Impact Factor : 0.548
- NLM ID: 101723284
- OCoLC: 999826537
- LCCN: 2017202541
Jimbili Patricia*, Yisa Benjamin Nma, Flora O Ugoanyanwu, , Dennis Amaechi and Ini P Ekpe
Received: July 06, 2023; Published: July 17, 2023
*Corresponding author: Yisa Benjamin Nma, Biochemistry Department, Veritas University Abuja, Nigeria
DOI: 10.26717/BJSTR.2023.51.008126
Faro 61 rice starch is a Nigerian rice variety that has been underutilized due to low edible qualities. Consequently, the chemical modification process of carboxy methylation was contemplated for the improvement of the physicochemical and morphological properties, as native starch has been shown to be integrally unsuitable for industrial applications. In this study the physicochemical and morphological properties of native and modified FARO 61 rice starch were analyzed using standard methods. Scan electron microscope (SEM), Fourier transform infrared spectroscopy (FTIR), and thermogravimetric analyzer were utilized to decide the morphological properties of native and modified starch. The results showed significant increase (p<0.05) values in swelling capacity, water solubility, ethanol solubility, acidity, browning and charring temperature compared to the native starch. Significant decrease in moisture content, pH, gelatinization temperature, and amylose content in modified starch were observed compared to native starch. Further significant changes were also observed in morphology of the modified starch compared to the native. Thus, it can be concluded that carboxy methylation altered the physicochemical and morphological properties of native underutilized Nigerian FARO 61 rice, enhancing its industrial applications.
Keywords: Native FARO Rice Starch; Modification; Carboxymethylation; Photomicrograph; Physicochemical Properties
Rice starch is one of the most important but flexible food ingredients possessing value added attributes for innumerable industrial applications. Corn, potato, wheat, cassava, and rice are the common other sources of starch. The most developed cereal crop around the world is rice and integral to billions of people who lives around the world [1]. Rice is grown broadly for food and industrial uses. Rice serves as staple food for rural and urban ranges in Nigeria and has changed from being subsistent crop to cash crop. Federal Agriculture Research Oryza (FARO) rice may be modern breed obtained at Badegi, Niger State, Nigeria National Cereal Research Institute and cultivated predominately within Bida and its environs of Niger State, Nigeria. Pharmaceutical or food industries utilize starch to either influence or control features like, texture, moisture, consistency, and shelf soundness. Starch binds or disintegrates, expands, clarifies, attracts, or inhibits moisture. It produces either smooth or pulpy surfaces and soft or crisp coatings. Starch serves greatly as multifunctional settling in pharmaceutical industries [2]. Regardless of the source of native starch, it is undesirable for industrial applications due to failure to withstand situations as uncommon temperature, adverse pH, high shear rate, tall capacity to retrograde, mis-fortune thickness, syneresis affinity and control thickening due to cooking reduced pH [3].
To advance appealing valuable features and overcome its limitations, native starches are often modified. Modification is used to improve basic dejected physicochemical properties of native starch for industrial applications [2]. The chemical form of modification is used for the treatment of native starch with specific chemicals. In this way modified starch is sensible for canning food, surgical cleaning powder and other applications [2]. A long time after, significant advances have been achieved in getting non-conventional botanical source starch become valuable in their physicochemical properties [4].
Morphological and physicochemical properties for FARO rice variety have never been studied and on this basis, this research aimed at investigating effect of chemical modification by carboxy methylation of the variety of underutilized Nigerian FARO 61 rice starch.
Sample Collection
FARO 61 rice used for this work was obtained from the National Cereal Research Institute Badegi, Bida, Niger State, Nigeria. Extraction of FARO Rice Starch
The method of Sodhi& Singh(2003) was used for starch extraction. Place 20 grams of the rice flour in 200ml of 0.05Msolution of sodium hydroxide for 3 hours. The steep liquor was drained off and re-diluted with 0.05M sodium hydroxide solution. The process continued until the supernatant became clear and gave a negative Biuret test. The starch residue was centrifuged at 3500 rpm and oven dried at 50˚C overnight. The dried starch was sieved with 100 mm sieve and stored at -40C in plastic pack.
Carboxymethylation of Faro 61
The native starch (70g) was suspended in 2-propanol (100ml) using the standard preparation method and aqueous sodium hydroxide solution was added (3% W/v (10ml). The mixture was stirred at regulated temperature of 300C for 10 minutes, after which sodium mono chloro acetate was added and the stirring was continued until the timer went off. The PH was adjusted to 5.0 after the addition of 50% glacial acetic acid. The carboxymethylated starch was sieved through a 100-mesh sieve.
Physicochemical Tests
pH Determination: Five grams each of native and modified (phosphorylated) starch samples were put into 15ml distilled water and mixed separately. Boiling distilled water was used to make up the slurries to 100ml. Each slurry pH was taken after cooling and recorded. The procedure was triplicated for each sample.
Percentage of Moisture Loss: The strategy portrayed by AOAC [5] was utilized to decide the moisture content of native and phosphorylated FARO rice starches. Five grams of each starch was dried to constant weight at 1050C. The procedure was triplicated for each sample. Weight reduction recorded and percentage moisture calculated as follows:

Ash Content Determination
The method portrayed by AOAC [5] was utilized to decide the ash content of the samples. One and half (1/2) gram of each sample was put into crucible of known weight separately. The crucibles were heated in a furnace for 4hrs at 400˚C, crucibles removed and cooled to room temperature and reweighed. The procedure was triplicated for each sample.

Acidity Level Determination
About ten grams of native and modified starch samples were incorporated into 70%v/v alcohol separately using phenolphthalein solution as indicator. A rotary shaker was used to shake the mixtures for an hour; 50ml filtrate from each sample was pipetted and titrated against 0.1M NaOH solution. The method was triplicated for each sample.
Solubility: The strategy of [6] was used to determine the solubility of each sample. Two grams each of the starch was dissolved in 10 ml cold distilled water separately and drained overnight. About 5ml of the clear supernatant was taken for each and heated to dryness on water bath. The method was triplicated for each sample and the formula below was used to determine the solubility.

Swelling Capacity Determination
Daramola & Osanyinlusi [7], (procedure was used to determine swelling capacity. About 0.1 g of native and modified sample was weighed into separate test tubes, 10 ml distilled water added to each and the mixtures warmed at50°C for 30 min with ceaseless shaking on water bath. On conclusion, each mixture was centrifuged at 1500 rpm for 20 min, supernatant decanted and starch paste weighed. The method was triplicated for each sample and the formula below was used to determine the swelling capacity.
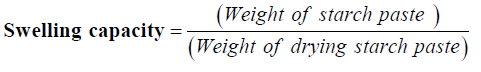
Gelatinization Temperature Determination
The method of Nnamani & Attama [8] was used. One gram of both native and modified starch sample was put into 20ml beaker separately and 10ml distilled water added to each. Each slurry has a thermometer inserted into it and heated with hot plate to obtain their gelatinization temperature. The method was triplicated for each sample.
Browning and Charring Temperature Determination
Builders, et al. [9] method was utilized. A few quantities of native and modified FARO 40 rice starch sample were separately put into a capillary tube, using a melting point apparatus title Electrothermal 9100; browning and charring temperature for each sample was determined and recorded. The method was triplicated for each sample.
Viscosity Determination
Viscosity was determined utilizing the procedure [10]. The viscosity of each starch mucilages concentration was done with rotational viscometer utilizing shaft 4 at 20 revolutions per minute at room temperature. Starch formulated through the suspension of10 grams of each starch separately in equal volume of distilled cold water, 250ml distilled boiled water was added to each slurry, mixed properly and mixtures heated to 700C on ther mostated water bath until translucent mucilage formed. The procedure was triplicated for each sample and their viscosities recorded.
Amylose/Amylopectin content Determination
Colorimetric method of Williams was used. One hundred mg of each sample was transferred into a 100 mL volumetric flask separately. 1 ml of 95% ethanol and 9 ml of 0.1N NaOH were added to each mixture separately and the samples were heated for 10 minutes to gelatinize on boiling water bath, cool and make up to volume with distilled water. Pipette 5ml of native and modified starch sample into different 100-ml volumetric flask, add 1 ml of 1N acetic acid and 2 ml of iodine solution to each differently and make each up to 100 mL with distilled water. Shake for 20 min on a rotary shaker and take absorbance at 620 nm utilizing UV/VIS spectrometer. Replicate the procedure for both samples.
Morphological Properties
Scan electron Micrographs for both native and modified sample were obtained with a scanning microscope (JOEL 6060LV version). The starch samples images were captured within the amplification ranges of 300 and above and 15KV working voltage.
Fourier Transform Infrared (FTIR) Spectra
The Instrument was turned on and allowed to warm-up for 10- 15 minutes and likewise the computer system for initialization. Double click on ‘Micro Lab PC window’ icon and wait for it to open. Click on the start button to initiate the sampling operation and select the method i.e., Absorbance or Transmittance to be used. Clean the Crystal with organic Solvent and click next to Check the Crystal and Collecting Background. The sample of about 10-15mg was then placed. For solids sample close and press to make a pellet on top of the crystal. For sample Alignment check for blue line from Red to Green region for proper sampling and put the sample identity for coding. Click next for sampling. Right click for picking the peaks and select peaks for labeling by dragging to acquire the wavenumbers as well as Transmittance or Absorbance.
Thermogravimetric Analysis
Ten(10) mg of each sample was put into a TGA dish separately. Along these lines, the samples experience warming at 25°C to 850°C with warming rate of 10 K•min−1 and nitrogen gas streams at 20 mL•min−1. A record for each sample was taken and their charts drawn.
Method of Data Analysis
The obtained data were analyzed as mean ± standard deviation utilizing SPSS factual computer program, form 23.0. The pointer level of significance difference was set at p<0.05.
(Table 1 & Figures 1-6)
Note: Values are Mean ± standard error of mean of triplicate determinations. P-values < 0.05 indicates significant difference between the mean values.
The result reported on Table 1 showed decreased in pH of modified FARO 61 rice starch after carboxymethylation. The reduction in pH for carboxy methylated FARO rice starch could be due to the carboxymethylation, dilution with HCl solution and the washing steps applied. The pH for both native and modified FARO rice starches obtained from this study were within the recommended range sutilized by most pharmaceutical and food industries as reported by Omojolaet al and Nuwmanya. It is therefore important that the pH of starch tends towards neutrality to maximise its use in industries where products pH changes are undesirable [11,12].
Table 1 showed modified FARO 61 rice starch having decreased moisture content compared to native starch. The reduction observed for modified starch could be due to alteration and changes within the molecular structure of the starch granules caused by sodium mono chloro acetate. The result showed no significant difference at (p<0.05) for modified FARO 61as compared to native starch. This reduction trend observed in this work is comparable to reduction trend observed by Dolas, et al. [13], on impact of modification on physicochemical uses and basic features of cassava starch (Manihotesculenta Crantz) which shows reduction in moisture content of modified starchbecause off expanded clarity of modified starches. Therefore, reducing the moisture content of modified starch prolonged the shelf stability during storage and helps to keep the quality of the starch by preventing the growth of moulds during storage [14].
Table 1 shows the outcome of swelling capacity of both native and modified FARO 61 rice starch. The result revealed an increase in swelling capacity for modified starch than native starch. The increase observed for modified starch could be due to alteration within the glucose granular structure that makes up the starch which allows water to enter easily. Additionally, it could also be ascribed to tight squeezing of molecules within the granule. Significant difference at (p<0.05) in swelling capacity for modified FARO rice starch was observed as compared to native starch. Carmona Garcia, et al., Chatakanonda, et al., Bea-Young and Yoo, et al., [15-17] had observed that cross-linked starch has higher swelling capacity than native starch at increased temperatures.
The gelatinization temperature of modified FARO 61 rice starch was observed to be lower when compared to native starch as shown in Table 1. This might be due to weakening intermolecular qualities and introduction of carboxy methyl groups into the starch structure. A significant difference was observed between modified and native FARO starch. This result is comparable to work done [18-20] on modification effect on starches of yam and sweet potato. Therefore, the stronger the intermolecular bond between the starch molecules the more heat is needed to break it and the higher the gelatinization temperature. Thus, carboxymethylation was effective in starch modification.
Browning temperature of modified starch for FARO 61was higher than that of the native starch as shown in Table 1. A significant difference was observed between native and modified starch for FARO 61. The result shows that modified starch indeed can be heated to higher temperature without change in colour thereby making the starch better for industrial utilizations at higher temperatures. This agrees with the result of Omojola, et al. [11] on preparation and physicochemical characterization of icacina starch citrate a potential industrial starch.
Table 1, presents amylose content for native FARO 61 rice starch which was lower than that of native starch and significantly different at (p<0.05) from the native starch of FARO 61. The decrease in amylose content for carboxy methylated FARO rice starch may be related to an expanded formation of dextrins and presentation of carboxymethylated groups within the course of corrosive treatment. This agrees with previous work done by [20], for phosphoric acid impact on physico chemical properties of yam (Dioscorea rotundata) starch, which reveals reduction in its blue value and amylose content. Amylose content is significant in almost all starches because higher amylose starches have increased retrogradation tendencies.
FARO 61 rice starch for native and modified scan electron microscopy results are shown on Plate 1 and 1a, respectively. Starch images for native FARO rice starch show clustered, smooth, and specific granular shapes as compared to modified FARO 61 starch which has rough and cruel surface. The changes observed on starch granule surfaces for modified starch could be due to acid anhydride and sodium mono chloro acatetate reactions. Adewoyin, et al. [20] in previous research revealed similar surface roughness on maize and potato starch granules for phosphoric destructive alteration. Besides, Sha, et al. [21] explained that change on surface of acetylated rice starch granules was mild compared to native starch, because of crystalline regions that control certain mishapin granular structure amidst the reaction. In this manner, granule shape of starch impacts their valuable characteristics which influences their applications. Starch granule shape contributes to starch gelatinization, swelling and thickening rat.
Native and modified FARO 61starch FTIR results are shown in Figures 1 & 2. Native starch discernable peaksare observed at 3261.4 which came because of hydroxyl (O-H) and C-H bunches vibration, exclusively comparable to Rivas [22] native starch results. OH, contributes to the inter-molecular and intra-molecular interactions which are a characteristic of starch structure. Another peak was observed at 2929.7 as C-H stretch (Alkanes).
The C=O stretching vibration at 1641 cm−1 represented a carbonyl group which may be either from carboxylic acid, aldehyde or ketone groups and carbon-carbon double bond. Other peaks between 1148 and 1077.2 corresponds to C-O amplify and finally another top band seen on FTIR chart at 708.2cm-1 for native starch as C-H bend.
After modification intensities of absorption bands for O-H,C-O, C=C, C=O and C-H for modified starch were all increased when compared to native starch for FARO 61 rice. Absorption bands intensity increase could be due to collapsing of the C-O, C=C and C-H bands resulting in changes within starch structures [23,24].
FARO 61 rice starch for native and modified were thermos gravimetrically inspected for thermal stability and decomposition characteristics. Thermogravimetric analysis investigated starch-based materials utilized for industrial purposes degradation and thermal behaviour. Thermo gravimetric graphs and derivative thermogravimetric graphs are shown in Figures 1 & 2 for native and modified rice starches. The graphs show similar trends, indicating thermal breakdown and mass loss for native and modified starch occurring at different phases representing particular event during heating. Thermal action occurs first between 1000C and 1500C for native and modified starch which results in mass loss due to moisture and water fragments elimination by evaporation. In this phase, higher moisture content leads to higher mass loss.
Continuous heating leads to another loss in weight between 250°C and 460°C. At this weight lost, carboxymethylated starch requires higher temperature compared to native starch within the region of 3900C. This phase reduction in weight is related to depolymerization and degradation of starch structure carbon chain.
Weight loss finally occurs within the range 460°C and 700°C. Weight lost at these temperatures are not prominent compared to the one between 250°C and 460°C due to evaporation of less materials or water from native and modified starches which shows that modification hasn’t altered thermal stability of modified of starches. Isotton, et al. [25], had a similar report on effect of different plasticizers which they declared rate of degradation between 290.9°C and 295.4°C [26].
The moisture content, pH, amylose content, and gelatinization temperature of the native starches were generally higher than those of the modified starch while acidity, browning temperature, swelling capacity, water solubility an ethanol solubility of modified starch were significantly lower than that of the native starch. The physicochemical properties of the native starch were significantly affected by the modification processes. The carboxymethylated starch had good physicochemical properties, indicating that it will be good quality starch with potential industrial uses.


