Impact Factor : 0.548
- NLM ID: 101723284
- OCoLC: 999826537
- LCCN: 2017202541
Odangowei Inetiminebi Ogidi1* and Marcella Tari Joshua2
Received: September 23, 2023; Published: September 28, 2023
*Corresponding author: Odangowei Inetiminebi Ogidi, Department of Biochemistry, Faculty of Basic Medical Sciences, Bayelsa Medical University Yenagoa, Nigeria
DOI: 10.26717/BJSTR.2023.53.008345
In Nigeria, there is a prevalent marketing practice wherein polyherbal concoctions are promoted as panaceas for various ailments. Due to their numerous health benefits, these drugs are extensively employed and prevalent in numerous households across Nigeria. Ethnomedical herbal medicine has demonstrated efficacy in the prevention, diagnosis, control, and treatment of numerous disorders. Hence, the primary objective of this study was to examine the phytochemical compositions of specific polyherbal formulations (Ruzu, Goko, Yoyo, and Jalin) in order to substantiate their extensive pharmacological assertions. The herbal items underwent a qualitative and quantitative phytochemical analysis, which was conducted using the standard method established by the Association of Analytical Chemist (AOAC). The formulations underwent qualitative phytochemical screening, which revealed the presence of several compounds including alkaloids, phenols, saponins, tannins, flavonoids, terpenoids, cardiac glycosides, and steroids. The quantitative analysis revealed that the concentrations of saponin, phenol, alkaloid, and saponin were highest in Ruzu, Yoyo, Jalin, and Goko bitters, respectively, with values of 0.86±0.01, 13.2±0.02, 5.7±0.02, and 18.86±0.02. Conversely, the concentrations of terpenoid, flavonoid, steroid, and terpenoid were lowest in Ruzu, Yoyo, Jalin, and Goko bitters, respectively, with values of 0.03±0.01, 0.15±0.02, 0.36±0.02, and 0.02±0.02. The results of our study indicate that poly-herbal formulations consist of secondary metabolites that possess significant therapeutic qualities with potential for disease prevention.
Hence, the findings of this study suggest potential advantages of the formulations in relation to human health, thereby warranting their utilization in the design and implementation of relevant pharmacological investigations.
Keywords: Poly-Herbal Formulations; Phytochemicals; Pharmacology; Traditional Medicine
Abbreviations: AAOAC: Association of Analytical Chemist; TTC: Total Tannin Content; TAC: Total Alkaloid Content; TSC: Total Saponin Content; TFC: Total Flavonoid Content; TSC: Total Steroid Content; TPC: Total Phenol Content; TTC: Total Terpenoid Content
Plants are utilized as significant resources for sustenance and pharmaceutical purposes, contributing to the avoidance of diseases and the preservation of human well-being. In the context of Nigeria, a significant number of native plant species are extensively utilized for dietary purposes and as traditional treatments, particularly in the alleviation or control of prevalent ailments. The significance of plants in the field of medicine continues to be highly relevant, particularly in light of the ongoing global trend towards sourcing medications from plant-based origins. Consequently, there has been a notable focus on exploring the therapeutic properties of herbal treatments, with particular attention given to their safety, efficacy, and economic viability [1]. The World Health Organization has underscored the importance of implementing contemporary methodologies and appropriate standards to assure quality control of plant products in several decisions [2]. The therapeutic properties of particular plants have been attributed to the presence of certain bioactive compounds found in different plant sections and their corresponding extracts. The active compounds referred to as phytochemicals have the ability to elicit distinct physiological effects on specific species [3,4]. Plants are known to synthesize a wide array of phytochemical compounds, which are classified as secondary metabolites [5]. The secondary metabolites exhibit distinct mechanisms of action and find direct or indirect use within the pharmaceutical business. Phytochemicals possess the capacity to function as antioxidants, thereby impeding cellular damage often induced by free radicals, including those implicated in the development of heart disease and cancer.
Phytochemicals are recognised for their capacity to disrupt enzyme activity. For example, it has been observed that indoles have the ability to activate enzymes that reduce the potency of oestrogen, perhaps leading to a decrease in the susceptibility to breast cancer [6]. Phytochemicals have been identified as having the capacity to inhibit bacterial growth or induce bacterial destruction. One example of a phytochemical that has been identified as having antibacterial effects is allicin. Phytochemicals play a crucial role in inhibiting the adherence of pathogens to cellular membranes, hence contributing to the prevention of pathogenic infections. One example of a beneficial property exhibited by proanthocyanins is their anti-adhesion capabilities, which contribute to the mitigation of urinary tract infections and the improvement of dental health. The investigation and utilization of the diverse phytochemical constituents, along with their distinct mechanisms of action, have the potential to facilitate the identification of novel substances and active compounds that exhibit efficacy against microorganisms that are seemingly resistant to conventional drugs [7-9]. Simultaneously, the utilization of natural antimicrobials has been found to mitigate certain adverse effects commonly associated with synthetic antimicrobials [10,11]. According to (Ogidi, et al. [12]), phytochemicals have the potential to confer health advantages that extend beyond the nutritional content they possess, thereby aiding in the prevention or postponement of chronic diseases in both people and animals.
In Nigeria, there is a prevalent marketing practice of promoting polyherbal concoctions as panaceas for various ailments. Due to their numerous health benefits, these drugs are extensively employed and prevalent in numerous households across Nigeria. Ethnomedical herbal medicine has demonstrated efficacy in the prevention, diagnosis, control, and treatment of numerous disorders. Hence, the primary objective of this research was to examine the phytochemical constituents of specific poly-herbal formulations from Nigeria that are frequently employed in traditional medicinal practices within the country. The aim was to evaluate the potential therapeutic benefits associated with these formulations.
Collection of Samples
The Nigerian Herbal formulations utilized in this study were procured from Cynflac Pharmacy, located on Imiringi road in Yenagoa, Bayelsa State. The specific formulations included Dr Iguedo Goko Cleanser® (registered with NAFDAC under the number A7-0804L), Yoyo Cleanser Bitters (registered with NAFDAC under the number A7-1051L), Ruzu herbal Bitters (registered with NAFDAC under the number A7-1102L), and Jalin herbal mannex (registered with NAFDAC under the number A7-2077I). These formulations were subsequently transported to the laboratory for the purpose of conducting phytochemical screening and analysis.
Qualitative Screening of Phytochemicals of Poly-Herbal Formulations
The polyherbal formulation samples under investigation were subjected to phytochemical assays to screen for bioactive chemical ingredients. These tests were conducted following the established protocols as previously outlined by (Ogidi, et al. [13]).
Quantitative Analysis of Phytochemical Contents of Poly-Herbal Formulations
The Total Tannin Content (TTC) was estimated using a slightly modified version of the Folin and Ciocalteu method. A volume of 0.5 ml of the samples was combined with 3.8 ml of distilled water, a solution containing 35% sodium carbonate, and 0.25 ml of the Folin Phenol reagent. The measurement of absorbance was conducted at a wavelength of 725 nm. Standard solutions of tannic acid were prepared by diluting concentrations ranging from 0 to 0.5 mg/ml. The quantification of tannins is often reported in milligrammes of tannic acid per milliliter of the sample. The determination of Total Alkaloid Content (TAC) was conducted. A volume of forty milliliters (40 mL) of acetic acid solution containing ten percent (10%) concentration in ethanol was introduced to one milliliter (1 mL) of the polyherbal formulation samples. The mixture was then covered and left undisturbed for a duration of four hours. Subsequently, the filtrate was subjected to concentration on a water bath, resulting in a reduction to one-fourth of its initial volume. The sample was subjected to incremental addition of concentrated ammonium hydroxide till precipitation reached its conclusion. The entire solution was permitted to undergo sedimentation, and the resulting precipitate was subjected to a washing process using a solution of diluted ammonium hydroxide. Subsequently, the precipitate was filtered. The residual substance was subjected to a drying process and subsequently measured in terms of weight. The Total Flavonoid Content (TFC) was estimated using the Aluminum chloride technique.
After a duration of 6 minutes, the polyherbal formulations concentrate was combined with a solution of NaNO2, distilled water, and a solution of AlCl3. The mixture was permitted to remain undisturbed for a duration of 6 minutes, after which a solution of NaOH and distilled H2O was introduced to get the desired final volume. The mixture was vigorously vortexed and allowed to stand for a further 15 minutes. The optical density of the mixture was measured at a wavelength of 510 nm. Rutin was employed as a reference compound for the assessment of total flavonoid content. The quantification of total flavonoids was conducted by employing a standard curve, and the results were reported as rutin equivalent in milligrams per gramme of the sample. The determination of Total Saponin Content (TSC) was conducted. A mixture was prepared by dissolving 2 ml of an 80% methanol and 72% sulphuric acid solution, along with vanillin in ethanol solvents. To this mixture, 1 ml of the polyherbal formulation samples was added and properly mixed. The resulting mixture was then heated on a water bath at 60°C for a duration of 10 minutes. Subsequently, the absorbance at a wavelength of 554 nm was measured against a reagent blank. Diosgenin was employed as the standard material and subjected to assay in order to compare its potency with Diosgenin equivalents. The Total Terpenoid Content (TTC) was estimated by combining 1 ml of polyherbal formulation samples with 50 ml of ethanol and afterwards filtering the mixture. A 2.5 ml sample of a 5% aqueous solution of phosphomolybdic acid was mixed with concentrated H2SO4 and allowed to stand for 30 minutes.
The resulting mixture was diluted with 12.5 ml of ethanol solvent, and the absorbance at a wavelength of 700 nm was measured. The determination of the total steroid content (TSC) in the polyherbal formulations was conducted using the methodology outlined by (Ogidi, et al [12]). A 2 ml aliquot was extracted from a solution containing 2.5 g of powdered plant material that had been produced in 50 ml of distilled water and subjected to rapid agitation for a duration of 1 hour. The extract solution underwent a washing process using 3 ml of 0.1M NaOH with a pH of 9. Subsequently, it was combined with 2 ml of chloroform and 3 ml of acetic anhydride at a low temperature. Finally, two drops of concentrated H2SO4 were added with caution. The spectrophotometer was used to measure the absorbance of both the sample and the blank at a wavelength of 420nm.The Total Phenol Content (TPC) was estimated by combining 0.1 ml of the extract with 0.5 ml of Folin-Ciocalteu reagent in a test tube, followed by incubation at room temperature for a duration of 3 minutes. A volume of 2 mL of a sodium carbonate solution with a concentration of 20% was introduced into a tube, which was subsequently placed in a water bath at boiling temperature for a duration of 1 minute. The resultant blue hue exhibited a wavelength of 650 nm. The utilization of gallic acid as a reference compound was employed in the construction of a calibration curve.
Statistical Analysis
The data were reported as the mean ± standard deviation, and each analysis was conducted in triplicate.
Phytochemical Compounds of Selected Nigerian Poly-Herbal Formulations
The formulations underwent qualitative phytochemical screening, revealing the presence of alkaloids, phenols, saponins, tannins, flavonoids, terpenoids, cardiac glycosides, and steroids. The quantitative findings revealed that Ruzu, Yoyo, Jalin, and Goko bitters exhibited varying levels of saponin (0.86±0.01), phenol (13.2±0.02), alkaloid (5.7±0.02), and saponin (18.86±0.02) respectively. Conversely, Ruzu, Yoyo, Jalin, and Goko bitters demonstrated the lowest levels of terpenoid (0.03±0.01), flavonoid (0.15±0.02), steroid (0.36±0.02), and terpenoid (0.02±0.02) respectively, as presented in (Tables 1 & 2).
Note: (- Absent, + present, ++ Abundance, +++ More abundance).
Table 2: Quantitative Phytochemical Compositions of some selected Nigerian Poly-herbal Formulations.
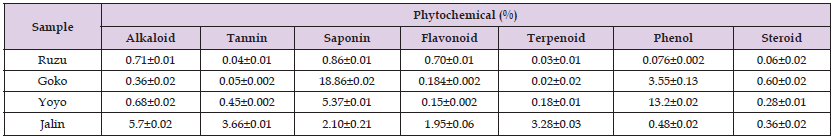
Note: Values are Mean ± SD of triplicate determination.
The present investigation has identified the existence of qualitative phytochemicals that are recognised as active pharmaco-chemical ingredients, including alkaloids, tannins, saponins, flavonoids, terpenoids, glycosides, steroids, and phenols refer to (Table 1). In terms of quantitative analysis, it was observed that Jalin exhibited the highest amounts of alkaloids (5.7 ± 0.02 %) among the various polyherbal formulations that were examined. According to (Ogidi, et al. [14]), it has been documented that alkaloids consistently elicit a bitter sensation. Bioactive chemicals found in plants are considered to be highly efficient in terms of their medicinal significance. The fundamental medical qualities of pure isolated alkaloids and their synthetic derivatives, such as analgesic, antispasmodic, and antibacterial effects, make them valuable as basic therapeutic agents [15]. The authors of a recent study [16] have identified the potential benefits of these interventions in mitigating stress and alleviating symptoms of depression. The Jalin variety exhibited the greatest tannin level, measuring at 3.66 ± 0.01%. The bioactive compound described in the study by (Ogidi, et al. [16]) has been found to accelerate the healing process of both wounds and inflamed mucous membranes. According to (Ogidi, et al. [13]), tannins have been recognised for their potential in cancer prevention and the management of inflamed or ulcerated tissues. The findings of this study are consistent with previous research conducted by other scholars in the same discipline [17,18]. The study conducted by (Ogoun, et al. [19]) revealed that Goko bitters exhibited the highest saponin content (18.86 ± 0.02 %) among the tested formulations. This finding implies that the active ingredients present in Goko bitters, namely Vernonia amygdalina, Allium sativum, Saccharum officinalis, and Cajanus cajan, hold potential as fertility agents and may also be beneficial in the treatment of hypercholesterolemia. This observation implies that saponins may exert their antidiabetic effects via disrupting the absorption of cholesterol in the intestines [20,21]. Flavonoids function as agents that scavenge free radicals, as demonstrated by studies conducted by (Hertog, et al. [22,23]). The formulations exhibited a maximum flavonoid content of 1.95 ± 0.06% in the case of Jalin. There is a body of epidemiological and clinical research that suggests a potential association between flavonoids and a reduced risk of various diseases, including coronary heart disease, cardiovascular disease, Alzheimer's disease, neurodegenerative diseases, diabetes, osteoporosis, and lung cancer [24].
This association is believed to be mediated by the anti-oxidative properties of flavonoids and their ability to modulate the functions of multiple proteins. The Jalin sample exhibited the highest terpenoid concentration, with a mean value of 3.28 ± 0.03. Terpenoids have a diverse array of biological properties, encompassing anticancer, antibacterial, anti-inflammatory, antioxidant, and antiallergic effects [25]. The concentration of phenol in Yoyo bitters was found to be the highest at 13.2± 0.02, as reported by (Radford, et al. [26]). Yoyo bitters belong to a significant category of naturally derived medications that are known for their therapeutic effects in managing congestive heart failure. The findings are consistent with the research conducted by (Ogoun, et al. [27]). The Goko bitters exhibited the highest concentration of steroid levels, measuring 0.60±0.02. Steroids are cyclic chemical molecules composed of carbon atom rings that serve crucial activities in several physiological processes, encompassing growth, development, energy metabolism, homeostasis, and reproduction [25].
Polyherbal formulations refer to extracts derived from many medicinal herbs. Indigenous tribes have historically integrated the utilisation of herbal items for the purpose of treating or mitigating the effects of prevalent illnesses. These formulations are commonly utilised in Nigerian households owing to their reputed health advantages. Nevertheless, a significant portion of these formulations has not undergone rigorous scientific examination. The findings indicate that the poly-herbal formulations consist of secondary metabolites (such as alkaloids, phenols, saponins, tannins, flavonoids, terpenoids, cardiac glycosides, and steroids) that possess therapeutic characteristics for the prevention and treatment of diseases. Hence, it can be inferred that the pharmacological activities attributed to the polyherbal formulation in this investigation are supported by the presence of secondary metabolites.


