Abstract
Background: APOE4 have been considered as the risk of AD, but the predict value of MCI progressing to AD was not clear.
Objective: The aim of the current study was to investigate the predictive-value of APOE4+ in the development of Mild Cognitive Impairment (MCI) to Alzheimer’s Disease (AD) using a meta-analysis.
Methods: At PubMed, Elsevier Science Direct, Scihub and Google Scholar we searched all the previous cohort studies on APOE4+ genotype associated with the risk of MCI-to-AD dementia, published before January 1, 2019. Stata Meta-DiSc (version 1.4) software was used to pool the APOE4+ prognosis data to examine the sensitivity, specificity and the summary receiver operating characteristic curve (SROC) in predicting the risk MCI-to-AD dementia; Stata software, to calculate the relative risk (RR) and 95% CIs.
Results: For the meta-analysis were involved 43 previously reported studies, where it was found that in MCI people aged ≥70, who had progressed to AD dementia within 5 years, the APOE4+ predictive sensitivity was 0.71; the specificity, 0.71; and AUC, 0.78. Moreover, the results showed that RR was 1.49 and 1.56, respectively, for MCI people in general and for MCI people aged ≥70 and with the risk of APOE4+-to-AD. Particularly, the RR was 2.24 for the individuals aged ≥70 and with APOE4+ who progressed to AD dementia within 5 years.
Conclusion: The findings strongly suggested that it could take less than 5 years for MCI people aged ≥70 and with the gene of APOE4 to progress to AD dementia.
Keywords: APOE; Alzheimer’s Disease; Mild Cognitive Impairment; Receiver Operating Characteristic Curve; Relative Risk
Introduction
Alzheimer’s Disease (AD) is a neurodegenerative disease which has high morbidity and mortality. The world Alzheimer’s report in 2018 showed that globally there were about 500 million patients afflicted with AD, with its mortality being approximately 4.5%, leading to one of the top five causes of death in the world [1,2]. Mild Cognitive Impairment (MCI) is a significant cognitive deficit, but not qualified as a dementia reference, while in older patients MCI progresses to dementia at a rate of 60-100% in 5-10 years [3,4]. Furthermore, a meta-analysis found that 5%-15% of amnestic MCI (aMCI) patients aged over 65 developed AD dementia every year; however, a part of MCI kept stable or even reverted to normal [5]. Therefore, it is important that we have a differentiation of the MCI which has a tendency to progress to AD, which is beneficial to clinical prevention and therapy. Cheng et al discovered that the cortical thinning in the temporal region reflected a cognitive change in the MCI patients, which could be of a useful prediction of MCI progressing to AD dementia [6]. A review provided a critical examination of MCI’s clinical concept, stressing an increased focus on the impact of Cerebrovascular Disease (CVD) and CVD risk during the prodromal period of AD dementia [7].
Many risk factors have been reported to be associated with AD such as Aβ and tau [8,9]. The other investigations have found ε4 allele of the apolipoprotein E gene (APOEε4+) as a risk genetic factor for AD dementia and MCI [10-12]. However, it was found to be a significant risk factor for AD dementia rather than for MCI [13]. A previously reported investigation on a Chinese MCI population discovered that both aMCI patients and normal ageing people who carried APOE ε4+ had a high risk of MCI progressing to AD dementia, the hazard ratio 2.0 and 5.3, respectively [14]; the differentiation indicated that the risk of progressing to AD dementia was lower in MCI with APOE ε4+ than in normal ageing with APOE ε4+. Given the relationship between APOE ε4+ and MCI/ AD, the predicting value of APOE ε4+ progressing to MCI or to AD dementia has become one of research focuses on the field recently [15,16]. But the contradictory results still exist based on different experiments. Elias-Sonnenschein and Li conducted a meta-analysis so as to assess the different ORs in different experiments on MCI patients with APOE ε4+ progressing to AD dementia [17,18], which pooled all the reports before 2008 and 2014, respectively. The review’s outcomes turned out to be that that those with MCI and APOEε4+ presented a high risk for AD dementia progression.
Oveisgharan S et al found the evidence that APOE ε2ε4 genotype in older adults was associated with MCI risk, as a greater burden of AD pathology [19]. On the contrary, APOE ε4+ was reported to be incapable of predicting the conversion MCI to AD dementia without using biomarkers [20], and APOE ε4+, to be not associated with the development of MCI and AD [21]. The diagnostic value of APOE ε4+ for MCI-to-AD dementia still remains unknown in terms of sensitivity and specificity. Thus, it is necessary that we reassess the diagnostic-value of the APOE ε4+ for MCI-to-AD. The aim of the current meta-analysis was to reassess the diagnostic and prognostic value of APOE ε4+ for MCI-to-AD dementia in different subgroups.
Methods
Search Strategy and Selection Criteria
The relevant literature ranging from January 1, 1987 to May 1, 2019 was systematically pursued at the PubMed, Elsevier Science Direct, Scihub and Google Scholar, with the searching key words as Apolipoprotein E/APOE, mild cognitive impairment/MCI, Alzheimer’s disease/AD/dementia. Some papers were traced via a reference link in the relevant literature.
Inclusion and Exclusion Criteria
The studies and investigations on the association of APOE ε4 allele with MCI progressing to AD dementia were included, the criteria of which were as follows:
a) Original studies
b) Reported in English
c) Petersen and co- workers criteria used for MCI [22],
NINCDS-ADRDA criteria for AD [23]
d) Complete description of the MCI group without AD
progress and of the MCI group with
e) APOE ε4 allele recorded
f) Case-control study or cohort study in nature
g) Full text available, or the requisite information from the
authors.
The studies and investigations were excluded if the study
sample included:
a) Abstract, literature review, case report, seminar
b) Other languages except English
c) Criteria of MCI and AD not explicitly described
d) Not clearly described MCI group with and without MCI-to-
AD dementia
e) Recorded APOE ε4 allele not well founded
f) Not a case-control study or a cohort study
g) Full text not available.
Literature Quality Assessment
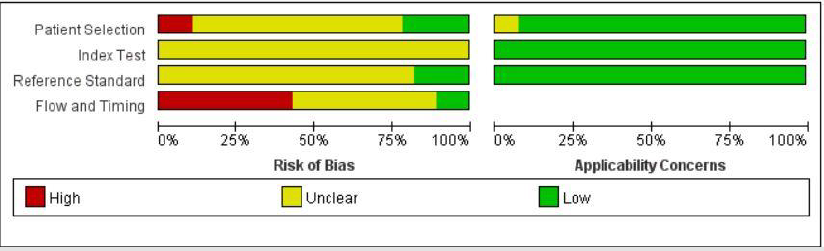
Each study was read by two coauthors (Juan Yang and Xiaohui Zhao), who would have a discussion over a contentious point, if any, before reaching a consensus. Study quality was assessed with the Quality Assessment of Diagnostic Accuracy Studies 2(QUADAS-2) for studies [15].
Data Extraction
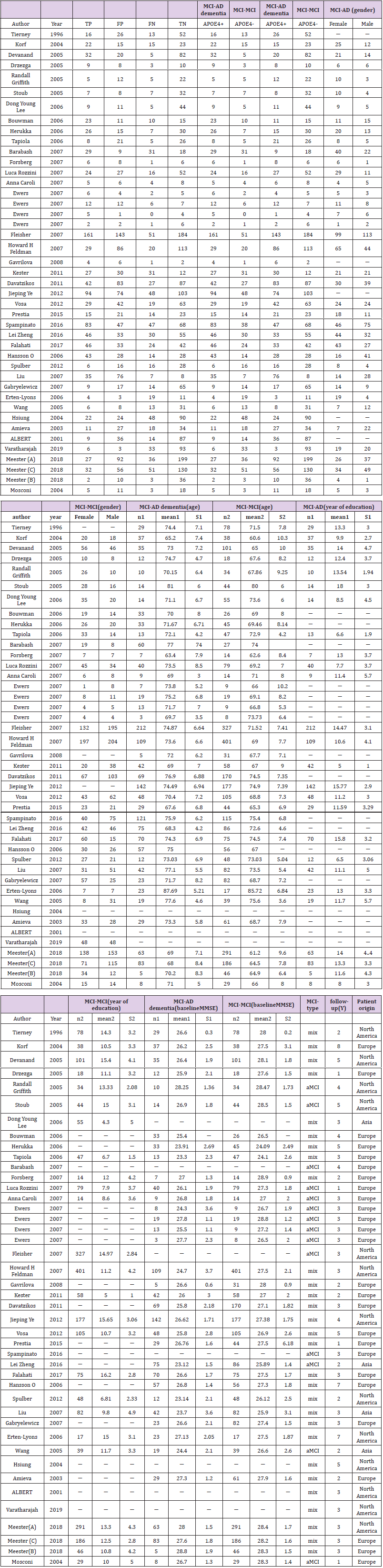
APOE with and withoutε4 allele was recorded as APOE4+ and APOE4-, respectively. NINCDS-ADRDA criteria for AD were considered as the goldenstandard, and APOEε4, as the diagnostic maker. The number of True Positive (TP), True Negative (TN), False Positive (FP) and False Negative (FN) were extracted from all the reports. Otherwise, tabulated were the authors, publication date, country of the study population, age, gender, MCI subtype, years of follow-up, baseline score of MMSE and years of education.
Statistical Analysis
Meta-DiSc (version 1.4, Unit of Clinical Biostatistics team of the Ramón Cajal Hospital in Madrid, Spain) software was used to analyze the sensitivity and specificity and the summary receiver operating characteristic curve (SROC). Sensitivity (TP rate) referred to the proportion of MCI to AD dementia, correctly identified as APOE4+; specificity (1-FP rate), to the proportion MCI to AD dementia, correctly identified as APOE4-. Heterogeneity was assessed using I² and Tau²; publication bias was analyzed and represented by a funnel plot, and funnel plot symmetry was assessed with Egger’s test [24]. Stata12.0 software (Stata Corp LP, College Station, TX, USA) was used to pool the relative risk (RR), and the RR and 95%CIs, to summarize the risk of MCI-to-AD with APOE4+. Statistical significance was defined as P < 0.05.
Results
Search Results
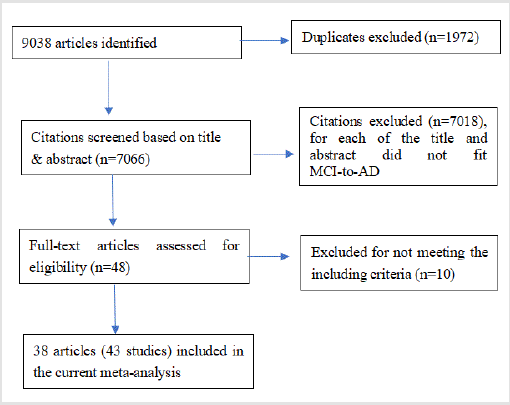
A total of 9038 studies were indexed in the primary screen and search, 1972 of which were duplicates. It was found from the screening that 7028 articles did not meet the relation of APOE ε4 allele to MCI-to-AD dementia to be excluded, and that 10 articles failed to meet our including criteria to be excluded. Consequently, a total of 43 studies [14,25-61] were enrolled for the current metaanalysis. As indicated in Figure 1, a full description was made of the search strategy.
Study Characteristics
As indicated in Table 1, a list was made of the characteristics of 38 articles included for the meta-analysis. A total of 5,040 subjects with MCI were characterized by a period of 1-8 year clinical follow-up observation, at the end of which, the number of TP, FP, FN and TN were 1,039, 1,221, 728 and 2,052, respectively. The mean age of MCI-AD dementia and MCI-MCI was 72.69±8.12 and 70.0±7.31 years, respectively, the females of MCI-AD dementia and MCI-MCI accounting for 51.11% and 54.36%, respectively. The mean educational years of MCI-AD dementia and MCI-MCI were 11.61±3.47 and 11.31±3.36, respectively. The mean baseline- MMSE-score of MCI-AD dementia and MCI-MCI was 26.12±1.94 and 27.28±1.94, respectively. Furthermore, MCI-classification in all studies was classified as aMCI and mix-MCI (not defined as aMCI). Three geographic regions were defined: China (including Taiwan) pertained to Asia; Netherlands/America, to North America; and Canada/Sweden/Italy/Germany, to Europe.
Literature Quality Assessment
Each of the 43 included studies was evaluated using the QUADAS-2. Review Manager 5.2 was used to assess the literature quality (Figure 2).
Meta-Analysis Results
With 38 articles enrolled for this meta-analysis, the predictive value of APOE4+ for MCI-to-AD dementia was analyzed as a whole as well as in different subgroups. In general, it was not statistically significant (sensitivity: 0.59, 95%CI: 0.56-0.61; specificity: 0.63, 95%CI: 0.61-0.64; AUC: 0.62; I squared: 55.8%; tau squared: 0.24).
APOE4+ and Ages
The predictive value of APOE4+ for MCI people aged ≥70 progressing to AD dementia was higher than that of those aged <70. (sensitivity: 0.60, 95%CI: 0.62-0.67; specificity: 0.60, 95%CI: 0.58- 0.62; AUC 0. 65; sensitivity 0.50, 95%CI: 0.45-0.55; specificity 0.64, 95%CI: 0.60-0.67; AUC: 0. 56, respectively).
APOE4+ and Progressing Interval Time
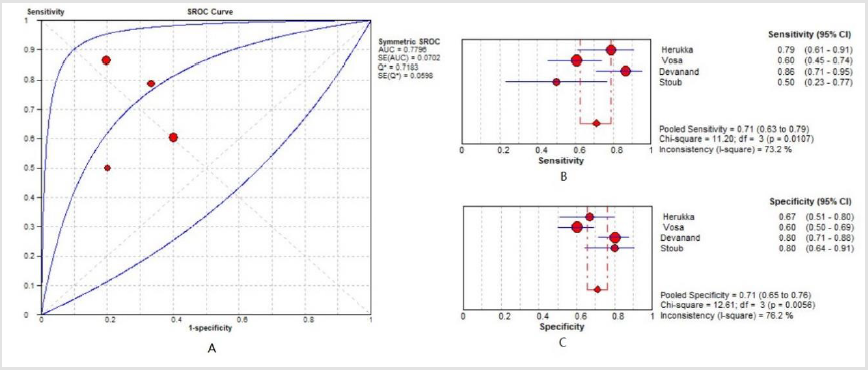
Figure 3: The diagnostic value of APOE4+ for MCI people aged 70 and with internal time of 5 years to AD dementia.
As indicated in Figure 3, it was 5 years as the interval time for MCI people with APOE4+ progressing to AD dementia, with SROC 0.78, the pooled sensitivity 0.57 (95%CI: 0.50-0.64) and specificity 0.73 (95%CI: 0.68-0.77). Furthermore, the predictive value of APOE4+ for MCI-to-AD was higher in MCI people aged ≥70 progressing to AD dementia in 5 years than that in other subgroups (sensitivity: 0.71, 95%CI: 0.63-0.79; specificity: 0.71, 95%CI: 0.65-0.76; AUC: 0.78). It was insignificant about other progressing intervals such as 1 year (sensitivity: 0.60, 95%CI: 0.49-0.70; specificity: 0.61, 95%CI: 0.53-0.68; AUC: 0.63); 2 years (sensitivity: 0.50, 95%CI: 0.43-0.56; specificity: 0.59, 95%CI: 0.54-0.64; AUC: 0.54); 3 years (sensitivity: 0.61, 95%CI: 0.57-0.64; specificity: 0.62, 95%CI: 0.60-0.64; AUC: 0.63); 4 years (sensitivity: 0.62, 95%CI: 0.55, 0.68; specificity: 0.60, 95%CI: 0.54-0.66; AUC: 0.64); and over 5 years (sensitivity: 0.59, 95%CI: 0.50, 0.68; specificity: 0.61, 95%CI: 0.48-0.67; AUC: 0.63). Legend: APOE4+ diagnostic value for MCI people aged ≥70 who trended to progress to AD at the time interval of five years (A: AUC; B: sensitivity; C: specificity).
APOE4+ and Geographic Area
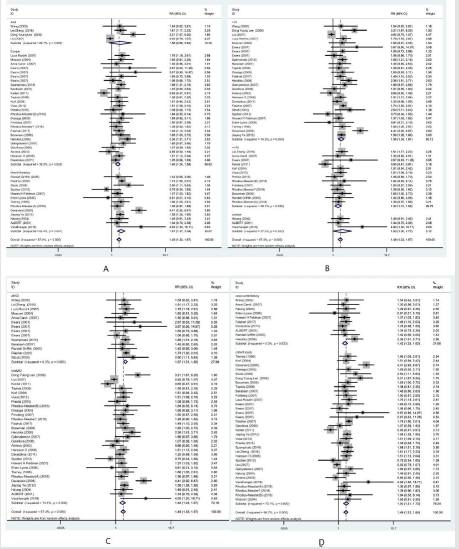
Among different geographic areas the predictive-value of APOE4+ for MCI-to-AD dementia was significantly. In Asia, the sensitivity was 0.64, 95%CI, 0.55-0.71; specificity, 0.52, 95%CI, 0.46-0.58; and AUC, 0. 63. In North America, the sensitivity was 0.57, 95%CI, 0.53-0.61; specificity, 0.67, 95%CI, 0.65-0.69; and AUC, 0. 67. In Europe, the sensitivity was 0.59, 95%CI, 0.56-0.63; specificity, 0.60, 95%CI, 0.57-0.62; and AUC, 0. 62.
APOE4+ and MCI Type
The predictive value of APOE4+ was higher in aMCI than in mix- MCI progressing to AD dementia (sensitivity: 0.66, 95%CI: 0.62- 0.69; specificity: 0.61, 95%CI: 0.58-0.65; AUC: 0. 67, vs. sensitivity: 0.55, 95%CI: 0.52-0.58; specificity: 0.63, 95%CI: 0.61-0.65; AUC: 0.62).
APOE4+ and Educational Years
The predictive value of APOE4+ for MCI-to-AD dementia was higher in those with ≥12 years of education than those with <12 years of education (sensitivity: 0.63, 95%CI: 0.59-0.66; specificity: 0.64, 95%CI: 0.62-0.67; AUC: 0. 67, vs. sensitivity: 0.56, 95%CI: 0.51-0.61; specificity: 0.56, 95%CI: 0.53-0.59; AUC: 0. 58).
APOE4+ and Multi-Factors
As to APOE4+, the predictive value of MCI-to-AD dementia was not improved when plus all relative predictive factors, such as MCI people aged ≥70 plus being North American (sensitivity: 0.66, 95%CI: 0.62-0.70; specificity: 0.61, 95%CI: 0.58-0.64; AUC: 0. 68); MCI people aged ≥70 plus being of aMCI type (sensitivity: 0.67, 95%CI: 0.62-0.71; specificity: 0.61, 95%CI: 0.57-0.65; AUC: 0. 68); and MCI people aged ≥70 plus ≥12 years of education (sensitivity: 0.68, 95%CI: 0.64-0.72; specificity: 0.62, 95%CI: 0.59-0.65; AUC: 0. 70).
The Risk of APOE4+ for MCI -to -AD
As indicated by the results, an association was observed between APOE4+ and MCI-to-AD dementia (RR: 1.49; 95%CI: 1.33- 1.67). The subgroup analysis showed that the heterogeneity was clinic heterogeneity caused by age, geographicarea, MCI type and research design (Figure 4). In the age subgroup, those who were aged ≥70 (RR: 1.56; 95%CI: 1.36-1.78) had a higher risk than those who were aged <70 (RR:1.25; 95%CI: 0.99-1.59). As to the progressing interval time, different intervals were found to be correlated with the different risks of APOE4+ for MCI-to-AD dementia. The risk was higher at the time interval of 5 years (RR: 2.24; 95%CI: 1.43-3.50) than the others such as 1 year (RR: 1.50; 95%CI: 1.17-1.93); 2 years (RR:1.13; 95%CI: 0.83-1.55); 3 years (RR:1.47, 95%CI: 1.25-1.74);4 years (RR:1.57; 95%CI:1.31-1.88); 7 years (RR:1.46, 95%CI: 1.09- 1.96); and 8 years (RR:1.51; 95%CI: 0.94-2.42). As indicated by the results of APOE4+ in the geographic area subgroup, Asian’s RR was 1.58, and 95%CI, 0.85-2.92; North American’s RR was 1.67, and 95%CI, 1.37-2.04; and European’s RR was 1.40, and 95%CI, 1.24- 1.58. In the MCI type subgroup, aMCI’s RR was 1.67, and 95%CI, 1.51-1.85; and mix-MCI’s RR was 1.44, and 95%CI, 1.24-1.67. In the education-year subgroup, ≥12years of education showed that the RR was 1.69, and 95%CI, 1.40-2.04, while <12 years of education showed that the RR was 1.32, and 95%CI, 1.06-1.63. In the research design subgroup, the case-control studies showed that the RR was 1.42, and 95%CI, 1.25-1.62, while the cohort studies indicated that the RR was 1.50, and 95%CI, 1.31-1.73. Legend: A. demonstrated subgroup (European, Asia and North American), B. demonstrated age subgroup (≥70 & <70), C. demonstrated subgroup (aMCI & mixMCI), C. demonstrated research design subgroup (case-control & cohort studies). To all case-control studies was applied OR, RR the estimator for OR.
Publication Bias
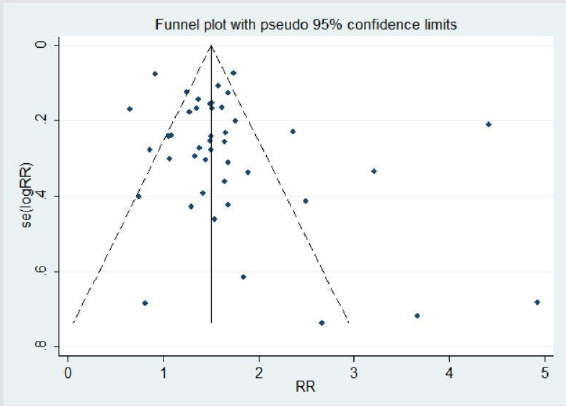
The funnel plots appeared to be symmetric, showing no evidence of publication bias sensitivity analysis. The Begg’s test results indicated that the Kendall’s score(P-Q)=95, Z=0.99, and P=0.32, and Egger’s test results showed P=0.11, both of them indicating no publication bias (Figure 5).
Discussion
Although there were two meta-analyses [17-18] which had calculated the effect of APOE4+ on MCI-to-AD dementia, the results were significantly different between the two studies, with the OR 2.29 (95% CI: 1.88-2.80) vs. 1.84 (95%CI:1.59-2.14). No metaanalysis was reported on the predictive value of APOE4+ for MCto- AD. In the current study, we analyzed the predictive value of APOE4+ for MCI-to-AD dementia in different subgroups. Although as a whole the predictive value of APOE4+ for MCI-to-AD was not statistically significant, it was in the different subgroups. In the MCI people aged ≥70, APOE4+ had a high predictive value for MCI-to- AD dementia. Previous studies [19,62,63] had indicated that there was a correlation between APOE ε4+ and cognition decline in older people, but the exact age was unclear. The results of our metaanalysis indicated that the age of 70 was the cut-off point, and that the APOE4+ predictive sensitivity was high in those who were aged ≥70, which suggested that APOE4+ was valuable for MCI people aged ≥70 to predict the progression of MCI to AD dementia.
In the current study, we discovered that predictive value of
APOE4+ for MCI-to-AD was significantly different in different
geographic areas, as indicated by the evidence that the risk of
MCI-to-AD dementia was higher in North America than in Europe
and Asia. The difference was reported to be caused by APOE gene
which variated among different geographic regions. APOE4+
showed a more significant increasing tendency in North European
populations than in Asian and Oceanian ones [64,65], which
suggested that APOE4+ could be more valuable for MCI people in
North America to predict MCI-to-AD dementia. It was also found
that the predictive value of APOE4+ for aMCI progressing to AD
dementia was high. Some literatures have demonstrated that aMCI
is a high risk of MCI-to-AD dementia [64,66], which agreed with
our conclusion. In fact, some studies [67-69] have testified that
APOE4+ is correlative with aMCI, likely to modulate the large-scale
brain network in aMCI subjects, as elucidated in a recently study
reporting that the risk of memory decline was associated with Aβ
and APOE4+ at each age. Therefore, our findings suggested that
APOE4+ was more valuable for aMCI people to predict MCI-to-AD
dementia. Moreover, the predictive value of APOE4+ for MCI-to-AD
dementia was high in those who had ≥12 years of education. Some
studies [70,71] have found that a higher education level may delay the progression of MCI to AD dementia. Our findings indicated that
the predictive value of APOE4+ was high in MCI people with more
years of education. Although a previously reported study found that
APOE ɛ4 might not be associated with years of education [72], our
findings still suggested that APOE4+ was valuable for MCI people
who had ≥12 years of education to predict MCI-to-AD dementia.
Furthermore, we found that the time interval for MCI-to-AD
dementia could be 5 years, especially for MCI people with APOE4+
and aged ≥70. The risk estimated by RR of APOE4+ for MCI-to-AD
dementia changed at different time intervals, with the highest at the
5th year, which suggested that APOE4+ was more valuable for MCI
people aged ≥70 to predict MCI-to-AD dementia at the time interval
of 5 years. Additionally, the predictive value of APOE4+ showed
no significance for MCI-to-AD dementia when the multiple-factor
superposition involved MCI people aged ≥70 plus being North
American, or plus being aMCI, or plus ≥12 years of education. This
suggests that a part of variable may not help make the incremental
effect when we predict the progression of MCI to AD dementia by a
multiple-factor superposition.
Limitations
There was high heterogeneity in our meta-analysis, which needs to be overcome with a bigger collection of relevant literatures in the future. Additionally, the high risk of bias in literature quality is a limitation, too.
Conclusion
The predictive value of APOE4+ for MCI-to-AD dementia was valuable for MCI people in North America, the subtype of aMCI and ≥12 years of education. As a strong implication, it may take less than 5 years for MCI people aged ≥70, who carried the gene of APOE4+, to progress to AD dementia (sensitivity: 0.71, 95%CI: 0.63-0.79; specificity: 0.71, 95%CI: 0.65-0.76, AUC: 0.78).
Conflict of Interest
The authors declare no conflict of interests.
Disclosure Statement
The authors have no actual or potential conflicts of interest.
Acknowledgement
This study was supported by Science and Technology Development Fund of Shanghai Pudong New Area (No. PKJ2018-Y12) and Science and Technology Development Fund of Shanghai (No. PW2018B-58) and Training Program for Academic Pioneers in Pudong New Area (No. PWRd2018-15).
Author Contribution
ZL, WXQ conceived and designed the project. YJ, XHZ collected data. YJ wrote the manuscript. YJ, LXY and SHJ prepared Figures 1-5. All authors reviewed the manuscript.
References
- Aisen PS, Cummings J, Jack CR, Morris JC, Sperling R, et al. (20147) On the path to 2025: understanding the Alzheimer's disease continuum[D]. Alzheimers Res Ther 9(1): 60.
- Arora P, Boyne D, Slater JJ, Gupta A, Brenner DR (2019) Bayesian Networks for Risk Prediction Using Real-World Data: A Tool for Precision Medicine. Value Health 22(4): 439-445.
- Shen T, Li Y, Wu P, Zuo C, Yan Z (2018) Decision Supporting Model for One-year Conversion Probability from MCI to AD using CNN and SVM. Annu Int Conf IEEE Eng Med Biol Soc 2018: 738-741.
- Xue J, Li J, Liang J, Chen S (2018) The Prevalence of Mild Cognitive Impairment in China: A Systematic Review. Aging Dis 9(4): 706-715.
- Sanford AM (2017) Mild Cognitive Impairment. Clin Geriatr Med 33(3): 325-337.
- Cheng CP, Cheng ST, Tam CW, Chan WC, Chu WC (2018) Relationship between Cortical Thickness and Neuropsychological Performance in Normal Older Adults and Those with Mild Cognitive Impairment. Aging Dis 9(6): 1020-1030.
- Bondi MW, Jak AJ, Delano Wood L, Jacobson MW, Delis DC, Salmon DP (2008) Neuropsychological contributions to the early identification of Alzheimer's disease. Neuropsychol Rev 18(1): 73-90.
- Chen TB, Lee YJ, Lin SY, Chen JP, Hu CJ, et al. (2019) Plasma Aβ42 and Total Tau Predict Cognitive Decline in Amnestic Mild Cognitive Impairment. Sci Rep 9(1):13984.
- Peña Bautista C, Vigor C, Galano JM, Oger C, Durand T, et al. (2019) New screening approach for Alzheimer's disease risk assessment from urine lipid peroxidation compounds. Sci Rep 9: 14244.
- Al Obeidi E, Al Tahan S, Surampalli A, Goyal N, Wang AK, et al. (2018) Genotype-phenotype study in patients with valosin-containing protein mutations associated with multisystem proteinopathy. Clin Genet 93(1): 119-125.
- Shi Z, Yu H, Wu Y, Ford M, Perschon C, et al. (2019) Genetic risk score modifies the effect of APOE on risk and age onset of Alzheimer's disease. Clin Genet 95(2): 302-309.
- Veiga S, Rodríguez Martín A, Garcia Ribas G, Arribas I, Menacho Román M (2020) Validation of a novel and accurate ApoE4 assay for automated chemistry analyzers. Sci Rep 10: 2138.
- Hendrie HC, Murrell J, Baiyewu O, Lane KA, Purnell C, et al. (2014) APOE ε4 and the risk for Alzheimer disease and cognitive decline in African Americans and Yoruba. Int Psychogeriatr 26(6): 977-985.
- Wang PN, Hong CJ, Lin KN, Liu HC, Chen WT (2011) APOE ε4 increases the risk of progression from amnestic mild cognitive impairment to Alzheimer's disease among ethnic Chinese in Taiwan. J Neurol Neurosurg Psychiatry 82(2): 165-169.
- Hojjati SH, Ebrahimzadeh A, Khazaee A, Babajani Feremi A (2017) Alzheimer’s Disease Neuroimaging Initiative. Predicting conversion from MCI to AD using resting-state fMRI, graph theoretical approach and SVM. J Neurosci Methods 282: 69-80.
- Shen T, Jiang J, Li Y, Wu P, Zuo C (2018) Decision Supporting Model for One-year Conversion Probability from MCI to AD using CNN and SVM. Conf Proc IEEE Eng Med Biol Soc 2018: 738-741.
- Elias Sonnenschein LS, Viechtbauer W, Ramakers IH, Verhey FR, Visser PJ (2011) Predictive value of APOE-ε4 allele for progression from MCI to AD-type dementia: a meta-analysis. J Neurol Neurosurg Psychiatry 82: 1149-1156.
- Li JQ, Tan L, Wang HF, Tan MS, Tan L, et al. (2016) Risk factors for predicting progression from mild cognitive impairment to Alzheimer's disease: a systematic review and meta-analysis of cohort studies. J Neurol Neurosurg Psychiatry 87(5): 476-484.
- Oveisgharan S, Buchman AS, Yu L, Farfel J, Hachinski V, et al. (2018) APOE ε2ε4 genotype, incident AD and MCI, cognitive decline, and AD pathology in older adults. Neurology 90(24): e2127-e2134.
- Gupta Y, Lama RK, Kwon GR (2019) Alzheimer's Disease Neuroimaging Initiative. Prediction and Classification of Alzheimer's Disease Based on Combined Features from Apolipoprotein-E Genotype, Cerebrospinal Fluid, MR, and FDG-PET Imaging Biomarkers. Front Comput Neurosci 13: 72.
- Banning L, Ramakers I, Deckers K, Verhey F, Aalten P (2019) Apolipoprotein E and affective symptoms in mild cognitive impairment and Alzheimer's disease dementia: A systematic review and meta-analysis. Neurosci Biobehav Rev 96: 302-315.
- Petersen RC, Morris JC (2005) Mild cognitive impairment as a clinical entity and treatment target. Arch Neurol 62(7): 1160-1163.
- Yazar T, Olgun Yazar H (2019) The prevalence of sarcopenia and dynapenia according to stage among Alzheimer-type dementia patients. Ideggyogy Sz 72(5-6): 171-179.
- Altobelli E, Del Negro V, Angeletti PM, Latella G (2017) Low-FODMAP Diet Improves Irritable Bowel Syndrome Symptoms: A Meta-Analysis. Nutrients 9(9): 940.
- Albert Ms, Moss Mb, Tanzi R, Jones K (2001) Preclinical prediction of AD using neuropsychological tests. J Int Neuropsychol Soc 7(5): 631-639.
- Amieva H, Letenneur L, Dartigues JF, Rouch Leroyer I, Sourgen C, et al. (2004) Annual rate and predictors of conversion to dementia in subjects presenting mild cognitive impairment criteria defined according to a population-based study. Dement Geriatr Cogn Disord 18(1): 87-93.
- Barabash A, Marcos A, Ancín I, Vázquez Alvarez B, Gil P, et al. (2009) APOE, ACT and CHRNA7 genes in the conversion from amnestic mild cognitive impairment to Alzheimer's disease. Neurobiol Aging. 30(8): 1254-1264.
- Bouwman FH, Schoonenboom SN, van der Flier WM, Kok A, Barkhof F, et al. (2007) CSF biomarkers and medial temporal lobe atrophy predict dementia in mild cognitive impairment. Neurobiol Aging 28(7): 1070-1074.
- Caroli A, Testa C, Geroldi C, Nobili F, Barnden LR, et al. (2007) Cerebral perfusion correlates of conversion to Alzheimer's disease in amnestic mild cognitive impairment. J Neurol 254(12): 1698-1707.
- Davatzikos C, Bhatt P, Shaw LM, Batmanghelich KN, Trojanowski JQ (2001) Prediction of MCI to AD conversion, via MRI, CSF biomarkers, and pattern classification. Neurobiol Aging 32(12): e19-27.
- Devanand DP, Pradhaban G, Liu X, Khandji A, De Santi S, et al. (2007) Hippocampal and entorhinal atrophy in mild cognitive impairment: prediction of Alzheimer disease. Neurology 68(11): 828-836.
- Drzezga A, Grimmer T, Riemenschneider M, Lautenschlager N, Siebner H, et al. (2005) Prediction of individual clinical outcome in MCI by means of genetic assessment and (18)F-FDG PET. J Nucl Med 46(10): 1625-1632.
- Erten Lyons D, Howieson D, Moore MM, Quinn J, Sexton G, et al. (2006) Brain volume loss in MCI predicts dementia. Neurology 66(2): 233-235.
- Ewers M, Buerger K, Teipel SJ, Scheltens P, Schröder J, et al. (2007) Multicenter assessment of CSF-phosphorylated tau for the prediction of conversion of MCI. Neurology 69(24): 2205-2212.
- Falahati F, Ferreira D, Muehlboeck JS, Eriksdotter M, Simmons A, et al. (2017) Monitoring disease progression in mild cognitive impairment: Associations between atrophy patterns, cognition, APOE and amyloid. Neuroimage Clin 16: 418-428.
- Feldman HH, Ferris S, Winblad B, Sfikas N, Mancione L, et al. (2007) Effect of rivastigmine on delay to diagnosis of Alzheimer's disease from mild cognitive impairment: the InDDEx study. Lancet Neurol 6(6): 501-512.
- Fleisher AS, Sowell BB, Taylor C, Gamst AC, Petersen RC, et al. (2007) Clinical predictors of progression to Alzheimer disease in amnestic mild cognitive impairment. Neurology 68(19): 1588-1595.
- Forsberg A, Engler H, Almkvist O, Blomquist G, Hagman G, et al. (2008) PET imaging of amyloid deposition in patients with mild cognitive impairment. Neurobiol Aging 29(10): 1456-1465.
- Gabryelewicz T, Styczynska M, Luczywek E, Barczak A, Pfeffer A, et al. (2007) The rate of conversion of mild cognitive impairment to dementia: predictive role of depression. Int J Geriatr Psychiatry 22(6): 563-567.
- Gavrilova SI, Fedorova YB, Roshchina IF, Korovaitseva GI (2008) Prognosis of mild cognitive impairment syndrome: data from a two-year clinical follow-up study. Neurosci Behav Physiol 38(2): 129-134.
- Griffith HR, Netson KL, Harrell LE, Zamrini EY, Brockington JC (2006) Amnestic mild cognitive impairment: diagnostic outcomes and clinical prediction over a two-year time period. J Int Neuropsychol Soc 12(2): 166-175.
- Hansson O, Zetterberg H, Buchhave P, Londos E, Blennow K (2006) Association between CSF biomarkers and incipient Alzheimer's disease in patients with mild cognitive impairment: a follow-up study. Lancet Neurol 5(3): 228-234.
- Herukka SK, Helisalmi S, Hallikainen M, Tervo S, Soininen H (2007) CSF Abeta42, Tau and phosphorylated Tau, APOE epsilon4 allele and MCI type in progressive MCI. Neurobiol Aging 28(4): 507-514.
- Hsiung GY, Sadovnick AD, Feldman H (2004) Apolipoprotein E epsilon4 genotype as a risk factor for cognitive decline and dementia: data from the Canadian Study of Health and Aging. CMAJ 171(8): 863-867.
- Kester MI, Verwey NA, van Elk EJ, Blankenstein MA, Scheltens P (2011) Progression from MCI to AD: predictive value of CSF Aβ42 is modified by APOE genotype. Neurobiol Aging 32(8): 1372-1378.
- Korf ES, Wahlund LO, Visser PJ, Scheltens P (2004) Medial temporal lobe atrophy on MRI predicts dementia in patients with mild cognitive impairment. Neurology 63(1): 94-100.
- Lee DY, Youn JC, Choo IH, Kim KW, Jhoo JH, et al. (2006) Combination of clinical and neuropsychologic information as a better predictor of the progression to Alzheimer disease in questionable dementia individuals. Am J Geriatr Psychiatry 14(2): 130-138.
- Liu HC, Wang PN, Wang HC, Lin KN, Hong CJ, et al. (2007) Conversion to dementia from questionable dementia in an ethnic Chinese population. J Geriatr Psychiatry Neurol 20(2): 76-83.
- Mosconi L, Perani D, Sorbi S, Herholz K, Nacmias B, et al. (2004) MCI conversion to dementia and the APOE genotype: a prediction study with FDG-PET. Neurology 63(12): 2332-2340.
- Prestia A, Caroli A, Wade SK, van der Flier WM, Ossenkoppele R, et al. (2015) Prediction of AD dementia by biomarkers following the NIA-AA and IWG diagnostic criteria in MCI patients from three European memory clinics. Alzheimers Dement 11(10): 1191-1201.
- Rhodius Meester H, Liedes H, Koikkalainen J, Wolfsgruber S, Coll Padros N, et al. (2018) Computer-assisted prediction of clinical progression in the earliest stages of AD. Alzheimers Dement (Amst) 10: 726-736.
- Rozzini L, Chilovi BV, Conti M, Bertoletti E, Delrio I, et al. (2007) Conversion of amnestic Mild Cognitive Impairment to dementia of Alzheimer type is independent to memory deterioration. Int J Geriatr Psychiatry 22(12): 1217-1222.
- Spampinato MV, Langdon BR, Patrick KE, Parker RO, Collins H, et al. (2016) Gender, apolipoprotein E genotype, and mesial temporal atrophy: 2-year follow-up in patients with stable mild cognitive impairment and with progression from mild cognitive impairment to Alzheimer’s disease. Neuroradiology 58(11): 1143-1151.
- Spulber G, Niskanen E, Macdonald S, Kivipelto M, Padilla DF, et al. (2012) Evolution of global and local grey matter atrophy on serial MRI scans during the progression from MCI to AD. Curr Alzheimer Res 9(4): 516-524.
- Stoub TR, Bulgakova M, Leurgans S, Bennett DA, Fleischman D, et al. (2005) MRI predictors of risk of incident Alzheimer disease: a longitudinal study. Neurology 64(9): 1520-1524.
- Tapiola T, Pennanen C, Tapiola M, Tervo S, Kivipelto M, et al. (2008) MRI of hippocampus and entorhinal cortex in mild cognitive impairment: a follow-up study. Neurobiol Aging 29(1): 31-38.
- Tierney MC, Szalai JP, Snow WG, Fisher RH, Tsuda T, et al. (1996) A prospective study of the clinical utility of ApoE genotype in the prediction of outcome in patients with memory impairment. Neurology 46(1): 149-154.
- Varatharajah Y, Ramanan VK, Iyer R, Vemuri P (2019) Alzheimer’s Disease Neuroimaging Initiative. Predicting Short-term MCI-to-AD Progression Using Imaging, CSF, Genetic Factors, Cognitive Resilience, and Demographics. Sci Rep 9: 2235.
- Vos S, Van RI, Burns L, Knol D, Scheltens P, et al. (2012) Test sequence of CSF and MRI biomarkers for prediction of AD in subjects with MCI - Neurobiology of Aging. 33(10): 2272-2281.
- Ye J, Farnum M, Yang E, Verbeeck R, Lobanov V, et al. (2012) Sparse learning and stability selection for predicting MCI to AD conversion using baseline ADNI data. BMC Neurol 12: 46.
- Zheng L, Kong X, Cui Y, Wei Y, Zhang J (2016) Conversion from MCI to AD in patients with the APOE ε4 genotype: Prediction by plasma HCY and serum BDNF. Neurosci Lett 626: 19-24.
- Jia J, Wang F, Wei C, Zhou A, Jia X, et al. (2014) The prevalence of dementia in urban and rural areas of China. Alzheimers Dement 10(1): 1-9.
- Rao D, Luo X, Tang M, Shen Y, Huang R, et al. (2018) Prevalence of mild cognitive impairment and its subtypes in community-dwelling residents aged 65 years or older in Guangzhou, China. Arch Gerontol Geriatr 75: 70-75.
- Di Domenico F, Pupo G, Giraldo E, Lloret A, Badia MC, et al. (2016) Autoantibodies Profile in Matching CSF and Serum from AD and aMCI patients: Potential Pathogenic Role and Link to Oxidative Damage. Curr Alzheimer Res 13(2): 112-122.
- Singh PP, Singh M, Mastana SS (2006) APOE distribution in world populations with new data from India and the UK. Ann Hum Biol 33(3): 279-308.
- Chasles MJ, Tremblay A, Escudier F, Lajeunesse A, Benoit S, et al. (2019) An Examination of Semantic Impairment in Amnestic MCI and AD: What Can We Learn from Verbal Fluency? Arch Clin Neuropsychol. 35(1): 22-30.
- Chen GQ, Sheng C, Li YX, Yu Y, Wang XN, et al. (2016) Neuroimaging basis in the conversion of aMCI patients with APOE-ε4 to AD: study protocol of a prospective diagnostic trial. BMC Neurol 16: 64.
- Lim YY, Kalinowski P, Pietrzak RH, Laws SM, Burnham SC, et al. (2018) Association of β-Amyloid and Apolipoprotein E ε4 With Memory Decline in Preclinical Alzheimer Disease. JAMA Neurol 75(4): 488-494.
- Wang L, Pan X, Fei G, Wang C, Wan W, et al. (2019) Decreased Function of Delayed Recall in Non-demented Elderly Subjects with Apolipoprotein E ε4 Allele. Front Aging Neurosci 11: 71.
- Garibotto V, Borroni B, Kalbe E, Herholz K, Salmon E, et al. (2008) Education and occupation as proxies for reserve in aMCI converters and AD: FDG-PET evidence. Neurology 71(17): 1342-1349.
- Hoenig MC, Bischof GN, Onur ÖA, Kukolja J, Jessen F, et al. (2019) Level of education mitigates the impact of tau pathology on neuronal function. Eur J Nucl Med Mol Imaging 46(9): 1787-1795.
- Ramanan VK, Castillo AM, Knopman DS, Graff Radford J, Lowe VJ, et al. (2019) Association of Apolipoprotein E ɛ4, Educational Level, and Sex with Tau Deposition and Tau-Mediated Metabolic Dysfunction in Older Adults. JAMA Netw Open 2(10): e1913909.

 Research Article
Research Article