Impact Factor : 0.548
- NLM ID: 101723284
- OCoLC: 999826537
- LCCN: 2017202541
Dan Prat1,3, Asaf Bloch1,3,4,6*, Tami Barazani-Brutman4, Or Benifla4, Eran Avivi1, Dina Orkin2 and Amit Zabtani1,3,4,5
Received: November 10, 2022; Published: November 16, 2022
*Corresponding author: Asaf Bloch, Department of Orthopedic Surgery, Chaim Sheba Medical Center, affiliated with the Sackler Faculty of Medicine, Tel Aviv University, Ramat Gan, Israel, Sackler Faculty of Medicine, Tel Aviv University, Tel Aviv, Israel, The 3D Medical Solutions Center, Sheba Medical Center, Tel Hashomer, Arrow Project, Sheba Medical Center, Tel Hashomer, Ramat Gan, Israel
DOI: 10.26717/BJSTR.2022.47.007456
Isolated talus osteomyelitis is exceedingly rare [1]. Effective treatment of chronic osteomyelitis includes thorough debridement of necrotic tissue, systemic antibiotics, and high dose locally delivered antibiotics [5]. Antibiotic-loaded cement spacers deliver high concentrations of antibiotics directly to infected tissues, which is of most importance in poorly vascularized bones such as the talus. Cement spacers can be articulating or non-articulating. Articulating spacers mimic joint congruency and allow joint motion and weight-bearing. In recent years, the availability of CT scanbased 3D modelling and printing technology facilitated the use of patient specific prosthetics [3,4]. Total talus replacement has been previously described for salvage of severe fracture dislocations of the talus, talar avascular necrosis, and end stage peritalar arthritis [4,5,6]. 3D printing technology was described scarcely in the literature to create patient specific molds but never using internal supports. Here, we describe a novel technique for making a 3D modelled patient-specific, Kirshner-wire reinforced, antibioticimpregnated total talus articulating spacer for the treatment of persistent osteomyelitis of the talus [7]. 13 months after the procedure the patient underwent a 2nd stage surgery implanting a permanent personalized titanium coated 3D printed talar implant matching the talar anatomy and tibial polyethylene part of a Cadence total ankle system. To our knowledge, there is no published literature describing this technique to treat hematogenous talar osteomyelitis [7].
In this case, an overweight 56-year-old male presented to the emergency department with chronic right ankle pain lasting for three months. The patient was partially treated with oral antibiotics in an outside hospital for presumed cellulitis. Diagnostic arthrocentesis from his ankle yielded less than 1cc of fluid. Cultures revealed methicillin-sensitive staph aureus (MSSA). Plain radiographs and a CT scan demonstrated osteomyelitis of the talus. (Figure 1) During hospitalization, the patient underwent surgical debridement of the ankle joint and bone biopsy from the talus that confirmed the diagnosis. IV cefazolin was started, endorsed by infectious diseases specialists. After 18 days of hospitalization, the patient was discharged and continued home IV treatment for a total of 6 weeks.
Three months postoperatively and 6 weeks after cessation of the IV antibiotics, the patient presented to the emergency department with pain and swelling in his right ankle. A new CT scan showed progression in the severity of the osteomyelitis, represented by enlarged diffuse cysts (Figure 2). Ankle arthrocentesis revealed pus and cultures were positive for MSSA.
A multidisciplinary team consisted of foot and ankle orthopaedic surgeons and infectious diseases specialists decided that the next step in management would require excision of the infected bone. The substantial extent of infected bone in the talus led us to opt for talectomy and reconstruction. This therapeutic route could simultaneously offer eradication of the infection and a functional articulating joint spacer. The multidisciplinary team considered bone grafting as treatment but given the patient state and risk of nonunion it was a considerable risk. Our hospital operates an inhouse 3D laboratory that provides tools for complex modelling and reconstructions. In collaboration with our 3D lab, we designed a patient specific antibiotic-impregnated bone cement articulating spacer using a 3D printed mold replicating the talus of the patient.
Image acquisition: DICOM files of the CT scan were extracted from the PACS. Model resolution is highly dependent on the original CT scan quality and slice thickness. In this case we used 0.9 mm slices. Ideally, 0.6 to 1.0 mm slices can be used. Segmentation was conducted using D2P® software (3D Systems Inc, Rock Hill, SC, USA). The software allows separation of the bone by adjusting the threshold of the voxels’ Hounsfield units (HU). The 3D model conversion was performed using an automated process in the D2P® software, the segmented bone was converted into a virtual 3D model. Later we applied a smoothing algorithm to eliminate inconsistencies. The model was reduced to 95% of its original size to facilitate the spacer insertion into the articular space. We used Fusion 360 CAD software (Autodesk, San Rafael, CA,USA) to design the mold. We created a hollow shell consisted of 4 separate plates (Figure 4). The multiple building plates allowed extraction of the spacer with minimum effort. Alignment of the plates was achieved by six key bolts. The mold was designed to integrate four 1.6 mm Kirschner-wires (K-wires) between the building plates for reinforcement of the cement after its hardening. The 3D model was then extracted as a native 3D printing file (STL format) and transferred to our lab’s printers: Ultimaker 3 extended (Ultimaker BV, Utrecht, Netherlands) and Formlabs Form-3 (Formlabs, Somerville, MA, USA). We then performed a trial with the printed mold with bone cement to ensure the accuracy of the mold, comparing it a pre-printed model of the talus. The final version of the mold was printed in sterilization-safe Nylon PA12 on an HP MJF 580 printer (Palo Alto, CA, USA). We decided to use nylon PA12 due to its flexibility, which ease extraction of the model from the mold (see supplementary video 3).
Figure 4:
a) Articulating spacer mold assembled after injection of medical cement in OR.
b) Image from the designing process of the mold in the virtual working environment.

We used a standard anterior approach to the ankle joint. Purulent joint effusion and infected capsular tissue were excised. We used saw cut at the talar neck to ease the talus removal.
The articulating spacer was prepared on the back table. The mold was coated with bone wax prior to assembly to ease the extraction of the hardened cement. K-wires were introduced in the designed grooves. Finally, the antibiotic-loaded (4 grams of vancomycin and 320 mg of gentamycin) bone cement (2-packs of 40 gr SMARTSET, DePuy Synthes, Warsaw, IN, USA) was injected into the mold. After complete hardening, the spacer was extracted and manually smoothed around the edges of the gaps between the mold’s parts and at injection site (Figure 5). The articular spacer was then introduced into the peritalar space. Accurate reduction of the joint was confirmed under fluoroscopy (Figure 6). Proper joint motion was visually confirmed. (video 1) A surgical drain and a short leg splint were applied.
The patient was discharged after 5 days, upon receiving the final wound cultures confirming MSSA infection. The patient was treated with IV cefazolin 2g 3 times a day for 6 weeks. Weight bearing in a controlled ankle motion (CAM) boot was initiated at 2 weeks postoperatively. At 3 months follow-up the patient reported going back to work and daily activities. The patient used a cane intermittently. Physical examination showed no signs of active infection. Ankle range of motion was measured to 0-20 with minimal subtalar motion. The patient resumed suppressive treatment with oral Cephalexin for 3 months postoperatively, endorsed by infectious diseases specialists. CRP values normalized to 0.6 mg\L (normal reference values: 0-5). Radiographs at 3 months follow-up showed no signs of implant collapse or subluxation at full weight bearing (Figures 7a, 7b). At 6 months postoperatively, the patient reported activity related pain. He continued to ambulate with a CAM boot and a cane. Passive ankle range of motion was painless but limited to 0-20 degrees. Subtalar motion was minimal and painful. No other clinical or radiographic signs of infection were noted (Figure 8a, 8b). The CRP value was 0.45 mg\L. At 10 months postoperatively, CT scan showed damage to the tibial plafond (Figure 9a, 9b). Joint replacement surgery was discussed with the patient as the next step in management. At 13 months after the insertion of the cement spacer, the patient underwent total ankle replacement with Cadence Total Ankle System, Smith and Nephew, Memphis, TN) along with a total talar replacement (Figure 10 a,10b). A personalized titanium coated 3D printed talar implant was made to match the patient’s original anatomy and the tibial polyethylene of the Cadence. (Figure 11 a-d & Figure 12). Tissue and bone cultures from the last operation were negative.
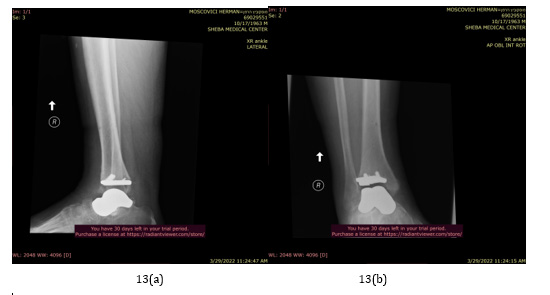
At the last follow-up, 6 weeks after the total ankle replacement surgery, the patient remained infection and pain free and with passive ankle range of motion of 0-20 degrees. The patient was recommended to start walking again without the assistance of controlled ankle motion (CAM) boot and continue with the intense physiotherapy (Figure 13)
Figure 7:
a) 3 months follow-up lateral weight bearing radiographs.
b) 3 months follow-up anterior-posterior weight bearing radiographs
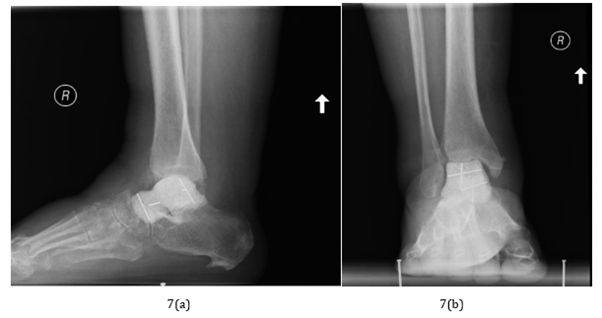
Figure 8:
a) Six months follow-up lateral weight bearing radiograph
b) Six months follow-up anterior-posterior weight bearing radiographs.
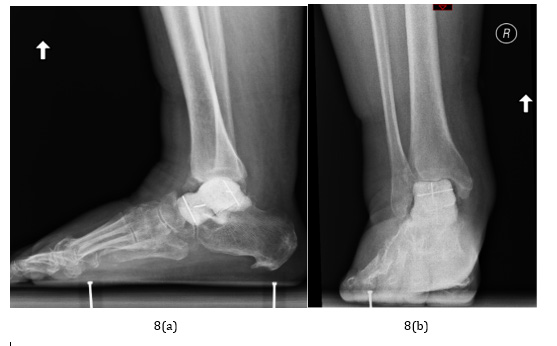
Figure 9:
a) 10 month follow-up lateral CT shows damage to the tibial platfond
b) 10 month follow-up anterior-posterior CT shows damage to the tibial platfond
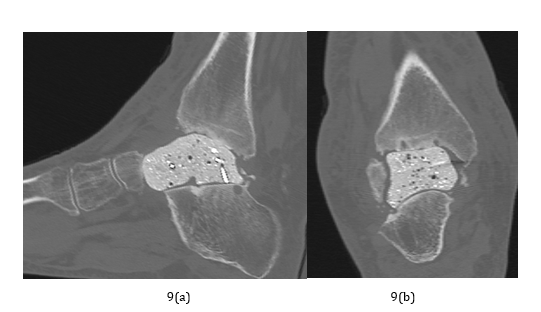
Figure 10:
a) 2nd surgery. Total talar replacement procedure using Cadence total ankle system
b) 2nd surgery. Removal of the individualized cement spacer and total ankle replacement.
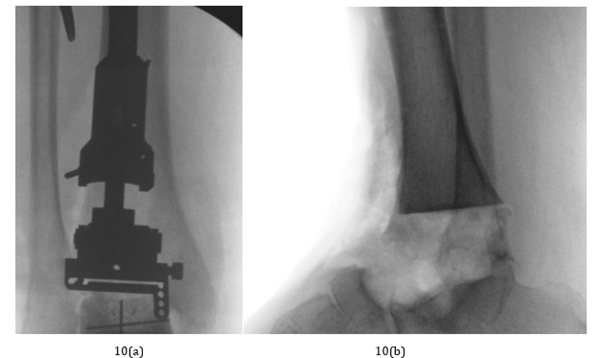
Figure 11:
a) 3D printed titanium coated individualized talar implant in 2nd surgery, lateral view.
b) 3D printed titanium coated individualized talar implant in 2nd surgery, lateral view in full dorsiflexion.
c) 3D printed titanium coated individualized talar implant in 2nd surgery, anterior posterior.
d) 2nd surgery, Cadence system and individualized implant inserted in patient, in OR.
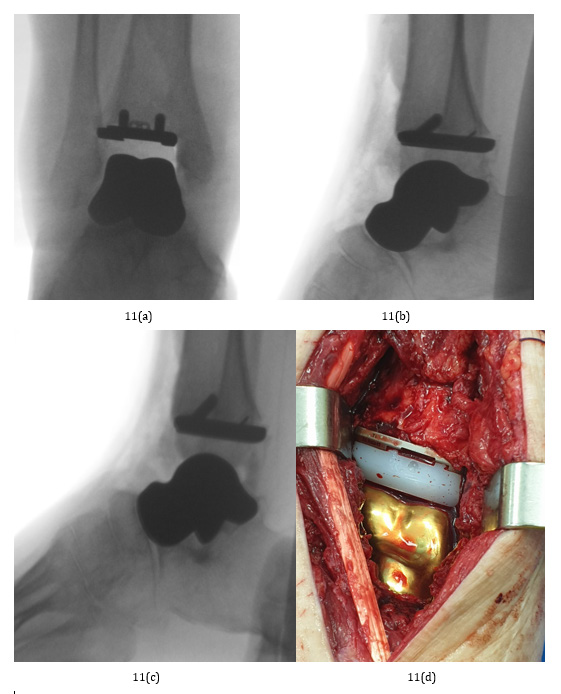
Figure 12: Comparison between the individualized cement spacer, individualized titanium implant and a plastic model.
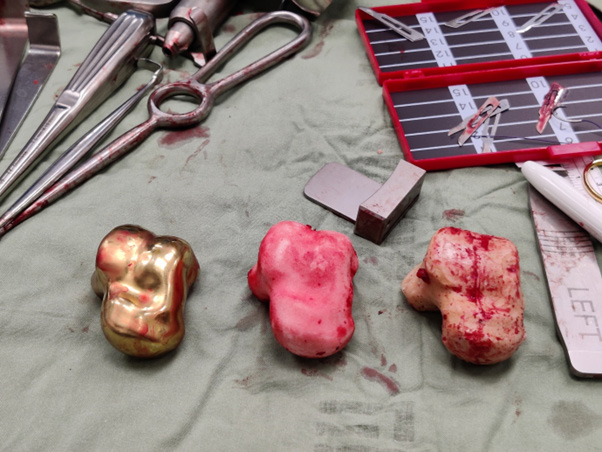
Figure 13:
a) . 6 weeks follow-up from 2nd surgery, weight bearing anterior-posterior image.
b) Fig 13b. 6 weeks follow-up from 2nd surgery, weight bearing lateral image.
Isolated osteomyelitis of the talus is exceedingly rare in adults. A small number of case reports, most of them in children, described various pathogens. No case MSSA were described.3 Chronic Osteomyelitis is a challenging orthopaedic condition. Biofilm development and avascular necrosis of the bone render systemic antibiotics ineffective. Debridement of necrotic tissue with highly concentrated locally delivered antibiotics in the form of antibioticimpregnated bone cement is the gold standard of treatment. 5 Talectomy without reconstruction, of with tibial calcaneal fusion have been described as salvage procedure for severe fracture dislocations of the talus, talar avascular necrosis, and end stage peritalar arthritis [1,9 ] On the other hand, total talus replacement is growing in popularity as an alternative to talectomy [6]. Commonly, the prosthesis was made from stainless-steel, ceramics, titanium, or cobalt-chrome. The implants can be combined with a tibial prosthesis or used in isolation. The advantages of replacing the talus include preservation of joint movement, shorter period of restricted weight bearing, faster pain relief, and preservation of limb length [4,6]. In the present case, we harnessed 3D modelling and printing technology to combine the advantages of total talus replacement and antibiotic-impregnated cement spacer.
Another novelty in our case is the use of K-wires for reinforcement of bone cement. This technique has been previously utilized for intramedullary nail exchange in long bone infection, but never for short bones such as the tarsal bones. Broughton et al reported a successful similar solution without using K-wires but the patient was lost to follow-up 4 months post operatively, not allowing for analysis of the spacer. Our experience showed that Reinforcement with K-wires creates a composite material in which the cement’s relatively low tensile strength and ductility are compensated by the inclusion of high tensile strength and ductility K-wires. This is equivalent to the use of steel bars in reinforced cement concrete and is intended to enhance the durability of the weight bearing articulating spacer. Indeed, the spacer has maintained its shape and remained intact for the 13 months it was implanted.
Articulating spacers as a modified one-stage revision after prosthetic joint replacement have been suggested in the past [8]. Nonetheless, the long-term implications of a partial joint replacement with bone cement are virtually unknown. Most articulating spacers function as a temporary total joint replacement, leaving joint cartilage uninvolved. The rough surface of bone cement is much higher compared to a polished metal surface, such as in hip or shoulder hemiarthroplasty heads. Hence, we expected that the cartilage wear will be greater in the cement-cartilage interface. After reevaluation with new CT scans, The surgical options included total talus replacement alone, a combination of peritalar fusion and replacement (i.e., subtalar fixation to a total talus and a total ankle replacement), or talectomy and tibiocalcaneal fusion if other options fail.8 These surgical options are not valid in case of any residual infection. 13 months after the cement spacer implantation, the cartilage wear was indeed great enough in the cement-cartilage interface to require a more permanent solution. We integrated a similar solution to the one presented by broughton et al. and used personalized titanium coated 3D printed talar implant.1 The implant was made to match the patient’s original anatomy and the polyethylene tibial platfond part of the Cadence total ankle system (Supplementary video 1,2,3,4).
The described personalized cement spacer can be utilized to replace any short bone or irregular bone with unsalvageable infection or tumor. Tarsal bones, carpal bones, vertebrae and other short and irregular bones can be replicated with 3D technology. It is paramount to discuss the associated costs with access to 3D printing technology. There are yearly or one-time investments such as buying a 3D printer or software licenses, and the per case associated costs such as workhour invested and printing materials. This case required about 16 work hours of our 3D lab designer and about 2 hours of consultation with the surgeon. The total printing costs for the cement spacer mold were 150$ for both trial and sterilizable outsourced Nylon PA12 material. The one-time cost associated with purchasing a 3D printer for trials is about 3,500$, the d2p software lifetime license with a cost of about 11,200 $ and license for Fusion 360 CAD software at about 500$ a year. The titanium coated personalized implant required CT images of the cadence total ankle system, 2 workhours of the lab designer. Printing costs were about 2,250$. Costs may vary depending on multiple factors such as region, specific deals with individual hospitals etc. We intend to continue our research on the outcome of patient-specific 3D printed molds as individualized treatment for complicated cases.
As in this rare case of isolated talus osteomyelitis, patient specific 3D-printed antibiotic-impregnated bone cement spacer and 3D printed titanium coated permanent implant are a step forward towards personalized treatments for complex osteomyelitis cases and prosthetic joint infections.


