Impact Factor : 0.548
- NLM ID: 101723284
- OCoLC: 999826537
- LCCN: 2017202541
Jesús Cuéllar Álvarez1*, Melva García Martínez2 and Yaquelin Gómez Morales3
Received: July 08, 2022; Published: July 18, 2022
*Corresponding author: Amy Throckmorton, Associate Professor, School of Biomedical Engineering, Science and Health Systems Drexel University, Bossone Research Enterprise Center 3141 Chestnut Street, Rm. 718 Philadelphia
DOI: 10.26717/BJSTR.2022.45.007168
There continues to be an unmet therapeutic need for an alternative treatment strategy for respiratory distress and lung disease in pediatric and adult patients. We are developing a portable cardiopulmonary support system that integrates an implantable oxygenator with a hybrid, dual-support, continuous-flow total artificial heart (TAH). The hybrid TAH has a centrifugal flow pump domain that is rotating about an axial flow pump. By attaching the hollow fiber bundle of the oxygenator to the base of the TAH, we aim to establish a new cardiopulmonary support technology that permits a patient to be ambulatory during usage and that can assist adult and pediatric patients with lung disease and cardiovascular compromise. In this study, we investigated the design and improvement of the blood flow pathway from the inflowto- outflow of four oxygenators using a mathematical model and computational fluid dynamics (CFD). Pressure loss and gas transport through diffusion were examined to assess oxygenator design efficiency. All of the oxygenator designs led to a resistancedriven pressure loss range of 25-35 mmHg for flow rates of 1-7L/min. All of the designs met requirements. The configuration having an outside-to-inside blood flow direction was found to have higher oxygen transport. Based on this advantageous flow directional determination, two designs (Model 1 and 3) were then integrated with the axial-flow impeller of the TAH into a CFD simulation. Flow rates of 1-7L/min and axial rotational speeds of 10,000-16,000 RPM were analyzed. Blood damage studies were performed, and Model 1 demonstrated the lowest potential for hemolysis. Future work will focus on developing and testing a physical prototype for integration into the new cardiopulmonary assist system that we are designing for pediatric and adult patients.
Keywords: Pump Oxygenator; Respiratory Failure; Right Ventricular Failure; Respiratory Assist Device; Right Ventricular Assist Device
Abbreviations:: ARDS: Acute Respiratory Distress Syndrome; TAH: Total Artificial Heart; CFD: Computational Fluid Dynamics; LVAD: Left Ventricular Assist Device; RVAD: Right Ventricular Assist Device; CAD: Computer Aided Design; DLC: Dual Lumen Cannula
Lung disease is the third leading cause of death that claims the lives of an estimated 150,000 Americans [1], and acute respiratory distress syndrome (ARDS) affects approximately 190,000 in the U.S. each year [2]. Despite advances in clinical diagnosis and pharmacotherapy, lung disease and ARDS impose high mortality (22%-55%) and long-term adverse effects on patient quality-of-life [2,3]. ECMO and protective ventilation strategies are the standards of care, and current forms of pharmacotherapy are limited and are typically contraindicated after several days of administration [4,5]. Studies have demonstrated advantages to the use of ECMO over conventional ventilation strategies for patients with ARDS [6]. Current ECMO and protective lung ventilation strategies, however, limit the potential treatment efficacy for the disease and generally result in longer hospital stays. ECMO is also limited by the complexity of the circuit, increased risk of hemorrhage and thrombosis, restricted patient ambulation, and an oxygenation threshold potential that is defined by perfusion levels [7-9]. Hence, there continues to be an unmet therapeutic need that requires a new solution or technology as an alternative treatment strategy for ARDS and lung disease in pediatric and adult patients. To address this unmet need, we are developing a novel implantable oxygenator that will be integrated into a hybrid total artificial heart (TAH) [10]. The continuous flow TAH is designed to provide left-sided systemic, right-sided pulmonary, and/or biventricular cardiac support. The TAH has a centrifugal flow pump domain that is rotating about an axial flow pump. By attaching the hollow fiber bundle of the oxygenator to the base of the TAH, we aim to establish a new cardiopulmonary support technology that permits a patient to be ambulatory during usage and that can assist adult and pediatric patients with lung disease, ARDS, cardiovascular compromise, or a combination of cardiopulmonary failure states.
In this study, we concentrate our efforts on the oxygenator and its coupling to the TAH. Four potential oxygenator geometries with differing blood flow paths were investigated using computational fluid dynamics (CFD). We employed the standard, 1-D model of mass transport to predict oxygen and carbon dioxide transport. For this initial design, we used characteristics of a commercially available oxygenator for the hollow fiber bundle. We also validated our approach against published computational predictions and experimental data [11,12]. The oxygenator was connected to the magnetically-levitated axial flow pump to assess performance.
We are developing a hybrid-design, continuous-flow, implantable or extracorporeal, magnetically levitated, TAH for pediatric and adult patients. The TAH is a hybrid design since it incorporates two pumps into one medical device; therefore, it integrates both an axial and centrifugal blood pump within a single, compact pump housing. The axial flow pump is positioned into the center of the centrifugal pump, a location that is generally occupied by a hub spindle. The centrifugal pump rotates around the separate axial pump domain, and both impellers rotate around a common central axis. This TAH has only 2 moving parts: an axial impeller to drive blood flow into the pulmonary circulation and a centrifugal impeller to drive blood flow into the systemic circulation. Figure 1A illustrates the conceptual design of this novel device [10]. The TAH implements a combined magnetic suspension-motor drive system to levitate and induce impeller rotation. The left ventricular assist device (LVAD) or centrifugal pump consists of an inlet volute, exit volute, and magnetically levitated impeller. In addition, the design of the axial-flow pump or right ventricular assist device (RVAD) has:
1) An impeller with blades helically wrapped around the rotor surface, which spins to impart rotational kinetic energy to the blood,
2) An inducer region with stationary blades mounted to the pump housing to eliminate pre-rotation of the blood entering the impeller,
3) A set of diffuser blades mounted to the pump housing that are designed to redirect the blood leaving the impeller and convert this energy into pressure, and
4) A straightener region at the outlet to reduce exiting flow vorticity. The target performance for the axial-flow blood pump is to produce flow rates of 1-7L/min for pressure rises of 3-200mmHg at 8,000-16,000RPM.
The integrated TAH and oxygenator are shown in Figure 1B. In this study, we investigate the performance and placement of a hollow fiber bundle of the oxygenator at the base of the TAH. The axial flow pump will provide blood flow through the oxygenator. The outlet of the oxygenator may either be connected to the pulmonary artery for partial cardiopulmonary support or to the inlet of the centrifugal pump to provide full cardiopulmonary support. The outlet of the centrifugal pump is connected to the aorta to provide oxygenated blood to the systemic circulation.
A fiber bundle for an oxygenator consists of hollow fiber membranes, which are composed of woven mats of thousands of hollow fibers. Hollow fibers are made out of several materials with polypropylene being the most common. These hollow fibers have an outer diameter of 200-400 micron with a side wall of 20-50 micron, having pores in the side of the fibers. Oxygen-rich sweep gas flows through the fibers with blood flowing through the interstitial space between and around the hollow fibers receiving oxygen. The arrangement of fibers within the fiber bundle further improves diffusion due to the passive convective mixing of blood around the fibers [13,14]. Four oxygenator geometries were created using a three-dimensional computer aided design (CAD) software package (SolidWorks 2014, Dassault Systems, Inc. Concord, MA USA). The hollow fiber bundle is designed as an annulus with an inner and an outer radius. Blood flow is permitted in either direction through the annulus along the path length. Design and dimensions for the hollow fiber bundle are based on a commercially available oxygenator, the Medtronic Affinity (Medtronic, Minneapolis, MN) as described by Zhang et al. [11]. Figure 2 illustrates the four oxygenator geometries: a) Models 1 and 3 (Figure 2A & 2C) facilitate blood flow moving from the inside of the fiber bundle to the outside, and b) Models 2 and 4 (Figure 2B & 2D) are based on the reverse of the aforementioned designs. Model 2 has a design with the axial pump outlet at the bottom of the oxygenator, and an external cannula is used to redirect the blood flow toward to the TAH to the inlet of the centrifugal pump. These four oxygenator geometries are the result of a series of initial CFD simulations to provide a streamlined inflow and outflow pathways to and from the hollow fiber bundle. The outflow of the axial flow pump was connected to the inlet of the oxygenator. We also incorporated slanted inflow and outflow connections on either side of the fiber bundle.
Figure 1: Hybrid Continuous-Flow Total Artificial Heart with Oxygenator.
A. TAH components consisting of an axial-flow pump for the pulmonary circulation and a centrifugal-flow pump for the systemic circulation [10];
B. TAH with integrated oxygenator. Blood flow is moved through the oxygenator as driven by the axial-flow pump.

Figure 2: Oxygenator Designs Under Consideration.
A. Model 1;
B. Model 2;
C. Model 3;
D. Model 4;
E. Axial- flow Blood Pump;
F. Model 1 with axial-flow pump;
G. Model 3 with axial-flow pump.
B. TAH with integrated oxygenator. Blood flow is moved through the oxygenator as driven by the axial-flow pump.

This feature was included in order to gradually reduce the surface area on the inlet side to move flow into the fiber bundle using a funnel which aids in reducing the pressure loss at the fluid fiber bundle interface by increasing the flow velocity. A second funnel was added on the outlet side to collect the oxygenated blood with a gradual expansion to maintain pressure as blood flowed toward the outlet by decreasing the flow velocity. In these oxygenator designs, we ensured that the inlet ports to the oxygenator were located for ease of integration with the axial-flow pump and to minimize energy loss. These simulations were performed to provide a seamless transition of the flow distribution circumferentially into the hollow fiber lumen and to minimize irregular flow patterns including regions of flow stagnation and flow vortices.
The oxygen mass transfer model of Mockros and Leonard [15] and also as described in Svitek et al. [16] was utilized to predict the one-dimensional mass transfer across the blood flow path length between the inlet and outlet side of the fiber bundle. The partial pressure of oxygen with respect to the blood path length (Equation 1) is determined using the values in Table 1. Due to the non-linearity of the oxyhemoglobin saturation curve, Equation 1 is solved using ODE45 in MATLAB (MATLAB 2018a, The Mathworks, Natick, MA).

Where a and b are empirical constants derived from the research of Hewitt et al. [17] and referenced by Zhang et al. [11] for the hollow fiber bundle geometry of the Affinity oxygenator; p denotes the porosity of the hollow fiber bundle, D is the molecular diffusivity of oxygen in blood, λ(P) represents the slope of oxyhemoglobin dissociation curve, PG is the partial pressure of oxygen at the surface of the fibers, υ is the kinematic viscosity of blood, Qb denotes the blood flow rate at the oxygenator inlet, Af signifies the frontal area of the hollow fiber bundle, d represents the fiber outer diameter. The frontal area varied based on the direction of flow through the fiber bundle (i.e. between inside to outside or outside to inside depending upon the design). The frontal area remained constant with a unidirectional flow through the fiber bundle (i.e. between inside to outside of the bundle). The Hill Equation was used to describe the oxygen dissociation curve through the use of the Hill constant (n) and partial pressure for 50% oxyhemoglobin (P50), shown in Equation 2. Table 1 defines the related model parameters for this equation. P50 is dependent upon pH and temperature and was calculated from equations in Mockros et al. [15]

Table 1: Oxygenator Design Characteristics.

Note: Values are expressed as the mean ± SD, number (percent) or median (interquartile range). eGFR, estimated glomerular filtration rate (MDRD). This table is generated using empower stats (www.empowerstats.com) and R software.
Based on a hematocrit of 36%, pH of 7.4, temperature of 37 °C, the Hill constant was determined to be 2.7 and the value of P50 was calculated to be 26.6mmHg. The partial pressure of oxygen at the inlet of the fiber bundle examwas ined at 70%, 80%, 90%, and 95% saturation for a wide range of flow rates. Hence, the predicted oxygen (O2) transfer rate (𝑉𝑂̇2) was calculated according to Eqn. 3:

where ΔPO2 and ΔSO2 are differences in O2 partial pressure and oxyhemoglobin saturation between the inlet and outlet of the fiber bundle. The design point for the oxygenator was selected to have an oxygen transfer rate of 150mL/min at a flow rate of 5L/min to provide full respiratory support. Eqn. 1 and Eqn. 3 were used to determine the pressure loss across the oxygenators and oxygen transfer rate.
The carbon dioxide mass transfer model described in Svitek et al. [16] was utilized to predict the one- dimensional mass transfer across the blood flow path length between the inlet and outlet side of the fiber bundle. The partial pressure of carbon dioxide with respect to the blood path length (Eqn. 4) is determined using the values in Table 1. The carbon dioxide dissociation curve 𝜆̅𝐶̅𝑂̅ is assumed to be linear for partial pressures of CO2 in the range of 20- 50mmHg. This equation is able to be solved analytically and plotted using MATLAB.

Where a and b are empirical constants derived from the research of Hewitt et al. [17] for the hollow fiber bundle geometry of the Affinity oxygenator; DCO2 is the molecular diffusivity of carbon dioxide in blood, DHCO3 is the
diffusivity of bicarbonate. The predicted oxygen (CO2) transfer rate (VCO2) was calculated according to Eqn. 5:

where ΔCCO2 is the change in the concentration of CO2.
We utilized a commercially available CFD software (ANSYS 15.0, ANSYS, Inc., Canonsburg, PA) to simulate blood flow conditions in the oxygenator geometries. The CAD models of the oxygenator were meshed using CFX-Mesh with hybrid tetrahedral mesh of more than 12 million elements. A grid density and convergence study were performed to ensure mesh independence. Since it is impractical to directly model individual fibers within the hollow fiber bundle due to the complexity of micro-scale flow patterns that are encountered in and around individual fibers [14-15], we implemented the approach that is described in Mazaheri et al. [12]. In this study, researchers demonstrated that the porous media model is acceptable for modeling a compact hollow fiber bundle. CFD studies by Gage et al. [18] and Schlanstein et al. [19] have also successfully used this approach to model the hollow fiber bundle as porous media. The porous media model includes the addition of a source term Si to the momentum equation. This new source term accounts for momentum losses in the ith-coordinate direction, according to Eqn. 6.

where t reflects the time, ρ is the fluid density, xixj are directional coordinates, gi is gravitational force, and τij denotes the shear stress tensor. The source term was defined per Eqn. 7 in accordance with an isotropic and homogenous porous medium with uniform permeability using Ergun correlation. This is frequently used to model the pressure drop through a fiber bundle according to simultaneous viscous and kinetic energy losses.

The variables are k1 which represents the permeability to fluid flow and k2 which is the inertial loss coefficient, μ is the dynamic viscosity, and 𝑣𝑖 is the superficial velocity in the -i,-j,-k, coordinate system, defined as the volumetric flow rate divided by the fiber bundle cross-sectional area [18]. V is the magnitude of the superficial velocity and ρ is the fluid density. The permeability and inertial loss coefficient were estimated using Eqn. 8 and 9 where ε is the porosity of the fiber bundle and dp is the equivalent spherical diameter.


A model of the Affinity oxygenator was created to validate our approach. A set of simulations were run with dp =d0 or the outer diameter of one hollow fiber in the fiber bundle. It has been shown by Zhang et al. [11] and Jones et al. [20] that the Affinity oxygenator is modelled effectively using this method. We also examined the conventional approach presented by Pacella et al. [21] and set dp=1.5d0. Blood was modeled as Newtonian fluid with a dynamic viscosity of 2.9 cP and density of 1050kg/m3.
Boundary Conditions – Oxygenator Fiber Bundle: The hollow fiber bundle was modeled as a continuous volume by combining the blood volume and fibers together as a porous medium. The oxygenator characteristics are available in Table 1. Three domains were defined: an inlet fluid domain for flow into the fiber bundle, a porous domain, and an outlet domain to collect flow exiting the fiber bundle with each region separate by a fluid-porous interface condition. Blood was modeled as Newtonian fluid with a dynamic viscosity of 3.5 cP and 1,050kg/m3 in line with physiologic conditions. A uniform mass flow rate was defined at the inlet to the oxygenator, and a blood flow range of 1-7L/min was evaluated. The outlet pressure was set to be a static and physiologic pressure boundary condition.
Blood oxygen transport is dependent on several factors, including the diffusion and convection along with velocity and blood residence time. Flow residence time is required for red blood cells to stay in the vicinity of the hollow fiber tubules, in order for mass transport to occur as well as to establish favorable hemodynamic conditions without stagnant regions of blood flow. In order to assess the flow patterns and inside of the oxygenator models, streamlines and velocities were assessed. A total of 678 massless particles were released at the inlet to oxygenator model and were tracked through the hollow fiber bundle to the outlet.
Boundary Conditions – Axial Flow Pump: Figure 2 displays the computational model of the axial-flow blood pump and the oxygenator. A steady mass flow rate was defined at the inlet to the axial-flow pump. A no-slip condition was enforced for all stationary walls with fluid velocities at these locations being set to zero. A stationary wall boundary condition was applied to the inducer, housing, diffuser and flow straightener regions. The motor stator and the impeller hub were specified as being in the rotating reference frame in the clockwise direction of rotation. A frozen rotor interface linked the regions of the axial flow pump with differing reference frames along the length of the impeller. The outlet of the axial flow pump was connected to the inflow region of the oxygenator. The element number for axial- flow blood pump was approximately 15 million elements. It is expected that turbulent flow conditions dominate in the axial-flow pump domain. We calculated the global Reynolds number in the pump domain by using the impeller diameter (D) as the characteristic length as Re=ρωD2/μ, where ρ is the fluid density, ω is the angular velocity of the impeller, μ denotes the dynamic viscosity of the fluid. The Reynolds numbers were found to range from 6,350-10,000 for a rotational speeds above 10,000 RPM. As a second indicator of turbulence, we calculated the Taylor number (Ta) as 1,350-2,220. According to Schlichting [22], experiments have demonstrated that flow conditions become turbulent for Taylor numbers greater than 400. Thus, in the combined pump-oxygenator model, we selected the k-ε turbulence model, which has been successfully utilized by our group for simulating axial-flow blood pump [23]. Numerical simulations were performed for a flow range of 1-7L/min rotational speeds at 10,000-16,000 RPM.
In addition to the design and mass transfer considerations, fluid stresses in the axial-flow pump and the oxygenator may damage red blood cells, leading to a release of hemoglobin. Scalar stress has been widely employed as a predictive tool for considering the potential for hemolytic blood damage in blood pumps and oxygenators from elevated scalar stress. Several mathematical models exist for modeling fluid stresses within blood pumps [24- 26]. Eqn. 10 was used to calculate the scalar stress, which includes the components of the stress tensor and represents the level of stress experienced by a red blood cell [27].

Using the scalar stress and fluid residence time, we estimated a blood damage level. Blood damage is calculated, according to Eqn. 11:

where Hb is the predicted hemoglobin content, dHb is the change in hemoglobin content as a result of blood trauma, σ is the accumulated scalar stress, and T is the corresponding exposure time to the scalar stress. The coefficients α, β, and C are empirically derived constants [28]. We assess a maximum scalar stress value of 425 Pa for 600ms as the design cutoff level in the development of the axial-flow pump [23,29]. The fluid streamlines generated by the impeller and passing through the inlet domain of the oxygenator were examined to determine the particle residence times and scalar stresses. A power law relationship according to Eqn. 12 was utilized in this blood damage analysis to relate the scalar stresses and residence time in the estimation of a blood damage index:

where ‘Inlet’ and ‘Outlet’ represent the inflow and outflow faces of the pump domain and the fluid inlet region to the bundle, D is the damage index, σ signifies the scalar stress and t is the exposure time [28]. This provides a statistical estimation of the probability for blood damage to occur from the inlet to the outlet of the pump and up to the inlet region of the fiber bundle. The numerical constants in Eqn. 12, relating the stress to the exposure time, were obtained by regression of experimental data in a Couette viscometer with an exposure time of 0.0034-0.6 seconds for fluid stresses of 40-700 Pa as described by Heuser et al. [25]. This range of investigation is comparable to the flow conditions and scalar stress levels that are found in blood pumps [25,30]. We seek a blood damage index below 2% as the design criterion for blood contacting devices [23,28]. A total of 678 particles were released at the inlet to the axial flow pump and were tracked through the pump. Since the fiber bundle was modeled as porous media, the blood damage index may only be examined in the pump and fluid inlet region to bundle. Although blood damage is known to occur in the hollow fiber bundle, this cannot be effectively modeled due to known and significant computational limitations. Data analysis was performed using a MATLAB code that computed the blood damage index. These estimations were performed for flow rates of 3 and 5L/min at 10,000 and 12,000RPM.
We initially validated the hollow fiber bundle of the Affinity oxygenator against existing computational and experimental data [11,31]. The results of these studies are shown in Figure 3 and good agreement is shown between the computational and experimental data from Zhang, et al. [11]. We also investigated the definition of the equivalent particle diameter under the approximations of dp=d0 as the fiber outer diameter and compare it to dp=1.5*d0. The approximation of dp = d0 showed a strong agreement to the computational and experimental data presented by Zhang et al. [11]. The percent error between our computational data and the accepted computational and the experimental data was 5% and 12%. The approximation of it to dp=1.5*d0 underpredicted the pressure loss on the flow through the porous media. The percent error was 39% for the computational data and 36% for the experimental data. We also validated the model using experimental data from the manufacturer and found strong agreement [31].
Figure 4 displays the estimated pressure loss across four oxygenator geometries for flow rates of 1-7L/min. Each data point reflects the steady state solution for a particular operating condition. All four geometries showed a linear trend of pressure loss with increasing flow rate. The simulations for Model 1 demonstrated a pressure loss of 3-25mmHg. This can be compared with a pressure drop of 3-27mmHg for Model 2. The second set of simulations revealed a pressure loss of 5-35 mmHg and 5-37 mmHg for Models 3 and 4, respectively. The linear slope of the pressure loss across the bundles follows expected trends due to Darcy’s Law.
Figure 4: Computational Estimation of Pressure Loss Across the Oxygenator Designs.
A. Pressure drop across all models. A steady flow rate range of 1-7L/min was examined;
B. Model 1 with the axial flow pump;
C. Model 3 with the axial flow pump.

Figure 5 shows the streamlines in the four oxygenator geometries at a flow rate of 5L/min. The streamlines show that the flow path of massless particles vary considerably in different regions of the models, which will affect the blood residence time for mass transfer to occur. The residence times were obtained by releasing 678 particles in the oxygenator geometries and allowing the particles to flow from the inlet to the outlet. In Model 1 (Figure 5A), the blood flow from the inlet down to the funnel which spreads the flow radially into inside flow gap and interface of the fiber bundle. Flow moves radially through the hollow fiber bundle and blood is collected in the outside gap between the outside shell wall and the outer face of the hollow fiber bundle. Blood then moves out of the outside gap circumferentially and is recollected and directed to the outlet port. The streamlines show the flow uniformity without significant flow stagnation. Model 2 (Figure 5B), has the flow enter the fiber bundle circumferentially. Blood flow is redistributed in a funnel and enters the outside flow gap between the shell wall and outer surface of the hollow fiber bundle. Flow traverses the fiber bundle to the inside flow gap where it is collected and recombined to flow into the outflow pipe. A stagnant region of flow is visible at the base of the model since the flow exits above the base of the hollow fiber bundle. As a result of the poor performance, this model was removed from further consideration in the design process. Model 3 (Figure 5C), has similar blood flow path to Model 1 with the blood flow being redistributed radially and redirected upwards inside the flow gap and interface of the fiber bundle. Flow moves through the hollow fiber bundle and blood is collected in the outside gap between the outside shell wall and the outer face of the hollow fiber bundle. Blood flow is then merged into the outflow duct. The streamlines illustrate the uniformity of the flow traverse without regions of flow stagnation. In Model 4 (Figure 5D), blood flow is distributed in a flow gap below the hollow fiber bundle and is then redirected upward through the outside flow gap between the shell wall and the hollow fiber bundle.
Figure 5: Streamline Plots for Flow Across the Oxygenator Designs. Full Volume:
A. Model 1;
B. Model 2;
C. Model 3;
D. Model 4; Cross-sectional View:
E. Model 1;
F. Model 2
G. Model 3;
H. Model 4; Flow rate is 5L/min.
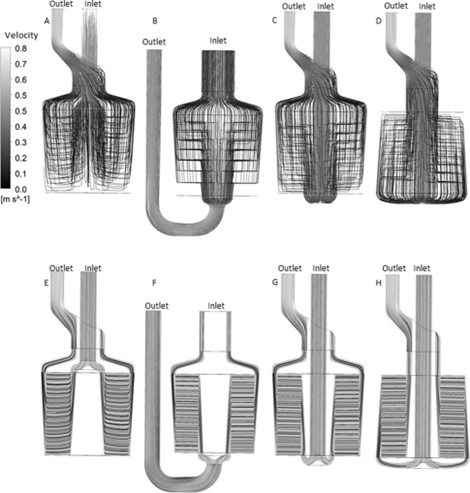
Blood flow then passes through the hollow fiber bundle and flows into the inside flow gap. Blood flow from this region is recombined and directed to the outlet port. The cross-sectional views of the models (Figure 5E-5H) show the uniformity of the flow traverse. These results show that the blood flow is uniform from top to bottom in all 4 models. Particle residence times were in good agreement with the particle streamline plots. Model 1 showed a large distribution of particle residence times with the maximum being 2 seconds and a range of 0-6.8 seconds. Model 2 had a maximum of 3.8 seconds with a range of 4.2 seconds. Model 3 had a maximum of 3.2 seconds with a range of 3-3.8 seconds. Model 4 has a maximum of 3 seconds with a range of 3-3.6 seconds.
Figure 6: Carbon Dioxide Modeling.
A. Plot of CO2 concentration versus CO2 partial pressure and Plot of the slope of the CO2 dissociation curve versus partial pressure.
B. CO2 transfer rate versus the blood flow rate comparison. The error between our predicted curve and the published curve was less than 8%. The prediction for the carbon dioxide mass transfer rate of 550mL/min at 7L/min of blood flow is reassuring.

Carbon Dioxide Mass Transport: Figure 6 illustrates the results of our carbon dioxide model development. A plot of CO2 concentration versus CO2 partial pressure and plot of the slope of the CO2 dissociation curve versus partial pressure, according to the Svitek model, are provided and correlate with expected findings. We also graphed the CO2 transfer rate as a function of blood flow rate per the Svitek model, experimental data, and the results of our model. The deviation between our predicted curve and the published results was less than 8%. Figure 7A displays a plot of CO2 partial pressure versus the fiber bundle radius for a flow rate of 3L/ min, while Figure 7B shows a plot of the CO2 transfer rate versus the blood flow rate for the current fiber bundle. The CO2 model predicted a carbon dioxide mass transfer rate of between 170mL/ min at a flow rate of 1L/min and 550mL/min at a 7L/min flow rate.
Figure 7: Estimations of Carbon Dioxide Transfer Rate Across the Hollow Fiber Bundle.
A. Predicted carbon dioxide transfer rate with blood flow through the hollow fiber bundle for a flow rate of 3L/min;
B. Predicted oxygen saturation at the outlet for an inlet saturation of 70% for both model geometries.

Oxygen Mass Transport: Figure 8 illustrates the estimation of oxygen transfer across the bundles based on inlet oxygen saturations of 70%, 80%, 90%, and 95%, over a range of blood flow rates. Figure 8A displays the oxygen transport from inside to the outside of the fiber bundle, while Figure 8B shows the oxygen transport from outside to inside the hollow fiber bundle. The transfer rate findings demonstrate similar trends for all of the oxygenators. The design having the outside-to-inside flow direction, however, had higher oxygen transport. All of the designs met the performance criterion of a transfer rate of 150mL/min at a blood flow rate of 5L/min.
Integrated Pump-Oxygenator Performance: Models 1 and 3 (Figure 4) were selected for integration with the axial flow pump. Using an oxygenator geometry that moves flow from the inside of the annulus to the outside would permit a more compact design. The hydraulic performance of the axial pump was consistent across all flow rates for each of the oxygenator designs. The axial pumps delivered pressure generations of 2-200mmHg (Model 1) and 1-200mmHg (Model 3), respectively, for flow rates of 1-7L/min at 10,000-16,000RPM.
Figure 8: Estimations of Oxygen Transfer Rate Across the Hollow Fiber Oxygenators.
A. Predicted oxygen transfer rate with blood flow through the inner and outer surfaces of the hollow fiber bundle for both model geometries;
B. Predicted oxygen saturation at the outlet for an inlet saturation of 70% for both model geometries.

Blood Damage Analysis: A blood damage analysis was completed for the oxygenator geometries that were selected for integration with the axial-flow pump. For both geometries, an operating speed of 10,000 and 12,000RPM and at flow rates of 3 and 5L/min were used. Table 2 shows the results of this analysis. The scalar stress distribution for the axial flow pump and both pump-oxygenator models is shown in Figure 9 for the 10,000RPM case at5 L/min flow rate. For the axial-flow pump, the mean scalar stress value was found to be 24.9Pa with a maximum value of 333.2Pa. For Model 1, the mean scalar stress value was estimated at 23.0Pa with a maximum value of 327Pa. For Model 3, the mean scalar stress was determined to be 23.4Pa with a maximum value of 335Pa. Using 678 particle streamlines, we estimated a blood damage index for the blood flow through the axial blood pump and inflow of the oxygenators. In general, higher rotational speeds and higher flows led to higher damage indices. At a rotational speed of 10,000RPM and a 3L/min flow rate, flow through Model 1 led to a mean damage index of 0.021% with a maximum value of 0.239%, while Model 3 had a mean damage index of 0.022% with a maximum value of 0.336%. For the 10,000 RPM case at 5L/min flow rate for Model 1, the mean value for the damage index was found to be 0.030% with a maximum value of 0.375%, while for Model 3 the mean value for the damage index was found to be 0.027% with a maximum value of 0.443%. For the 5L/min flow rate for Model 1 at 12,000RPM, the mean value for the damage index was found to be 0.037% with a maximum value of 0.579%. Similarly, for Model 3, the mean value for the damage index was found to be 0.037% with a maximum value of 0.624%, a higher finding than at the lower rotational speeds.
Figure 9: Scalar Stress and Streamline Distribution of the Axial Flow Pump and Oxygenators for 5 L/min at 10,000 RPM.
A. Streamline Plot for the Axial-flow impeller;
B. Streamline Plot for Model 1 with the axial flow pump;
C. Streamline Plot for Model 3 with the axial flow pump;
D. Scalar Stress Plot for the axial flow impeller;
E. Scalar Stress Plot for Model 1 with the axial flow pump;
F. Scalar Stress Plot for Model 3 with the axial flow pump.

A combination, single replacement technology for more than one organ is not a new concept. Extracorporeal life support (ELCS) systems combining pump with the oxygenator probably serve as the best example. Recently, the pump and the oxygenator of such ECLS systems were integrated into one compact unit, available commercially (Cardiohelp life support system, Maquet). Besides the added convenience of a more compact system that includes ease of transport, no major functional differences were found compared to a standard ECLS circuit [32]. Beyond ECLS, similar attempts have been made to combine a decarboxylation device (extracorporeal CO2 removal or ECCO2R) with a hemofiltration system [33], and such a system has been found to be effective [34]. Commercialization of such a combined low-flow ECCO2R-hemofiltration unit would be beneficial. To date, however, no implantable cardiopulmonary replacement system, or any combined organ replacement system, has been proposed. The primary hurdle to such devices would be the limited life span of the hollow fiber oxygenators which, in a matter of days to weeks, experience accumulating thrombus formation resulting in high resistance. Wearable, “paracorporeal” devices are under development; however, no current oxygenator technology is suitable for implantable, durable use [35]. As a corollary to that, efforts have been lacking to design a device that would combine a pump and an oxygenator into a compact, durable, fully implantable unit. However, progression of the oxygenator technology, including advancements in the hollow fiber oxygenators and microfluidic oxygenators [36] promise further improvement in the field [37], likely to result in full implantability and long-term usage. As such, fully implantable cardiopulmonary replacement would be feasible.
Our study represents the first such effort to integrate an oxygenator into a continuous flow TAH for pediatric and adult patients [10]. By attaching the hollow fiber bundle of the oxygenator to the base of the TAH, we have thereby created a new cardiopulmonary support technology that permits a patient to be ambulatory during usage and that can assist patients with lung disease, ARDS, cardiovascular compromise, or a combination of cardiopulmonary dysfunction and failure states. The TAH is designed to support a wide range of patients from children to adults. For adults and larger pediatric patients, the device is intended to be fully implantable with cannulae connecting to the internal vessels for intracorporeal placement. There has been a shift toward using less invasive implantation techniques for the extracorporeal respiratory support. We designed our oxygenator geometries with the inlet and outlet on the same side of the device for ease of access and to prevent kinking of tubing or grafts. This feature also permits integration with a dual lumen cannula (DLC) such as the Avalon Elite bi-caval DLC (Maquet, Rastatt, Germany), which provides minimally-invasive deployment for extracorporeal support.
In this study, we investigated four oxygenator geometries with varying fluid flow pathways using CFD. The oxygenator fiber bundle geometry was assessed for gas exchange using a one dimensional mass transport model that is available in the literature [38]. The oxygenator is uniquely configured to seamlessly integrate the axial- flow pump and provide a streamlined blood flow path. Radial distribution of the flow from the axial-flow pump has been specifically designed into the inflow region of the oxygenator to minimize the prospect of blood damage. Flow from the pump is radially distributed which permits the flow to circumferentially interact with the hollow fiber bundle. Regions of flow stagnation and recirculation regions were minimized in our initial CFD studies in order to provide a streamlined flow path in the inlet and outlet regions of the oxygenator. We used the mass transfer model to predict the oxygen exchange from either the inner surface-to-outer surface of the bundle or vice versa at 4 different venous saturations. The presence of the inner versus the outer direction affected the gas transfer efficiency, as theoretically expected. The predicted curves were acceptably validated against data from the Affinity oxygenator published in [11]. The Affinity oxygenator was used as a starting point for the oxygenator design as it met our design criteria of an oxygen transfer rate of 150mL/min at a flow rate of 5L/min for the optimal design point to provide full respiratory support. The Affinity oxygenator is capable of producing greater than 250mL/ min of oxygen transfer. However, the oxygenator surface area is 2.5m2, and this surface area can be reduced during future design iterations without compromising oxygen transport efficiency. The fiber bundle characteristics must be altered to facilitate this by increasing the void ratio (porosity) for fluid flow around the microtubules. The challenge will be promoting active mixing of blood in the fiber bundle using the axial-flow pump to augment the gas transfer efficiency while minimizing the resistance to flow through a small surface area.
In evaluating the oxygenator designs, we found that Models 1 and 2 had low pressure drops compared to models 3 and 4. This is due to resistance in the inlet and outlet pathways and conduits. Particle distribution and streamline plots were used to make determinations on the uniformity of the flow traverse as this has the potential to impact oxygen diffusion. The flow distribution in the hollow fiber bundles was in the radial direction in all models with a uniform flow profile. Model 2 exhibits a stagnant region of flow at the base of the oxygenator and was removed from further study. Graefe et al. [39] attempted to optimize flow uniformity in hollow fiber bundles by testing various inlet and outlet port designs according to particle residence time, but this approach does not provide additional information regarding the actual distribution of flow in the fiber bed. Due to the porous media assumption that is used in the CFD studies, the particle residence times that are calculated may underestimate the true flow travel time to pass through a fiber bundle [12]. Given these findings and the geometric constraints associated with using an axial-flow blood pump, we selected Models 1 and 3 for the CFD analysis. Model 4 was discarded due the large pressure drop incurred in the inflow pathway as the flow must spread out circumferentially. These Models 1 and 3 provide a smaller internal area by which to distribute flow from the axial flow pump and lead to a lower pressure loss at the inlet translating to a lower pressure drop across the hollow fiber bundle. These findings also correlate well with theoretical expectations and clinical observations using the Affinity oxygenator. The pressure rises across the oxygenator geometries met expectations and trends, with a decrease in pressure generation as a function of increasing flow rate for each pump rotational speed.
Blood damage predictions showed that probability for blood damage would be highest in the pump domain. The highest scalar stresses were encountered at the impeller blade tips. This is to be expected due to the fluid motion over the tips of the impeller blades. Scalar stresses was predicted throughout the entire oxygenator geometries, however the most important regions were from the pump inlet to the inlet of the hollow fiber bundle. It is important to note that the hemolysis index model that is used in this study has been shown to over predict the incidence of hemolysis. Based on the hydrodynamic and hemolytic predictions, it was determined that Model 1 provided the superior design performance when integrated with the axial flow pump. This geometry provides a streamlined flow path into the fiber bundle inlet. The outflow region of the pump propels fluid into oxygenator. Model 3, however, provides a more compact design by placing the axial-flow pump at the bottom of the oxygenator. The positioning in this configuration requires a 180° bend that leads to irregular flow patterns and elevated shear stresses; of course, future design improvement with iterative optimization could be performed to make the configuration of Model 3 more functional than Model 1.
This study has several limitations to be addressed in future work. Design improvement and iterative optimization of the oxygenator are needed to reduce the size of the bundle and improve the oxygen transfer efficiency. A physical prototype of the oxygenator and TAH must be built and experimentally tested to facilitate measurement of the pressure loss across the bundle, pressure generation of the axial pump, and hemolytic- thrombotic levels. Numerical methods, other than CFD, have been used to advance the design and optimization of oxygenators, and these approaches will provide additional insight into the flow physics and mass transfer properties16. We used a one-dimensional mathematical model in this study, and an analysis of the full threedimensional characteristics of the integrated oxygenator and TAH would add value. Blood residence time is linked to the local fluid velocity and is important for oxygen diffusion from the hollow fibers to the red blood cells suspended in the blood plasma. A longer residence time provides more time for mass transport to occur while a faster velocity affects convective mixing in the hollow fiber lumen. Further investigation is required to experimentally measure the oxygen transfer rate and oxygen transfer efficiency. This study is a pilot study using a proven oxygenator configuration, and future work will focus on developing and testing a new oxygenator for integration into the new cardiopulmonary assist system that we are designing for pediatric and adult patients.
This new oxygenator has demonstrated promising results at this stage of development. The CFD-derived computational data demonstrated the ability of the integrated pump oxygenator to generate pressures and flows that would be comparable for mechanical circulatory and respiratory support. Further development and physical testing are necessary to address limitations and to advance the oxygenator design in the development of a new implantable hybrid and full cardiopulmonary assist systems.
The authors wish to acknowledge the financial support as provided by the American Heart Association Postdoctoral Fellowship (S.G. Chopski), a 2022 U.S. Department of Education GAANN Fellowship (E. Garven), the 2016 Individual Biomedical Research Award from The Hartwell Foundation, and the National Heart, Lung, And Blood Institute of the National Institutes of Health, under Award Number: R01HL153536. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
All authors contributed equally to the development of this manuscript. Full patent application is pending with approval imminent. No conflicts of interest; no disclosures.
a) Study Conception – AT, SC;
b) Clinical Significance & Motivation – AT, SC, RS, VT;
c) CFD Simulation & Mathematical Modeling – AT, SC, KG, AM, EG;
d) Manuscript Composition & Revision – All


