Abstract
Primary ovarian leiomyosarcoma (OLMS) is an extremely rare and aggressive neoplasm. By 2018, only 73 clinical cases of ovarian leiomyosarcoma have been published in the English medical literature. We present a right-sided OLMS in a 77–year-old woman. A total abdominal hysterectomy with bilateral adnexectomy and partial omentectomy was performed. Moderately differentiated (G2) leiomyosarcoma of the right ovary is diagnosed on the basis of morphological and immunohistochemical (IHC) examination. According to the FIGO classification, it refers to an early IA clinical stage corresponding to pT1aN0M0. Despite the early clinical stage, after 6 months of diagnosis, a local recurrence with paravertebral soft tissue metastases were diagnosed, and after 6 months- disease progression with distant bone, lung and liver metastases werе observed. The article emphasizes the importance of immunohistochemical analysis for the diagnosis of soft tissue sarcomas with organ localization, unfavorable prognosis and the need for complex oncological treatment, despite the early clinical stage. In principle, the main treatment is radical surgery, but an individual assessment of adjuvant chemo-radiotherapy (Ch-RT), depending on the sarcoma volume and adverse pathohistological tumor characteristics, is required.
Keywords: Primary Ovarian Leiomyosarcoma; Immunohistochemistry; Surgery; Adjuvant Chemo-Radiotherapy; Complex Oncological Treatment
Abbreviations: POS: Primary Ovarian Sarcomas, OLMS: Ovarian Leiomyosarcoma, IHC: Immunohistochemical, TD: Total Dose, DD: Daily Dose
Introduction
Primary Ovarian Sarcomas (POS) are rare neoplasms that are diagnosed in less than 2% of all ovarian malignancies [1,2]. The ratio between the incidence of ovarian sarcomas and carcinomas is 1:40 [3]. Primary ovarian Leiomyosarcoma (OLMS) is extremely rare - less than 0.1% of all ovarian sarcomas [2,4-6]. The most commonly diagnosed pathohistological POS variants are fibrosarcoma, stromal cell sarcoma, or rhabdomyosarcoma [2,7]. The immunohistochemical (IHC) analysis is required to clarify the pathogenesis of sarcoma cells [8,9]. Due to the rare incidence of primary ovarian leiomyosarcoma, the optimal complex treatment has not yet been determined.
Clinical case: A 77-year-old woman with low abdominal pain is presented. Ultrasound of the abdominal organs and small pelvis revealed a cystic formation in the area of the right adnexa with a diameter of about 6 cm. Radical surgery - total abdominal hysterectomy with bilateral adnexectomy and partial omentectomy - was performed.
Pathohistological Examination-Macroscopic Characteristics of The Right Adnexa: Tumor formation with a diameter of 6.5cm adjacent to the fallopian tube; in section a whitish tumor with necrotic areas - most likely ovarian sarcoma. Gefrir - Mesenchymal tumor with high cellularity, nuclear polymorphism and necrotic areas - most likely ovarian sarcoma.
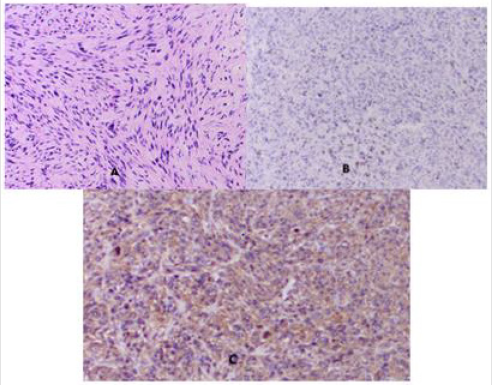
Microscopic Characteristics: In the right ovary a tumor formation of 65 mm in diameter was found. A high-cellular neoplasm composed of drained atypical cells arranged in bundles is reported. Tumor cells contain vacuolated polymorphic eosinophilic cytoplasm. Elongated-shaped hyperchromic nuclei were observed, as well as high mitotic activity of up to 10 mitoses per high magnification field (Figure 1A & 1B). Hypocellular areas and extensive areas with necrosis were reported. In the tumor periphery a shaped capsule with a growth of tumor cells was reported. The fallopian tube has peritubular serous cysts. The morphological characteristic corresponds to primary ovarian sarcoma. The IHC analysis to clarify the histological subtype is needed.
Figure 1: Photomicrography of ovarian leiomyosarcoma: A / Diffuse infiltration of atypical spindle cells / H&E x20; B / IHC of the proliferative index Ki67 in the nuclei of tumor cells (17.4%) x20; C/ The characteristic corolla-like distribution of sarcoma cells in diffuse IHC expression to SMA x20.
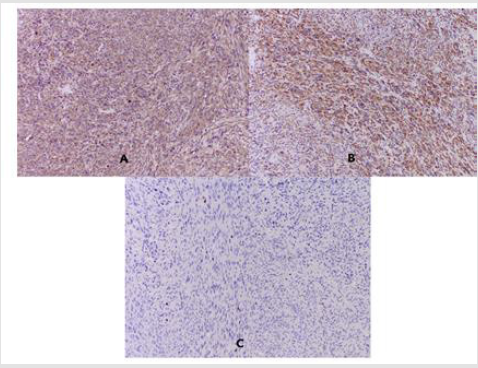
Figure 2: Photomicrography of IHC: A / SMA, clone 1A4- Diffuse distinctly strong positive reaction in the cytoplasm of tumor cells x20; B / Desmin, clone D33- Diffuse distinctly strong positive reaction in the cytoplasm of tumor cells x20; C /S-100- Focal positive reaction in single tumor nuclei x20.
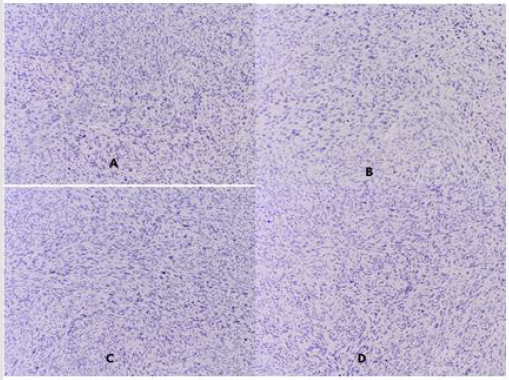
Immunohistochemical Analysis: SMA, clone 1A4- Diffuse distinctly strong positive reaction in the cytoplasm of tumor cells (Figure 2A); Desmin, clone D33- Diffuse distinctly strong positive reaction in the cytoplasm of tumor cells (Figure 2B); S-100 protein- Focally positive reaction in single tumor nuclei (Figure 2C). Negative IHC expression for Estrogen and Progesterone receptor in the nuclei of tumor cells was reported (Figure 2A & 2B); for Cytokeratin, clone AE1 / AE3 in the cytoplasm of tumor cells (Figure 3C); for CEA125 in tumor cells (Figure 3D); for CD 34 in tumor cells / but positive in the walls of blood vessels at the border of the tumor (Figure 3A) and for Myogenin A- in the nuclei of tumor cells (Figure 3B).
Figure 3: Photomicrography of negative IHC expression A / Estrogen receptor; B / Progesterone receptor; C / Cytokeratin, clone AE1 / AE3; D / CEA125.
Conclusion
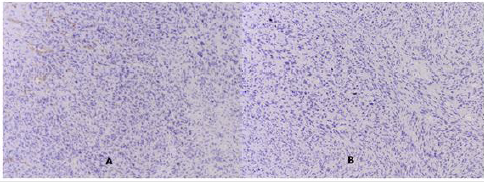
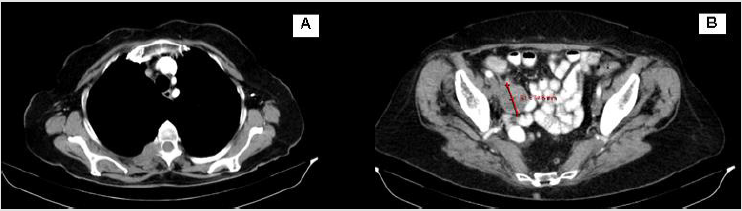
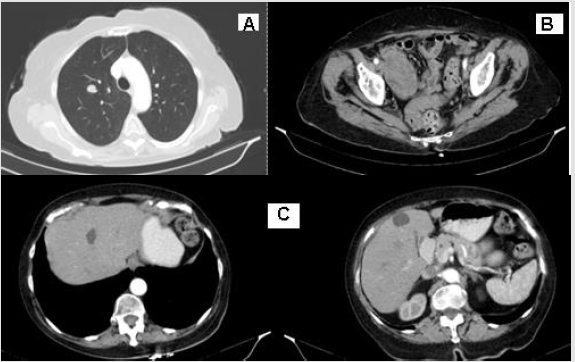
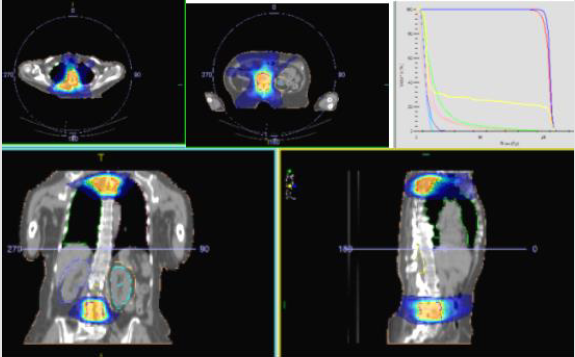
Based on the performed morphological and immunohistochemical examination, primary moderately differentiated (G2) leiomyosarcoma of the ovary is diagnosed. In the presented clinical case, the following unfavorable pathohistological tumor characteristics: tumor formation with a diameter of more than 5 cm, high mitotic activity (Ki 67-17.4%), extensive areas with tumor necrosis, tumor capsule in places overgrown with tumor cells were reported. Those data led us to offer adjuvant chemo-radiotherapy (Ch-RT), despite the early clinical stage and radical surgery. The patient refused Ch-RT and remained on dispensary observation. Despite the early clinical stage, after 6 months of diagnosis, a local recurrence with paravertebral soft tissue metastases were diagnosed (Figure 4 A/B), and after 6 months- disease progression with distant bone, lung and liver metastases (Figure 5A-5C) werе observed. In the area of the metastatic vertebrae (Th 3-4 and L1-3), palliative Intensity Modulated Radiotherapy using the VMAT technique up to total dose (TD) 20 Gy with daily dose (DD) 4 Gy was performed (Figure 6). Regarding liver and lung metastases, the patient underwent systemic chemotherapy.
Figure 4: Photomicrography of IHC expression: А/ СD 34- negative expression in tumor cells, but positive in the walls of blood vessels at the border of the tumor; В/ Myogenin A- negative expression in the nuclei of tumor cells.
Figure 6: Intensity Modulated Radiotherapy using the VMAT technique up to total dose (TD) 20 Gy with daily dose (DD) 4 Gy.
Discussion
Primary OLMS is a rare neoplasm of teratoma or non-teratoma
origin [10]. Non-teratoma OLMS is very rarely diagnosed and
is defined as “true” leiomyosarcoma [11]. Leiomyosarcomas of
teratoma origin are unilateral and occur at a young age, and nonteratomas
are diagnosed mainly in adults [3]. The pathogenesis of
OLMS is unclear, but theories include malignant degeneration of
smooth muscle cells in the mesonephrotic elements surrounding
the ovary and in the walls of blood vessels, or migration with
malignant transformation of uterine leiomyoma in the ovary
[1,2,3]. However, malignant transformation of leiomyoma is rarely
seen [3]. Migration of subserous uterine leimiosarcomas with
secondary ovarian involvement is possible [12]. Macroscopically,
tumors are unilateral ovarian solid masses with or without cystic
degeneration, depending on the size of the formation [5,8,9].
Microscopically, the tumor cells are spindle-shaped with abundant
eosinophilic cytoplasm and elongated nuclei [3] (Figure 1A).
These cells are arranged corolla-like (Figure 1C) with nests of
polymorphism, nuclear mitosis, and necrosis [7]. Often, sarcoma
cells are organized into tangled or intertwined bundles with areas
of pleomorphism [3]. IHC markers for the diagnosis of ovarian
leiomyosarcoma include desmin, vimentin, smooth muscle actin
(SMA), and S-100 [13,14]. We report the strongly positive IHC
expression of sarcoma cells to desmin and SMA, as well as focal
positive to S-100 (Figure 2). In OLMS, some authors reported
positive expression of estrogen receptors, progesterone receptors,
p53 and bcl-2 [1,13,15]. In the presented clinical case, estrogen and
progesterone receptors are negative (Figure 3A & 3B).
Differential Diagnosis (DD)
Pathomorphological and IHC analysis of OLMS to differentiate undifferentiated sarcomas, mixed Mueller tumors, sarcomoid form of sex-cord stromal tumors and Krukenberg tumors is required. Leiomyosarcomas are distinguished from benign tumors by hypercellularity and increased mitotic activity / above 10 on a field with magnification x10 [16]. DD for other rare ovarian sarcomas such as chondrosarcoma, malignant schwannoma, angiosarcoma, and rhabdomyosarcoma is required [5]. In the presented case we performed DD with the epithelial tumors of the ovary through the negative IHC expression of the tumor cells to Cytokeratin and CEA125 (Figure 3C & 3D), as well as with angiosarcoma and rhabdomyosarcoma through the negative IHC expression to CD 34 and Myogenin A (Figure 4A & 4B).
Prognosis
Primary OLMS were associated with an unfavorable prognosis [9,17]. In a large number of cases the disease has a poor prognosis with a fatal outcome one year after diagnosis [3]. In the presented clinical case we take into account the following prognostic adverse pathohistological characteristics: tumor formation with a diameter above 5cm, high mitotic activity (Ki67-17,4%), extensive areas with tumor necrosis, tumor capsule with growth from tumor cells.
Treatment
Due to the rare OLMS diagnosis, the standard treatment approach has not yet been specified [6,9]. The most commonly used treatments include total abdominal hysterectomy, bilateral salpingo-oophorectomy, excision of a residual tumor, and possibly total tumor resection [1]. The main treatment method is surgery, which varies from surgery to preserve fertility to total abdominal hysterectomy, bilateral salpingo-oophorectomy and removal of the sarcoma mass [8]. OLMS requires total tumor surgical resection, which significantly improves the prognosis [1,3,13,15,17-19]. In large tumor formations, Dixit et al recommend postoperative chemotherapy to improve local tumor control and to suppress distant metastasis, although their adjuvant effect has not been sufficiently demonstrated [8,13,19,20]. Shakfeh and Woodruff present 2 clinical cases of OLMS, of which one patient exited after 12 months without specific treatment, and the other has a 4-year survival after total hysterectomy with bilateral salpingooophorectomy followed by postoperative radiotherapy alone [21]. To increase survival in patients with advanced OLMS, maximally radical surgery, followed by combined chemo-radiotherapy as well as immunotherapy to prevent distant metastasis is required [3,6].
Conclusion
Primary ovarian leiomyosarcoma is an extremely rare aggressive systemic neoplasm. For diagnosis, strict pathomorphological and immunohistochemical analysis is required. In general, the prognosis is unfavorable. The main treatment is radical surgery. To assess the need for postoperative adjuvant chemo-radiotherapy, it is necessary to clarify a number of malignant pathohistological features. Improving survival in patients with early clinical stage requires complex treatment, including radical surgery followed by combined chemo-radiotherapy, as well as immunotherapy to prevent distant metastases.
References
- Bouie SM, Cracchiolo B, Heller D (2005) Epithelioid leiomyosarcoma of the ovary. Gynecol Oncol 97(2): 697-679.
- Kurian RR, Preethi J, Remadevi AV (2005) Leiomyosarcoma of ovary- a case report. Indian J Pathol Microbiol 48(1): 19-20.
- Dixit S, Singhal S, Baboo HA (1993) Leiomyosarcoma of the ovary. J Postgrad Med 39(3): 151-153.
- Anderson B, Turner DA, Benda J (1987) Ovarian sarcoma. Gynecol Oncol 26(2): 183-192.
- Rosai J (1989) Female reproductive system, ovary. In: Ed Ackerman's Surgical Pathology, vol 2, 7th edn Washington DC: CV Mosby Co pp. 1163.
- Asuka Tanaka, Ai Miyoshi, Serika Kanao (2018) Case Report of a Primary Ovarian Leiomyosarcoma Diagnosed by H-Caldesmon Staining. Journal of Clinical Gynecology and Obstetrics 7(1): 26-29.
- Friedman HD, Mazur M (1991) Primary ovarian leiomyosarcoma. An immuno-histochemical and ultrastructural study. Arch Pathol Lab Med 115(9): 941-945.
- J Vijaya Kumar, Anil Khurana, Paramjeet Kaur (2015) A rare presentation of primary leiomyosarcoma of ovary in a young woman. Ecancermedicalscience 9: 524.
- Elizabeth Jayne Goodall, Thumuluru Madhuri, Simon Butler Manuel (2011) The Management Dilemma of Leiomyosarcoma of the Ovary. World J Oncol 2(5): 265-266.
- Prat J, Fox H (1987) Mesenchymal tumour of the ovary. In: Fox H, editor. Hainjps and Taylor Obstetrics and Gynaecology, vol 2, 2nd edn. Edinburgh: Churchill Livingstone pp. 701.
- Won Sang Park, Seong Beom Lee, Jung Yong Lee (1996) Primary Ovarian Leiomyosarcoma: A case report. The Korean Journal of Pathology 30(6): 548-550.
- Novak E, Woodruff JD (1977) Novak's Gynecologic and Obstetric Pathology, Philadelphia: WB Saunders pp. 455.
- Taşkin S (2007) Primary ovarian leiomyosarcoma: A review of the clinical and immunohistochemical features of the rare tumor. Obstet Gynecol Surg 62(7): 480-488.
- Balaton A, Vaury P, Imbert MC (1987) Primary leiomyosarcoma of the ovary: a histological and immunocytochemical study. Gynecol Oncol 28(1): 116-1120.
- Rasmussen CC, JS Skilling, JI Sorosky, DJ Lager, RE Buller (1997) Stage IIIC Ovarian Leiomyosarcoma in a Premenopausal Woman with Multiple Recurrences: Prolonged Survival with Surgical Therapy. Gynecol Oncol 66(3): 519-525.
- Russel P, Banntype P, Solomen HJ (1992) Malignant Mullerian and miscellaneous mesenchymal tumours of the ovary, In: Copplesonn M, editor. Gynaecologic Oncology, vol 2, 2nd edn Edinburgh: Churchill Livingstone pp. 971.
- Arslan OS (2011) A rare tumor of the female genital tract: primary ovarian leiomyosarcoma. Arch Gynecol Obstet 283(Suppl 1): 83-85.
- Inoue J, Gomibuchi H, Minoura S (2000) A case of a primary ovarian leiomyosarcoma. J Obstet Gynaecol Res 26(6): 401-407.
- Dai Y (2011) Primary sarcoma of the ovary: clinicopathological characteristics, prognostic factors and evaluation of therapy. Chin Med J (Engl) 124(9): 1316-1321.
- Monk BJ, Nieberg R, Berek JS (1993) Primary leiomyosarcoma of the ovary in a perimenarchal female. Gynecol Oncol 48: 389-393.
- Shakfeh SM, Woodruff JD (1987) Primary Ovarian sarcoma, report of 46 cases and review of literature. Obstet Gynecol Survey 42: 331-349.

 Clinical Image
Clinical Image