Abstract
Objectives: To assess the incidence of pressure ulcers associated with lower extremity fractures splinted in a pediatric emergency department (ED) and to identify factors involved in pressure ulcer development.
Methods: A retrospective chart review of 249 pediatric patients’ charts from 2016- 2019 conducted at a level 2 trauma pediatric ED. Inclusion criteria included patients who had any lower extremity injury splinted in a regional ED with follow-up with a pediatric orthopedist at our institution. Patients over the age of 11.0 years old at the time of injury were excluded. Variables gathered included age, weight, gender, mechanism of injury, bone(s) fractured and location of fracture, whether they were seen at our pediatric hospital ED or an outside ED, time from injury to orthopedic specialist visit, pressure ulcer location and grading, method of splinting and patient comorbidities. Statistical analyses were performed using SPSS version 22.0 (IBM).
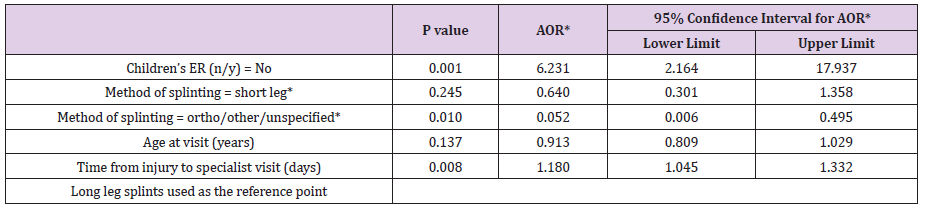
Results: Univariate analysis showed age, weight, time from injury to specialist visit, and the number of days in a splint were highly correlated to the occurrence of pressure ulcers. Multiple logistic regression demonstrated that for every 1-day increase in time from initial injury to the follow-up orthopedic visit, the odds of a pressure ulcer increased by 18% (p <0.05), and that patients who received their splint at an outside ED were 6.23 times likely to develop pressure ulcers.
Conclusion: Splinting of the lower extremity injuries at an outside, more rural ED and increased time to specialist follow-up were both correlated with increased rates of pressure ulcer formation.
Short Communication
A common complication of leg immobilization via splinting is ulcer formation. Ulcer formation is caused by several interrelated factors, including moisture, friction, temperature, shear forces, and pressure [1-3]. Ulcers most commonly form over prominent bony surfaces, putting the medial/lateral malleoli and heel at risk with lower extremity splints. Inadequate padding over bony prominences resulted in increased focal cast pressure. If cast pressure exceeds local capillary pressure it will decrease perfusion of the local tissues, eventually leading to tissue death and necrosis [4-7]. Certain medical conditions can contribute to the development of pressure ulcers when mobility and sensation are inhibited. Conditions such as cerebral palsy, paraplegia, or pediatric patients being treated in the intensive care unit have been shown to increase the risk of pressure sore development [8-10].
Management of such patients requires routine movement and body positioning to avoid pressure ulcer development. In spite of the advances in medicine and technology the prevalence and incidence of pressure ulcers continues to affect health outcomes and increase healthcare costs. Pressure ulcers can vary significantly in severity and are graded on a scale of I-IV, with stage I ulcers involving intact skin with non-blanchable erythema and stage IV ulcers involving exposure of underlying connective tissue, including bone, muscle or tendon. The average cost of treatment incurred by a stage IV ulcer and its complications is almost $130,000 [1]. Current literature shows that pressure ulcer management has a significant impact on healthcare costs for a seemingly preventable ailment [11-13]. This study examines the incidence of pressure ulcer formation among pediatric patients who have been placed in a lower extremity splint, with the goal of identifying modifiable factors that can be addressed to reduce the risk of pressure ulcer formation.
Methods
A retrospective chart review of 249 pediatric patients’ charts from 2016-2019 was conducted utilizing a level 2 trauma pediatric trauma center’s electronic medical record. Patients who had a lower extremity injury splinted in any ED and followed up with a pediatric orthopedist at our pediatric institution were included. Patients over the age of 11.0 years old at the time of injury were excluded as these patients were hypothesized to be able to more appropriately specify prodromal pain before an ulcer formed and therefore skew the incidence. Patients were identified using ICD 10 codes ranging from S82.301-S89.149 for lower extremity fractures in the ED. Variables recorded included age, weight, gender, mechanism of injury, bone(s) fractured and location of fracture, whether they were seen in the pediatric ED or an outside ED, time from injury to orthopedic specialist visit, time in splint before seeing the orthopedic specialist, pressure ulcer location and grading, and method of splinting.
Patients were noted as having a pressure ulcer if chart review revealed mention of a “pressure ulcer” or “pressure sore” in any physician notes or if the presence of a pressure ulcer was otherwise described in the notes in any way. Grading of pressures sores was performed using a standard four stage grading system employed by many clinicians. An example being an area of “nonblanchable erythema” over a boney prominence consistent with a stage 1 pressure ulcer. Statistical analyses were performed using SPSS version 22.0 (IBM) with a priori significance level of α = 0.05, Bonferroni-corrected for multiple comparisons on the same data set. Categorical variables were compared by either Chi-Square/ Fisher Exact Test while nonparametric data was analyzed using the Kruskal-Wallis Test with multiple comparisons.
Results
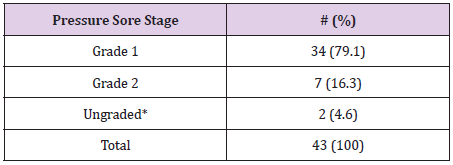
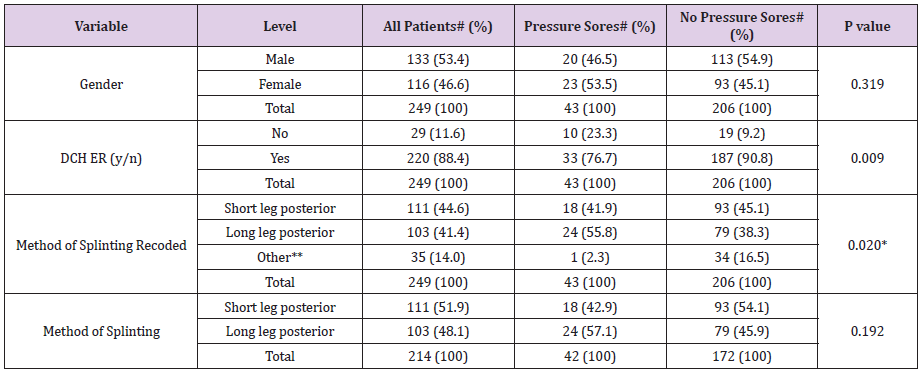
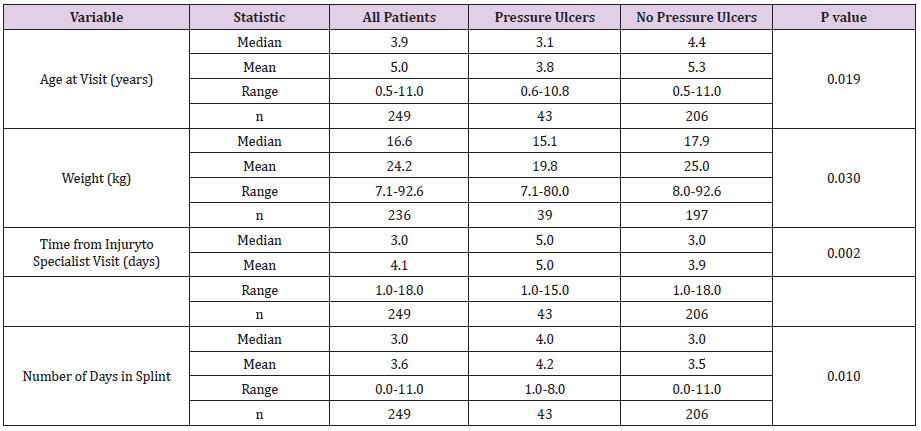
Forty-three of the 249 patients reviewed presented with pressure ulcers (Table 1). A majority of these pressure ulcers were reported as grade 1 and were found over the heel (Table 2). Younger age and decreased weight, increased time from injury to orthopedic specialist follow up, and increasing number of days spent in a splint were all correlated with increased incidence of pressure ulcer formation in a univariate analysis. Patients who were splinted at an outside ED also experienced a significantly higher rate of pressure ulcer formation compared to those initially splinted in our institutions pediatric ED (Tables 3 & 4). There was no difference when comparing a long leg splint versus a short leg splint, however, off-the-shelf immobilization devices (ortho boots, Aircasts, and foam ankle splints) were found to have a significantly lower pressure ulcer formation, when compared with long leg splints. Multiple logistic regression showed that for every 1-day increase in time from initial injury to the follow-up orthopedic visit, the odds of a pressure ulcer increased by 18% (p <0.05). Patients who were initially splinted at an outside ED, were 6.23 times more likely to develop pressure ulcers (95% CI 2.2-17.9) after controlling for other significant variables (Table 5). Splinting method, when comparing long leg splints versus other (not including short leg splints) was again found to be statistically significant. Weight and the number of days in a splint were not included in the multiple logistic regression model as they were highly correlated to age and time from injury to specialists follow up.
Table 2: Pressure Ulcer Staging.
Presence of a pressure ulcer was noted upon chart review but no descriptors were given to allow for staging.
Table 3: Categorical Variables
The difference between groups that is statistically significant (P<0.05) using Bonferroni correction for multiple comparisons is: Long
leg posterior vs. Ortho boot/other/unspecified.
**Includes ortho boot, Aircast, CAM boot, foam ankle splint, and unspecified splints.
Discussion
Consensus among the literature supports the notion that in order to prevent ulcer formation, forces must be unloaded at the most vulnerable sites. For lower extremity splints, unloading pressure at the heel is paramount in reducing pressure ulcer formation as it is a bony prominence and bears the most pressure in a recumbent individual. The majority of pressure ulcers found during our study had formed over the heel (Table 1), which is consistent with current literature [1-4]. Several techniques have been proposed to counteract this in a bid to decrease the overall incidence of pressure ulcer formation. One technique described in literature has the splint formed in such a way that a gap is created behind the heel, thereby alleviating any pressure and improving ventilation, preventing both pressure- and moisturedependent ulcer formation [3-4]. However, support for this method is anecdotal. A meta-analysis of different types of dressings noted that foams, hydrocolloids, and polyurethane films used in addition to standard care all decreased the incidence of pressure ulcers [1]. The foams were the most effective and had the added functionality of absorbing moisture, thereby preventing maceration.
Forni, et al. describe using a polyurethane foam placed inside a plaster cast at the heel. The foam used in this technique had originally been shown to be a cost-effective technique in the prevention of bed ulcers. The work by Forni, et al. revealed a decreasing amount of pressure exerted on the foot and reduction in ulcer formation in a significant proportion of two at-risk populations: those with previous heel skin ulcers (grade 1 ulcers) and those undergoing antiblastic chemotherapy [5].Our study revealed the majority of pressure ulcers documented were of Grade 1 quality (Table 2). While speculative, perhaps the method described by Forni, et al. is most suitable for our population of patients. Future studies will aim to investigate which technique provides the best prevention of ulcer formation in our cohort of patients. A significant modifiable factor revealed by our study was the duration of time from injury to follow up with an orthopedic specialist. Multiple logistic regression revealed that the average time, in days, spent in a splint resulting in pressure ulcer formation was 5 days, compared to 3.9 in those who did not develop pressure ulcers.
This is consistent with current literature noting that increased duration of pressure applied to a bony prominence precipitates ulcer formation as it causes both shear forces and interrupts local blood flow. In a meta-analysis by Geten, et al., researchers reviewed and examined the development of pressure ulcers in three different models-in vitro, animals and clinical studies. From their research, pressure ulcer development tends to develop across all three models from 1 hours to 4 hours of sustained loading [6-8]. This is consistent with the finding that there is a delay in follow-up care, assuming patients remained in their splints aside from bathing and dressing, they are at increased risk of pressure formation [1-5].An additional finding was that patients splinted at an outside ED were more likely to develop pressure ulcers than those initially splinted at our pediatric hospital. Of the 249 patients in our study, 220 were seen in our pediatric ED initially with only 14.3% of this group developing pressure ulcers. Of the 29 who were not seen initially at our institution’s ED, 10 patients, representing 34.5% of that group, developed pressure ulcers.
The pediatric hospital performed much better in terms of pressure ulcer incidence compared to outside, more rural, facilities, though the 95% confidence interval is vast (95% CI 2.2-17.9) due to the small number of patients seen at outside facilities.Plausible reasons for this include the lower volume of pediatric fractures at smaller facilities as well as a lack of orthopedic techs to apply splints. When comparing long leg splints versus other forms of less frequently applied splints, including CAM boots, air casts, and foam ankle splints, the long leg splints had a higher frequency of pressure ulcer formation. Of the 35 patients who fit into the “other” splinting category, only 1 developed a pressure ulcer (Table 2). This may be due to the fact that these alternative splints are used for less severe injuries, come with standard amounts of padding, and can be more easily removed for periodic relief. Certain comorbidities also increase a patient’s risk of pressure ulcer formation. This includes those who cannot communicate effectively, those under general or localized anesthesia, the very young and developmentally delayed, and those with impaired sensitivity [1-6]. All of these conditions prevent effective communication between the patient and his or her caregiver, and this breakdown in communication can ultimately lead to complications such as pressure ulcers.
With this in mind, younger age was hypothesized to be a predictor of ulcer formation as younger children are unable to effectively communicate the location and causes of pain, allowing for the insidious development of a pressure ulcer before it is noticed by a caregiver [2-8]. Although this was supported in the univariate analysis, subsequent multiple logistic regression showed that age itself cannot account for pressure ulcer formation when controlling for other variables that are also affected by age, particularly weight.Finally, splinting material, often plaster or fiberglass, is also projected to play a large role in pressure ulcer development because of its relationship with pressure formation [14-20]. Plaster is a heavy material compared to fiberglass, but it is more easily molded to a patient’s limb. Plaster’s pliable nature allows it to be more readily molded into a position that deposits less strain on bony prominences, thus helping to reduce the incidence of pressure ulcers [18-21]. A limitation of our study was the retrospective nature, of which we were unable to determine splint material. However, this represents an interesting possible future direction for pressure ulcer incidence associated with splinting.
Conclusion
Pressure ulcer formation is multifaceted and can be challenging to patients and providers in managing rehabilitation and progress for the patient. While our study is not inclusive of all possible factors, it should be appreciated that increasing the time spent in a lower extremity splint between the initial injury and specialist follow-up increases the likelihood of developing a pressure ulcer. Other factors, such as in which institution the splint is applied, the type of splint applied and the material it is comprised of, and certain patient comorbidities also play a role. With identification of these modifiable factors, future studies will consist of education and technique modification where needed with the aim of reducing pressure ulcer incidence among this cohort. We hope this study will stress to providers the importance of proper splinting technique and of managing and monitoring the amount of time patients spend in lower extremity splints in order to prevent ulcer formation.
References
- Huang L, Woo K, Liu L, Wen R, Hu A, et al. (2015) Dressings for preventing pressure ulcers: A meta-analysis. Adv Skin Wound Care 28(6): 267-273.
- Bou JETI, López JR, Camañes G, Narvaez EH, Blanco JB, et al. (2009) Preventing pressure ulcers on the heel: a Canadian cost study. Dermatology Nurs 21(5): 268-272.
- Bots TC, Apotheker BFG (2004) The prevention of heel pressure ulcers using a hydropolymer dressing in surgical patients. J Wound Care 13(9): 375-378.
- Russell L (1998) Physiology of the skin and prevention of pressure sores. Br J Nurs 7(18): 1084.
- Cooper KL (2013) Evidence-based prevention of pressure ulcers in the intensive care unit. Crit Care Nurse 33(6): 57-66.
- Stechmiller JK, Cowan L, Whitney JD, Phillips L, Aslam R, et al. (2008) Guidelines for the prevention of pressure ulcers. Wound Repair Regen 16(2): 151-168.
- Maklebust J (1987) Pressure ulcers: etiology and prevention. Nurs Clin North Am 22(2): 359-377.
- Gefen A (2008) How much time does it take to get a pressure ulcer? Integrated evidence from human, animal, and in vitro Ostomy Wound Manage 54(10): 26-28.
- Mc Cord S, Mc Elvain V, Sachdeva R, Schwartz P, Jefferson LS (2004) Risk factors associated with pressure ulcers in the pediatric intensive care unit. J Wound Ostomy Continence Nurs 31(4): 179-183.
- Curley MA, Razmus IS, Roberts KE, Wypij D (2003) Predicting pressure ulcer risk in pediatric patients: the Braden Q Scale. Nursing Research 52(1): 22-33.
- Quigley SM, Curley MA (1996) Skin integrity in the pediatric population: preventing and managing pressure ulcers. J Spec Pediatr Nurs 1(1): 7-18.
- Inman KJ, Sibbald WJ, Rutledge FS, Clark BJ (1993) Clinical utility and cost-effectiveness of an air suspension bed in the prevention of pressure ulcers. Jama 269(9): 1139-1143.
- Philbeck JT, Whittington KT, Millsap MH, Briones RB, Wight DG, et al. (1999) The clinical and cost effectiveness of externally applied negative pressure wound therapy in the treatment of wounds in home healthcare Medicare patients. Ostomy Wound Manage 45(11): 41-50.
- Brilli RJ, Mc Clead RE, Crandall WV, Stoverock L, Berry JC, et al. (2013) A comprehensive patient safety program can significantly reduce preventable harm, associated costs, and hospital mortality. The Journal of Pediatrics 163(6): 1638-1645.
- Halanski M, Noonan K (2008) Cast and splint immobilization: complications. J Am Acad Orthop Surg 16(1): 30-40.
- Boyd A, Benjamin H, Asplund C (2009) Principles of casting and splinting. Am Fam Physician 79(1): 16-22.
- Hipps H (1964) Prevention of cast pressure-sores on the heel. Southwest Med 45: 19-22.
- Forni C, Loro L, Tremosini M, Mini S, Pignotti E, et al. (2011) Use of polyurethane foam inside plaster casts to prevent the onset of heel sores in the population at risk. A controlled clinical study. J Clin Nurs 20(5-6): 675-80.
- Hsu R, Lareau C, Born C (2013) Novel posterior splinting technique to avoid heel ulcers. Orthopedics 36(1): 31-32.
- Castro Aragon O, Rapley J, Trevino S (2009) The use of a kickstand modification for the prevention of heel decubitus ulcers in trauma patients with lower extremity external fixation. J Orthop Trauma 23(2): 145-147.
- Konda S, Jordan C, Davidovitch R, Egol K (2011) A novel technique for reduction and immobilization of tibial shaft fractures: the hammock. J Orthop Trauma 25(6): 385-390.

 Short Communication
Short Communication