Abstract
To cerebral ischemic (CI) in patients, frequently thrombotic or embolic strokes occur in the area of the middle cerebral artery (MCA). The intraluminal middle cerebral artery occlusion (MCAO) model is quite relevant for stroke studies. The aim of this work is to introduce technical improvements in acute cerebral ischemia-reperfusion model by endovascular occlusion of the middle cerebral artery in the rat. Male Sprague-Dawley rats were used for this experiment. They were divided into two groups: Sham (without CI) and CI. To CI group, surgical procedure was used blocking blood flow in the MCA, introducing an intraluminal filament through the internal carotid artery. The nylon filament tip was rounded to the flame and coating with nail varnish for four mm long and 0.35 mm thick. The anesthetics were Ketamine/Xylazine at a dose of 70/10 mg/Kg. The occipital artery, the superior thyroid and the external carotid artery were permanently ligated using unraveled silk thread, during 60 minutes of occlusion. In sham group, surgery was performed except the introduction of the filament. Brains were obtained to evaluate the infarct area using 2,3,5-triphenyltetrazolium chloride staining. To evaluate the effect of IC, sensory and motor responses were analyzed. The results show that the infarct zone was 18% more than in the sham animals and the survival percentage was 65% using Ketamine/Xylazine at a dose of 70/10 mg/Kg as anesthetic. in IC animals, it was found that 90% presented neurological alterations such as: loss of gait, crawling in circles and retraction of the right foot. In both sensory and motor evaluation, a significant decrease was observed in the animals with IC compared to sham group. The results show that the modifications made to the MCAO model in rat, can be useful and easily accessible to obtain the expected effects of the CI.
Keywords: Cerebral Artery Occlusion; Cerebral Ischemia; Adhesive Tape Task; Neurological Test
Abbreviations: CI: Cerebral Ischemic; MCA: Middle Cerebral Artery; MCAO: Middle Cerebral Artery Occlusion; TTC: 2,3,5-Triphenyltetrazolium Chloride; ACA: Anterior Cerebral Artery; BA: Basilar Artery; CCA: Common Carotid Artery; C. Willis: Circle of Willis; ECA: External Carotid Artery; ICA: Internal Carotid Artery; MCA: Middle Cerebral Artery; PCA: Posterior Communicating Artery; PPA: Pterygopalatine Artery.
Introduction
Strokes are the third cause of death in industrialized countries,
after heart disease and cancer. Among cerebrovascular accidents,
cerebral ischemia (CI) occupies 80% of the cases, and the remaining
20% corresponds to intracerebral or subarachnoid hemorrhage,
with a mortality of 25% of all cases [1]. In 2016, 80 million people
around the world were affected by a stroke, of which 67 million were
ischemic events [2]. The CI is one of the most serious neurological
disorders. Most of the survivors suffer permanent disabilities,
including motor, communication, cognitive, learning and memory
problems, reducing quality of daily life [3]. IC is the temporary or
permanent interruption of the blood supply to the brain, which
reduces the flow of molecular oxygen (O2) and important nutrients
such as glucose. In most cases, it is caused by the occlusion of a
cerebral artery either by an embolus or by local thrombosis [1]. The
reduced blood supply decreases to levels insufficient to maintain
normal metabolism and the adequate function of brain cells [4].
The reduction in cerebral perfusion is limited to the territory
supplied by the affected artery (focal cerebral ischemia). As a
consequence of intense and/or prolonged ischemia, an irreversible cell loss occurs with a localized destructive lesion, which could lead
to a cerebral infarction [5]. To mimic ischemic stroke in patients,
frequently thrombotic or embolic strokes occur in the area of the
middle cerebral artery (MCA), the intraluminal middle cerebral
artery occlusion (MCAO) model is quite relevant for stroke studies.
They models were first developed in rats by Koizumi, et al. [6] and
Tamura, et al. [7]. It has previously been reported that physical
properties of the occluder such as tip diameter, length, shape, and
flexibility are critical for the reproducibility of the infarct volume
in rodents [8-11]. The aim of this work is to introduce technical
improvements in acute cerebral ischemia-reperfusion model by
endovascular occlusion of the middle cerebral artery in the rat.
Material and Methods
Animals
Male Sprague-Dawley rats aged 8 to 12 weeks, weighing 270 to 330 g were used for this experiment. They were divided into two groups: Sham (without CI, n= 12) and CI (n=12). The rats were provided by the animal farm of the Autonomous University of Aguascalientes and maintain at 22°C with a 12-hour light-dark cycle and continuous access to food (purine chow) and water. All surgeries were performed under general anesthesia. They were used and treated in accordance with the Institutional Welfare Standards (Universidad Autónoma de Aguascalientes) and every effort was made to avoid suffering and to avoid unnecessary testing.
Surgery of MCAO
In the CI group, surgical procedure consists of blocking blood flow in the MCA, introducing an intraluminal filament through the extracranial internal carotid artery to reduce blood flow, described by Longa, et al. [12] with modifications (Figure 1). The rats were anesthetized with Ketamine/Xylazine/70/10 mg/kg. With the aid of a stereoscope microscope, an incision is made in the midline of the neck, the glandular tissue is retracted, and a triangle formed by the omohyoid, the digastric muscles (easily identifiable by their bright white tendinous portion), and the sternomastoid is observed. Using blunt scissors, dissect the center of the triangle to expose the common carotid artery, which is evident. The vagus nerve is separated and isolated. The occipital artery, the superior thyroid and the external carotid artery are permanently ligated using unraveled silk thread.
Figure 1: Scheme of the occlusion of the middle cerebral artery using nylon filament coated with nail varnish.
A. Simplified scheme of rat brain and cerebral arteries showing sutures and clip to introduction of filament.
B. The position of filament through the circle of Willis is represented. The filament is introduced into ICA via ECA to occlude
the base of the MCA.
A twenty five mm long nylon filament is used (made in our laboratory). The filament tip is rounded to the flame and coating with nail varnish for four mm long and 0.35 mm thick. It is introduced through the left internal carotid artery and after 60 minutes, it is removed allowing reperfusion. In the rats of the SHAM group the internal, external, common, occipital and superior thyroid arteries were manipulated and exposed without performing any ligation. All animals were injected intradermally with 5 ml of sterile saline solution for fluid recovery and with penicillin (penprocycline 5,000 IU i.m.). They were placed in a recovery box (thermal mattress at 37°C and continuous oxygen) for 2 to 3 hours and so that they would not suffocate, a steel wire mouthpiece to keep its mouth open was used.
Measurement of the Infarct Area
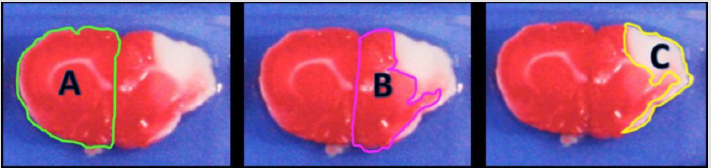
To evaluate the percentage of damage produced after cerebral ischemia, at 72 h post-reperfusion, the animals are anesthetized with sodium pentobarbital and intracardiac perfusion with 0.9% cold saline solution. The brains are removed and placed in a Petri dish and freeze for 5 minutes. Serial coronal cuts 2 mm thick were made with a microtome blade and placed in a 1% of 2,3,5-triphenyltetrazolium chloride (TTC) solution at 37°C for 20 minutes. The healthy area is stained red, while the infarcted area is white. Photographs were taken with a stereoscope microscope of each slice of tissue and measurements of the infarct area were made using Image J software. The infarct area (mm2) was calculated by measuring the area of the contralateral hemisphere minus the healthy area of the ipsilateral hemisphere for each slice.
Neurological Scale Evaluation
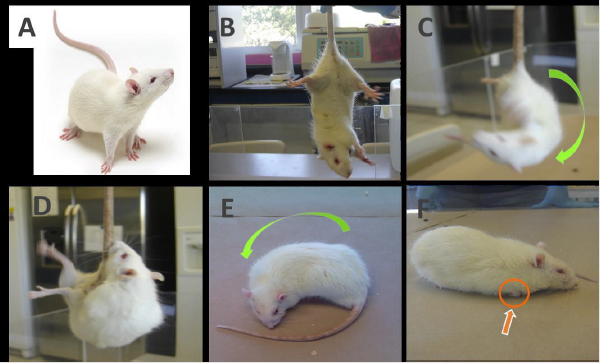
The first test is a neurological assessment scale to identify alterations in the motor response of the rat. Neurological deficits make it possible to evaluate the success of MCAO after reperfusion, and to calculate the degree of severity of the injury. The rat is scored according to the clinical signs on the scale grade 0-5 [9] (Figure 2). This test was performed 24 after surgery.
Figure 2: Images of rats with different degrees of injury evaluated with neurological score.
A. Normal, grade 0;
B. Grade 1;
C. Grade 2; rat turns ipsilaterally when it grabs its tail and tries to climb its tail
D and E. Grade 3; the rat turns on an ipsilateral side with its nose almost reaching its tail,
F. Grade 4; loss of gait, crawling in circles and retraction of the right foot is observed.
Adhesive Tape Removal Test
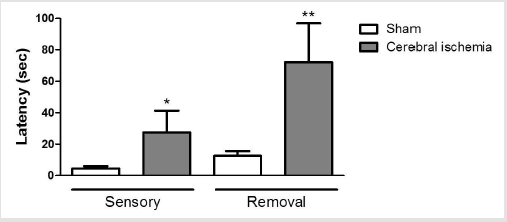
This test was considered as a parameter of sensory-motor function. Adhesive tape of one cm2 was trimmed. The anterior feet are cleaned with 70% ethanol. The two squares of tape are placed on each foot using the same pressure on both feet. Once the two tapes are in place, the rat is placed in the evaluation box and recording begins, the video is stopped at the moment that rat removes two adhesive tape. The animal will react to the presence of the adhesive tape, either by shaking the paws or directly bringing them to the mouth. This behavior is marked as “first contact” and is defined as the point at which the animal reacts to the tape. The “removal time” is the time that ends when the rat removes both tapes. Animals with ischemia will take significantly longer to feel and remove the tape.
Statistical Analysis
The data obtained were analyzed using the GraphPad Prism version 5 software. The data were expressed as the mean ± standard error of the mean (mean ± SEM) and were analyzed using one-way ANOVA and the Tukey test of multiple comparison to compare the means of different groups. A significance value of p <0.05 was considered.
Results
The results show that areas of infarction were obtained (Figure 3) with 0.15 mm diameter nylon filaments coated with nail varnish until obtaining a diameter of 0.35 to 0.39 mm (since the diameter of the internal carotid artery is ≈ 0.35 mm). When analyzing both the infarct percentage and the survival of the animals, it was found that the infarct zone was 18% more than in the sham animals and the survival percentage was 65% 24 h after surgery. All animals were anesthetized with Ketamine/Xylazine at a dose of 70/10 mg/ Kg. Neurological deficits were observed 24 h after of the MCAO reperfusion surgery. In 90% of the animals, a grade 4 was found on the evaluation scale: loss of gait, crawling in circles and retraction of the right foot was observed. In the group of CI rats, it is observed that at 24 h it was approximately 7 times longer to recognize the tape on the foot contralateral to the damage compared to latency of sham group (27.4 ± 13.8 vs 4.5 ± 1.4) and six times in the time of removal of the adhesive tape compared to latency of the sham rats (72.1 ± 24.8 vs 12.6 ± 3) (Figure 4).
Figure 3: Representative coronal sections of rat brain stained with TTC after 72 h post-reperfusion. Infarct area appears in white (unstained by TTC). The injury zone (white area) was delimited.
Figure 4: Figure 4: Evaluation of sensory and motor damage with the adhesive tape test in rats with cerebral ischemia induced by MCAO. Latency time for feeling the tape adhesive on the right foot. Adhesive tape removal time on the right foot.
Data are expressed as the mean ± SEM; n = 12 per group. * p <0.04, ** p <0.02 compared to their respective sham group.c
Discussion
CI causes a temporary or permanent reduction in cerebral
blood flow that can lead to acute or chronic dysfunctions in the
central nervous system. The rat is a widely used animal model
for CI due to its relatively low animal husbandry costs and to the
similarity of its cerebral circulation to that of humans. In humans,
the middle cerebral artery (MCA) is most commonly affected in
stroke syndromes and multiple techniques of MCAO have been
reported to mimic this clinical effect in rodent models. Because
recanalization commonly occurs following an acute stroke in the
human, reperfusion after a period of occlusion has been included
in many of these models. There is considerable risk in CI during
anesthesia and surgery in these models. Anesthetic agents have
been shown to have a profound effect on the pathophysiology of
CI. Some authors have used different anesthetics and various doses
during the analysis of the volume of infarction produced by IC, such
as halothane, Ketamine/Xylazine or isoflurane [8,10,11].
In this study, the best results in terms of the percentage of
infarction obtained and survival were obtained with an occlusion
time of 60 minutes and using Ketamine/Xylazine 70/10 mg/kg
as anesthetic. In other experiments, trials were performed with
occlusion times of 30 and 45 minutes, in which no infarct area was
observed, it was also tested with a 90-minute time in which the
infarction area was approximately 50%, but mortality was also very
high, about 80% (data no shown). In the present work, a twenty
five mm long nylon filament was used (made in the laboratory)
to occlude blood flow. The filament tip was rounded to the flame
and coating with nail varnish for four mm long and 0.35 mm thick.
A great advantage is that this filament reduces the risk to induce
subarachnoid hemorrhages. There are commercial filaments such as silicone coated but the costs are higher and the same result is
obtained [9,10]. Neurological deficits were observed 24 h after of
the MCAO reperfusion surgery. In 90% of the animals, a grade 4 was
found on the evaluation scale. Similar effects have been observed by
other researchers [9,10].
Conclusion
According to the results obtained, it can be concluded that IC can be induced by occlusion-reperfusion of the MCA. The effects of IC can be assessed by the infarct area using TTC staining, and both sensory and motor neurological tests, including those with adhesive tape. In this rat model, good infarct area and survival results can be obtained by different considerations:
a) The filament tip is rounded to the flame and coating with nail
varnish for four mm long and 0.35 mm thick,
b) Ketamine/Xylazine at a dose of 70/10 mg/Kg,
c) The occipital artery, the superior thyroid and the external
carotid artery are permanently ligated using unraveled silk
thread, during the 60 minutes of occlusion,
d) Two stitches were temporarily placed on the skin of the neck
to prevent the filament from coming out and
e) A steel wire mouthpiece to keep its mouth open.
Disclosure
The authors report no conflict of interest.
References
- Dirnagl U, Iadecola C, Moskowitz M (1999) Pathobiology of ischaemic Stroke an integrated view. Trends Neurosci 22(9): 391-397.
- Benjamin EJ, Muntner P, Alonso A, Bittencourt MS, Callaway CW, et al. (2019) Heart Disease and Stroke Statistics-2019 Update: A Report From the American Heart Association. Circulation 139(10): e56-e528.
- Knecht S, Hesse S, Oster P (2011) Rehabilitation After stroke. Dtsch Arztebl Int 108(36): 600-606.
- Montaner J (2007) Physiopathology of Cerebral Ischemia. Marge Books, Barcelona.
- García J (1984) Experimental ischemic stroke: a review. Stroke 15(1): 5-14.
- Koizumi J, Yoshida Y, Nakazawa T, Ooneda G (1986) Experimental studies of ischemic brain edema, I: a new experimental model of cerebral embolism in rats in which recirculation can be introduced in the ischemic area. Jpn J Stroke 8(1): 1-8.
- Tamura A, Graham DI, Mc Culloch J, Teasdale GM (1986) Focal cerebral ischaemia in the rat: 1. Description of technique and early neuropathological consequences following middle cerebral artery occlusion. J Cereb Blood Flow Metab 1(1): 53-60.
- Lin TN, He YY, Wu G, Khan M, Hsu CY (1993) Effect of brain edema on infarct volume in a focal cerebral ischemia model in rats. Stroke 24(1): 117-121.
- Rousselet E, Kriz J, Seidah NG (2012) Mouse Model of Intraluminal MCAO: Cerebral Infarct Evaluation by Cresyl Violet Staining. J Vis Exp 69: 4038.
- Uluç K, Miranpuri A, Kujoth GC, Aktüre E, Başkaya MK (2011) Focal Cerebral Ischemia Model by Endovascular Suture Occlusion of the Middle Cerebral Artery in the Rat. J Vis Exp 48: 1978.
- Yrjänheikki J, Keinänen R, Pellikka M, Hökfelt T, Koistinaho J (1998) Tetracyclines inhibit microglial activation and are neuroprotective in global brain ischemia. Proc Natl Acad Sci U S A 95(26): 15769-15774.
- Longa EZ, Weinstein PR, Carlson S, Cummins R (1989) Reversible middle cerebral artery occlusion without craniectomy in rats. Stroke 20(1): 84-91.

 Research Article
Research Article