Impact Factor : 0.548
- NLM ID: 101723284
- OCoLC: 999826537
- LCCN: 2017202541
Toshihiko Nakamura1,2*, Jin Xiuri3, Shingo Yamada3 and Haruo Goto4
Received: June 01, 2023; Published: June 07, 2023
*Corresponding author: Toshihiko Nakamura, Department of Neonatology, Japanese Red Cross Musashino Hospital, 1-26-1 Kyonan-cho, Musashino city, Tokyo 180-8610, Japan
DOI: 10.26717/BJSTR.2023.50.008024
Background: Neonatal bacterial infection is diagnosed when the acute phase reactants score (APR-Sc) is positive for 3 items of CRP, α1-acid glycoprotein (AGP), and haptoglobin (Hp), and 2 points are positive for 2 items, CRP and AGP. However, the mechanism by which the presence or absence of an increase in Hp, which is the difference between the 3 points and the 2 points, is not known.
Objective: This study aimed to examine differences between APR-Sc, which is used to evaluate neonatal infectious status by cytokine profiles, of 3 versus 2 points.
Subjects: Inborn infants whose APR-Sc was measured over time from the early neonatal period were studied. They included 10 infants with APR-Sc of 3 points, 10 Hp-negative infants with APR-Sc of 2 points, and 18 infants with APR-Sc of 0 or 1 point.
Methods: Cytokine profiles (IL-1α, IL-1β, IL-2, Il-4, IL-6, IL-8, IL-10, and TNFα) were measured by ELISA. APR-Sc was measured over time at least 5 times by day 3 of age, using residual serum obtained early after birth.
Results: IL-1β and IL-6 were the only cytokines showing a significant difference between the APR-Sc 3-point group and APR-Sc 2-point group. Furthermore, a weak positive correlation was found between IL-1β and IL-6. A common pattern was confirmed in all infants in which the cytokine profile and the three APR, CRP, α1-acid glycoprotein, and Hp, increased once from birth in the early neonatal period and then decreased sharply thereafter.
Discussion: We confirmed that APR-Sc of 3 points and 2 points are suitable not only for indicating infectious diseases but also patients showing a strong inflammatory reaction. These findings suggested that a difference between the two groups may occur depending on the presence or absence of an increase in Hp according to the degree of increase in IL-6, whose production is promoted by IL-1β.
Keywords: Acute Phase Reactants Score; Haptoglobin; Interleukin-1β; Interleukin-6; Neonates
Abbreviations: Hp: Haptoglobin; AGP: Acid Glycoprotein; CRP: C-Reactive Protein; FIRS: Fetal Inflammatory Reaction Syndrome; APR-Sc: Acute Phase Reactants Score; PPH: Primary Pure Hepatocytes
Since the 1970s, we have proposed an acute phase reactants score (APR-Sc) that uses an increase in blood APR as an index for screening tests for neonatal infections [1-3]. Here, APR refer to three acute phase reaction proteins: C-reactive protein (CRP), α1-acid glycoprotein (AGP), and haptoglobin (Hp). Then, depending on the presence or absence of an increase in the levels of these three proteins, 0 or 1 point is scored for each substance, and a total APR-Sc of 0 to 3 points is calculated. Based on clinical diagnosis including that from autopsy, the APR-Sc is calculated to be 3 points when all three APR are increased and to be 2 points when CRP and AGP are increased. Either of these two-point value groups are diagnosed as a state of bacterial infection. In the group with elevation of AGP and Hp, the APR-Sc is also 2 points, but this group is diagnosed as a state of post-infection. The group with an increase in AGP only receives an APR-Sc of 1 point and is evaluated as a state of infection healing [1,2]. In terms of the usefulness of the APR-Sc, when compared with the pro-inflammatory cytokines that produce and secrete these APR in the liver, the latter is superior in early diagnosis in terms of the reaction rate. However, we have already reported that the APR-Sc is more useful in assessing when to end antibiotic treatment [4]. The pathophysiology of fetal inflammatory reaction syndrome (FIRS), which is important to differentiate from neonatal infections, has been attracting attention for a long time [5,6]. Furthermore, the pathophysiology of fetal/ neonatal asphyxia has been investigated from the aspect of an inflammatory reaction called ischemia-reperfusion injury [7].
In other words, a situation in which an inflammatory reaction with an APR-Sc of 3 points is strong is not necessarily limited to infectious diseases and especially bacterial infectious diseases. Therefore, we examined the changes in the APR-Sc over time, but it was not possible to clearly distinguish infectious diseases from these other conditions alone [4,8,9]. As a result, to comprehensively interpret the clinical picture of an infant, physicians currently must accurately distinguish maternal information, placental findings, and peripheral blood leukocyte count (WBC) of the infant and its fraction. This time, we had the opportunity to examine the difference between an APR-Sc of 3 points, which is considered to indicate infection, and an APR-Sc of 2 points, in infants with no increase in Hp, from the cytokine profile. All studies on the mechanism of promoting APR production by cytokines, mainly IL-1β and IL-6, are in vitro studies using hepatocytes of various species. To the best of our knowledge, there are no in vivo studies of the association between cytokines and APR in newborns.
The subjects were previously reported inborn infants admitted to the Nishisaitama Chuo Hospital Perinatal Center from whom residual blood serum was obtained at the time of admission within 1 hour after birth [4]. The study period spanned one year from January to December 2008. These newborns had been judged by attending physicians to require daily blood analysis for close monitoring during the first week of life, and any serum remaining after these tests was stored. In total, 438 newborns were admitted to the center during the 2008 calendar year. The subjects were divided into 3 groups. Group A (Gr-A) consisted of those whose APR-Sc indicated an infectious state (3 points: CRP, AGP, and Hp positive). Group B (Gr-B) consisted of those whose APR-Sc indicated an infectious state (2 points: CRP and AGP positive). Group C (Gr-C), as a control, consisted of those whose APR-Sc did not indicate an infectious state (neither 3 points indicating an infectious state nor 2 points for being CRP and AGP positive) at any time during the first week of life. We used data from infants in whom the APR-Sc was measured at least 5 times from 0 to 3 days of life because this is the time when the APR-Sc changes the most. The changes in the measured values from those at the time of admission and the concentrations of various cytokines in the early postnatal period were examined. The Q-PlexTM human inflammatory cytokine array (QUANSYS Bioscience Inc., Logan, UT, USA) was used for cytokine profile analysis. Each well of a 96-well plate contained 9 types of inflammatory cytokine (IL-1α, IL-1β, IL-2, IL-4, IL-6, IL-8, IL- 10, INFγ, and TNFα) capture antibodies.
The test is based on the immobilized ELISA method. After the antigen-antibody reaction was performed, various cytokine concentrations were calculated by the brightness of black and white images using a CCD camera at Sagamihara R&D Center, Shino-Test Corporation, Sagamihara, Japan. The measurement ranges in pg/mL were as follows: IL-1α: 9.38 to 600, IL-1β: 10.94 to 700, IL-2: 7.81 to 500, IL-4: 4.69 to 300, IL-6: 8.59 to 550, IL-8: 10.94 to 700, IL-10: 7.81 to 500, INFγ: 5.47 to 350, and TNFα: 9.38 to 600. If the upper limit of the measurement range was exceeded, the serum was diluted 2-fold, 5-fold, 10-fold, and 100-fold and re-measured in consideration of the residual serum volume. A serum volume of 30 μL was used for each measurement [4]. The LatesieTM from Shino-Test Corporation (Sagamihara) was used to measure APR and calculate the APR-Sc. CRP is measured with a latex agglutination reaction turbidimetry method, and AGP and Hp use the immunoturbidimetry method. The sample volume required is 10 μL each, and the total measurement time is less than 3 minutes, so the results can be obtained within a short time [2]. This study was approved by the Ethics Committees of the National Hospital Organization Nishisaitama Chuo National Hospital, and written informed consent was obtained from the parents or guardian of the hospitalized newborns. Tests of significance between each group were performed by one-way analysis of variance using of StatMate VTM (ATMS, Tokyo, Japan) statistical software. The χ2 test was used to compare the diseases. For the correlation between cytokines, the Pearson correlation coefficient was tested. A significant difference was indicated when p <0.05. A correlation coefficient of r >|0.2| indicated a correlation.
Thirty-eight inborn neonates were included. Gr-A included 10 neonates, Gr-B 10 neonates, and Gr-C 18 neonates. Their gestational ages (mean ± SD) were 37.1 ± 3.9 weeks, 39.6 ± 1.3 weeks, and 37.7 ± 2.3 weeks, respectively, and there was no significant difference between the groups. Similarly, the birth weights were 2555 ± 731 g for Gr-A, 2959 ± 389 g for Gr-B, and 2848 ± 473 g for Gr-C, and the difference between them was not significant. The main diagnoses for Gr-A and Gr-B infants were classified into infectious disease, FIRS, and respiratory disorders associated with asphyxia (amniotic fluid aspiration syndrome, meconium aspiration syndrome) and were not significantly different between the two groups (p = 0.161). In the measurement of cytokines, the upper limit of the measurement range was exceed in 1 sample for IL-4, 9 samples for IL-6, 9 samples for IL-8, 1 sample for IL-10, and 1 sample for INFγ, and these samples were appropriately diluted and then re-measured. When the value was below the lower limit of the measurement range, the following measurement limit values in pg/mL were adopted as the measured values for the convenience of statistical processing: IL-1α: 2.5, IL-1β: 4.5, IL-2: 6.5, IL-4: 2.0, IL-6: 3.5, IL-8: 4.5, IL-10: 4.0, INFγ: 3.0, and TNFα: 6.5. The results are shown in (Table 1) together with the WBC values. Only serum IL-1β and IL-6 showed a significant difference between Gr-A and Gr-B. The relative strength of the inflammatory response was compared by determining the ratio of the inflammatory cytokines IL-1β and IL-6 as the activator IL-8 to the anti-inflammatory cytokine IL-10.
Table 1. Cytokine levels of 9 cytokines and white blood cell counts in blood of 38 infants early neonatal periods.
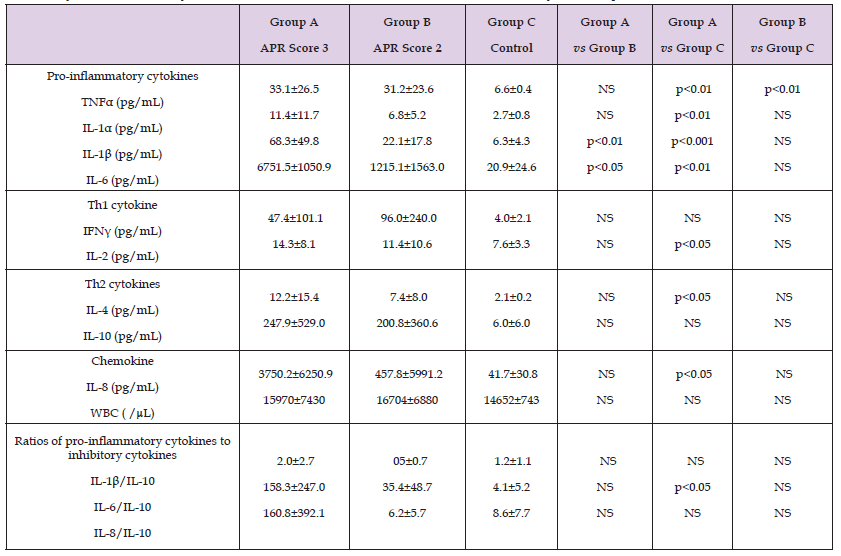
Note: Mean±SD.
Table 2. Comparisons of day 0 and maximum APR values between each group during early neonatal period.
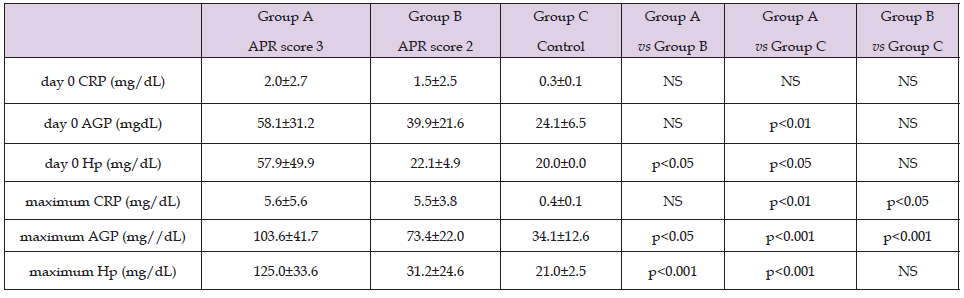
Note: Mean±SD.
However, no significant difference was observed between Gr-A and Gr-B. The day 0 values and the maximum values of each of the three APR in the early neonatal period can be compared over time in (Table 2). A significant difference between Gr-A and Gr-B was revealed not only for Hp but also for AGP. However, there was no significant difference for CRP. Time courses are shown in (Figure 1) and (Figure 2) for each of three representative cases from Gr-A and Gr-B. Changes in the cytokines over time in both groups peaked early after birth and declined sharply thereafter. This was a common pattern both in infectious diseases and in FIRS, meconium aspiration syndrome, and asphyxia. In particular, the high serum concentration of IL-6 was conspicuous. In Gr-A, APR increased with the decrease of cytokines, and CRP decreased slightly ahead of AGP and Hp. CRP and AGP showed similar changes in Gr-A and Gr-B, except that Hp was negative in Gr-B. In determining the correlation coefficients between cytokines, a weak but significant positive correlation was found between IL-1β and IL-6 (r = 0.487, p <0.001). All samples with values below the lower limits of the measurement range were excluded (Figure 3).
It has been about 45 years since Goto, one of our research team members, first proposed the APR-Sc as a diagnostic method for neonatal infectious diseases [1,2]. He selected three APR, CRP, AGP, and Hp, and repeated their measurements using sera from early neonates. These data were classified according to days after birth and birth weight, and reference values were evaluated. The APR-Sc use a method of adding 1 point when the standard value is exceeded and scoring ranges from 0 to 3 points. A vast amount of clinical data was analyzed, and the group with an APR-Sc of 3 and the group with positive CRP and AGP values, that is, a group with a negative value only for Hp, were considered to have infectious diseases and were treated with antibiotics. Of course, it is not necessary to make an immediate decision based on the result of only 1 point, and it is best to consider the pros and cons of treatment based on the subsequent clinical symptoms and changes in the APR-Sc. Currently, testing to determine an APR-Sc is being performed at bedside using a very small amount of serum (total of 30 μL: 10 μL for each APR). Even preterm infants and low birth weight infants in the early postnatal period can be measured by collecting blood in only one capillary tube. In terms of measurement time, if the reagent is prepared, the measured values and APR-Sc can be automatically calculated in less than 3 minutes, so it is possible to respond in real time. These APR are mainly produced in the liver and released into the blood, where they are measured.
Stimulation of hepatocyte production with inflammatory cytokines is essential for the production and release of each substance in the liver. So far, three inflammatory cytokines, IL-1β, IL-6, and TNFα, have been considered important for enhancing the production of these APR [4,10]. As mentioned above, when an infectious disease is regarded as one of the causes of inflammation, the inflammation includes the spillover of the inflammatory reaction to the infant due to the intrauterine infection called FIRS and the inflammatory reaction in the ischemia-reperfusion injury called fetal and neonatal asphyxia. Each type of inflammation can result in an increase in APR [9,11,12]. Differentiation of these inflammation types requires comprehensive interpretation. In the differentiation in the present study, we focused on neonates with an APR-Sc of 3 points and Hp-negative neonates with an APR-Sc of 2 points, both of which are considered to indicate infectious diseases. Then, what is the difference between an APR-Sc of 3 points, which is judged to indicate a high possibility of infectious disease, and that of 2 points with Hp being negative, when focusing on pre-inflammatory cytokines that are said to control APR production? Many studies have been reported on cytokines that regulate the production of APR. In vitro studies have often used hepatoma cells, which are tumor cells [13]. The main reason is that a small amount of contamination is indispensable for culturing pure hepatocytes, and if Kupffer cells are allowed to grow as contamination, the reaction of sufficient hepatocytes is mainly hindered [14-16].
Gradually, as culture technology developed, it became possible to conduct research on primary pure hepatocytes (PPH). As a result, it was also found that the production mode of APR differs between normal hepatocytes and tumor cells. In addition, hepatocyte responses in mice [17-19], rats [20], cattle [21,22], chickens [23], and humans are considered to be species specific and should be interpreted with caution. According to the study using PPH by Kramer et al., IL- 1β stimulates nuclear factor κB and members of the CCAAT box/ enhancer binding protein (C/EBPβ)-dependent autocrine IL-6 loop. Furthermore, the IL-6 loop promotes IL-6 production by itself [24]. As a result, a high concentration of IL-6 can promote the production of CRP, AGP, and Hp. The purpose of this study was not to compare the APR-Sc based on the presence or absence of infection but to examine the difference between an APR-Sc of 3 points judged to indicate an infectious disease state and that of 2 points where there is no increase in Hp. However, there was no significant difference in the infectious diseases and asphyxia, meconium aspiration syndrome, and FIRS seen in Gr-A compared with those seen in Gr-B with no increase in Hp. Our initial expectation was that the difference between the two groups, that is, the presence or absence of an increase in Hp, would be caused by the difference in the strength of the inflammatory response. According to previous reports [16,25], CRP and AGP are promoted in hepatocyte production by IL-1α, IL-1β, TNFα, and TGFβ, which are considered to be IL-1–type cytokines.
Hp is said to be promoted by IL-6, the main cytokine of IL-6–type cytokines [26]. In a relatively recent report, IL-1β and TNFα were said to be involved in the production of APR in hepatocytes via the IL-6 loop, which is considered to be autocrine production of IL-6. Although it has been reported that IL-1–type cytokines have an inhibitory effect on IL-6–type cytokines, our study results showed that IL-1β in Gr-B was significantly lower than that in Gr-A, supporting the former. Our study has also shown that even if the pre-inflammatory cytokine level is high in the early postnatal period, it will continue to decrease sharply thereafter. The magnitude and duration of the inflammatory response, including intra-uterine infection, result in different births. In addition, differences in postnatal vaginal delivery, emergency cesarean section, elective cesarean section, and transvaginal delivery with interventions for conditions such as fetal distress add to the degree of hypoxic stress in the infant before and after birth. This affects the production of cytokines after birth, and finally, the production of APR is promoted. There were significant differences in the concentrations of both Il-1β and IL-6 between the Gr-A and Gr-B groups, and a slightly weak but significant positive correlation was found between IL-1β and IL-6, suggesting that the strength of the inflammatory response varies greatly among individuals and that a considerable IL-6 concentration is required to promote Hp production [27]. A weak synergistic effect of IL-6 and IL-1β was observed in this study as in the in vitro study using human hepatocytes.
Gr-A had significantly higher maximum values of Hp and AGP than Gr-B, which took 24 hours to be produced and secreted into the blood by cytokine stimulation to the liver before reaching their maximum blood concentrations. Contrastingly, CRP reached its highest value in a short time of about 6 hours, but no significant difference was observed between the two groups. Because the onset time of inflammation was different, it is easy to understand that the range of results is wide. In addition, as the length of the period from the origin of the inflammatory reaction to the birth of the neonate is different in each infant, it is possible that the peaks of inflammation do not match even in the early postnatal period. That is, when comparing the cytokine concentrations in the samples obtained in the early postnatal period, it was difficult to determine the strength of the inflammatory response based on the cytokine concentrations of the neonate at the peak of inflammation and the neonate at the stage when concentrations are decreasing. The limitation of this study is that there were few subjects in Gr-A and Gr-B in whom to compare the concentrations of various cytokines within each group classified according to the pathophysiology of infection, asphyxia, and FIRS. In the future, it will be necessary to increase the number of each pathological condition in each group and examine the APR-Sc and cytokine profile for each disease.
As a result of cytokine profile measurements centered on proinflammatory cytokines in neonate groups with APR-Sc of 3 points and APR-Sc of 2 points indicating the presence of infectious states, the serum concentration of IL-1β of the APR-Sc 3-point group was significantly higher than that of the APR-Sc 2-point group. This suggested that the difference in IL-6 production due to the IL-6 loop mediated by IL-1β may cause a difference in Hp production. A weak but significant positive correlation was found between IL-1β and IL-6. From the above, the presence or absence of an increase in Hp in the infectious state as indicated by the APR-Sc was considered to be due to the strength of the inflammatory reaction.
Cytokine measurement was performed by the Department of Development at Shino-Test Corporation. We appreciate the kind advice on English proofreading from Rise Japan LLC.
The authors declare no conflict of interest.
The manuscript was written by TN. HG, JX and SY provided advice on the validity of the content of the paper. TN collected and analyzed data. All authors read and approved the final manuscript.

