Impact Factor : 0.548
- NLM ID: 101723284
- OCoLC: 999826537
- LCCN: 2017202541
Rajiv Kumar1* and MP Choudhary2
Received: March 21, 2025; Published: April 01, 2025
*Corresponding author: Rajiv Kumar, Faculty of Sciences, University of Delhi, New Delhi 110007, India
DOI: 10.26717/BJSTR.2025.61.009564
The extracellular matrix (ECM) consists of three layers: the epimysium, endomysium (basal lamina), and perimysium, which serve as a connector in the cell microenvironment, govern tissue development, and control the physiological functions of muscle cells. These three layers of the ECM directly participate in and influence the progression of mechanical force transmission, the formation of neuromuscular junctions, and the regeneration of muscle fibers [1]. Physiological signaling pathways alter the mechanical properties of the ECM components and stimulate contractility [2]. Hence, a comprehensive understanding of the structure and function of the ECM is of great concern and plays various roles in connecting tissues of the body, skeletal muscle, and overall physiological function [3]. Here, the authors discuss the importance of the ECM in the physiology of skeletal muscle regeneration and tissue. Furthermore, the application of the ECM in biomedical engineering is explored through the aforementioned constituents. Recently published articles featured various research papers highlighting the importance of the ECM in force transmission and tissue structure maintenance, especially in bone, muscle, tendons, and ligaments. The biological processes of collagen synthesis and degrading metalloproteinase enzymes have been linked with the mechanical loading and physical activity of the ECM (Figure 1).
Other activities, such as the systemic release of growth factors, transcription, and post-translational modifications, promote the previously mentioned activities of the ECM. To understand the pathways of tissue overloading, a detailed comprehension of the relevant physiological routes is necessary; that is why the constitutive modeling of the extracellular matrix is a critical aspect of vascular biology. The membrane matrix determines the structural and organizational stability of the ECM, as well as membrane-type matrix metalloproteinases that exhibit a signal within it [4]. Other functions of the ECM, including the regulation of various physiological processes such as proliferation, survival, migration, and invasion, are also examined. At this stage, the role of mechanical guidance forces is crucial in controlling organizational cues whenever cell-to-cell contact fails. Each time there is a change in the ECM proteins (collagen and elastin), the processes responsible for stiffness in the arterial wall are initiated. These abnormal pathways were activated when a failure occurred in ECM reorganization settings. Likewise, the authors stressed the necessity of using experimental and computational analysis to define the roles of ECM proteins in the mechanical properties of arteries [5]. These elucidation methods further reveal the pathways of opening and highlight other features, such as how the microenvironment and mechanotransduction influence arterial adaptation [6].
The interpretation of the micro-mechanical topographies generated in muscle can be analyzed to identify the presence of neuromuscular diseases. Such research will be beneficial in identifying the symptoms of severe diseases like cerebral palsy or muscular dystrophy in their initial stages. The analysis of the mechanical behavior of whole muscles will aid in selecting the appropriate materials that exhibit novel properties for optimal medical applications to treat muscle deformation and strain energy-density distributions. Recently, synthetic 3D extracellular matrices have been developed and applied in cell studies, drug discovery, and regenerative medicine [7]. This onion offers a clear view of needs of the constitutive modeling of the extracellular matrix of muscle tissue response and resemblance [8]. These insights have exploited bioresponsive cell microenvironments that can induce specific cell fates. Moreover, synthetic strategies have been emphasized for creating functional 3D ECM mimics. Additionally, the fundamentals of mechanical interpretation versus mathematical predictions of ECM are discussed, covering the chemical and structural features of scaffolds, which can generate ECM mimics and be useful in signaling biomolecules as needed.
The deformation gradient F(Χ,t), its determinant J (Χ,t), and the right Cauchy–Green tensor C(Χ,t) are defined as;
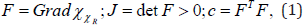
where the dependence on place and time is understood; and all symbols and notations are having their usual meanings [9].
Recently the researchers have been studied an anisotropic hyperelastic
material characterized by n reinforcing families of fibers
whose direction is specified by the unit vector 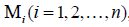 the
strain–energy density function (SEDF) must be a scalar isotropic tensor
function of C and , Μi where
the
strain–energy density function (SEDF) must be a scalar isotropic tensor
function of C and , Μi where 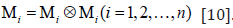 .
Applying the properties of the representation theorems for isotropic
scalar-valued functions of symmetric tensors, we can represent the
SEDF in the following form,
.
Applying the properties of the representation theorems for isotropic
scalar-valued functions of symmetric tensors, we can represent the
SEDF in the following form,
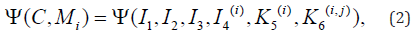
Where all symbols and notations are having their usual meanings [11]. Further, the materials with fibers dispersed symmetrically around the 𝑛 directions, generalized structure tensors Ηp have been studied by the researchers and can be defined as follows;
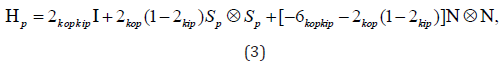
Where all symbols and notations are having their usual meanings. Also, [0,1/ 2] kip ∈ and [0,1/ 2] kop ∈ represent in-plane and outof- plane dispersion parameters, respectively [12]. Now the SEDF can then be represented in the following form;

Researchers have been drawing motivation from the experimentally observed muscle ECM microstructure, its composition and distribution in the muscle tissue, and its resemblance with the network structures found in other tissues such as skin, cornea, and arterial walls, the ECM was modeled based on the generalized structure tensor (Figure 2) [14]. There are particularly two local mean fiber directions as given below;
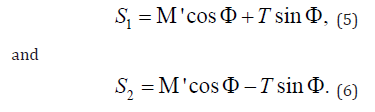
Where all symbols and notations are having their usual meanings. This concept was combined with the hyperelastic variant of the soft tissue model and the corresponding SEDF [15].
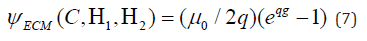
Where all symbols and notations are having their usual meanings. As per currently available experimental data observed by the researchers on ECM pointed at a significant influence of shear across the layered ECM, which is in line with the hypothesis that the transmission of lateral forces across muscle fibers in muscle tissues is largely facilitated through shearing the ECM [16,17]. Hence, the previous SEDF is supplemented by a term that depends on a simple kinematic invariant related to the shear across ECM layers.
The author (Rajiv Kumar) gratefully acknowledges his younger brother, Bitto, for his motivation. The author also thanks BioRender for providing the facility to illustrate the diagrams (Figure 1). Furthermore, he thanks BioRender for adapting and creating with permission [biorender.com] as per instructions.
Wherever necessary, relevant citations are included in the reference section. The author divided the reference section into two parts: scientific references and non-scientific references, to avoid any confusion.
The author has declared that no competing interest exists.

