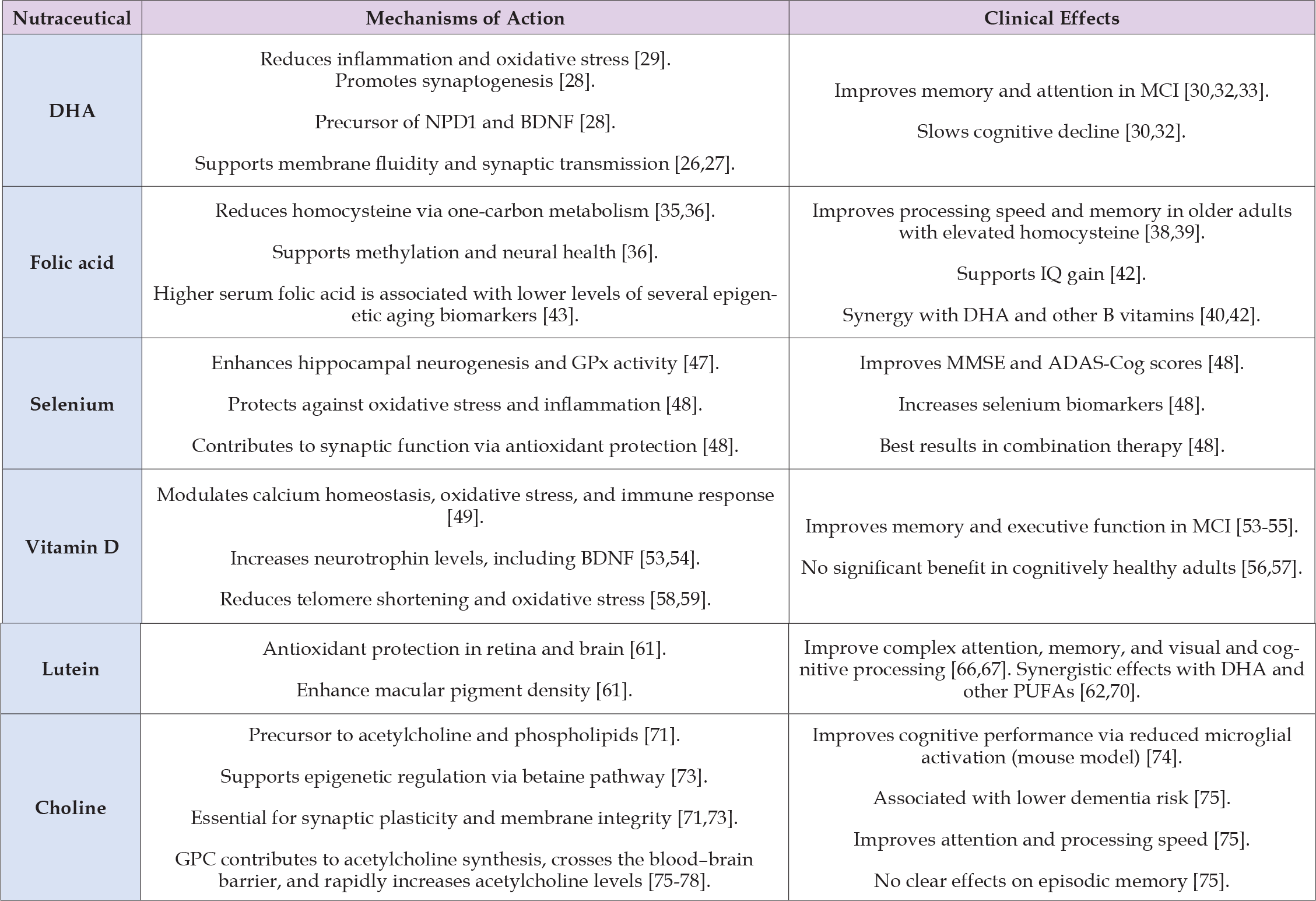
Note: Summary of key biological mechanisms and clinical evidence from selected studies evaluating the cognitive effects of nutraceuticals. The references correspond to the full-text citations included in the manuscript. ADAS-Cog: Alzheimer’s Disease Assessment Scale–Cognitive subscale; BDNF: Brain-Derived Neurotrophic Factor; DHA: Docosahexaenoic Acid; GPC: glycerophosphocholine; GPx: Glutathione Peroxidase; MCI: Mild Cognitive Impairment; MMSE: Mini-Mental State Examination; NPD1: Neuroprotectin D1.
Figure 1: Pathophysiological mechanisms contributing to cognitive impairment and the modulatory effects of selected nutraceuticals
Similarly, nutrient combination formulations containing lutein, zeaxanthin, and PUFAs (DHA, EPA, and arachidonic acid) have been shown to improve verbal, visual, and composite memory scores [62,70]. These findings highlight the potential of multicomponent interventions to address the multifactorial nature of cognitive aging. From a public health perspective, nutritional epidemiology supports the relevance of these interventions. The ANIBES study reported widespread deficiencies in omega-3 fatty acids, folate, and vitamin D among older adults in Spain [18], nutrients that are consistently associated with better cognitive outcomes in clinical trials [39,49,66]. The combination of frequent nutrient gaps and consistent evidence of benefit highlights the value of nutritional support in cognitive aging. Nevertheless, clinical evidence is not uniform. While some randomized trials show improvements in memory, executive function, or processing speed [30,33,42,53], others report limited or null effects, especially in cognitively healthy individuals [56,57]. These discrepancies may reflect differences in study design, intervention duration, baseline nutrient levels, and sensitivity of cognitive tests employed [25].
Indeed, data from DHA and folate trials suggest that individuals with lower baseline levels of these nutrients are more likely to benefit from supplementation [32,75]. Folate provides a clear example of this variability. While FACIT and VITACOG used high-dose supplementation (800 μg/day) to lower homocysteine and improve cognition [38,40], other studies have shown that even standard doses, such as 400 μg/day, may be beneficial even in individuals with inadequate dietary folate intake [34,39]. These findings raise important questions about dose individualization and the role of nutritional screening in clinical decision-making [36]. Similarly, Se supplementation has shown promise in improving MMSE and ADAS-Cog scores, particularly when combined with other nutrients [48]. However, its narrow therapeutic window and limited long-term data necessitate further research [46]. Vitamin D has also shown selective benefits in individuals with MCI but not in cognitively intact subjects, suggesting a threshold effect or a need for targeted use [51,53,54]. Taken together, these findings support the exploration of personalized nutritional strategies. Future interventions may benefit from stratifying participants by baseline nutrient status, genetic polymorphisms, or clinical phenotype to better predict response [75].
Rather than focusing exclusively on single nutrients, nutrient combination formulations designed to reflect common dietary deficiencies in aging populations may offer broader and more consistent cognitive benefits [42,70]. While nutraceuticals are not substitutes for pharmacological or behavioural therapies, their favourable safety profile and accessibility make them appealing as adjunctive tools in preventive care, particularly in early stages of decline when interventions are most effective [21,22]. Although not substitutes for established interventions, nutraceuticals may represent a complementary strategy worth exploring in the context of early cognitive decline, especially as further evidence emerges. However, this review presents certain methodological limitations. As a narrative synthesis, it lacks a predefined search protocol, which may introduce selection bias and limit reproducibility. However, this format allowed for greater flexibility in selecting and interpreting evidence, including mechanistic and observational studies, not always eligible for inclusion in systematic reviews, that contribute to a broader understanding of nutraceuticals and cognitive health. Given the heterogeneity of the available data, a flexible, narrative approach was considered the most appropriate to explore the state of the science and identify directions for future research.
In conclusion, this review summarizes the promising potential of nutraceuticals such as DHA, folic acid, Se, vitamin D3, lutein, and choline to support cognitive function and their beneficial role particularly in early cognitive decline, although heterogeneity in study designs and populations warrants cautious interpretation. Based on these results and the safety profile and accessibility of nutraceuticals, they can serve as valuable complementary tools within a holistic strategy for cognitive health, and personalized, nutrient combination interventions tailored to individual nutritional status may enhance their effectiveness. Further well-designed, long-term clinical trials are essential to clarify optimal formulations, dosing, and target populations.
The authors would like to acknowledge Meisys for their support in the preparation of this manuscript.
All authors contributed to the study. GJ and TP wrote the outline of the review. OJ revised and approved the outline. TP reviewed the literature and wrote the primary draft. GJ and OJ reviewed the literature and approved the primary draft. All authors read and approved the final manuscript.
This study was funded by ITF Research Pharma, S.L.U.
Not applicable.
Ethics Approval and Consent to Participate
Not applicable.
Competing Interests
GJ is an employee of ITF Research Pharma, S.L.U., Madrid, Spain. TP is an employee of Meysis and has received compensation for medical writing support related to this manuscript. OJ has received honoraria for participation in and review of the present manuscript.
Consent for Publication
Not applicable.

