Impact Factor : 0.548
- NLM ID: 101723284
- OCoLC: 999826537
- LCCN: 2017202541
Oluwafemi Emmanuel Ekun1*, Augustine Olusegun Olusola1, Chris Junior Iyalla1, Racheal Gift Ekun2, Joseph Adaviruku Sanni1, Oluwafemi Shittu Bakare1 and Feyisayo Ishola1
Received: October 02, 2023; Published: October 26, 2023
*Corresponding author: Oluwafemi Emmanuel Ekun, Department of Biochemistry, Faculty of Science, Adekunle Ajasin University, Akungba Akoko, Ondo State, Nigeria
DOI: 10.26717/BJSTR.2023.53.008408
This study evaluated the inhibitory activities of peptide fractions obtained from papain proteolysis of Moringa oleifera seed protein on carbohydrate-hydrolyzing enzymes. M. oleifera seeds were defatted using n-hexane, after which protein was extracted from the resulting seed meal via alkaline solubilization followed by precipitation in acidic medium. The extracted protein was hydrolyzed using papain and the resulting hydrolysates were separated into molecular weight peptide fractions F1 (<1 kD), F2 (1-3 kD) and F3 (3-5 kD) by gel filtration chromatography and comparison with elution volumes of standards of known molecular weights. The peptide fractions obtained were investigated for their abilities to inhibit α-amylase and α-glucosidase. Their mechanisms of inhibition were also determined. Results revealed that the peptide fractions inhibited α-amylase such that fractions F1 and F3 displayed 71.222 ± 2.437% and 72.088 ± 2.711% α-amylase inhibitory activities respectively. Fraction F3 demonstrated 90.605 ± 0.926 % inhibition of α-glucosidase, which was significantly higher (p<0.05) than the activities of the other two fractions. Kinetic analysis of selected fractions indicated mixed inhibition of both carbohydrate-hydrolyzing enzymes, with fractions F1 and F3 having enzyme – inhibition dissociation constants of 0.122 mg/mL and 1.367 mg/mL for α-amylase and α-glucosidase respectively. It is concluded that the subjection of Moringa oleifera seed proteins to papain hydrolysis could yield bioactive peptides with immense potential, which could be harnessed for the development of novel anti-diabetic agents.
Keywords: Hydrolysates; Peptides; Papain; Diabetes Mellitus; α-Amylase; α-Glucosidase
Abbreviations: MSPH: M. oleifera Seed Protein Hydrolysate; F1: Papain fraction 1(< 1kD); F2: Papain fraction 2(1-3 kD); F3: Papain fraction 3 (3-5 kD)
Protein hydrolysates are products obtained from the proteolysis of proteins into peptides of varying sizes via chemical or enzymatic hydrolysis. These protein digests, commonly called hydrolysates, have varying bioactivities which are dependent upon several factors such as protein source, method of hydrolysis, choice of proteolytic enzyme, degree of hydrolysis, hydrolysis time, composition and sequence of aminoacyl residues in the resulting hydrolysate (Arise, et al. [1,2]). Enzymatic methods of hydrolysis have largely supplanted chemical means of hydrolysis over time. Enzymes commonly used in generating bioactive peptides include pepsin, chymotrypsin, alcalase, Proteinase K, trypsin, among other proteases (Arise, et al. [1,3,4]). In addition, fractionation of protein hydrolysates has contributed significantly to their functional properties in various in vitro and in vivo models, such that some reports have demonstrated that peptide fractions of defined sizes (especially shorter-length peptide fractions) possess potent bioactivities (Awosika, et al. [5]), whereas other studies argued in favour of long chain peptides in eliciting certain bioactivities (Ekun [6]). In the last decade, there has been renewed interest in the exploration of these peptide products for their value-added benefits. Protein hydrolysates and their peptide fractions have been reported to possess a wide range of therapeutic and industrial applications (Lien, et al. [7]).
In several studies, they have been demonstrated to possess blood pressure-lowering potentials (Yamada, et al. [8,9]), antioxidant properties (Udenigwe, et al. [10-12]), antidiabetic potentials (Vilcacundo, et al. [13,14]), antiplasmodial effects (Perez-Picaso, et al. [15]), antineoplastic potentials (Vlieghe et al. [16,17]), among other disease-ameliorating properties. Diabetes mellitus is a disease of metabolic dysregulation, most notably a dysregulation of glucose metabolism, accompanied by long-term vascular and neurological complications. Diabetes has several clinical forms, each of which has a distinct etiology, clinical presentation, and course (Rhoades, et al. [18]). It is the most common endocrine disorder, as it adversely affects carbohydrate, protein and fat metabolism (Arise, et al. [1]). Symptoms usually include frequent urination (polyuria), increased thirst (polydipsia), increased food consumption (polyphagia), and unexplained weight loss (Vasudevan, et al. [19]). The elevated glucose levels (hyperglycemia) also spills into the urine, causing glycosuria. In diabetes mellitus, triacylglycerol hydrolysis, fatty acid oxidation, gluconeogenesis, and ketone body formation are accelerated, and, in a condition termed ketoacidosis, which represents the hallmark of uncontrolled diabetes mellitus, ketone body levels in the blood become abnormally high (Raju, et al. [20]). Since ketone bodies are acids, their high concentration puts a strain on the buffering capacity of the blood and on the kidney, which controls blood pH by excreting the excess hydrogen ions into the urine.
This unusually high excess H+ excretion is accompanied by electrolyte and water excretion, causing severe dehydration and a decrease in blood volume, which are ultimately life-threatening situations (Voet, et al. [21]). Complications arising from Diabetes mellitus include neuropathy, nephropathy, cardiovascular disease, stroke, renal impairments, among others (Deshmukh, et al. [22]). Therapeutic interventions have been aimed at controlling plasma glucose levels and these include a combination of pharmacologic and non-pharmacologic approaches. Non pharmacologic interventions include lifestyle changes such as the avoidance of foods with high glycemic index and physical exercise (Arise, et al. [1]). Pharmacologic approaches involve the use of specific drugs which affect glucose absorption, transport, metabolism and excretion (Katzung, et al. [23]). As a result, several classes of hypoglycemic agents exist, which help to bring down glucose concentration in patients. Key enzymes and proteins commonly targeted in diabetes mellitus therapy include amylase, glucosidase, glucose transporters, dipeptidyl peptidase (iv), among other pharmacologic targets (Katzung, et al. [4,24]). Moreover, many pharmacologic interventions can cause unwanted side effects which could lead to damage to internal organs in some cases. In addition, they care cost-intensively and can be difficult for patients and their families to procure. As a result, there is a growing need to explore other alternatives. Peptide products from plant proteins have been one of such alternatives that have been explored for their therapeutic potentials (Lopez-Barrios, et al. [12]).
One plant whose proteins have been excellent sources of bioactive peptides is Moringa oleifera. This plant is indigenous to peoples of Asian and African descent, and also grown in other parts of the world (Madubuike, et al. [24]). Its seeds have been found to be abundant in oils, proteins, carbohydrates as well as fibre (Kwaambwa, et al. [25]). The protein profile of M. oleifera leaves and seeds showed that it is abundant in albumins, prolamins, globulins as well as glutellins. Furthermore, amino acid analysis revealed that its seeds are especially rich in aspartic and glutamic acids, proline, arginine, leucine, threonine, but were limited in sulfur-containing amino acids (Mune-Mune, et al. [26]). Various extracts of M. oleifera leaf and seeds have been reported to possess numerous health promoting benefits (Divi, et al. [27,28]). M. oleifera extracts have been shown to demonstrate antioxidant properties, (Singh, et al. [29]) hypoglycemic effects (Ndong, et al. [30]), antihypertensive potentials (Anwar, et al. [31]), antimicrobial properties (Divi, et al. [27]) among other potentials, owing to the presence of physiologically active secondary metabolites. Also, investigations have been made in exploring the bioactive properties of peptides obtained from M. oleifera proteins. Several enzymes have been used to digest M. oleifera seed proteins and which have in turn yielded potentially bioactive protein hydrolysates. Also, fractionation of these peptides is essential to further enhance biologic activity (Awosika, et al. [5]) Commonly used proteases to hydrolyze M. oleifera seed proteins have been trypsin (Ekun [6]) pepsin (Ekun [32]), and chymotrypsin (Ekun, et al. [4]), and they have been used to generate antidiabetic protein hydrolysates and peptides in earlier studies.
However, information on the use of papain as a routine hydrolytic enzyme to digest proteins and yield potentially bioactive peptide fractions have been scarce. As a result, this study focused on investigating the carbohydrase-inhibitory effects of hydrolysates and peptide fractions obtained from papain-digested M. oleifera seed proteins.
Materials
oleifera seeds were obtained from farms in Akungba-Akoko, Ondo State, and was authenticated by the Department of Plant Science and Biotechnology, Adekunle Ajasin University, Akungba Akoko. Voucher samples were deposited, and number assigned. Enzymes used were papain (from Carica papaya) α-amylase (from yeast), α-glucosidase (bovine) were products of Sigma-Aldrich Laboratories, Co-Artrim, United Kingdom. All other reagents used in this study were of analytical grade and were also products of Sigma-Aldrich laboratories, Co-Artrim, United Kingdom.
Methods
Preparation of Defatted Seed Meal: The method described by (Arise, et al. [33]) was employed with slight modifications, such as the use of cold extraction method as against soxhlet extraction. Briefly, the seeds were dried, ground into powder and stored in an air-tight container at 4oC. This was then defeated using n-hexane. The meal was extracted three times with n-hexane using a meal/solvent ratio of 1:10 (w/v). The meal was then dried at 40oC in a vacuum oven and ground again to obtain a fine powder, and referred to as defatted seed meal, which was stored at -10oC.
Protein Extraction from Defatted Seed Meal: The protein component of the defatted meal was extracted using the method described by (Alashi, et al. [11]). One hundred grams (100 g) of the defatted seed meal was suspended in 1000 mL of 0.5 M NaOH solution, pH 12.0, and continuously stirred for an hour to achieve solubilization in alkali. The resulting suspension was spinned at a speed of 3000 g and a temperature of 18°C for 10 minutes. Two more extractions of the residue from the centrifugation were carried out using the same volume of sodium hydroxide and the resulting supernatants were pooled together. The supernatant pH was adjusted to 4.0 using 0.5M HCl solution in order to bring about acid-induced precipitation of Moringa seed protein, and the precipitate formed was recovered by centrifugation. The precipitate was washed with distilled water, adjusted to pH 7.0 using 0.5 M NaOH, freeze-dried and the protein isolate was then stored at -20°C until required for further analysis.
Preparation of oleifera Seed Protein Hydrolysates: The protein isolate was hydrolysed using the methods described by (Olusola, et al. [34]). Five grams (5g) of the protein isolate was dissolved in 100 mL of phosphate buffer solution pH 6.0 and at a temperature of 60ºC. The enzyme papain was added to the suspension at an enzyme-substrate ratio of 1:50. Hydrolysis was carried out at the specified conditions for six hours as the mixture was continuously stirred. The enzyme was then inactivated by boiling in water bath at 1000C for 15 minutes and proteins which were undigested were precipitated from the solution by pH adjustment to 4.0 with 2M HCl. This was followed by centrifugation at 7000 g for 30 minutes. The supernatant containing the peptides were then collected. The protein content of the samples was then determined using the Biuret assay method.
Fractionation of oleifera Seed Protein Hydrolysates: The Moringa oleifera seed protein hydrolysates were separated into molecular weight fractions using gel filtration chromatography as described by (Ekun, et al. [4]). Briefly, 5 mL of the clear supernatant resulting from protein hydrolysis, at a protein concentration of 10 mg/mL was filtered, suspended in 50 mM phosphate buffer pH 7 and passed into a sephadex G25 chromatographic column of dimensions 30cm x 4 cm which had earlier been equilibrated with the buffer. The same phosphate buffer was used to elute the separating fractions, and the elution peaks were monitored at 400 nm according to (Prasad, et al. [35]) The separating fractions eluted under the same elution peak were collected, pooled and their molecular weights were determined by comparison with the graph of the logarithm of molecular weights against elution volumes of known standards. The eluates, according to their molecular weights, were then sorted into <1 kD, 1-3 kD and 3-5 kD ranges. Peptide fractions of molecular weights higher than 5 kDa were removed and discarded. The collected peptide fractions were stored at -200C for further analysis.
Determination of Degree of Hydrolysis: The method described by (Olusola, et al. [2]) was employed. Aliquot amount (1) ml of protein hydrolysate was added to 1 ml of 20% TCA to produce 10% TCA soluble material. The mixtures were left to stand for 30 minutes to allow for precipitation, followed by centrifugation at 4000 g for 20 min. The supernatants were then analyzed for protein content using Biuret assay method with bovine serum albumin (BSA) as standard. The degree of hydrolysis (DH) was computed by dividing the amount of peptide soluble in 10% TCA by the total protein content of isolate and the result expressed in percentage.
Determination of Peptide Yield: The percentage peptide yield was determined using the method described by (Girgih, et al. [36]). The peptide yields (%) of Moringa oleifera seed protein hydrolysates were calculated as the ratio of peptide content of lyophilized hydrolysate/fraction to the protein content of unhydrolyzed protein isolate.
Determination of α-amylase Inhibition: An α-amylase-inhibitory assay was performed according to the method reported by (Oboh, et al. [37]). Briefly, 125 µL of the hydrolysate/peptide fraction (0.5 to 2.0 mg mL–1) was placed in test tubes and 125 µL of 20 mM sodium phosphate buffer (pH 6.9, with 6mM NaCl) containing α-amylase solution (0.5 mg/mL) added. The content of each tube was pre-incubated at 25°C for 10 min, after which 125 µL of 1% starch solution in 20 mM sodium phosphate buffer (pH 6.9, with 6 mM NaCl) was added at timed intervals. The reaction mixtures were incubated at 25°C for 10 min. The reaction was terminated by adding 250 µL of dinitro salicylic acid (DNS) colour reagent and further incubated in boiling water for 5 min and cooled to room temperature. The content of each test tube was diluted with 500 µL distilled water and the absorbance measured at 540 nm. A control was also prepared using the same procedure except that the hydrolysate was replaced with distilled water. The α-amylase-inhibitory activity was calculated as shown:
% Inhibition = ( A control – A sample) / A control × 100.
The concentration of hydrolysate resulting in 50% inhibition of enzyme activity (IC50) was determined from a plot of percentage inhibition against hydrolysate concentrations using GraphPad Prism version 6.0 (GraphPad Software, San Diego, CA, USA).
Determination of Kinetics of α-amylase Inhibition: The kinetic study of α-amylase inhibition was conducted according to the method described by (Olusola, et al. [34]). Aliquot volume (125 µL) of the hydrolysate was pre-incubated with 125 µL of α-amylase solution for 10 min at 25°C in a set of tubes. In another set of tubes, 250 µL of phosphate buffer (pH 6.9) was also pre-incubated with 125 µL of α-amylase solution. Starch solution (125 µL) of increasing concentrations (1.0 to 8.0 mg mL–1) were added to both sets of reaction mixtures to initiate the reaction. The mixture were then incubated for 10 min at 25°C, and then boiled for 5 min after the addition of 250 µL of dinitro salicylic acid (DNS) reagent to stop the reaction. The amount of reducing sugars released was determined spectrophotometrically from a maltose standard curve and converted to reaction velocities as shown below:
Specific Activity (μmol mg protein –1) min –1) = Maltose released / Incubation time × enzyme amount (in mg / mL) in reaction mixture
where maltose concentration is in µmol mL–1; Incubation time = 10 min;
A double reciprocal plot (1/V versus 1/[S]), where V is reaction velocity and [S] is substrate concentration was plotted. The mode of inhibition and the kinetic parameters of α-amylase inhibition by hydrolysates were determined by analysis of the double reciprocal plot. The inhibition constant (Ki) was determined using a secondary plot known as the Dixon plot (Palmer [38]), by plotting a graph of inverse of initial velocities on the y-axis against inhibitor concentrations on the x-axis, at fixed substrate concentration.
Determination of α-Glucosidase Inhibition: The effect of the hydrolysates on 𝛼-glucosidase activity was determined according to the method described by (Kim, et al. [39]). The substrate solution p-nitrophenyl glucopyranoside (pNPG) was prepared in a 20 mM phosphate buffer, and pH 6.9. 100𝜇L of 𝛼 glucosidase from Saccharomyces cerevisiae (1.0 U/mL) was pre-incubated with 50𝜇L of the different concentrations of the hydrolysates for 10 min. Then 50𝜇L of 3.0 mM (pNPG) as a substrate dissolved in 20 mM phosphate buffer (pH 6.9) was added to start the reaction. The reaction mixture was incubated at 37∘C for 20 min and stopped by adding 2mL of 0.1 M Na2CO3 The 𝛼-glucosidase activity was determined by measuring the yellow-colored para-nitrophenol released from p-NPG at 405 nm. The results were expressed as percentage of the blank control. Percentage inhibition was calculated as:
% Inhibition = ( Acontrol – Asample) / Acontrol × 100.
Determination of Kinetics of α-Glucosidase Inhibition
The kinetic parameters of 𝛼-glucosidase by the hydrolysates were determined according to the modified method described by (Ali et al. [40]). Briefly, fifty (50) 𝜇L of the (5 mg/mL) hydrolysate was pre-incubated with 100 𝜇L of 𝛼-glucosidase solution for 10 min at 25∘C in one set of tubes. In another set of tubes, 𝛼-glucosidase was pre-incubated with 50𝜇L of phosphate buffer (pH 6.9). Fifty (50) 𝜇L of pNPG at increasing concentrations (0.5–2.0 mg/mL) was added to both sets of reaction mixtures to start the reaction. The mixture was then incubated for 10 min at 25∘C, and 500 𝜇L of Na2CO3 solution was added to stop the reaction. The amount of reducing sugars released was determined spectrophotometrically at 405nm using a para nitrophenol standard curve and converted to reaction velocities. A double reciprocal plot (1/V versus 1/[S]) where V is reaction velocity and [S] is substrate concentration was plotted. The mode of inhibition of the hydrolysates on 𝛼-glucosidase activity was determined by analysis of the double reciprocal (Lineweaver-Burk) plot using Michaelis Menten kinetics. The inhibition constant (Ki) was also determined using the Dixon plot (Palmer [38]), by plotting a graph of inverse of initial velocities on the y-axis against inhibitor concentrations on the x-axis, at fixed concentration of substrate.
Statistical Analysis: The results were presented as means± standard deviation of triplicate determinations. One Way Analysis of Variance (ANOVA) and Tukey’s multiple range tests were used in statistical analysis of the results obtained. Differences among means were considered significant at p<0.05. The software package used was GraphPad Prism version 6.0 (GraphPad Software, San Diego, CA, USA).
Degree of Hydrolysis and Peptide Yield
The degrees of hydrolysis of Moringa oleifera seed protein isolate (MSPI) with papain was 51.590 ± 3.891 %, whereas the peptide yield was 54.880 ± 4.132 %.
Alpha-Amylase Inhibitory Activity
The percentage α-amylase inhibitory activity of papain-derived Moringa oleifera seed protein hydrolysate and its fractions in relation with acarbose are illustrated in (Figure 1). The inhibitory activities of the hydrolysates and fractions at all study concentrations were significantly (p<0.05) lower than those of acarbose. All peptide fractions demonstrated higher (p<0.05) inhibitory activity than the unfractionated hydrolysate. The hydrolysate fractions demonstrated inhibitory activities above 50% at all concentrations in the study, such that papain fractions F1 and F3 showed higher (p<0.05) inhibitory effects at 0.2 mg/mL, 0.4 mg/mL and 1.0 mg/mL than F2. F1 and F3 attained maximal inhibitory extent of 71.222 ± 2.437 % and 72.088 ± 2.711 % at a final concentration of 1.0 mg/mL which was significantly (p<0.05) higher than that obtained by F2, which attained maximum inhibitory activity of 67.836 ± 1.169% at 0.8 mg/mL. The unhydrolyzed papain hydrolysate, MSPH, had lower (p<0.05) α-amylase inhibitory activity when compared to its peptide fractions. Comparing all hydrolysates and their fractions, chymotrypsin hydrolysate fraction F3 had the highest (p<0.05) α-amylase inhibitory activity, followed by F1 and F2. The IC50 values of α-amylase inhibition by M. oleifera seed protein hydrolysates and their peptide fractions in relation to the standard, acarbose are depicted in (Figure 2). Acarbose had an IC50 value of 0.151 ± 0.027 mg/mL. The hydrolysate and its fractions F1, F2 and F3 had IC50 values of 0.220 ± 0.062 mg/mL, 0.448 ± 0.029mg/mL and 1.183 mg/mL respectively. Bars are expressed as means ± standard error of mean of triplicate determinations (n=3). Values within the same concentration but with different letters are significantly different (p<0.05). Values at different concentrations of the same hydrolysate with different symbols are also significantly different (p<0.05). Bars carrying the same letter or symbol are not significantly different from one another (p<0.05). Bars are expressed as means ± standard error of mean (SEM) of triplicate determinations (n=3). Bars with the same letters do not differ significantly while bars with different letters are significantly different (p<0.05) from one another.
Kinetics of α-Amylase Inhibition
The peptide fraction with the least IC50 (F1) was evaluated for its effect on the catalytic activity of α-amylase in converting starch to maltose. The result is presented in (Figure 3). Kinetic parameters determined from the Lineweaver-Burk plot in the absence and presence of two different concentrations of the selected peptide fraction is summarized in (Table 1). Inhibition of α-amylase activity increased with increasing concentrations of the peptide fractions, such that the KM of the enzyme increased while Vmax and catalytic efficiency, CE, of α-amylase were reduced in the presence of the peptide fractions. The enzyme-inhibitor dissociation constant, KI, of α-amylase inhibition by fraction F1 was found to be 0.122 mg/mL. F1 showed an uncompetitive subtype of mixed inhibition at 0.5 mg/mL, while exhibiting a simple mixed inhibition at 1.0 mg/mL.
Table 1: Kinetic Parameters of α-amylase-catalyzed hydrolysis of starch in the presence and absence of an inhibitory peptide fraction.
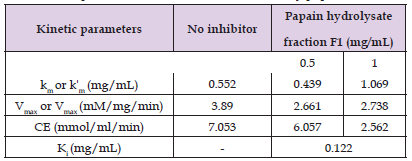
Note: 1. Km or Kˈm: Michaelis constant in the absence or presence of the inhibitory hydrolysate fraction. 2. Vmax or Vˈmax: Maximum velocity in the absence or presence of inhibitory hydrolysate fraction. 3. CE: Catalytic Efficiency. 4. Ki: Enzyme-Inhibitor dissociation constant.
α-Glucosidase Inhibitory Activity
The inhibitory activities of the M. oleifera seed protein hydrolysate and fractions on α-glucosidase–catalyzed hydrolysis of p-nitrophenyl glucopyranoside at varying concentrations in comparison to acarbose (control) is summarized in (Figure 4) while their IC50 values are displayed in (Figure 5). Hydrolysates and fractions obtained from papain digestion showed α-glucosidase inhibition, which was concentration dependent, but only in the case of unfractionated papain and fraction F3 (Figure 4). Fractions F
and F2 did not display significant (p<0.05) increases in α-glucosidase inhibition as concentrations were increased. The unfractionated hydrolysate and its fractions F1, F2 and F3 achieved maximal inhibitory activities of 30.555 ± 0.825 %, 19.992 ±2.366 %, 24.848 ± 2.891 % and 90.605 ± 0.926 % respectively at a final concentration of 1.0 mg/mL. They all had lower (p<0.05) inhibitory activity when compared to acarbose. However, fraction F3 had the highest (p<0.05) inhibitory activity when compared to other fractions and the unfractionated papain hydrolysate. The IC50 values of α-glucosidase inhibition by papain hydrolysate and F1, F2 and F3 displayed in (Figure 5) showed that they inhibited α-glucosidase to a 50% extent at concentrations of 1.506 ± 0.066 mg/mL, 0.702 ± 0.056mg/mL, 0.357 ± 0.085 mg/mL and 0.772 ± 0.041 mg/mL. These are significantly higher than acarbose, but F2 had lower IC50 value when compared to other papain hydrolysates and fractions. Bars are expressed as means ± standard error of mean of triplicate determinations (n=3). Values within the same concentration but with different letters are significantly different (p<0.05). Values at different concentrations of the same hydrolysate with different symbols are also significantly different (p<0.05). Bars carrying the same letter or symbol are not significantly different from one another (p<0.05). Bars are expressed as means ± standard error of mean (SEM) of triplicate determinations (n=3). Bars with the same letters do not differ significantly while bars with different letters are significantly different (p<0.05) from one another.
Kinetics of α-Glucosidase Inhibition
The effect of a selected M. oleifera seed protein hydrolysate fraction (F3) on the kinetics of α glucosidase–catalyzed hydrolysis of p-nitrophenyl glucopyranoside, p-NPG, to p-nitrophenol is illustrated in (Figure 6). The kinetic parameters from the resulting Line-weaver Burk plots are summarized in (Table 2). In the absence of inhibitor, the Michaelis constant, KM of α- glucosidase for its substrate was determined to be 0.297 mg/mL p-NPG, while maximum velocity, Vmax, was 270.27 mM/mg/min. All hydrolysate fractions caused decreases in the Vmax and catalytic efficiency, CE of the enzyme. Fraction F3 caused a concentration dependent increase in the apparent KM of α- glucosidase for its substrate. The selected peptide fraction also had an enzyme-inhibitor dissociation constant (kI)of 1.367mg/mL. The fraction F3 displayed a mixed type of inhibition at both study concentrations (0.5 mg/mL and 1.0 mg/mL).
Table 2: Kinetic Parameters of α-Glucosidase-catalyzed reaction in the Presence and Absence of M. oleifera Seed Protein Hydrolysate Fractions.
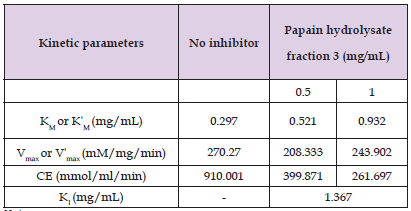
Note: 1. Km or Kˈm: Michaelis constant in the absence or presence of inhibitory hydrolysate fractions. 2. Vmax or Vˈmax: Maximum velocity in the absence or presence of inhibitory hydrolysate fractions. 3. CE: Catalytic Efficiency. 4. Ki: Enzyme-Inhibitor dissociation constant.
The degree of hydrolysis (DH) is a measure of the number of cleaved peptide bonds in a protein. DH can affect the molecular sizes and amino acid compositions of the peptides and thereby affect the biological activities of the peptides formed during hydrolysis. Consequently, the degree of hydrolysis is essential in determining the functional properties of protein hydrolysate preparations (Jamdar, et al. [2,41]). The degree of hydrolysis obtained for papain hydrolysates (51.18 ± 2.46 %) at an enzyme-substrate ratio of 1:50 was higher than the duo of 27.00 ± 0.50 % and 28.80 ± 0.57 % degrees of hydrolysis determined for Cryptozona bistrialis protein hydrolysates at E/S ratios of 1:100 and 1:50 respectively (Ulagesan, et al. [3]). However, it was not different from 53.48 ± 0.49 % obtained for freshwater catfish protein hydrolysates (Seniman, et al. [42]) at an enzyme – substrate ratio of 1:50. It was also lower than the DH of 71.98% obtained for another study on catfish protein hydrolysates at an enzyme-substrate ratio of 6% (that is, 3:50) (Baehaki, et al. [43]) The DH of papain proteolysis could be due to the relative non-specificity of the endo protease (when compared to proteases such as pepsin, trypsin and chymotrypsin), which in turn leads to hydrolysis of more peptide bonds, producing potential biologically active peptides. In addition, (Bagul, et al. [44]) had previously shown that an increased E/S ratio also raises the degree of protein hydrolysis by papain.
Peptide yield gives an estimate of the amount, in percentage, of peptides generated relative to the whole protein subjected to enzymatic proteolysis; thus, it represents an important index in determining the efficiency of the overall process (Alashi, et al. [11]), as these enzymes degrade the proteins into several peptides of varying lengths and sizes. It therefore follows that a high peptide yield is indicative of an increased proteolysis and resultant peptide release (Girgih, et al. [36]). Peptide yield is also correlated with the degree of hydrolysis. This is such that, a higher peptide yield indicates that the protein is more susceptible to hydrolytic cleavage by a given protease into peptides of shorter chain lengths, and a corresponding higher degree of hydrolysis (Arise, et al. [1]). The enzyme α-amylase in mammals is an important component of both saliva and pancreatic juice, and it catalyzes the hydrolysis of α-(1-4) glycosidic bonds of polysaccharides, releasing glucose and maltose in the process (Voet, et al. [21]). In recent times, protein hydrolysate preparations and peptides from some plant and animal sources have been demonstrated to inhibit α-amylase activity, with potential implications for alternate therapies to the management of diabetes mellitus (Arise, et al. [1,5,45]). In this study, the hydrolysate and peptide fractions demonstrated lower α-amylase inhibitory activities when compared to acarbose, and this is not unexpected because acarbose is a synthetic inhibitor of α-amylase.
Also, the peptide fractions exhibited better α-amylase inhibition than the unfractionated hydrolysate, and this could be that the fractionation process improves bioactivity, by allowing for more bioactive peptides to gain access to the enzyme active site, causing inhibition of the enzyme. This is also consistent with the reports of (Malomo, et al. [46]) that hydrolysates do contain large molecular weight peptides that possess antagonistic effects to enzyme inhibition. Among all of the peptide fractions, those with the highest inhibitory activities at a maximum concentration of 1.0 mg/mL are papain hydrolysate fractions F3 and F1 with values of 72.088 ± 1.169% and 71.222 ± 2.437% respectively. This could be a direct result of the nature of the peptides released by these enzymes during proteolysis. It is known that papain, owing to its relative non-specificity, could release a myriad of shorter and biologically active peptides that could potentially inhibit α-amylase. Reports from previous studies stated that phenylalanine, leucine, proline and glycine residues are required for the inhibition of α-amylase (Yu, et al. [47,48]). Similarly, (arise, et al. [1]) also suggested that α-amylase binds to peptides containing cationic residues such as Lys and branched chain residues such as Phe, Tyr and Trp. Thus, these peptides obtained from enzymatic proteolysis could contain these specific amino acid residues that locks into sites on the enzyme, inhibiting its activity in the process. The Lineweaver-Burk plot was used to determine the mode of α-amylase inhibition by varying concentrations of selected peptide fractions obtained from M. oleifera seed protein hydrolysates in this study.
Also, the kinetic parameters determined from the double-reciprocal plots were summarized in (Table 2), suggesting that the Michaelis constant, km of α-amylase (from Saccharomyces cerevisiae) in the absence of inhibitory hydrolysates is 0.552 mg/mL of starch. The presence of increasing amounts of the hydrolysate fraction increased the apparent Km of the enzyme for its substrate, while also reducing both maximal velocity, Vmax, and catalytic efficiency, CE of α-amylase. Papain hydrolysate fraction F1 exhibited a mixed type of inhibition, and this suggests that the peptides in this fraction are capable of binding α-amylase in both its free form and in its starch-bound forms, creating dead-end complexes on both occasions. (Arise, et al. [1]) had reported a mixed type of inhibition of α-amylase for peptic, tryptic and alcalase hydrolysates of Citrullus lanatus seed protein hydrolysates. The KI value of 0.122 mg/mL obtained for F1 in this study is higher than that obtained for peptic fraction 1 from M. oleifera seed protein hydrolysate (0.029 mg/mL) in another kinetic study of amylase inhibition (Ekun [32]) but lower than chymotryptic fraction F2 of the same hydrolysate reported in earlier studies (Ekun, et al. [4]). It appears that binding affinities of these peptides to amylase are inversely correlated with average molecular weights of peptides, and this position is also corroborated by (Awosika, et al. [5]) that peptides with low molecular weights have more propensities to access and inhibit substrate binding sites and/or allosteric sites required for α-amylase activity.
The enzyme α-glucosidase is commonly found on the brush border membranes of the intestinal mucosa and participates in carbohydrate digestion by hydrolyzing glucose residues from oligosaccharides (Voet, et al. [21]). Thus, the modulation of the activity of this enzyme represents one of the key strategies in the control of blood glucose levels in the management of diabetes mellitus (Qaisar, et al. [49]). All hydrolysates and their fractions demonstrated lower α-glucosidase inhibitory activities than control (acarbose). This is because acarbose happens to be a dual inhibitor of α-amylase and α-glucosidase, with higher binding affinity for α-glucosidase (Katzung, et al. [23]). Among the papain hydrolysate fractions, the F3 fraction which comprises peptides having molecular weights of between 3 and 5 kD was shown to have high inhibitory activities against α-glucosidase. This could be that the peptides in that fraction exhibited synergistic effects in α-glucosidase inhibition. Also, papain being a relatively non-specific proteinase may have caused the cleavage and release of several peptides of varying structures, lengths and sizes (Baehaki, et al. [43]), resulting in their interactions with substrate binding sites on α-glucosidase, inhibiting its activity in the process. When compared to kinetics of α-amylase inhibition, there is paucity of information in the literature regarding kinetic analysis of α-glucosidase inhibition by protein hydrolysate fractions obtained from papain proteolysis. The kinetic parameters obtained from the double - reciprocal plots of α-glucosidase inhibition by a selected peptide fraction (F3) in (Figure 6), is summarized in (Table 2).
Kinetic analysis of papain peptide fraction F3 revealed that it inhibited α-glucosidase via a mixed inhibition mode, that is, these peptides were able to bind and inhibit the enzyme, both in its free form and when it has bound the substrate, p-nitrophenyl glucopyranoside, p-NPG. This is evident by the increases in the apparent K/M, and the reduction in the maximal velocity, Vmax, when compared with the parameters in the absence of inhibitor. This correlates to what was obtained with the <1 kD fraction of chymotryptic digests of yellow field pea protein hydrolysate (Awosika, et al. [5]) as they inhibited α-glucosidase via a mixed type of inhibition mechanism. (Ibrahim, et al. [14]) had stated that aminoacyl residues such as tyrosine, lysine, arginine and serine are needed at terminal ends of a peptide for α-glucosidase inhibitory activity, and owing to the moderate specificity of the enzyme papain (as it hydrolyzes peptide bonds of basic aminoacyl residues, glycine and leucine), proteolysis could have released peptides having these residues at their terminals (Voet, et al. [21]) hence the α-glucosidase inhibitory activities observed by peptide products obtained from the proteolytic action of papain.
This study concludes that proteins from M. oleifera seeds, on hydrolysis with papain and subsequent fractionation produced peptide fractions that elicited antidiabetic potentials by inhibiting carbohydrate hydrolyzing enzymes – α-amylase and α-glucosidase. Also, this implies that M. oleifera seed proteins could be potential sources of bioactive peptides possessing antidiabetic potential. In addition, it is recommended that structural identification, modifications, and possible chemical synthesis of peptides responsible for the observed bioactivities should be considered in further research.

