Impact Factor : 0.548
- NLM ID: 101723284
- OCoLC: 999826537
- LCCN: 2017202541
Györgyi Szabó1*, Domokos Csukás1, János Hanics2, Katalin Dezső3, Ramin Talebian1, Krisztina Juhos1, Daniella Fehér1, József Sándor1 and Andrea Ferencz1
Received: October 04, 2023; Published: October 13, 2023
*Corresponding author: Györgyi Szabó, Department of Surgical Research and Techniques, Heart and Vascular Center, Semmelweis University, 1085 Budapest Üllői út 26, Hungary
DOI: 10.26717/BJSTR.2023.53.008377
At first sight, the esophagus is a tube that connects the pharynx and the stomach, but its structure and function are more complex. It secretes mucus that helps the food pass, and its wall has multiple layers with different tissues that protect the esophagus and help the bolus move toward the stomach. The human esophagus has four main histological layers: tunica mucosa, tela submucosa, tunica muscularis (or muscularis propria), and adventitia. These layers are similar but not the same in different mammal species. Seven laboratory animal species were used for our anatomical examination: mouse, rat, guinea pig, rabbit, cat, dog, and pig. The tissue samples were taken from 3 zones, the upper, middle, and lower parts of the esophagus, and all layers were examined. The extension and structure of the layers showed significant differences comparing the species and even the zones in the same animal. Esophageal diseases are ranged from minor mutations to adenocarcinoma. Finding the best way to prevent or treat a reliable animal model is inevitable. Three aspects are considered to find a suitable model: practical, scientific, and ethical aspects. This research provided data for the scientific aspect. An ideal model that fulfils almost all perspectives would be pigs, due to their anatomical and dietary similarities and the related ethical and practical aspects.
Keywords: Esophagus Anatomy; Laboratory Animals; Comparative Study; Animal Model; Esophageal Diseases
Abbreviations: BE: Barrett’s Esophagus; CM: Circular Muscle; GERD: Gastroesophageal Reflux Disease; LEM: Lamina Epithelialis Mucosae; LES: Lower Esophageal Sphincter; LM: Longitudinal Muscle; LMM: Lamina Muscularis Mucosae; LPM: Lamina Propria Mucosae; MALT: Mucosa-Associated Lymphoid Tissue; PAS: Periodic Acid-Schiff; SMGs: Submucosal Glands; STRP: Stratum Planocellulare; TSM: Tela Submucosa; TM: Tunica Muscularis; UES: Upper Esophageal Sphincter
At first sight, the esophagus is a tube that connects the pharynx and the stomach, but its structure and function are not so simple. It secrets mucus that helps the food pass, and its wall has multiple layers with different tissues that protect the esophagus and help the bolus move towards the stomach. Functional return is very poor after damage because the blood supply is weak, and no serosa holds the structure [1]. There are two sphincters that form a border between the neighbouring organs, the upper esophageal sphincter (UES), also called the pharyngoesophageal sphincter, and the lower esophageal sphincter (LES), called cardiac sphincter. This ’tube’ has an important function in passing the food toward the stomach via peristaltic contraction. The primary peristalsis initiates from the pharynx, helping the bolus pass through the UES and enter the stomach. If the primary peristalsis is not enough, the secondary peristalsis helps the bolus move forward. LES has an important role in preventing stomach content from entering the esophagus [2]. In the human esophagus, three regions can be identified according to their position: the cervical, thoracic, and abdominal regions. The cervical segment begins at the level of the cricopharyngeal muscle and ranges to the level of the suprasternal notch. It runs behind the trachea. The highest point of the thoracic region can be found on the left side of the sixth cervical vertebra. The lowest point is at the diaphragm. The abdominal region extends from the diaphragm to the cardia of the stomach [2].
The main layer difference between the regions is the type of muscle present, as mentioned above. The human esophagus has four main histological layers: tunica mucosa, tela submucosa, tunica muscularis (or muscularis propria), and adventitia [3]. The tunica mucosa consists of three sublayers (laminae). The first inner sublayer (or lamina) is the lamina epithelialis that surrounds the lumen. It is a non-keratinized, stratified squamous epithelium with three cellular layers (or strata): stratum basale, stratum spinosum, and stratum planocellulare (STRP). The thickness of the epithelium is limited, as it has no direct blood supply. Its nutrition is supported only by diffusion from the vessels of the connective tissue in the mucosal sublayer, the lamina propria. If there are connective tissue papillae present, the blood supply can easier reach the cells so the thickness of this mucosal sublayer can be wider. The top layer of non-keratinized epithelium the STRP can be identified by flattened cells with oval nuclei [4]. On the surface of the epithelium, a mucous layer protects the esophagus from abrasion, bacteria, and chemical effects. This mucous layer consists of mucins, which are different types of glycoproteins that, due to the sulphur bindings, are resistant to proteases. The multi-layered squamous epithelium also serves as a permeability barrier, allowing no acidic material to diffuse into a cell. If the H+ level increases in the cells, ion transporters help increase the pH [5]. The basal layer of the epithelium is the proliferative zone, which is only one-sixth or even less of the total thickness [6].
The second sublayer of the mucosa is the lamina propria mucosae (LPM) placed under the epithelium as a separate loose connective tissue layer. Blood and lymphatic vessels can be found among the connective tissue fibers and cells. The third sublayer of the mucosa is the lamina muscularis mucosae (LMM) a thin smooth muscle layer with elastic components. The submucosa is a connective tissue consisting of collagen and elastic fibers. It has supporting roles with nerves, blood, and lymphatic vessels. The esophageal glands secrete here mucus, which, through its mucin content, can be detected in blue via PAS (Periodic Acid-Schiff) staining. The mucus also supports the passage of food in the lumen. Sometimes lymphatic follicles can be found nearby the glands. The LMP and the submucosal layer consist of mucosa-associated lymphoid tissue (MALT), which includes lymphocytes, plasma cells, and macrophages. The layer of muscularis propria consists of two muscular sublayers, which are perpendicular to each other. These two muscle sublayers enable peristaltic movements. The inner one is a thicker circular muscle layer. The outer sublayer is longitudinal. Between them, there is Auer Bach’s nervous plexus. The muscularis propria consists of different types of muscular tissues based on the topography of the esophagus’ length. In the proximal or upper region, the muscularis propria consists of striated muscle. There is a mixed muscle type in the middle region involving striated and smooth muscle, and in the distal part of the esophagus, only smooth muscle is present.
The ratio of the muscle units and types differs from person to person. The outermost layer is the tunica adventitia, a loose connective tissue. It binds the esophagus to the adjacent structures [3,7-9]. There are some data available on the esophageal layers of different species. These layers of the esophagus are adapted to the nutrition and lifestyle of each species. If we compare the different layers in the different species, we can see how the structure adapted to the circumstances and determine whether the species concerned is a suitable model for the studied esophageal disease. Our study aimed to compare the anatomical structure of the esophagus in some experimental animal species with different body sizes and lifestyles. Collecting the data and comparing them to the human esophagus helps to choose the best model animal.
Seven laboratory animal species were in focus: mouse, rat, guinea pig, rabbit, cat, dog, and pig. One animal from each species was used for sampling the esophagus except for pig and rabbit, where the first samples from the upper part were unfit for our purposes, so we used two animals for the examination. Each of the sampled animals was adult, so the size of the esophagus can be considered average. The animals were terminated for educational purposes or veterinary anatomical examinations; the esophagus samples were shared with us, and no animals were killed just for sampling. (The number of permission for education: PE/EA/2595-6/2016 approved by the National Food Chain Safety Office.) The esophagus was divided into three parts, and tissue samples were collected from the following sections: the upper, the middle, and the lower regions. Our examination focuses on the anatomical differences not only between the seven species but also in terms of the different parts of the esophagus in the same animal. The species with similar diets were compared. There were herbivorous (a guinea pig and two rabbits), carnivorous (a dog, and a cat), and omnivorous groups (two pigs, a rat, and a mouse). The histological samples were collected and stained with Hematoxylin-Eosin for a general overview. The size and the structure of different layers of the esophagus were determined. For evaluation and measuring, we used Panoramic Viewer (3DHistech) program. The wall thickness of the esophagus was determined in each species, in all three section levels.
The total diameter was not measured because of the distortion during the sectioning. One section was measured three times in different locations, and 2-4 sections were evaluated, so we determined the average value from 6 to 12 data entries. We measured the thickness of the mucosal sublayers (epithelium, lamina propria, and lamina muscularis mucosae), the submucosa. and the muscular layers. The total wall thickness was also determined. All data were published as a percentage (relative to the total wall thickness) to make it comparable between different species. The thickness and the extension of the cellular layers of the epithelium were measured. The extension of keratinization was determined in percentage relative to the total epithelial thickness in rodents. Where there was no keratinized layer, the thickness of STRP was determined, which is the top layer of the non-keratinized epithelium with flat, living cells. The thickness of the muscular sublayers and the type of the different muscle tissues were further determined. The presence or absence of striated muscle fibers and the smooth muscle cell layers were compared. The presence or the absence and the density of the glands in the submucosal layer were established. Lymphatic follicles as components of the MALT in the submucosal layer were also recorded. The presence and the locations of white adipose (fatty) tissue were also detected. We used trichrome stain to make better contrast between different layers. This staining method allowed us to better determine the thickness of the connective tissue and the presence of the glands.
The Thickness of the Esophageal Wall Layers
(Figure 1) shows the layers of the esophageal wall in different colours. The diameter of the lumen is not in the pictures, as because it could not be accurately determined due to the distortion caused by the sectioning. The difference is remarkable. The species were classified according to the diet of the animals in (Figures A-C), but there was no clearly visible correlation between the thickness of the layers and the diet. The tunica muscularis (TM) was a very thick layer in each species, thinner in carnivores. The ratio of the muscle layer to the total wall thickness in the different animals is in (Table 1). Rodents had the thickest muscle layer and carnivores had the thinnest. It is conspicuous that the tela submucosa (TSM) was very thick in the cat. Especially in the upper part of the cats’ esophagus, the TSM was thicker than the muscle layer. In the dog’s esophagus, the TSM without glands was the thinnest layer. TSM was also a thick layer in pigs, especially in the upper two-thirds of the esophagus. LMM was a very thin layer, the extension was higher in the lower esophagus of pigs (11.5%) and the middle and lower part of the cat’s esophagus (8.2%, 7.2%). In dogs, the LPM was the thickest layer in the upper part. In both carnivores, the lamina epithelialis mucosae (LEM) was very thin. In the dog, it did not exceed 4%. The thickest part of the LEM was 8.4% in the cat. The LEM was very thick in rodents: it was around 25% (Table 2).
Table 1: The percentage of the tunica muscularis of the total wall in different levels of the esophageal wall in seven species.
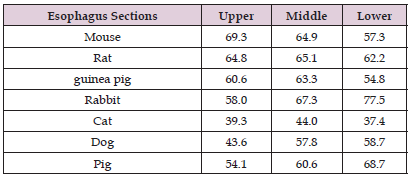
Table 2: The percentage of Lamina epithelialis mucosae in different levels of the esophageal wall in seven species.
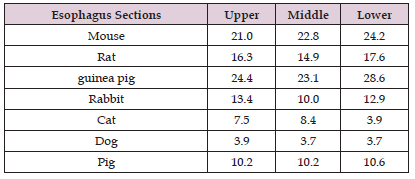
Lamina Epithelialis Mucosae (LEM)
The different cellular layers were well detectable. Stratum basale stained in dark with bigger cells. The flattened layers above were clearly traceable. The thickness was different, a very wide layer with many cell rows could be seen in the esophagus of rabbits and guinea pigs (Figure 2), and just a few layers in dogs, cats, rats, and mice. In guinea pigs, lower-section papillae cores could be detected in the epithelial layer.
Keratinization – Presence or Absence
There were only 3 species (rodents) that display keratinization. The extension of this area was different in the three sections of each species. In the mouse and the rat, the most extended keratinization was detectable in the lower part of the esophagus. It makes up 40.6% of the epithelial layer in the mouse and 50.3% of the epithelial layer in the rat. There was no difference between the thickness of the upper and middle layers. In all three sections of a guinea pig’s esophagus, the keratinization was around 25% of the epithelial layer, the extension is similar in each section. The border of the keratinized layer is clearly visible in the histological picture (rat lower section) (Figure 3). In the other four species, the extension of STRP was determined, and their epithelial layer showed no signs of keratinization. This layer was the thinnest in the pig in each section. It was around 20%. The ratios were similar in cats and dogs, but the thickest STRP was in the lower section of the cat, more than 44%, and in the upper section of the dog: 37.4%. STRP showed large differences in the rabbit. The upper section is one-third of the epithelial layer and heading downwards, it was getting thinner. In the lower section, it was only 9.3% of the LEM. STRP can be seen in the rabbit’s esophagus histological picture. The difference between the keratinization and the STRP layer is clearly comparable in histological pictures (Figure 3).
In the other four species stratum planocellulare is a typical layer above the epithelial cells. In the histological section (rabbit uppert) this layer can be well observed and marked with a blue line. The number and the width of the diagram show the percentage of keratinization or stratum planocellulare thickness of the total epithelial layer.
Submucosal Glands (SMGs)
Submucosal glands were found in only two of the studied species (pig and dog). There were no glands in herbivorous animals in any section of the esophagus. In the omnivorous group, the small rodents had no SMGs in any section. The pig had glands, but there were differences in the three sections. The upper section was full of SMGs. Some glands disperse throughout the submucosal layer in the middle section (Figure 4). There were no glands in the lower section of the pig esophagus. In some cases, lymphatic follicles could be discovered close to the glands. The extension was different. In other species, we could not observe this special position of the lymphatic follicles. Comparing carnivorous animals, a noticeable difference between the SMGs was visible. The submucosal layer of the dogs was rich in glands. Every section of the esophagus consisted of glands that fill most of the submucosal layer. It created a wide layer that sometimes narrowed the LPM (Figure 4). There were no SMGs in any section of the cat’s esophagus.
Tunica Muscularis (Muscularis Propria)
In each species, there is a circular and longitudinal muscle sublayer of tunica muscularis (TM). There were some differences between the species in terms of the type and distribution of the muscle tissue. In rodents, the dominant type was striated muscle. In rats and mice, both layers in each section included striated muscle. In mice, the circular (CM) and longitudinal (LM) muscle sublayers could not be separated. In guinea pigs, the circular sublayer was smooth muscle, and the longitudinal sublayer was striated muscle in the lower section. Above that, all muscle layers were formed by striated muscle. The upper part of the rabbit’s esophagus consisted of only striated muscle. The direction of the muscle fibers or cell layers was the same, making it difficult to separate the two-muscle sublayer. The middle and lower parts were mixed, and the smooth muscle layer is surrounded by striated muscle layers. In the pig, the smooth muscle layer was only present in the lower part. It formed the whole TM. The upper and middle parts consisted of striated muscles. There were differences between the two carnivore species. The dog had only striated muscle in each section of the TM. The muscle in the cat’s esophagus was more complex. The upper part was formed by striated muscle. The direction of the muscle fibers was the same, but we could separate TM and CM. The middle part was mixed, where the inner circular layer was smooth muscle, and the outer thinner longitudinal layer was formed by striated muscle. In the lowest part, only a smooth muscle layer was visible.
Fatty Tissue in the Esophagus
In some species, some fat tissue is present in the submucosa. In the dog and the rabbit, it filled the whole submucosa layer in the upper part of the esophagus in some sections, but in the guinea pig, fatty tissue formed bigger islands in some sections from the lower part of the esophagus (Figure 5).
Trichrome Staining
Trichrome staining is suitable for establishing visual contrast in histological specimens. The connective tissue fibers staining with blue colour, and the cellular components with red colour. By this method, we could easily identify the different layers, determined the extension of the connective tissue, observed the border of circular and longitudinal muscle sublayers, and we could better isolate the vessels in the connective tissue. In (Figure 6). The middle part of the esophagus of each species is presented.
Experimental animals are still used for developing prevention and treatment methods for different diseases that bring us closer to the solution. In planning an experiment, the balance between the ‘harm’ caused and the ‘benefit’ gained must be considered. For reducing the number of used animals and getting more reliable data an appropriate animal model is required. In our opinion a good animal model is based on the following:
1. Scientific aspects: e.g., anatomical, and physiological similarity
2. Practical aspects, e.g., the size, sensitivity, or even availability of the model species, and
3. Ethical and animal welfare considerations. The esophagus has different diseases, ranging from minor mutations to adenocarcinoma.
Some of them have no effects, and no signs, while others can reduce the quality of patient life of patients, and some of them can cause serious morbidity or even death. The wall of the esophagus can be damaged by the chemical effect originating from the stomach. Normally, sphincter control (which involves the diaphragmatic contraction), greater intra-abdominal than intragastric pressure, the peristalsis towards the stomach, and the anatomical structure involving the lower esophageal sphincter, help in avoiding serious damage [8]. Reflux is a normal physiological process that does not cause any damage. There are three preventive mechanisms that can reduce the effect of reflux: the anti-reflux barrier, the esophageal clearance mechanism (which reduces the duration of gastric acid staying in the esophagus), and the resistance mechanism that protects the mucous surface from damaging effects.
If the prevention mechanism damages the conditions are suitable for developing different diseases. Reflux esophagitis develops as acid, bile, pancreatic juice, or their mixture contact with the mucosa. Reflux can cause heartburn, and if it occurs frequently, it results in gastroesophageal reflux disease (GERD). The dysfunction of the LES and the reduction of clearance results in a more serious condition called Barrett’s esophagus (BE), where the squamous epithelium goes over a metaplasia change to the columnar epithelium, and the erosion can cause esophageal ulcer as well. Sometimes Barrett’s esophagus can progress to cancer, which is supported by smoking and drinking [10]. Attwood et al. mentioned three aspects that should be applied in GERD research: the genetic relevance, a conserved gastroesophageal junction appropriate to man, and a naturally occurring pathophysiological GERD [11].
The models could be:
1. Natural – if the disease can develop without any induction. Baboons are very similar to humans, they have natural reflux and Barret’s esophagus, but there are no reports of adenocarcinoma development [12]. In addition, using primates in experiments raises various ethical concerns.
2. Induced – because the disease does not occur naturally in some species, such as adenocarcinoma in rats or mice, or we would like to speed up the process by surgical or chemical methods [13-19].
3. Genetically modified animals – e.g., transgenic mouse models can determine the specific effect of a gene or a process [20].
4. Sometimes a negative model helps to determine the reasons that prevent the development of the specific disease.
The structure of different layers in the esophagus has a role to adapt to different lifestyles that are sometimes species-specific. The epithelial layer has a protective role in the esophagus. In rats and mice, the epithelium comprises 4-5 cell layers while in humans, it is about 5 times wider [21]. In humans, it is not keratinized, but in animals, especially ones that have a rough diet, keratinization can be identified in the epithelial layer. The keratinization results in a tough, non-living layer. The final step of epithelial cell maturation is when cross-linked cytokeratin accumulates in the cells. Keratin is wrapped in the residual plasma membrane. It adapts the surface to the abrasion effect. The keratinization of the epithelium depends on the diet of the animals. This can be detected in rodents, pigs, and ruminants. The keratinized layer protects the mucous surface of the esophagus in case of the consumption of roughage and high-fiber food. The epithelial layer is not keratinized in carnivores [1,22]. There is a study focusing on the keratinization of the esophageal epithelium in different species, classifying herbivores, omnivores, and carnivores. All layers of epithelium were examined to determine the distribution of keratins. The authors found keratins in all layers except in the str. spinosum in rats and cats. Disulphide bonds can stabilize the proteins, making the cells more resistant. The strongest reaction was observed in herbivores, and the highest sulphur contents were also measured in herbivores. High sulphur content could be detected in rats.
These results are in the context of the diet, herbivores and rats eat very hard materials that could cause damage without a stronger, more resistant protective histological solution [23]. We found that the main difference between the examined species is the thickness and keratinization of the epithelial layer. The rodents had a quite wide keratinized layer, and rats and mice have just a few squamous epidermal cell rows. In the guinea pig, besides the keratinization, the epidermal cells have as many rows as in humans. The other examined species have STRP on the top of the epidermal layer, just like in the human esophagus. The number and the location of the submucosal glands (SMGs) are not related to the type of diet. The SMGs develop during the postnatal period and are present in clusters in humans. They are also present in other species such as dogs, opossums, raccoons, and guinea pigs, but not in cats, rabbits, rats, and horses. SMGs are located below the muscularis mucosae sublayer, and acinar secretion is collected into a single duct. Their role is to secrete water, electrolytes, mucins, epidermal growth factor, and prostaglandins. The secretion is controlled by the vagus nerve [24]. Submucosal glands can be found in pigs and dogs in our sections. We could only detect lymphatic follicles in pigs but not in the lower part of the esophagus. The motility (peristalsis) of the esophagus through the muscle layer can be controlled at will. Central and peripheral mechanisms play a role in regulation. The muscle layer (muscularis propria) can be different among the species.
The muscular layer consists of striatal muscle in ruminants, dogs, mice, and rats, and is mixed in many species [1]. As experimental animals, dogs and rodents are used to study striated muscles; cats provide information about smooth muscles. The motility is under the control of the inhibitory and excitatory innervation. The authors found that there is a significant difference in the physiology of different species. For example, cholinergic influences are more prominent in human smooth muscle [25]. In humans, muscle type depends on the region. We observed, that in pigs, cats, rabbits, and guinea pigs, both types of muscle are present. Rabbits’ and pigs’ muscle structures are close to that of humans, but the distribution of the two types is a little bit different. We should remember the three aspects: practical, scientific, and ethical aspects. Rodents need a small living space; their maintenance has a cost advantage, and they can reproduce quickly, so it is easy to generate statistically valuable groups. The problem is their anatomical difference, so in most cases, esophageal diseases do not occur naturally, but we could induce them in different ways. In rats and mice, BE can be induced by surgical or chemical procedures [26]. The dog could be a good model, but ethical and some practical questions arise. Dogs were the first reflux model, but the problem is the long period of progression [22]. The most excluded species from our range is the cat, neither scientific, practical, nor ethical aspects are acceptable of this species.
From an anatomical perspective, primates e.g., baboons would be the best choice; esophageal diseases can occur naturally. They could be a model of BE because mucous gland metaplasia develops spontaneously [27], but the ethical aspect outweighs all other considerations. Earlier, opossums were quite frequently used as the animal model in esophageal research due to their anatomical similarity, and they were a reliable model in studying the role of submucosal glands and bicarbonate secretion [28,29]. The main problem is the limited availability of animals of this species. The most ideal model that fulfils almost all aspects would be pigs, due to their anatomical and dietary similarities and the related ethical and practical aspects (Figure 7).
Esophagus has several diseases that ethiology and the way to prevent needs to be researched from minor abnormalities to adenocarcinoma. In literature, different species are used as an esophageal model. In our study, the aim was to collect anatomical information on the esophagus of different laboratory animal species to find the most reliable and relevant model for the study of esophageal diseases. For this aim, 7 species were used. The unique feature of our study is that we investigated 3 sections of the esophagus in each species. Summarizing our results with some earlier results we established that pigs could be the most compatible model for esophageal diseases not only of their omnivorus aspect but also esophageal anatomical structure. No keratinization, submucosal glands in the upper and middle sections, striated muscle in the upper and middle, and smooth muscle mainly in the lower section are the main characteristics of the pig’s esophagus that makes it similar to the human’s esophagus. These are important data because significant anatomical variation could result in false results from which bad consequences can be drawn.
There is no conflict of interest.


