Impact Factor : 0.548
- NLM ID: 101723284
- OCoLC: 999826537
- LCCN: 2017202541
Srinivasa rao pedada1,2, K Jagadeesh3, K Ganesh kadiyala3, D B N Suresh varma3, Nagendra sastry yarla4, Gururaja Rangaiah5* and P S Brahmanandam5
Received: September 12, 2022; Published: September 21, 2022
*Corresponding author: Gururaja Rangaiah, Department of Biochemistry, Sarada Vilas College, Mysore-570004, Karnataka, India
DOI: 10.26717/BJSTR.2022.46.007324
Secretory PLA2 (sPLA2) is an important enzyme that plays a crucial role in various inflammatory diseases, including cancer, and its inhibitors have been developed as preventive or therapeutic agents. In the present study, A novel hybrid series of 1, 2, 3 - triazole containing 1,3,4-oxadiazole hybrids 9(a-0) is synthesized and evaluated for their GIIA sPLA2 inhibitory activities. All compounds 9(a- o) showed significant sPLA2 inhibition activities. The compound 9o has shown a considerable inhibition activity (8.51±1.3μM) towards sPLA2. Further, the compound 9o was screened for anti-inflammatory activity in rats using the carrageenan-induced paw edema model and found promising results. In vitro anticancer studies of 9o were performed on various cancer cell lines, including MCF-7 breast, MIA PaCa-2 pancreatic, and DU145 prostate. Compound 9o inhibited the growth of breast MCF-7 at IC50 of 16.59±0.56 μM and prostate DU145 cancer cell lines at IC50 of 22.83±1.81 μM. The synthesized compounds exhibited substantial anticancer activities. The in vitro, in vivo, and silico results are encouraging and warrant detailed preclinical studies to develop GIIA sPLA2- inhibitory compound 9o as a novel therapeutic agent for various inflammatory disorders and cancer types.
Keywords: 1, 2, 3 – Triazole; Giia Spla2; Anti-Cancerous; Breast Cancer (Mcf-7 Cell Line); Prostate Cancer (Du145 Cell Line); Spla2 Inhibition; Anti-Inflammatory Activity; In Silico Studies
Phospholipase A2s (PLA2s) are an essential group of enzymes that cleave phospholipids specifically at the sn-2 position to liberate free fatty acids, primarily arachidonic acid (AA) and lysophospholipids (LPLs)1-3. Inhibition of PLA2 prevents the liberation of AA and LPLs and the production of eicosanoids. Hence, PLA2s have been considered good therapeutic targets than cyclooxygenase-2 and lipoxygenases (downstream enzymes) [1- 3]. The secreted PLA2S (13-55 kDa) is an essential group of PLA2 enzymes and is categorized into several groups4. Among all groups of the sPLA2, group IIA sPLA2 (GIIA sPLA2) is predominantly distributed. It plays a vital role in the pathophysiology of various inflammatory diseases, including arthritis, inflammatory bowel disease, atherosclerosis, psoriasis, and several cancers [1-4]. In this background, researchers have considered GIIA sPLA2 a promising therapeutic target in inflammatory and oncologic diseases. Realizing the role of sPLA2 in inflammatory and oncologic diseases, considerable efforts are being made by several investigators for the discovery and development of sPLA2 inhibitors as therapeutic or preventive agents2. Several sPLA2 inhibitors have been developed, and some of the sPLA2 inhibitors are currently under clinical trials for various inflammatory and oncologic diseases [2- 4]. Several investigators have reported triazoles and isoxazoles to possess multiple biological activities, including PLA2-inhibitory activities [5-9]. During the last several decades, various derivatives of triazoles and isoxazoles have been individually developed as GIIA sPLA2-inhibitory activities. Both triazole and isoxazoles are attractive chemical scaffolds for developing novel sPLA2 inhibitors. Considering this background, the present study aimed to synthesize triazole containing isoxazole 9(a-o) with potent sPLA2- inhibitory activity. Among all compounds, 9o showed substantial sPLA2-inhibitory activity. Further in vivo anti-inflammatory studies of 9o were performed in rats using an inflammogen (carrageenan)-induced paw edema model6. In vitro anticancer studies using sPLA2-inhibitory 9o were also performed on MCF-7 breast carcinoma and DU145 prostate cancer cell lines since sPLA2 exhibited a pro-carcinogenic role in these types of cancers [10-13].
General Information: All raw materials were purchased from Sigma-Aldrich and S.D. Fine Chem Ltd., India, and used for the experiments as such. 1H NMR and 13C NMR spectra were recorded in DMSO-d6, the chemical shifts are reported in δ ppm relative to tetramethyl silane. The 100 Perkin-Elmer FT-IR spectrometer was used for FTIR spectra [14-15]. Mass spectra were recorded on Agilent 1200 Series LC/MSD VL system. Buchi (B-545) melting point apparatus was used for determination of melting points. Melting points are uncorrected.
Chemicals and Reagents: Phosphate buffered saline (PBS), RPMI 1640, fetal calf serum (FCS), penicillin and streptomycin were purchased from Gibco BRL (Life Technologies) Rockville, MD, USA). MTT [3-(4,5 dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide], doxorubicin, λ-carrageenan and ursolic acid was purchased from Sigma-Aldrich Chemical Company, (St. Louis, MO, USA).
In vitro Secretory Phospholipase A2 Assay: sPLA2 assay was performed using type IIA sPLA2 enzyme activity kit (Cayman Chemical, Ann Arbor, Michigan, USA) according to the manufacturer’s instructions. Urosolic acid was used as a positive control [16-18]. The percent inhibition of enzyme activity was calculated using the following formula:
% inhibition = [(Absorbance of control – Absorbance of test/ Absorbance of control) ×100].
In vivo Anti-Inflammatory Activity by Carrageenan Induced Rat Paw Edema Model: Carrageenan-induced paw edema model is the most widely used for the evaluation of anti-inflammatory activity [16,19]. Animal experiments were conducted according to CPCSEA guidelines. Animal experimental protocol was approved by institutional animal ethical committee (IAEC no. GU/GIS/IAEC/ PROTOCOL NO.03/2013. The animals were divided into three groups (n = 6). Group I served as control and received NaCl saline at 1 ml/kg body weight (b.w) per os (p.o). Group II served as standard, received ursolic acid at a dose of 5 mg/kg and 10 mg/kg b.w, p.o. and group III served as test and received synthesized compound(s) at a dose of 5 mg/kg 10 mg/kg b.w, p.o, respectively. The animals were treated with synthesized compounds and ursolic acid before 1 h to inject (subcutaneous) 0.1 ml of 1% λ-carrageenan solution with saline into sub-plantar region of left hind paw of each rat. The right hind paw of same rat was treated with 0.1 ml of saline alone in manner as control. Before induction of edema, the dorsiventral thickness of both the paws of each was measured using mercury displaced glass paleothermometer and also the measurements were taken at 4th hour after carrageenan injection since edema is high at this stage. The results are expressed in percent edema inhibition. Ursolic acid was used as a positive control.
Cancer Cell Lines: MCF-7 breast cancer cell line was procured from National Cancer Institute (Frederick, MD, USA). Human prostate carcinoma cell line DU145 was obtained from American Type Culture Collection (Manassas, VA, USA). These cancer cells were maintained in RPMI medium supplemented with 10%FCS, 100 units penicillin and 100 μg of streptomycin per ml of medium. The cells were maintained in 95% humidity with CO2 at 37 0C temperature.
Cell Proliferation by MTT Assay: MTT assay was performed to assess the antiproliferative activity of compounds [20]. Cancer cells were treated with isolated compound at various concentrations for 48 h. 20 μl of freshly prepared MTT reagent was added to cells and incubated at 37 °C for 2 h. The supernatant growth medium was removed and replaced with DMSO (100 μl) to dissolve the formazan crystals. The optical density (absorbance) was read at 570 nm with reference wavelength of 620 nm.
In silico studies Drug like properties: The rule was formulated by Christopher A. Lipinski in 1997 [21]. The rule describes the molecular properties important for a drug’s pharmacokinetics in the human body, including their absorption, distribution, metabolism and excretion (ADME). The rule is important for drug development where a pharmacologically active lead structure is optimized stepwise for increased activity and selectivity, as well as druglike properties. Molinspiration tool was used in present study to demonstrate the druglike properties of compounds [22].
Molecular Docking Studies: All X-ray crystal structures of proteins were obtained from Protein Data Bank. human group IIA sPLA2 (PDB ID: 1DB5) was used in docking studies. Cocrystalized ligands and water molecules were removed from target protein using Argus lab. Ligands are prepared using chemoffice (Cambridge, UK). Energy minimization was done using molecular mechanics. Energy minimized ligands and receptors were used for docking studies using GEMDOCK (Generic Evolutionary Method for molecular DOCKING) software. PyMol was used for better visualization of interactions [23-25].
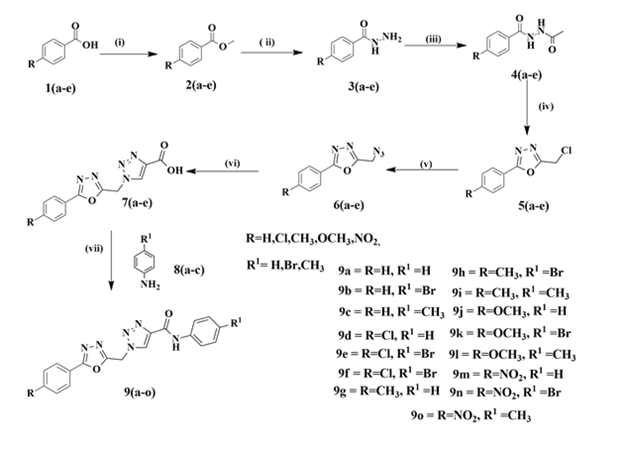
General Procedure for Synthesis 1,2,3-Triazole Containing 1,3,4-Oxadiazole: A series of novel 1,2,3-triazole containing 1,3,4-oxadiazole derivatives were prepared. Overall, fifteen molecules 9(a-o) were synthesized in nine steps using the linear synthesis approach. In synthesis, 4-substituted aromatic acids 1(a-e)were converted into corresponding esters 2(a-e), which were treated with hydrazine hydrate to obtain p-substituted phenyl hydrazides 3(a-e) compounds.The compound, 3(a-e) were further treated with chloroacetyl chloride in EtOAc under reflux conditions to afford compounds 4(a-e), which were cyclized in the presence of POCl3 at 80 °C to the corresponding 2-chloromethyl- 5-(p-substituted-phenyl)-1,3,4-oxadiazoles 5(a-e). The cyclized intermediate 5(a-e) reacted with sodium azide in DMF to obtain 6(a-e), which further reacted with propargylic acid to afford key intermediate 7(a-e). Finally, the target compounds 9(a-o) were synthesizedbycoupling p-substituted amine 8(a-c) with 7(a-e) using HATU as an amide coupling reagent in good yields.
General Procedure for the Synthesis of 9(a-o): To a solution of 1,2,3- triazole acid 7(a-e) (1.8 mmol) in DMF (5.0 ml) was added DIPEA (4.6 mmol) and HATU (2.2 mmol) under nitrogen and stirred for 45 min at room temperature. To the reaction mixture, 4-substituted amine 8(a-c) (1.8 mmol) was added, and the reaction mixture was stirred for another one hour at RT. After completion of the reaction, the reaction mixture was poured into ice water, and stirred for 30 min. The solid obtained was filtered, washed with water and dried to get desired compounds as solid (55–70% yield). The characterization data of target compounds 9(a-o) are given below (Scheme 1).
Synthesis of 1,2,3-Triazoles Containing 1,3,4-Oxadiazole Derivatives 9(a-o): N-Phenyl-1-((5-phenyl-1,3,4-oxadiazol-2- yl)methyl)-1H-1,2,3-triazole-4-carboxamide (9a): Yield: 63 %; Off white solid; mp: 187-189 ˚C; 1H NMR (400 MHz, DMSO-d6) δ:10.50 (s, 1H), 8.94 (s,1H), 7.99 (d, J=6.8Hz, 2H), 7.82 (d, J=8.0Hz, 2H), 7.66–7.60 (m, 3H), 7.36 (t, J=8.0Hz, 2H), 7.10 (t, J=7.6Hz, 1H), 6.24 (s, 2H); 13C NMR (100 MHz, DMSO-d6) δ:163.3, 161.6, 158.5, 143.7, 138.9, 132.8, 129.9, 129.0, 128.9, 127.1, 124.3, 123.3, 120.9, 44.7; IR (KBr) cm-1 :3333, 3132, 3063, 2993, 2368, 2337, 1604, 1519, 1388, 1211, 1072, 871; MS (ESI) m/z [M+H]+ 347.1.N-(4- Bromophenyl)-1-((5-phenyl-1,3,4-oxadiazol-2-yl)methyl)-1H- 1,2,3-triazole-4-carboxamide (9b): Yield: 66 %; Off white solid; mp: 245-248 ˚C; 1H NMR (400 MHz, DMSO-d6) δ:10.68 (s, 1H), 8.96 (s, 1H), 7.99 (d, J=6.8Hz, 2H), 7.82 (d, J=8.4Hz, 2H), 7.65-7.59 (m, 3H), 7.53 (d, J=8.4Hz, 2H), 6.24 (s, 2H); 13C NMR (100 MHz, DMSO-d6) δ:165.3, 161.6, 158.6, 143.4, 138.3, 132.7, 131.9, 129.5, 129.1, 127.1, 123.3, 122.8, 116.0, 44.7; IR (KBr) cm-1 :3317, 3140, 3063, 2985, 2955, 2368, 1589, 1450, 1303, 1211, 1018, 964, 887 ; MS (ESI) m/z [M+H]+ 427.0. 1-((5-Phenyl-1,3,4-oxadiazol-2-yl) methyl)-N-p-tolyl-1H-1,2,3-triazole-4-carboxamide (9c): Yield: 65 %; Off white solid; mp: 220-222 ˚C; 1H NMR (400 MHz, DMSO-d6) δ:10.40 (s, 1H), 8.92 (s, 1H), 7.99 (d, J=7.2Hz, 2H), 7.70 (d, J=8.4Hz, 2H), 7.65–7.59 (m, 3H), 7.14 (d, J=8.0Hz, 2H), 6.23 (s, 2H), 2.27 (s, 3H); 13C NMR (100 MHz, DMSO-d6) δ:165.3, 161.7, 158.3, 143.7, 136.3, 133.3, 132.7, 129.9, 129.4, 128.8, 127.1, 123.3, 120.9, 44.7, 20.9; IR (KBr) cm-1 :3379, 3140, 3063, 2924, 1597, 1450, 1404, 1296, 1211, 1041, 810; MS (ESI) m/z [M+H]+ 361.1.
1-((5-(4-chlorophenyl)-1,3,4-oxadiazol-2-yl)methyl)-Nphenyl- 1H-1,2,3-triazole-4-carboxamide (9d): Yield: 65 %; Light brown solid; mp: 268-270 ˚C; 1H NMR (400 MHz, DMSO-d6) δ:10.49 (s, 1H), 8.94 (s, 1H), 8.00 (d, J=8.4Hz, 2H), 7.82 (d, J=8.0Hz, 2H), 7.69 (t, J=8.4Hz, 2H), 7.34 (t, J=8.0Hz, 2H), 7.10 (t, J=7.6Hz, 1H) 6.24 (s, 2H); 13C NMR (100 MHz, DMSO-d6) δ:164.6, 161.8, 158.5, 143.6, 138.8, 137.5, 130.1, 129.0, 128.9, 124.3, 122.2, 120.9, 44.7; IR (KBr) cm-1 :3371, 3325, 3101, 2985, 2368, 2337, 1658, 1604, 1519, 1489, 1419, 1311, 1249, 1095, 1010, 964, 910, 841; MS (ESI) m/z [M+H]+ 381.1.
Scheme 1: Reagents and conditions:
(i) MeOH, H2SO4, reflux
(ii) NH2NH2H2O, MeOH
(iii) ClCH2COCl, EtOAc, reflux
(iv) POCl3, reflux
(v) NaN3, DMF, 60-65 °C
(vi) Propargyl acid, toluene, 60-65 °C
(vii) HATU, DIPEA, DMF, rt.
N-(4-Bromophenyl)-1-((5-(4-chlorophenyl)-1,3,4-oxadiazol-2- yl)methyl)-1H-1,2,3-triazole-4-carboxamide (9e): Yield: 65 %; Light brown solid; mp: 245-248 ˚C; 1H NMR (400 MHz, DMSO-d6) δ:10.68 (s, 1H), 8.96 (s, 1H), 8.00 (d, J=7.6Hz, 2H), 7.82 (d, J=8.8Hz, 2H), 7.69 (t, J=8.0Hz, 2H), 7.53 (d, J=8.4Hz, 2H), 6.23 (s, 2H); 13C NMR (100 MHz, DMSO-d6) δ:164.6, 161.8, 158.6, 143.4, 138.3, 137.5, 131.8, 130.1, 129.0, 128.8, 122.8, 122.2, 116.0, 44.7; IR (KBr) cm-1 : 3325, 3101, 2985, 2368, 2337, 1658, 1558, 1489, 1311, 1249, 1095, 964, 879; MS (ESI) m/z [M+H]+ 461.1. 1-((5-(4-Chlorophenyl)-1,3,4- oxadiazol-2-yl)methyl)-N-p-tolyl-1H-1,2,3-triazole-4-carboxamide (9f): Yield: 70 %; Light brown solid; mp: [23] 208-210 ˚C; 1H NMR (400 MHz, DMSO-d6) δ:10.40 (s, 1H), 8.91 (s, 1H), 8.00 (d, J=8.4Hz, 2H), 7.70 (m, 4H), 7.14 (d, J=8.4Hz, 2H), 6.23 (s, 2H), 2.28 (s, 3H); 13C NMR (100 MHz, DMSO-d6) δ:164.6, 161.8, 158.3, 143.7, 137.5, 136.3, 133.3, 130.1, 129.4, 128.9, 128.8, 122.2, 120.8, 44.6, 20.9; IR (KBr) cm-1 :3364, 3101, 2924, 2368, 1658, 1519, 1481, 1411, 1303, 1249, 1095, 1010, 879, 841; MS (ESI) m/z [M+H]+ 395.1. N-Phenyl-1-((5-p-tolyl-1,3,4-oxadiazol-2-yl)methyl)-1H-1,2,3- triazole-4-carboxamide (9g): Yield: 67 %; Light brown solid; mp: 210-212 ˚C; 1H NMR (400 MHz, DMSO-d6) δ:10.50 (s, 1H), 8.94 (s, 1H), 7.88 (d, J=7.6Hz, 2H), 7.82 (d, J=8.0Hz, 2H), 7.42 (d, J=8.0Hz, 2H), 7.34 (t, J=7.6Hz, 2H), 7.10 (t, J=6.8Hz, 1H), 6.22 (s, 2H), 2.39 (s, 3H); 13C NMR (100 MHz, DMSO-d6) δ:165.4, 161.4, 158.5, 143.6, 143.0, 138.9, 130.5, 129.3, 129.0, 128.9, 127.0, 124.3, 120.9, 120.6, 44.7, 21.5; IR (KBr) cm-1 :3904, 3317, 3140, 2360, 1666, 1597, 1558, 1504, 1311, 1242, 1180, 1087, 1041, 879, 825; MS (ESI) m/z [M+H]+ 361.1. N-(4-Bromophenyl)-1-((5-p-tolyl-1,3,4-oxadiazol- 2-yl)methyl)-1H-1,2,3-triazole-4-carboxamide (9h): Yield: 72 %; Light brown solid; mp: 233-236 ˚C; 1H NMR (400 MHz, DMSO-d6) δ:10.67 (s, 1H), 8.96 (s, 1H), 7.87 (d, J=7.6Hz, 2H), 7.82 (d, J=8.4Hz, 2H), 7.53 (d, J=8.0Hz, 2H), 7.42 (d, J=7.2Hz, 2H), 6.22 (s, 2H), 2.39 (s, 3H); 13C NMR (100 MHz, DMSO-d6) δ:165.4, 161.3, 158.6, 143.4, 143.0, 138.3, 131.8, 130.5, 129.1, 127.0, 122.8, 120.6, 116.1, 44.7, 21.5; IR (KBr) cm-1 :3888, 3317, 3140, 2360, 1674, 1589, 1458, 1396, 1303, 1242, 1080, 1010, 825; MS (ESI) m/z [M+H]+ 441.1.N-p-tolyl-1-((5-p-tolyl-1,3,4-oxadiazol-2-yl)methyl)-1H- 1,2,3-triazole-4-carboxamide (9i): Yield: 66 %; Light brown solid; mp: 210-213˚C; 1H NMR (400 MHz, DMSO-d6) δ:10.40 (s, 1H), 8.91 (s, 1H), 7.87 (s, 2H), 7.69 (s, 2H), 7.42 (s, 2H), 7.14 (s, 2H), 6.21 (s, 2H), 2.40 (s, 3H), 2.27 (s, 3H); 13C NMR (100 MHz, DMSO-d6) δ:165.4, 161.4, 158.3, 143.7, 143.0, 136.3, 133.3, 130.5, 129.4, 128.8, 127.0, 120.8, 120.6, 44.7, 21.5, 20.9; IR (KBr) cm-1 :3325, 3140, 2924, 2360, 1666, 1558, 1458, 1411, 1334, 1242, 1118, 1080, 1041, 817; MS (ESI) m/z [M+H]+ 375.1.
1-((5-(4-Methoxyphenyl)-1,3,4-oxadiazol-2-yl)methyl)-Nphenyl- 1H-1,2,3-triazole-4-carboxamide (9j): Yield: 65 %; Light brown solid; mp: 198-200 ˚C; 1H NMR (400 MHz, DMSO-d6) δ:10.50 (s, 1H), 8.93 (s, 1H), 7.93 (d, J=8.4Hz, 2H), 7.82 (d, J=8.0Hz, 2H), 7.34 (t, J=8.0Hz, 2H), 7.16–7.10 (m, 3H), 6.20 (s, 2H), 3.85 (s, 3H); 13C NMR (100 MHz, DMSO-d6) δ:165.3, 162.7, 161.0, 158.5, 143.6, 138.8, 129.0, 128.9, 128.8, 124.3, 120.9, 115.6, 115.4, 55.9, 44.7; IR (KBr) cm-1 :3333, 3140, 3070, 2978, 2839, 2569, 2206, 1666, 1604, 1504, 1381, 1303, 1265, 1180, 1026, 841; MS (ESI) m/z [M+H]+ 377.0. N-(4-Bromophenyl)-1-((5-(4-methoxyphenyl)- 1,3,4-oxadiazol-2-yl)methyl)-1H-1,2,3-triazole-4-carboxamide (9k): Yield: 70 %; Light brown solid; mp: 236-238 ˚C; 1H NMR (400 MHz, DMSO-d6) δ:10.67 (s, 1H), 8.95 (s, 1H), 7.92 (d, J=8.8Hz, 2H), 7.82 (d, J=8.8Hz, 2H), 7.53 (d, J=8.4Hz, 2H), 7.14 (d, J=8.8Hz, 2H), 6.20 (s, 2H), 3.85 (s, 3H); 13C NMR (100 MHz, DMSO-d6) δ:165.3, 162.7, 161.0, 158.6, 143.4, 138.3, 131.8, 129.0, 128.9, 122.8, 116.0, 115.6, 115.4, 55.9, 44.7; IR (KBr) cm-1 :3325, 3124, 2970, 2839, 2368, 2052, 1905, 1674, 1597, 1504, 1427, 1396, 1303, 1257, 1180, 1080, 956; MS (ESI) m/z [M+H]+ 457.0.
1-((5-(4-Methoxyphenyl)-1,3,4-oxadiazol-2-yl)methyl)-Np- tolyl-1H-1,2,3-triazole-4-carboxamide (9l): Yield: 65 %; Light brown solid; mp: 225-227 ˚C; 1H NMR (400 MHz, DMSO-d6) δ:10.40 (s, 1H), 8.91 (s, 1H), 7.92 (d, J=8.8Hz, 2H), 7.70 (d, J=8.0Hz, 2H), 7.15–7.13 (m, 4H), 6.20 (s, 2H), 3.85 (s, 3H), 2.27 (s, 3H); 13C NMR (100 MHz, DMSO-d6) δ:165.3, 162.7, 161.1, 158.3, 143.7, 136.3, 133.3, 129.4, 128.9, 128.7, 120.9, 115.6, 115.4, 55.9, 44.7, 20.9; IR (KBr) cm-1 :3333, 3140, 1666, 1604, 1558, 1458, 1334, 1265, 1180, 1087, 1026, 956, 879; MS (ESI) m/z [M+H]+ 391.1.
1-((5-(4-Nitrophenyl)-1,3,4-oxadiazol-2-yl)methyl)-N-phenyl- 1H-1,2,3-triazole-4-carboxamide (9m): Yield: 65 %; Light yellow solid; mp: 207-209 ˚C; 1H NMR (400 MHz, DMSO-d6) δ:10.50 (s, 1H), 8.95 (s, 1H), 8.43 (d, J=8.8Hz, 2H), 8.25 (d, J=8.8Hz, 2H), 7.82 (d, J=8.4Hz, 2H), 7.34 (t, J=7.6Hz, 2H), 7.10 (t, J=7.2Hz, 1H), 6.20 (s, 2H); 13C NMR (100 MHz, DMSO-d6) δ:164.0, 162.5, 158.4, 149.8, 143.6, 138.8, 129.0, 128.9, 128.8, 128.5, 125.1, 125.0, 124.3, 120.9, 120.7, 44.7; IR (KBr) cm-1 :3109, 2368, 1658, 1604, 1558, 1527, 1442, 1342, 1303, 1249, 1111, 1080, 1049, 910; MS (ESI) m/z [M-H]- 390.1. N-(4-Bromophenyl)-1-((5-(4-nitrophenyl)-1,3,4- oxadiazol-2-yl)methyl)-1H-1,2,3-triazole-4-carboxamide (9n): Yield: 55 %; Light yellow solid; mp: 228-230 ˚C; 1H NMR (400 MHz, DMSO-d6) δ:10.68 (s, 1H), 8.96 (s, 1H), 8.43 (d, J=8.4Hz, 2H), 8.25 (d, J=8.8Hz, 2H), 7.82 (d, J=8.4Hz, 2H), 7.53 (d, J=8.8Hz, 2H), 6.28 (s, 2H); 13C NMR (100 MHz, DMSO-d6) δ:164.0, 162.5, 158.5, 149.8, 143.4, 138.3, 131.8, 131.8, 129.1, 128.8, 128.6, 128.5, 125.1, 125.0, 122.3, 116.0, 44.7; IR (KBr) cm-1 :3109, 2368, 1658, 1589, 1519, 1458, 1396, 1342, 1303, 1242, 1180, 1080, 1010, 856; MS (ESI) m/z [M-H]- 470.0. 1-((5-(4-Bitrophenyl)-1,3,4-oxadiazol-2- yl)methyl)-N-p-tolyl-1H-1,2,3-triazole-4-carboxamide (9o): Yield: 58 %; Light yellow solid; mp: 220-222 ˚C; 1H NMR (400 MHz, DMSO-d6) δ:10.41 (s, 1H), 8.92 (s, 1H), 8.43 (d, J=8.8Hz, 2H), 8.25 (d, J=8.8Hz, 2H), 7.70 (d, J=8.8Hz, 2H), 7.14 (d, J=8.0Hz, 2H), 6.27 (s, 2H), 2.27 (s, 3H); 13C NMR (100 MHz, DMSO-d6) δ:164.0, 162.5, 158.2, 155.8, 149.8, 143.7, 136.3, 133.3, 129.4, 128.8, 128.5, 128.3, 125.1, 125.0, 120.8, 120.7, 44.7, 20.9; IR (KBr) cm-1 :3371, 3109, 2924, 2368, 1658, 1604, 1558, 1527, 1458, 1342, 1249, 1111, 1087, 1041, 856; MS (ESI) m/z [M-H]-404.1.
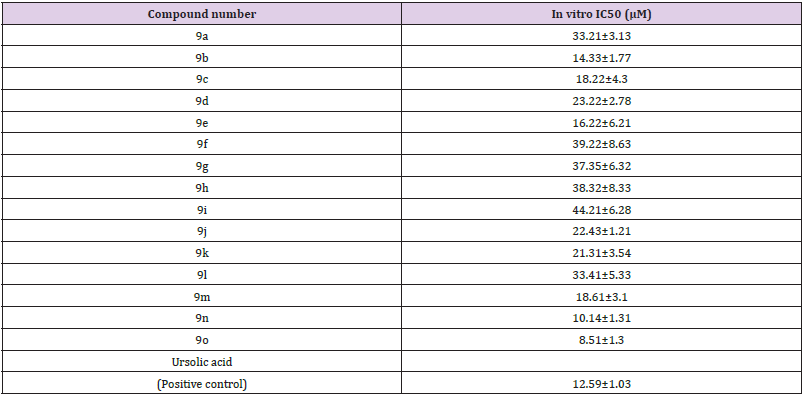
sPLA2 Inhibitory Studies of Novel 1,2,3-Triazole Containing with 1,3,4-Oxadiazole: A novel series of fifteen compounds 1,2,3-triazole containing 1,3,4-oxadiazole were tested for sPLA2 inhibition and binding affinity with crystal structure of sPLA2 (Table 1). In vitro PLA2 assay of compounds were performed using Cayman Chemical PLA2 assay kit. All compounds showed significant PLA2 inhibitory activity. Among all the tested compounds, 1-((5-(4-nitrophenyl)-1,3,4-oxadiazol-2-yl)methyl)-N-p-tolyl1H-1,2,3-triazole-4-carboxamide (9o) exhibited significant PLA2 inhibitory activity with IC50 of 8.51±1.3 μM, whereas Ursolic acid inhibited with IC50 of 12.59±1.03 μM (Figure 1). In vitro results are correlated with in silico studies. Compounds showed high binding affinity on PLA2 protein.
Table 1: GIIA sPLA2 inhibition activities of 1,2,3-trizole containing with 1,3,4-oxadiazole derivatives 9 (a-o).
Note: The positive (+) and negative (-) sign show the band intensity.
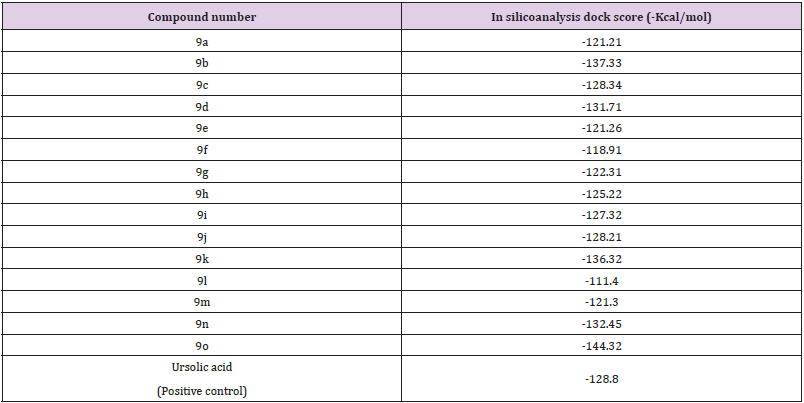
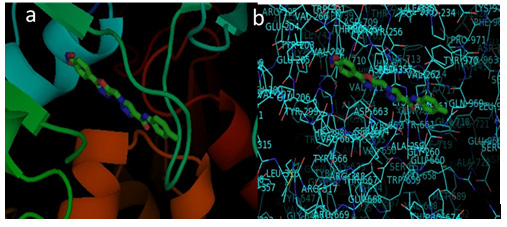
Molecular Docking Studies: Compound 9o binds with PLA2 crystal protein with binding energy of -144.32 Kal/mol(Table 2). Strong binding interactions with amino acids of PLA2 protein (Figure 2) may cause its inhibition activity whereas standard Ursolic acid bind with sPLA2 crystal protein with binding energy 12.59±1.03 Kal/mol.
Table 2: Docking score of compounds 9(a-o) on human GIIA sPLA2 crystal protein.
Note: The positive (+) and negative (-) sign show the band intensity.
Figure 2: Molecular docking studies of 9o on human GIIA sPLA2 crystal protein
(a) Binding orientation
(b) Molecular interactions of 9o on human GIIA sPLA2 crystal protein.
Anti-Proliferative Activities of Compound 9o: Antiproliferative effect of compound 9o was evaluated on breast cancer MCF-7 and prostate cancer cell lines DU145 by MTT assay. Compound 9o inhibited growth of breast MCF-7 at IC50 of 16.59±0.56 μM and prostate DU145 cancer cell lines at IC50 of 22.83±1.81 μM. Results are compared with Doxorubcine (positive control) showed anti-proliferative activity with breast cancer cell line MCF-7 (IC50 = 8.02±0.69) and prostate cancer cell lines DU145 (IC50 = 4.75±0.35 μM) (Figure 3).
Anti-Inflammatory Activities of Compound 9o: In vivo antiinflammatory activity of 9o (5 mg/kg, body weight) was tested group of evaluated using carrageenan-induced rat paw edema model and indomethacin (positive control) and untreated (vehicle control) [25]. 9o (5 mg/kg, body weight) inhibited 76.56±3.52% and 79.47±6.08% paw edema at 3rd and 4th hours respectivelyafter carrageenan-injection (Figure 4). The results were compared with that of Indomethacin.
Drug-Likeness Property of Compounds: In addition to sPLA2 inhibitory studies, the evaluation of drug-likeness properties of compounds according to Lipinski’s rule of five is done which states that, in general, an orally active drug has not more than 5 hydrogen bond donors (OH and NH groups), not more than 10 hydrogen bond acceptors (notably N and O), molecular weight under 500 g/mol, partition coefficient logP less than 5. Almost all compounds 9(a-o) obey lipinsky`s rule and is of drug gable nature (Table 3).
Oxadiazole and triazoles are important scaffolds for developing PLA2 inhibitors (Mouchliset al., 2016; Mouchliset al., 2016; Kai et al., 2013). In the above background, in the present study, we have synthesized a novel series of 1, 2, and 3-triazole containing 1, 3, 4-oxadiazole amide derivatives 9(a-o) with potent sPLA2- inhibitory activity. Among all fifteen compounds, 9o showed substantial sPLA2-inhibitory activity. The earlier basic and clinical studies demonstrated the role of PLA2 in cancer and inflammation, and PLA2 inhibitors can be good therapeutic agents for treating inflammatory-mediated diseases and cancer. In present study, in vivo anti-inflammatory studies of 1-((5-(4-nitrophenyl)- 1,3,4-oxadiazol-2-yl)methyl)-N-p-tolyl-1H-1,2,3-triazole-4- carboxamide(9o) were performed in rats using inflammogen (carrageenan)-induced paw edema model and found excellent activity(fig) by taking indomethacin as standard. In vitro anticancer studies using sPLA2-inhibitory 9o were also performed on MCF- 7 breast carcinoma and DU145 prostate cancer cell lines since sPLA2 exhibited a pro-carcinogenic role in these types of cancers and found significant activity(fig). The docking studies support the sPLA2 inhibition activity and anticancer studies of compound 9o (Table). The compound 9(a-o) has shown an excellent binding affinity toward the human GIIA sPLA2 crystal protein (table). But the docking score of 9o supports the anti-inflammatory and anticancerous activity. Present studies warranted further studies on 9o to develop it as an anti-inflammatory and anticancer agent.
A novel new hybrid series of molecules, i.e., 1, 2, 3-triazole containing 1, 3, and 4-oxadiazole compounds, showed significant PLA2 inhibitory activity based on in vitro and in silico results. among tested compounds,1-((5-(4-nitrophenyl)-1,3,4-oxadiazol-2- yl)methyl)-N-p-tolyl-1H-1,2,3-triazole-4-carboxamide 9o showed substantial PLA2 inhibitory activity. In further studies, 9o showed anti-inflammatory activity in rats and anticancer activity on breast MCF-7 and prostate DU145 cancer cells. Results are encouraging and require further studies to develop 9o as an anti-inflammatory (76.56±3.52% and 79.47±6.08% paw edema at 3rd and 4th hours, respectively)and anticancer drug[anti-proliferative activity with breast cancer cell line MCF-7 (IC50 = 8.02±0.69μM) and prostate cancer cell lines DU145 (IC50 = 4.75±0.35 μM)]. Compound 9o has shown a good docking score (-144.32 Kcal/mole) compared to the remaining compound 9(a-n). In molecular docking studies, the binding orientation and molecular interactions confirm the binding affinity of 9o towards the GIIA sPLA2 protein (Fig). Moreover, compounds 9(a-o) obey Lipinski`s rule of 5 and can be druggable.
Dr. KJ, Dr. KKG, Mr. DBN Suresh Varma, and Dr.PSB would also reciprocate the management of Shri Vishnu Engineering College for Women (A), Bhimavaram for providing infrastructure facilities, without which it would not have been possible to carry out this work.


