Research Article
Prostate Cancer Patient Reported Outcome After
Cyberknife Robotic Radiosurgery
Sebastian A Exner*, Felix K Behrens and Fabian Fehlauer
Author Affiliations
Strahlenzentrum and Cyberknife Center Hamburg, 22419 Hamburg, Germany
Received: August 14, 2020 | Published: September 01, 2020
Corresponding author: Sebastian A Exner, Felix K Behrens, Fabian Fehlauer, Strahlenzentrum and Cyberknife Center
Hamburg, 22419 Hamburg, Germany
DOI: 10.26717/BJSTR.2020.29.004875
Purpose:
The optimal management of localized Prostate Cancer (PC) should take
account consideration of patient and clinical risk factors, as well as patient preferences.
The objectives of this cohort study of men with newly diagnosed localised PC were to
document side effects with the International Prostate Symptom Score (IPSS), International
Consultation on Incontinence Questionnaire Short Form (ICIQ-SF), International
Index of Erectile Function Questionnaire (IIEF-5) and as well Patient-Reported Outcomes
version of the Common Terminology Criteria for Adverse Events Questionnaire
(PRO-CTCAE) following radiosurgery with the Cyberknife.
Materials and Methods: In this cohort analysis, 20 patient with localized prostate
cancer were analyzed who received Cyberknife radiosurgery (5 x 7 Gy) and completed
the IPSS (1-23), ICIQ-SF (0-17), IIEF-5 (6-24) and the German validated PRO-CTCAE
questionnaires.
Results:
Mean IPSS score was 6.9 (mild symptomatic), mean ICIQ-SF score was 3.3
(mild impairment of incontinence), mean IIEF-5 score was 17.0 (mild erectile dysfunction).
The highest score at PRO-CTCAE was for urinate frequently (one patient, 5%),
achieve and maintain erection (one patient, 5%), ejaculation problems (two patients,
10%) and decreased libido (one patient, 5%). There was no significant difference at follow-
up period or treated prostate volume (F/U: > 1 vs. < 1 year; Volume ml: >64 vs. <
64). Local control and overall survival were 100%. No patient developed metastasis after
radiosurgery. None of our patients died until now.
Conclusion:
Robotic Cyberknife radiosurgery for localized prostate cancer shows
good local control and overall survival with a very mild side effect profile reported by
patients.
Keywords: Cyberknife; Prostate Cancer;
PRO-CTCAE; Patient Reported Outcome;
Side Effects
Abbreviations: PC: Prostate Cancer, IPSS:
International Prostate Symptom Score,
HRQOL: Health-Related Quality of Life, PSA:
Prostate Specific Antigen
The optimal management of localized prostate cancer (PC)
should take account consideration of patient and clinical risk
factors, as well as patient preferences. Because of the high survival
for localized prostate cancer [1], the long-term effects on Health-
Related Quality of Life (HRQOL) are possibly the most important
base for the therapy decision [2,3]. Well-documented reports of
HRQoL effects are important in particular for patients to make
well informed and evidence-based treatment decisions. Patientreported
outcome (PRO) is the direct report from a patient
perspektive about the effect of disease or treatment that has not
been interpreted by anyone else and can be easily understood
from patient perspective [4]. Guidelines unfortunately do not
serve the increasing interest in the potential relationship between
treatment side effects and treatment decision [5]. This interest is
linked to the shared decision-making paradigm, in which a “good”
treatment choice is defined as one that fits the patients’ values and
preferences [6].
Recently, there has been a change toward the use of patientreported
outcomes (PROs), defined as the unfiltered direct report
of a given symptom toxicity by a patient, and considered to be the “gold standard” for the capture of symptomatic adverse events [7].
This change has been led by the release of the 2009 United States
Food and Drug Administration (FDA) Guidance for Industry on the
Use of PRO Measures in Medical Development to Support Labeling
Claims [8], which subsequently led to the NCI initiative to develop
a PRO version of the CTCAE (PRO-CTCAE) that will be used in
future U.S.-based clinical trials in oncology [9]. The objectives of
this cohort study of men with newly diagnosed localized PC were
to evaluate patient-reported outcome and side effects with IPSS,
ICIQ-SF, IIEF-5 and PRO-CTCAE questioner following Cyberknife
radiosurgery.
The observational, retrospective, monocenter study included
20 patients with histologically confirmed localized prostate cancer
treated with the Cyberknife® System (Accuray) treated after
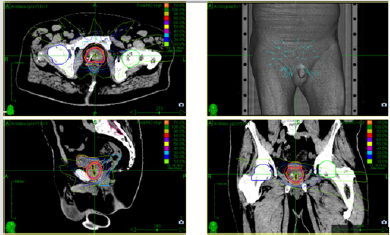
fiducial implants with 5 fractions of 7.0 Gy (35 Gy, 70 % isodose,
Figure 1) every second day (treatment protocol according PACE-B
study [10]). This type of stereotactic body radiotherapy (SBRT) is
well established and acknowledged by international guidelines
[11,12]. Patients who were treated between august 2012 and March
2020, were included in the analysis. Patients had newly diagnosed
clinically localised PC (cT1–2 and/or Gleason 6-7, Prostate-Specific
Antigen [PSA] < 20 ng/ml). All patients had to complete IPSS, ICIQSF,
IIEF-5 and the PRO-CTCAE questionnaire. The questions in the
IPSS questionnaire were as follows: question (Q)1, incomplete
emptying; Q2, frequency; Q3, intermittency; Q4, urgency; Q5, weak
stream; Q6, straining; Q7, nocturia; Q8, constancy and is utilized
to measure the severity of lower urinary tract symptoms [13].
The ICIQ was developed in the early 2000s to promote use of an
internationally uniform set of questionnaires [14]. The ICIQ-SF
consists of 3 scored items which evaluate the frequency, volume
of leakage, and overall impact of incontinence. The overall score
ranges from 0 to 21, with greater values indicating increased
severity.
Erectile function was prospectively assessed by a validated
self-administered questionnaire IIEF-5. This questionnaire is the simplified version of 5 items of the IIEF questionnaire, which was
developed by Rosen in 1997 [15]. It is recommended as a criterion of
effectiveness in clinical studies on ED. It enables to classify patients
into 5 classes such as: normal EF: > 21; mild ED: 17-21; mild to
moderate ED: 12-16; moderate ED: 8-11; severe ED: < 8. In total,
75 symptomatic toxicities (119 PRO-CTCAE items), corresponding
to the symptomatic adverse-events profile associated with the
regimens commonly used for prostate cancer treatment were
selected. Patients reported symptoms on a 5-point scale. The
IPSS, ICIQ-SF, IIEF-5 and PRO-CTCAE were used to evaluate
patient-reported disease-specific function. Patients were treated
at Strahlenzentrum Hamburg and completed questionnaires at
different time points after treatment. To limit study dropouts and
missing data, respondents were offered to re-call or e-mail the
authors for assistance, and patients received a telephone reminder
3 week after the questionnaire was sent. Study retention rates were
100% at three months.
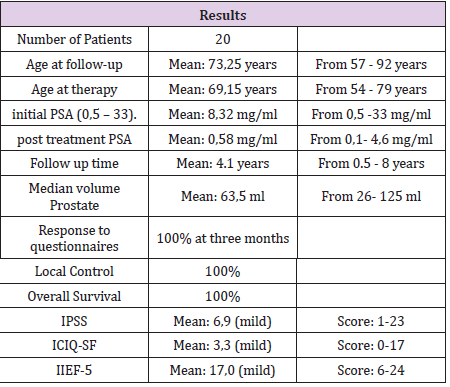
A total of 20 men were analyzed and completed the complete
questionnaire and at point after therapy (median age, 73 [57-92]
years). Median Age at time of therapy was 69.15 (54-79) years.
Median follow-up for was 4.15 (0.5-8) years. Median initial PSA was
8.32 mg/ml (0,5-33). Median post treatment PSA was 0.58 mg/ml
(0.1-4.6). Median volume of the treated volume with radiosurgery
was 63.5ml (26-125). Mean Score of IPSS was 6.9 (1-23). A Score
of 6.9 correlates to mild symptomatic. Mean Score of ICIQ-SF was
3.3 (0-17). A score of 3.3 shows a mild impairment of incontinence.
Mean Score of IIEF-5 was 17.0 (6-24). A score of 17 shows only
mild erectile dysfunction. The highest score at PRO-CTCAE was for
urinate frequently (one patient, 5%), achieve and maintain erection
(one patient, 5%), ejaculation problems (two patients, 10%) and
decreased libido (one patient, 5%). There was no significant
difference if the treatment was more or less than 1 year ago. There
was no significant difference if the treated volume was greater than
64 ml or less (Table 1),
Local Control and Overall Survival was 100% and no patient
developed metastasis after radiosurgery. None of the patients died
until now.
Using this approach (Cyberknife, SBRT), long-term data
with more than 10 years of follow-up are promising and show
a biochemical freedom from recurrence of> 90% for low and
intermediate-risk prostate carcinomas with significantly less
than 1% local recurrences. The effectiveness (PSA control) of the
radiosurgery was also with longer follow-up periods persistently
very high with low toxicity. In particular, the late toxicity appears
to be lower than with surgery or conventional external radiation.
Recently published results show slight side effects (grade 2) for SBRT
(5 treatments) below 5% in the urogenital area (GU} and below 2%
in the gastrointestinal tract (GI) with very rare serious side effects
(grade 3) of below 1% [10]. A German multicenter study using
this Cyberknife approach is open to confirm existing international
data for the national recommendations regarding long term effects
(ClinicalTrials.gov NCT03795337). In this limited cohort analysis,
Cyberknife radiosurgery for localized prostate cancer confirms low
adverse effect profiles determined by the patients themselves with
excellent local control and overall survival. The low incidence of
toxicity in the PRO-CTCAE questionnaire shows a very acceptable
and low adverse effect profile.
As well as with the good results of well-established scoring
systems like IPSS, ICIQ-SF and IIEF-5. Since curative therapy
options in man with localized prostate cancer (surgery: robotic
or conventional radical prostate resection; radiotherapy:
brachytherapy, conventional irradiation or radiosurgery) remain
iso- effective and with regard to recent publications [16], it is very
important to choose a treatment strategy with a low impact on
the health-related quality of life and high cure rate. Furthermore,
a short treatment interval (5 treatments only), minor side effects,
good quality of life, less follow-up therapies, no absence from
family and work, no hospitalization, no rehabilitation after therapy
and increasing reports might be cumulative reasons to decide
for Cyberknife treatment by man with localized prostate cancer
acknowledged by their families, international societies and health
care insurance companies.
None.
- Hamdy FC, Donovan JL, Lane JA,Richard M Martin, Jon Oxley, et al. (2016) 10-Year outcomes after monitoring, surgery, or radiotherapy for localized prostate cancer. N Engl J Med 375:1415-1424.
- Donovan JL, Hamdy FC, Lane JA (2016) Patient-reported outcomes after monitoring, surgery, or radiotherapy for prostate cancer. N Engl J Med375:1425-1437.
- Lardas M, Liew M, Van den, Bergh RC, De Santis M, et al. (2017) Quality of life outcomes after primary treatment for clinically localised prostate cancer: a systematic review. Eur Urol72(6):869-885.
- Talcott JA, Clark JA, Manola J, Mitchell SP (2006) Bringing prostate cancer quality of life research back to the bedside: translating numbers into a format that patients can understand. J Urol 176(4): 1558-1564.
- Hoffman RM, Lo M, Clark JA(2017) Treatment decision regret among long-term survivors of localized prostate cancer: results from the prostate cancer outcomes study. J Clin Oncol35(20):2306-2314.
- Christie DRH, Sharpley CF, Bitsika V (2015) Why do patients regret their prostate cancer treatment? A systematic review of regret after treatment for localized prostate cancer. Psychooncology 24(9):1002-1011.
- Chen RC, Chang P, Vetter RJ, Bryce B Reeve, Howard M Sandler, et al. (2014) Recommended patient-reported core set of symptoms to measure in prostate cancer treatment trials. J Natl Cancer Inst106:1-7.
- (2006) US Department of Health and Human Services FDA Center for Drug Evaluation and Research., U.S. Department of Health and Human Services FDA Center for Biologics Evaluation and Research., U.S. Department of Health and Human Services FDA Center for Devices and Radiological Health.Health Qual Life Outcomes4:79.
- Dueck AC, Mendoza TR,Sandra A Mitchell, Bryce B Reeve,Kathleen M Castro, et al. (2015) Validity and Reliability of the US National Cancer Institute's Patient-Reported Outcomes Version of the Common Terminology Criteria for Adverse Events (PRO-CTCAE).JAMA Oncol 1(8):1051-1059.
- Brand, Douglas HDodds D (2009) Intensity-modulated fractionated radiotherapy versus stereotactic body radiotherapy for prostate cancer (PACE-B): acute toxicity findings from an international, randomised, open-label, phase 3, non-inferiority trial. The Lancet Oncology 20(11): 1531-1543.
- NCCN Guidelines Version 2.2020 Prostate Cancer.
- Martin G,Sanda MD, Ronald C, Chenet al. (2017) American Urological Association (AUA) / American Society for Radiation Oncology (ASTRO) / Society of Urologic Oncology (SUO), Approved by the AUA Board of Directors April.
- Barry MJ, Fowler FJ, WK Mebust, AT Cockett(1992) The American Urological Association symptom index for benign prostatic hyperplasia. J Urol 148: 1549-1557.
- Donovan JL, Badia X, Corcos J (2002) Symptom and quality of life assessment. In Abrams P, Cardozo L, Khoury S, Wein A eds, Incontinence, (2nd edn)Chapt. 6. Plymouth, UK: Health Publication Ltd pp. 267-316.
- Rosen RC, Riley A, Wagner G, Osterloh IH, Kirkpatrick J, et al.(1997) The international index of erectile function (IIEF): a multidimensional scale for assessment of erectile dysfunction. Urology49(6):822-830.
- Hoffman KE, Penson DF (2020) Patient-Reported Outcomes Through 5 Years for Active Surveillance, Surgery, Brachytherapy, or External Beam Radiation with or Without Androgen Deprivation Therapy for Localized Prostate Cancer. JAMA323(2):149-163.

 Research Article
Research Article