Impact Factor : 0.548
- NLM ID: 101723284
- OCoLC: 999826537
- LCCN: 2017202541
Lester Raymundo Dominguez Huarcaya1-4* and María Fernanda Dominguez Ríos5,6
Received: May 22, 2023; Published: June 01, 2023
*Corresponding author: Lester Dominguez, Syneos Health del Peru, Novartis, Facultad de Farmacia y Bioquímica, Universidad Nacional Mayor de San Marcos, Lima, Pontificia Universidad católica del, Peru
DOI: 10.26717/BJSTR.2023.50.008004
Depression has the highest morbidity and disability rates of any mental disorder, and gastrointestinal issues are a common symptom. Recent research has linked the composition of one’s gut microbiome to a person’s susceptibility to developing depressive symptoms. The term “gut-brain axis” refers to a potential link between the CNS and the digestive system. It has lately been fashionable to treat illnesses by targeting bacteria in the digestive tract (via the use of probiotics, for example) and integrating the gut-brain axis mechanism. Together, we found that there is a robust connection between the composition of one’s gut microbiota and the rate at which people experience depression. Moreover, it has been hypothesized that the structural alteration of the gut microbiota system has both beneficial and deleterious regulatory effects on the development of depression. This article looked at the mechanics of the two-way communication between the gut and the brain, as well as the current methods of relieving symptoms and antidepressant drugs related to the gut microbiota. The field of probiotics has expanded quickly in recent decades thanks to an uptick in research into their medicinal potential. Probiotics-mediated microbiota modification along the microbiota-gut-brain axis (MGBA) has been proven to be beneficial in a number of health domains through pre-clinical and clinical investigations. Although probiotic research is just getting started in the realm of mental health, in vivo and in vitro studies have revealed potential effects of probiotics on a number of mental diseases, paving the way for translation of preclinical models to humans.
Keywords: Depression; Probiotics; Gut–Brain Axis; Gut Microbiota
Abbreviations: SSRIs: Selective Serotonin Reuptake Inhibitors; MGBA: Microbiota-Gut-Brain Axis; SSRIs: Serotonin Reuptake Inhibitors; SNRI: Serotonin-Norepinephrine Reuptake Inhibitors; GF: Germ-Free; SPF: Specific Pathogen-Free; CSDS: Chronic Social Defeat Stress; IRAQI: Isobaric Tag For Relative And Absolute Quantitation; MGBA: Microbiome-Gut-Brain Axis; MDD: Major Depressive Disorder; HSCL90: Hopkins Symptom Checklist–90; HADS: Hospital Anxiety And Depression Scale; SPT: Sucrose/Saccharin Preference Test; SPI: Sucrose/Saccharin Preference Index; TST: Tail Suspension Test; FST: Forced-Swim Test; OFT: Open Field Test; CRH: Corticotropin-Releasing Hormone; MIS: Mucosal Immune System; GI: Gastrointestinal; EECs: Enteroendocrine Cells; PYY: Peptide YY; GOS: Galactic-Oligosaccharides; IBS: Irritable Bowel Syndrome
Anxiety, sadness, and cognitive impairment may all benefit from further study of the microbiota-gut-brain axis [1-5]. Several hypotheses have been advanced to explain how the gut bacteria could influence brain activity. The composition of microbes in the gut can be altered, the immune system can be suppressed, the vagus nerve can be stimulated, tryptophan can be metabolized, the gut can respond with hormones, and microbes can produce neuroactive chemicals, cell wall carbohydrates, and other metabolites [5]. Evidence suggests a link between reduced diversity of the microbiome in the gut and neuropsychiatric diseases like maternal and infant sadness and anxiety. Mother-infant attachment-based therapy for prenatal depression may benefit from assessing the microbial ecosystems of both mother and child [6]. Targeting the gut-brain axis by modulation of the gut microbiota is one strategy for enhancing maternal mental health during the perinatal period. The microbiota-gut-brain axis equilibrium can be achieved through the use of probiotics and prebiotics.
Probiotics are “live microorganisms that, when administered in adequate amounts, confer a health benefit on the host” [7] according to a consensus statement of the International Scientific Association for Probiotics and Prebiotics. When consumed by the host microorganisms, prebiotics provide a health advantage [8]. Several disorders, including antibiotic and Clostridium difficile-associated diarrhea, irritable bowel syndrome, and inflammatory bowel disease, as well as anxiety, wound healing, and depression, may be treatable with probiotics, as shown by well-designed clinical trials and metaanalyses [9-14]. Recent clinical trials examining the effect of probiotics on the gut-brain axis have yielded encouraging findings, such as the amelioration of psychiatric and gut-barrier-associated traits [15] and cognitive functions [15] in patients with major depressive disorder, the reduction of anxiety symptoms in patients with anxiety disorders [16-19], and the enhancement of the mental state, sleep quality, and gut microbiota of healthy young adults under stress.
Since depression has been linked to suicidal behaviour, it has received a lot of attention from researchers and the general public. About 280 million individuals of all ages, or 5% of adults worldwide, are estimated to suffer from depression. The enormous global burden of disease severely limits the opportunities for individuals and communities to thrive. Anxiety and depression have become much more common as a result of the COVID-19 pandemic, as evidenced by 5683 separate data sources from the global burden of illness research in 2020 (COVID-19 Mental Disorders Collaborators, 2021). Current treatments rely heavily on the hypothesis that chemical imbalances in the brain are responsible for depression. Due to treatment-resistant depression and the delayed onset of first-line antidepressants like Selective Serotonin Reuptake Inhibitors (SSRIs) and serotonin-norepinephrine reuptake inhibitors (SNRIs) [20,21], the needs of many depression patients are not met despite the use of complementary therapies such as interpersonal psychotherapy and non-pharmacological therapy such as electroconvulsive therapy. One obstacle is that our current understanding of the pathophysiology of depression is incomplete. Depression’s underlying biological mechanisms remain poorly understood. Novel antidepressants may be developed from a new perspective that considers the interplay between the brain, the gut, and the microbiota [19].
Studies into the endocrine system of the gastrointestinal tract and its hormone communication with neurons and brain cells led, by accident, to the discovery of a gut-brain axis in the year 1980. Over the subsequent decades, researchers have expanded on this concept to account for the microbiome’s part in the gut-brain axis [22,23]. Currently, the term “brain-gut-microbiota” describes not only the axis but also the host’s CNS, endocrine chemical signal system, immune regulation, microbiota and metabolic effects, and barrier functions in the brain and gut (Figure 1). All of these factors need to balance out to keep a person healthy [24]. Depression and other mental health issues are just one of the many that can result from an imbalance in the brain-gut-microbiome axis. The microbiome is made up of trillions of microorganisms like bacteria, viruses, archaea, and fungus, and its genetic content comprises more than 100 times as many genes as the human genome. Therefore, there are approximately as many bacterial cells as human cells [25,26].
The gut microbiota secretes important vitamins including folic acid and vitamin K for overall bodily health, aids in digestion and nutrition absorption from food, and defends against pathogens. Recent studies have shown that the gut microbiota and host have a beneficial interaction that starts in the womb and continues through the mother-child bond, ultimately affecting the mental and cognitive health of the child. Recent research has shed light on the complex ways in which the gut microbiota communicates with the central nervous system, expanding our understanding of how the microbiome influences mental health and, in particular, depression [19].
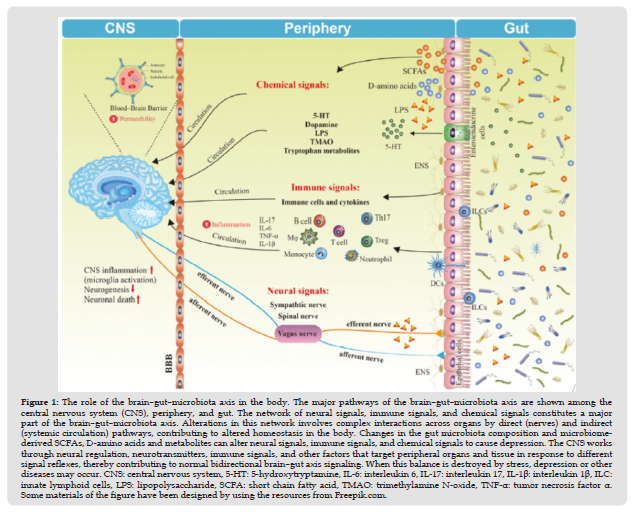
Figure 1 The role of the brain–gut–microbiota axis in the body. The major pathways of the brain–gut–microbiota axis are shown among the central nervous system (CNS), periphery, and gut. The network of neural signals, immune signals, and chemical signals constitutes a major part of the brain–gut–microbiota axis. Alterations in this network involves complex interactions across organs by direct (nerves) and indirect (systemic circulation) pathways, contributing to altered homeostasis in the body. Changes in the gut microbiota composition and microbiomederived SCFAs, D-amino acids and metabolites can alter neural signals, immune signals, and chemical signals to cause depression. The CNS works through neural regulation, neurotransmitters, immune signals, and other factors that target peripheral organs and tissue in response to different signal reflexes, thereby contributing to normal bidirectional brain–gut axis signaling. When this balance is destroyed by stress, depression or other diseases may occur. CNS: central nervous system, 5-HT: 5-hydroxytryptamine, IL-6: interleukin 6, IL-17: interleukin 17, IL-1β: interleukin 1β, ILC: innate lymphoid cells, LPS: lipopolysaccharide, SCFA: short chain fatty acid, TMAO: trimethylamine N-oxide, TNF-α: tumor necrosis factor α. Some materials of the figure have been designed by using the resources from Freepik.com.
The Link Between the Gut Microbiota and Behavior
Human gut microbiota weighs in at about 0.2 kilograms (recently updated estimate) [27] and contains roughly the same number of cells as human eukaryotic cells. It also has an estimated 150 times as many genes. Recent decades have seen a surge in research into the gut microbiome as its importance in metabolic and endocrine product creation and interactions with the host neurological and immunological systems has become more widely recognized. Inflammatory bowel illness and irritable bowel syndrome are associated with a significant co-morbidity of anxiety and depression disorders [28,29], which led researchers to speculate that the gut microbiota is linked with and may impact mood disorders. Individuals with anxiety or depression (even those in remission) have been demonstrated to have a different fecal microbiota composition compared to healthy controls in correlational investigations.
When comparing women with different levels of fecal Bacteroides abundance, we found that those with a higher abundance of Prevotella had a greater negative emotional response to unpleasant imagery and decreased hippocampal activity. The presence and makeup of gut bacteria has been proven experimentally to impact emotional behaviour in animals in several studies. In mice, anxiety-related behaviors like reduced exploration and greater behavioural inhibition were triggered by infections or inflammation in the digestive tract [29]. Anxiety and depression-like behaviors are altered in Germ-Free (GF) rats and mice (bred without any microbiota) but not in their Specific Pathogen-Free (SPF) counterparts. Certain probiotics have been shown to improve mental state. Those probiotics known to “confer mental health benefits through interactions with commensal gut bacteria” are called “Psychobiotics.” The term “Psychobiotics” [27,29] does not apply to all probiotics or prebiotics. Several explanations for the discrepancies in MGBA studies are provided below. Strain-specific aspects include differences in probiotics’ viability, mucosal adhesion, and bioactive chemical production [26].
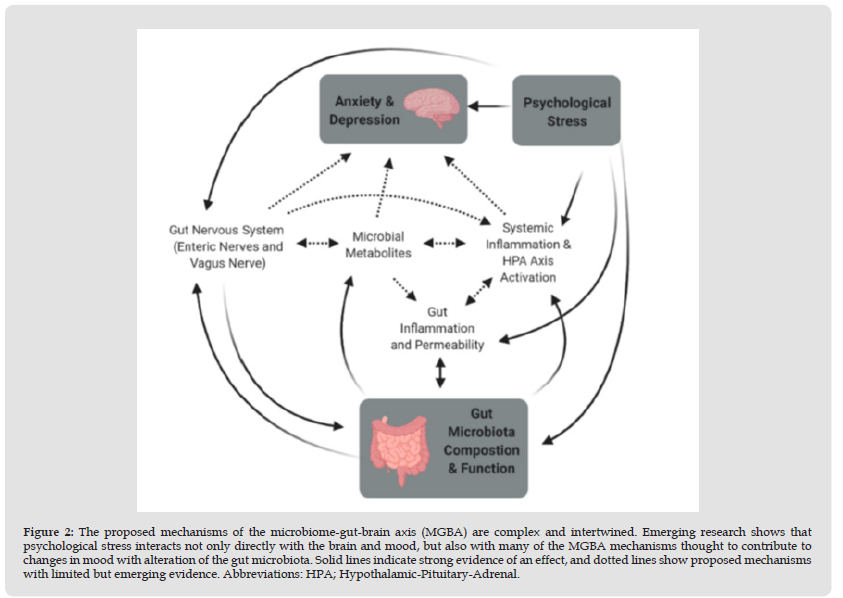
The dose is also crucial, and the optimal dosage may change depending on the specific probiotic strain being used. Changes in gut microbiota may or may not affect mood, as seen by the observed variation in this effect in GF rats, suggesting that it is the interactions of the microbiome with the host that are crucial. Mechanisms proposed for the MGBA (shown in (Figure 2) are intricate, mutually reinforcing, and two-way. Mood-altering mechanisms may be unique to each host species, subspecies, and even person based on factors such as food, stress levels, and level of physical activity. It is possible for multiple mechanisms to operate simultaneously [27]. It’s common knowledge that stress plays a role in the pathophysiology of depression and that resilience is mediated by adaptive changes in several circuits, such as brain-derived neurotrophic factor (BDNF), pro-inflammatory cytokines, and the spleen. Studies have shown that certain rodents are immune to LH and CSDS (chronic social defeat stress). Our group discovered that CSDS-resistant mice have significantly more Bifidobacterium than control and CSDS-susceptible mice.
Figure 2 The proposed mechanisms of the microbiome-gut-brain axis (MGBA) are complex and intertwined. Emerging research shows that psychological stress interacts not only directly with the brain and mood, but also with many of the MGBA mechanisms thought to contribute to changes in mood with alteration of the gut microbiota. Solid lines indicate strong evidence of an effect, and dotted lines show proposed mechanisms with limited but emerging evidence. Abbreviations: HPA; Hypothalamic-Pituitary-Adrenal.
Bifidobacterium orally administered significantly enhanced the number of CSDS-resistant mice compared to vehicle treatment, suggesting that Bifidobacterium confers resistance to CSDS. The relative abundances of Lactobacillus, Clostridium cluster III, and Anacrusis were likewise significantly higher in LH-vulnerable rats compared to control and LH-resistant rats. Mice with CSDS can keep up their resistance to anhedonia with the help of betaine administration, which has been demonstrated to have anti-inflammatory benefits. These findings corroborate one another in suggesting that the brain–gut–microbiome axis is involved in shaping both susceptibility and resistance to stress. Evidence from animal studies is mounting indicating the brain-gut-microbiota axis is crucial for the emergence of depressive symptoms in humans.
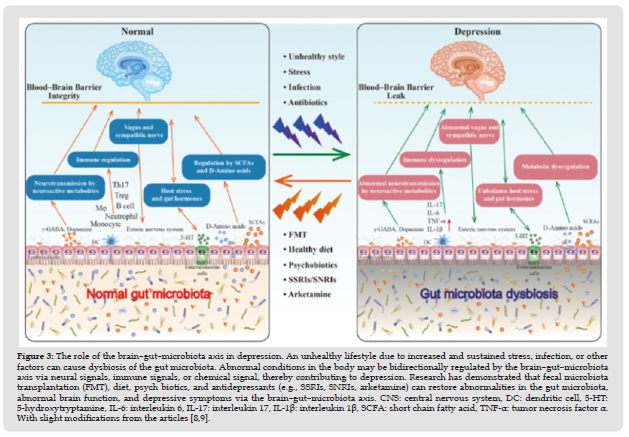
Abnormal levels of SCFAs and other metabolites like alanine, isoleucine, L-threonine, serine, and tyrosine associated to the gut microbiota have been linked to stress-induced depressionlike behaviors in rodents. Depressive-like phenotypes may also be influenced by these metabolites, which impact brain 5-HT levels. Evidence suggests the brain-gut-microbiota axis regulates depression in both directions, with administration of microbiota or their metabolites having opposite effects on depressive symptoms. In contrast, it has been shown that SCFA combination therapy, which includes acetate, butyrate, and propionate, can alleviate stressinduced depressive-like behaviors in rats. Proteomics studies using iTRAQ (Isobaric Tag for Relative and Absolute Quantitation) have demonstrated that FMT from depressed people causes changes in serum, prefrontal cortex, cecum, and liver proteins in a mouse model of depression. The microbiota-driven protein profile was considerably altered in these organs, suggesting a function for the gut-brain axis in depression. It was hypothesized that the changed protein profiles would contribute to metabolic regulation and the inflammatory immune response. Evidence for the brain-gut axis’ role in depression can be bolstered by learning more about the dramatic changes that take place either in the brain or the gut [30], Refer to (Figure 3).
Figure 3 The role of the brain–gut–microbiota axis in depression. An unhealthy lifestyle due to increased and sustained stress, infection, or other factors can cause dysbiosis of the gut microbiota. Abnormal conditions in the body may be bidirectionally regulated by the brain–gut–microbiota axis via neural signals, immune signals, or chemical signal, thereby contributing to depression. Research has demonstrated that fecal microbiota transplantation (FMT), diet, psych biotics, and antidepressants (e.g., SSRIs, SNRIs, arketamine) can restore abnormalities in the gut microbiota, abnormal brain function, and depressive symptoms via the brain–gut–microbiota axis. CNS: central nervous system, DC: dendritic cell, 5-HT: 5-hydroxytryptamine, IL-6: interleukin 6, IL-17: interleukin 17, IL-1β: interleukin 1β, SCFA: short chain fatty acid, TNF-α: tumor necrosis factor α. With slight modifications from the articles [8,9].
Evidence from rodent models suggests that gut flora may influence depression-like behaviour. Patients with depression are twice as likely to encounter digestive problems. One idea posits that depression, or at least some forms of this syndrome, is a microglial dysfunction [31], given that severe episodes of brain inflammation often accompany the onset of depression. Interestingly, minocycline, a tetracycline antibiotic, has been recommended as a potent antidepressant and demonstrated to improve sad behaviours in mice and people. It is reasonable to hypothesise that the microbiota influences microglial maturation and activation, which in turn influences depression processing, given recent findings on the function of the microbiome in microglial maturation and activation [32]. It should be noted, however, that it remains unclear whether minocycline’s antidepressant effects are due to the drug’s antibacterial qualities, its ability to suppress microglial activation, or some combination of the two. Patients with Major Depressive Disorder (MDD) have been demonstrated to have a much less diverse gut microbiome than healthy controls, with MDD-associated microbial groups including significantly more Actinobacteria and fewer Bacteroidetes, as shown by a recent study by Zheng et al. (2016).
Furthermore, the authors transplanted human control and MDD samples into GF mice, showing that the MDD samples caused recipients to exhibit depressive-like behaviours. These conclusions were corroborated by another investigation [33], which found a correlation between decreased bacterial richness and diversity in depression and the potential to transmit depressive-like features to rats by faecal transplantation. There is promising evidence from studies in mice and humans that the microbiota may play an active role in the onset of depressive-like behaviours, suggesting novel pathways for treatment.
Preclinical and clinical studies show that probiotic supplementation reduces anxious and depressive-like behaviours in mice, rats, zebrafish, and humans. The GF mouse model was used to examine the long-term effects of Lactobacillus plantarum PS128 (PS128) treatment on anxiety- and depression-like behaviours and on monoamine neurotransmitter levels in the striatum, prefrontal cortex, and hippocampus. No adverse effects were seen with chronic administration of live PS128. Live PS128 injection significantly increased the time mice spent in open arms and the distance mice travelled in the elevated plus maze test, but had no influence on the depressive-like behaviour of GF mice in the forced swim. Analysis of monoamine neurotransmitter levels demonstrated significantly increased serotonin and dopamine concentrations in the striatum following chronic live PS128 injection. No alterations were observed in the prefrontal cortex or hippocampal areas. Both Lactobacillus rhamnosus JB-1 and Bifidobacterium longum NCC3001 in mice with a chronic infection of the gut parasite Trichuris muris and Lactobacillus helveticus R0052 and Bifidobacterium longum R0175 in rats produced anxiolytic- and antidepressant-like effects or blocked anxiogenic-like effects.
Probiotics have also been demonstrated to increase monoamine neurotransmitters in the striatum of mice while decreasing BDNF and corticosterone. Preparations containing Lactobacillus rhamnosus or Lactobacillus helveticus enhanced gastrointestinal function, which normalised corticosterone levels in rats exposed to MS stress, as previously demonstrated by Gareau et al. Mice that were subjected to stress in the elevated plus-maze test exhibited less anxious behaviour after being treated with Lactobacillus reuteri, which was also associated with a reduction in plasma corticosterone levels.
Desbonnet et al [34] state that Bifidobacterium infantis can be given orally. Probiotics have been demonstrated to be useful in clinical research for treating mood disorders. One study found that patients who utilised Lactobacillus casei strain Shirota and had lower preprobiotic depression scores reaped the greatest benefits, as measured by post-probiotic mood outcomes. In addition, Messaoudi et al. found that patients who encounter low levels of stress may benefit from a long-term co-administration of Lactobacillus helveticus R0052 and Bifidobacterium longum R0175.
Over the course of 30 days, participants in the general population who took this probiotic formulation showed improvements on the Hopkins Symptom Checklist–90 (HSCL90) and the Hospital Anxiety and Depression Scale (HADS). As a result, this form of therapy is useful for helping patients with modest burdens control their stress reactions and anxious behaviours and boost their disposition. Focusing on the role of this probiotic combination in patients with the lowest urinary free cortisol levels at baseline, they concluded that the effectiveness of chronic co-administration of Lactobacillus helveticus R0052 and Bifidobacterium longum R0175 may vary depending on individual’s stress level. Preclinical research has suggested that Bifidobacterium and Lactobacillus in the gut had a beneficial affect on stress response and depression disorder, thus this study examined whether or not there was an association between the number of these strains and major depressive disorders.
They found that depressed people have significantly lower quantities of the germs Bifidobacterium and Lactobacillus compared to healthy people. In addition, a randomised, double-blind, placebocontrolled research conducted by Lew and coworkers demonstrated that the anti-inflammatory activities of Lactobacillus plantarum P8 were responsible for the amelioration of stress and anxiety symptoms. Recent clinical research also demonstrated a robust relationship between psychological factors and microbiota balance. Additionally, the benefits of targeted probiotic supplementation for promoting psychological health by reestablishing a healthy equilibrium in the gut-brain axis were highlighted [35]. The interconnectedness of the gut, the brain, and depressed symptoms Anhedonia, a hallmark of depression, is mirrored in the percentage of sucrose/saccharin solution consumed in the sucrose/saccharin preference test (SPT). Each animal in the SPT spent 48 hours in a tank with water and either 1% or 2% sucrose/saccharin solution, followed by 24 hours in two identical bottles with water and either 1% or 2% sucrose/saccharin solution. Water and sucrose/saccharin bottles were weighed before and after the experiment to determine which was preferred.
Decreased sucrose/saccharin consumption is indicative with a lower SP and a higher level of anhedonia. Antibiotic therapy in mice decreased SP, which may point to the significance of gut microbiota in depressive phenotypes. This is because drugs can eradicate as much as 90% of beneficial gut bacteria. Transplantation of fecal microbiota from anhedonia-prone rats significantly exacerbated depression-like behaviors in germ-free mice, as seen by a decrease in the sucrose/ saccharin preference index (SPI). Transplanting fecal microbiota from sad persons into rats with low microbiome levels results in depressivelike behaviors in the recipient animals. Another symptom of sadness is hopelessness, which can be measured in rats with the forced-swim test (FST) and the tail suspension test (TST). To test FST, for instance, mice were individually placed in a cylinder (25 cm in diameter and 35 cm in height) filled with 20 cm of water and maintained at a constant temperature of 23 _ 1 _C using the automated forced-swim device YHFST (Yihong Co., Ltd., Wuhan, China).
The five-minute interval during which the mouse was motionless was measured. About 2 centimeters from the end of the mouse’s tail, a little piece of adhesive tape was placed in the TST. Mice were hanged from a hook with a single hole punched in a piece of tape between them. The period of inactivity lasted 10 minutes and was filmed. The length of time a subject remains motionless throughout the FST or TST is used as a behavioural measure of desperation in these tests. The role of gut microbiota in regulating depressive behaviour was demonstrated by the fact that germ-free (GF) mice exhibited less immobility during the FST and TST compared to specific pathogenfree (SPF) animals and healthy controls. In contrast, GF mice that have been exposed to the microbiota of MDD patients show reduced central open field test (OFT) distance travelled and higher FST and TST immobility durations [36].
Figure 4 Intersections of Gut Microorganisms and Basic Developmental Processes Basic developmental processes driven directly or indirectly by gut microbes and their products. A) Gut microorganisms relay messages to the brain via various direct and indirect mechanisms. B) Basic neurodevelopmental processes are modulated because of colonization of GF animals or depletion of gut bacteria by antibiotics. Specifically, the following processes are modulates blood-brain barrier (BBB) formation and integrity, neurogenesis, microglia maturation and, myelination and expression of neurotrophies, neurotransmitters, and their respective receptors.
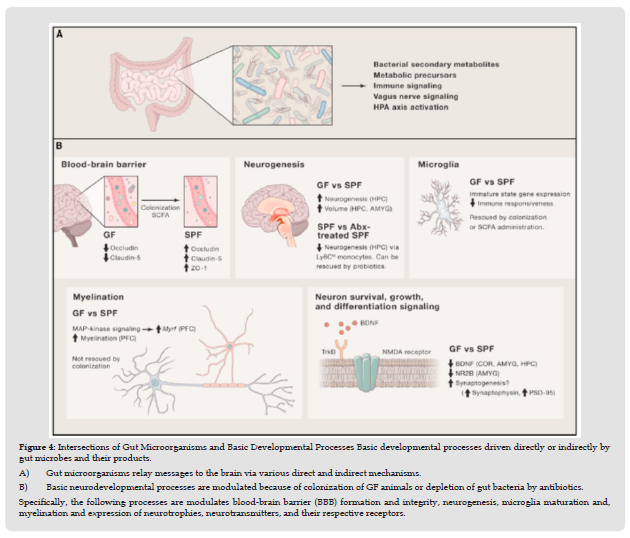
Mounting evidence alludes to a significant role for neurological, endocrine, immunological, and metabolic pathways in this communication (Figure 3), but the precise mechanism underpinning the microbiota-gut brain axis’ impact on depression is yet unknown. The article describes the two-way communication between gut microbes and the brain, which can affect depressive disorders through neuronal, endocrine, and immunological signaling pathways. The liver, because of its central location and blood supply, coordinates and participates in fundamental immune system activities (Figure 4). To regulate the development and plasticity of brain circuits important for mood and behaviour, intestinal flora regulates the expression of monoamine neurotransmitters, including 5-HT, DA, and GABA. Increased cortisol, adrenocorticotropin, and Corticotropin-Releasing Hormone (CRH) production indicates hyperactivity of the HPA axis, which can be prevented or treated by changing the function of the microbiota-gut-brain axis via gut microbiota.
The mucosal system provides the initial barrier against invading pathogens. Protecting against pathogens, blocking the entry of foreign antigens, promoting oral tolerance, and maintaining mucosal homeostasis are just few of the basic functions of the Mucosal Immune System (MIS). The liver is essential for a functional innate immune response because it receives direct access from microorganisms and their metabolites in the digestive tract via the portal vein. Alterations in gut microbiota can aid depressive symptoms by balancing out pro- and anti-inflammatory cytokines such IL-6, IL-1, and TNF-. The development of depressive symptoms may be influenced by alterations in the gut microbiota, which could lead to increased BDNF expression. The pathophysiology of depression involves all of these mechanisms [15]. Modulating the amount of brain-derived neurotrophic factor Brain-derived neurotrophic factor (BDNF) is a member of the neurotrophic family that has been connected to a variety of processes, such as cell differentiation, neuronal survival, synaptogenesis, and the development of neuroplasticity.
Lack of BDNF is associated with impaired neuroplasticity and the development of depressive symptoms. Multiple investigations have found that depressed patients’ serum BDNF levels are lower than those of healthy controls. Treatments for depression, such as ketamine, can improve mood and cognitive function by increasing BDNF activity via mTOR signaling. [19,20] Probiotic treatment shows potential as a prophylactic or therapeutic approach for addressing behavioural impairments by modifying the gut microbiota and BDNF expression. Bercik et al. found that administering oral antimicrobials to SPF mice caused a transient shift in their gut microbiome, which in turn increased BDNF levels in the hippocampus and protected the mice from developing depression. It has also been shown that pretreatment with probiotics (Lactobacillus helveticus R0052 and Bifidobacterium longum R0175) for two weeks increases hippocampal expression of BDNF in stressed mice, reversing synaptic dysfunction and ultimately improving performance in learning and memory tasks. In addition, Lactobacillus helveticus NS8 supplementation improved hippocampus BDNF mRNA expression and behavioural performance in a rat model of chronic stress-induced depression. Depressive symptoms may be influenced by changes in the gut microbiota, which regulate BDNF levels [21].
The etiology of gut dysbiosis is complex, with several possible risk factors. Antibiotic use has been linked to both short-term and longterm changes in the gut microbiome. Reproducible alterations to the gut microbiota have been linked to both obesity and high-fat, highsugar diets. It is also believed that gut dysbiosis can be affected by environmental variables at various stages of life. Birthing methods, infant feeding practices, and hospital settings have all been associated with altered microbiome diversity. Exposure to xenobiotics, such as heavy metals and pesticides, and social stressors are also associated with gut dysbiosis. Studies of identical twins have revealed that both genetic and environmental factors contribute to the formation of the gut microbiota [37].
There is constant bidirectional communication between the digestive system and the brain. Such communication may play a crucial role in mediating physiological effects ranging from gastrointestinal function to the brain and behaviour and may affect the sensation of visceral events as nausea, satiety, and pain. Both gastrointestinal (GI) secretions and motility are affected by stress. The clinical relevance of such two-way communication and shifts in gut flora composition is currently the subject of research. Intestinal (GI) tract sensory data may serve as the starting point for a bidirectional gut-brain connection that ultimately results in neuronal, hormonal, and immunological signals. These signals might be processed independently or in concert by the Central Nervous System (CNS). The neurological and immunological mechanisms are discussed in the paper where relevant. For in-depth analyses of the literature on neural and immune pathways, check out and. Furthermore, the gut microbiota-brain axis’s neurological and immunological pathways are depicted in a simplified form in (Figure 1), [23-25].
Gut peptides and other signaling chemicals, such as serotonin, are secreted by different EEC types all throughout the GI tract in response to food intake, particularly after the absorption of carbohydrates and fats, forming the gut endocrine system. The location and chemical class (including peptides) secreted by EECs determine their subtypes (Figure 2). Only around one percent of the epithelial cells lining the gut lumen are EECs. Enterochromaffins, or EECs, were recently shown by Bellono et al. in a stunning study to control serotonin release onto primary afferent nerve fibres that extend into intestinal villi and express 5-HT3 receptors, allowing them to sense and transfer information from the gut to the nervous system. Peptides are short, amide-bonded chains of amino acids (about 50 amino acid residues) that can operate as a signaling molecule in EECs by interacting with G protein-coupled receptors. Peptides are produced by some brain cells and are referred to as neuropeptides. Peptides can attach to receptors that are physically separated from the site of their release, and they are effectively degraded by endogenous enzymes to prevent tissue accumulation. There are more than a hundred peptides in mammals, with the ones most critical for the gut-brain link regulating satiety and digestion.
Peptides that control the gastrointestinal tract are found not only in EECs and the central nervous system, but also in the enteric nervous system. Intrinsic sensory neurons, for instance, express calcitonin gene-related peptide and tachykinins (also known as substance P). Controlling gastric acid production and intestinal motility, gastrin-releasing peptide triggers gastrin secretion from G cells in the stomach. To regulate the circadian cycle and suppress gastric acid production and intestinal lumen absorption, vasoactive intestinal peptide interacts with adjacent postsynaptic targets. Given the broad expression of peptides and their receptors in the brain and stomach, as well as their easier transit into the bloodstream, it is logical to expect that these peptides can act beyond their core signaling function. This may indicate (though indirectly) a function for gastrointestinal peptides in neuropsychiatric conditions. The dynamic profile of gut peptides includes their direct association with mood disorders, suggesting that this axis may be a target for Psychobiotics to prevent and cure such conditions [25-28], Take a peek at the diagram in (Figure 5).
“Probiotic” comes from the Greek “pro bios,” which means “for life.” The beneficial effects of lactic acid fermentation products on human health have been known for quite some time. Microbiologist and Nobel laureate Élie Metchnikoff is credited with creating the original theory. He has co-authored numerous seminal studies on the benefits of probiotic miscroorganisms. He suggests that the lactic acid bacteria normally found in the human colon may have life-extending effects. Nowadays, “live microorganisms that, when administered in adequate amounts, confer a health benefit on the host” is how people refer to probiotics. These are mostly made up of Lactobacillus and Bifidobactersium strains, with some Streptococcus and Enterococcus thrown in for good measure [38]. Antiallergic activity, improved intestinal health (by eliminating dysbiosis and sealing the intestinal epithelium), immune system stimulation, lactose intolerance inhibition, cancer prevention, and positive effects on mental health are just some of the benefits of probiotics [39-41] (Figure 6).
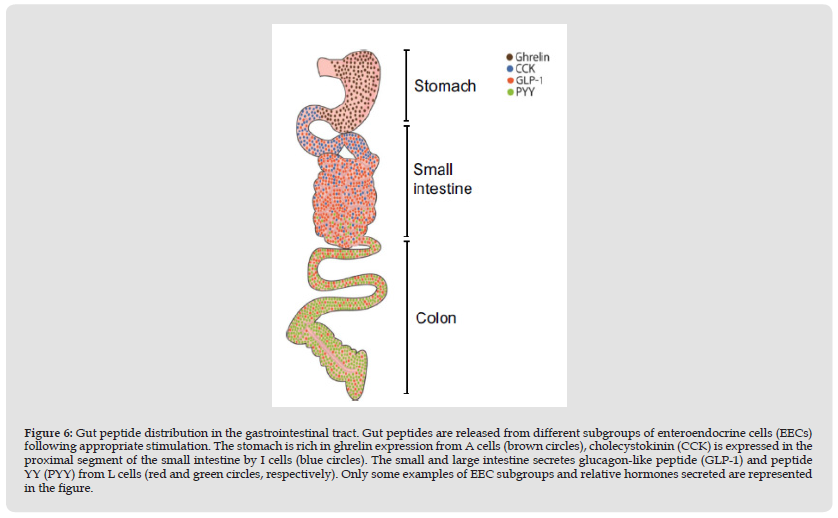
Figure 6 Gut peptide distribution in the gastrointestinal tract. Gut peptides are released from different subgroups of enteroendocrine cells (EECs) following appropriate stimulation. The stomach is rich in ghrelin expression from A cells (brown circles), cholecystokinin (CCK) is expressed in the proximal segment of the small intestine by I cells (blue circles). The small and large intestine secretes glucagon-like peptide (GLP-1) and peptide YY (PYY) from L cells (red and green circles, respectively). Only some examples of EEC subgroups and relative hormones secreted are represented in the figure.
It’s important to stress the fact that most bacteria are not naturally present in the human body and must be acquired by the mother before birth. Healthy people who eat well have a microbiome that is balanced. Additional probiotic bacterial administration might be considered in such a scenario. According to Dinan et al., patients with psychiatric disorders can reap health benefits from consuming certain strains of probiotics if they take them in large enough numbers. Animal and human studies show that Psychobiotics improve intestinal health and affect serum cytokine, cortisol, and brain neurotransmitter and protein levels, leading to behavioural changes. Important neuroactive metabolites, such as catecholamines, 5-hydroxytryptamine (5-HT), and Gamma-Aminobutyric Acid (GABA), may be under the influence of a wide variety of bacteria. GABA is an amino acid that inhibits synaptic conduction by hyperpolarizing the membranes of neuronal cells, hence reducing activity in the central nervous system. Mood disorders and damage to the GABAergic system go hand in hand. It has been shown that people with mood and anxiety problems have reduced amounts of the neurotransmitter gamma-aminobutyric acid in their brains [42,43].
Lactobacillus plantarum, Lactobacillus paracasei, Lactobacillus rhamnosus, and Lactobacillus brevis are all examples of foodderived Lactobacillus strains that regulate GABA levels [44-46]. Certain bacterial strains can also affect serotonin levels. Serotonin, a monoamine neurotransmitter, is synthesized from tryptophan. Most 5-HT is produced by enterochromaffin cells in the gut. Serotonin plays an extremely intricate role in the regulation of mood, cognition, and a few physiological systems. An imbalance in the serotonergic system is one of the fundamental causes of depression. Several bacterial species, including Escherichia coli, Klebsiella pneumoniae, Morganella morganii, Lactobacillus plantarum, Lactococcus lactis subsp. cremoris, and Streptococcus thermophilus, are known to disrupt the 5-HT pathway. Catecholamines, such as adrenaline, noradrenaline, and dopamine, are regulated by a number of bacteria, including Bacillus species, Escherichia coli, Staphylococcus aureus, Klebsiella pneumoniae, and Morganella morganii. Some of these strains have been incorporated into enhanced functional foods to increase health and have a substantial effect on neurometabolites control [45-48].
The original definition of a prebiotic was a “non-digestible food ingredient that benefits the host by specifically stimulating the growth and/or activity of one or a limited number of bacteria already resident in the colon.” However, as science and understanding have progressed, the term has broadened to include not just the induction of colonic bacteria but of other bacteria as well [49]. The International Scientific Association for Probiotics and Prebiotics defines prebiotics as “a substrate that is utilised by host microorganisms with a view to conferring a health benefit.” Many food and natural products contain prebiotics, which are nondigestible polysaccharides. Fructans (fructooligosaccharides, inulin), oligosaccharides, and galactooligosaccharides are all examples. Bacterial fermentation of prebiotic carbohydrates produces SCFAs like butyric acid, acetic acid, and propionic acid. Because of their immune-boosting properties, ability to fuel gut microbiota, and antagonistic effects on pathogenic gut bacteria, prebiotics and SCFAs are crucial for sustaining intestinal health. It has been shown in a number of laboratory studies that prebiotics can mitigate the symptoms of and even prevent diseases like schizophrenia, diabetes, irritable bowel syndrome (IBS), infectious infections, and colon cancer [50-52].
Prebiotics containing Galacto-Oligosaccharides (GOS) have been demonstrated to increase brain BDNF in the hippocampus of mice. Human blood BDNF levels have been demonstrated to be inversely linked with the degree of depression 86, and BDNF expression is assumed to be critical for continuous neurogenesis in the adult hippocampus [53]. After five weeks of feeding with the human milk oligosaccharide (HMO) 20-fucosyllactose, BDNF protein was likewise found to be raised in the hippocampus of rats. In this study, the HMOsupplemented rats learned the operant conditioning task in fewer sessions than the control group, suggesting that the HMO facilitated associative learning. Anxiety and depression-like behaviour in rodents can be altered by taking a prebiotic supplement. Mice that had been fed GOS for three weeks exhibited decreased anxiety in the LDB 24 hours after LPS administration compared to controls (water-only drinkers) [54]. Increased Bifidobacterium spp. in the gut and decreased IL- 1B and 5-HT2A receptor proteins in the prefrontal brain following LPS treatment were linked to the prebiotic’s anxiolytic impact. Both early-life stress caused by maternal separation and persistent mild stress have been demonstrated to have negative behavioural effects in mice, although prebiotics have been proven to alleviate these effects. This was demonstrated by an independent improvement in spatial learning in the Morris water maze as well as a reduction in anxiety and depressive-like conduct. Finally, it has been shown that GOS supplementation in rats after birth decreases anxiety-like behaviour in the EPM at the juvenile (3-week-old), adolescent (8-week-old), and adult (4-6-month-old) ages [55-57].
Lester Dominguez conceived this study, carried out this study, and drafted the manuscript. Maria Dominguez Rios designed the study, collected, and analyzed the data. Both authors were responsible for this manuscript and reviewed the article critically. All authors read and approved the final manuscript.
The authors declare no conflicts of interest.
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.

