Impact Factor : 0.548
- NLM ID: 101723284
- OCoLC: 999826537
- LCCN: 2017202541
Siddiqui Gulnaz1, Bawa Amandeep S2, Paal E3 and Nava Victor E1*
Received: August 08, 2022; Published: August 18, 2022
*Corresponding author: Nava Victor E, Department of Pathology, Department of Veterans Affairs Medical Center, Washington DC, United States
DOI: 10.26717/BJSTR.2022.45.007250
Strongyloides hyperinfection commonly causes pulmonary symptoms due to increased larval migration and entails high mortality secondary to sepsis due to enteric bacteria or fungi gaining access to the bloodstream as a result of parasite-driven gastrointestinal mucosal barrier breakdown. Here we present a severe Strongyloidiasis infection in an 83-year old black man with advanced prostate carcinoma receiving steroids, who presented with normocytic anemia, eosinophilia and mild cough. To the best of our knowledge, the association of Strongyloidiasis hyperinfection and advanced prostatic carcinoma has only been reported once before in PubMed.
Keywords: Strongyloides; Hyperinfection Syndrome; Prostate Carcinoma; Metastasis; Lung Mass; Eosinophilia
We present a case of Strongyloides hyperinfection in a patient with prostate carcinoma, which to the best of our knowledge has only been previously reported once [1]. Strongyloides hyperinfection is an accelerated autoinfection with increased larval migration and wide organ dissemination typically presenting in immunocompromised hosts. Most clinical manifestations including pulmonary involvement are attributed to increased larval migration. In our patient, both advanced prostate cancer and the use of steroids contributed to impairing adequate immunity precipitating a severe Strongyloidiasis infection [2,3]. This is the second case of hyperinfection by Strongyloides reported in PubMED in a patient with advanced prostatic carcinoma, which was also lethal1, and probably facilitated by steroid-induced immunosuppression [4,5].
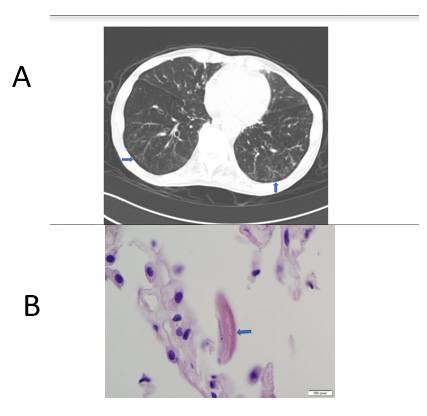
An 83-year-old African American male smoker with a history of prostatic carcinoma (Gleason score 8, diagnosed in 2002) status post-prostatectomy with positive margins was lost to follow up and consulted for urine incontinence in May 2018. Laboratory workup revealed markedly elevated PSA (389 ng/mL), rapidly reaching 957 ng/mL within one month. No hepatosplenomegaly, lymphadenopathy, or significant travel history was recorded. The patient did not reside in areas endemic for Strongyloides. Imaging studies, including computed tomography (CT) of the chest, revealed multiple sclerotic bone lesions concerning for metastatic prostate cancer, and treatment with a second dose of leuprolide and a 4-week course of bicalutamide was started in November 2018. Due to increasing PSA, a regimen of abiraterone and prednisone (5 mg PO daily) was initiated with partial compliance. Mild cough, normocytic anemia, and marked eosinophilia (10.2 K/cmm) developed prompting additional testing to rule out hematologic disorder and pulmonary eosinophilic syndrome.3 Repeated ova and parasites exams were negative. Chest radiography showed new bilateral lung patchy infiltrates, and chest CT showed a few areas of treein- bud nodularity (Figure 1A). Bronchoscopy with bronchoalveolar lavage (BAL) and lung biopsies were performed. BAL showed significant eosinophilia, and a transbronchial lung biopsy demonstrated eosinophilic infiltrates and few larvae consistent with Strongyloides species (Figure 1B). Bone marrow biopsy ruled out hematologic malignancy, and instead demonstrated metastatic prostatic carcinoma. Confirmatory serology for Strongyloides IgG was positive, allowing the diagnosis of hyperinfection syndrome, and the patient received Ivermectin (9 mg/day), resulting in rapid resolution of eosinophilia. Unfortunately, after discharge home following clinical improvement, Escherichia coli urosepsis developed, and he died in November 2019.
Figure 1:
A. Chest CT scan showing two areas of tree in-bud nodularity (arrows).
B. Hematoxylin & Esoin-stained lung section showing Strongyloides larvae (arrow) at 100X magnification.
Strongyloidiasis has a worldwide prevalence of 30 to 100 million cases and occurs predominantly in the tropics and subtropics, where it preferentially affects socioeconomically disadvantaged populations since it is mainly transmitted through contact with fecally contaminated soil. Strongyloides stercoralis filariform larvae penetrate intact skin or mucosae and migrate by various pathways gaining access to the small intestine, where they mature to adult forms. In the small intestine, only female adult Strongyloides reproduce parthenogenetically by laying eggs, which yield rhabditiform larvae that are passed in the stool or can cause autoinfection after molting into filariform larvae. Although the pulmonary-tracheal pathway is prominent in the life cycle, the filariform larvae can also randomly migrate through tissues during autoinfection. Factors predisposing to severe autoinfection and leading to hyperinfection are usually related to immunosuppression, which can be either primary or secondary to debilitating diseases or the iatrogenic effect of therapy, such as corticosteroids [3]. Interestingly, Strongyloides hyperinfection has been reported even after administration of low doses of steroids, which act not only as immunosuppressants but also as molting signals for the rhabditiform larvae to become infective filariform larvae. The mortality from Strongyloides hyperinfection can be as high as 60 to 86% due to sepsis secondary to enteric bacteria or fungi (residing on the cuticle of this nematode), which gain access to the bloodstream due to mucosal barrier breakdown provoked by the parasite. Fortunately, hyperinfection can be prevented with proper antihelminthic therapy. Unfortunately, the diagnosis is often very difficult to establish due to non-specific clinical manifestations and unawareness of pre-existing subclinical or oligosymptomatic lifelong Strongyloides infection. A low parasite burden may result in false-negative fecal tests. and be the source of hyperinfection decades after the primary infection. Therefore, a high level of suspicion is necessary for individuals with eosinophilia, especially in travelers or expatriates from endemic areas that present with abdominal or respiratory symptoms, accompanied by lung infiltrates on chest imaging (Loeffler syndrome). The identification of the parasite either in the stool or in biopsies (most commonly from lung or small bowel) allows a definitive diagnosis. However, due to the paucity of organisms, false negatives are common, and the diagnosis may require more sensitive immunologic or molecular methods 3-5 as exemplified by our case, where we excluded other pulmonary eosinophilic disorders. Regardless of the diagnostic approach, screening for Strongyloides is recommended for patients at risk of immunosuppression due to various debilitating diseases (viral infection, malnutrition, and malignancy) or exposure to immunosuppressive therapy.
Herein, a rare case of Strongyloidiasis hyperinfection syndrome associated with advanced prostatic carcinoma and corticosteroid treatment is presented. Sufficient compromise of the immune system could have resulted from the compound effect of malignancy and steroid treatment, allowing disseminated infection by Strongyloides, which was not suspected since the patient did not report gastrointestinal, respiratory or dermatologic symptoms in the past, and eosinophilia was absent before the acute episode. In summary, this is the second case of hyperinfection by Strongyloidiasis reported in PubMED in a patient with advanced prostatic carcinoma, which also had a fatal outcome1 probably facilitated by steroid-induced immunosuppression despite appropriate management. Acknowledgments, Financial Support, and Disclosures regarding real or perceived conflicts of interest are not applicable. The authors further confirm that the material is original, has not already been published, and has not and will not be submitted for publication elsewhere as long as it is under consideration by the SN Comprehensive Clinical Medicine.
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Not applicable.
Not applicable.
Not applicable.
Not applicable.
Not applicable.
Not applicable.
Gulnaz Siddiqui wrote the first draft and edited additional versions. Amandeep Bawa contributed to the clinical diagnosis and edited the manuscript. Edina Paal contributed to pathologic diagnosis and edited the manuscripts. Victor Nava designed the study, took micrographs, and edited the manuscript.


