Abstract
This study investigates the effect of G. kola on adrenal and testicular tissues oxidative and antioxidant activities and histology in normal Wistar rats. In a bid to achieve these objectives, 20 adult male Wistar rats were divided into 4 groups after accessing the oral acute toxicity of G. kola. Group A served as the control while groups B, C and D were treated on 1000, 1200 and 1400mg/kg of the constituted G. kola for 7 days. At the end of treatments, organs weights and histology as well as oxidative and antioxidant activities in tissue homogenates were determined. Using appropriate statistical analysis (ANOVA) the investigated variables were analyzed and results presented as means ± SEM with p<0.05 considered significant. The results showed that G. kola ingestions stimulates adrenal gland and testicular weights, SOD and CAT but inhibits MDA and protein in a dose dependent fashion. Histologically, there was no effect on the adrenal gland but there was dose dependent development of testicular germ cells. This study showed that G. kola reduces adrenal and testicular oxidative stress status via stimulation of tissue antioxidants activities.
Keywords: Adrenal Gland; Testis; Oxidative Stress; Antioxidants; Garcinia Kola
Introduction
Garcinia kola Heckel (family, Guttiferae); referred to as “bitter kola” or “male kola” because of its bitter taste and the perceived aphrodisiac activity respectively [1], is a plant found in moist forests grows up to a height of 12 meters [2]. It is traditionally regarded as a highly prized item and it is served to visitors in social gatherings as a mark of honour [2]. The plant is regarded as a wonder plant because every part of the plant (bark, leaves, root, and wood) has been found to be of medicinal value [3]. G. kola is reported safe when taken with or without other foods and has been documented to increase the absorption of the key ingredients when taking it an hour before or after meals [4]. The medicinal importance of bitter kola has been documented to be based mainly on the phytochemicals of the plant [5].
Phytochemical and biochemical analysis revealed G. kola seeds to be a complex mixture of sterols, terpenoids, flavonoids, glycosides, saponin, tannins, reducing sugar, triterpenoids [6,7], and phenolic compounds including biflavonoids, xanthones, benzophenones, cycloartenol and triterpenes [3,8,9], Kolaviron [10,11]. In term of percentage, nutritional composition of G. kola nut has been documented to be rich in Moisture, dry matter, crude fat, crude protein, ash, crude fiber and total carbohydrates [12-14]. Mineral composition of G. kola has been documented to include K, Ca, Mg, Fe, Zn and P as well as a rich source of vitamins [14]. Several studies have documented that plant-derived polyphenolic compounds possess antioxidative properties which can mitigate the damaging effect of ROS and by so doing promote good health in humans [15]. Consequently, a number of evidence-based studies have implicated a strong correlation between the ingestion of fruits, vegetables, and medicinal plant rich in polyphenols with antioxidant potential and the reduced risk of oxidative stress-related disorders [16,17]. Although reactive oxygen species are considered to be unavoidable by-products of normal aerobic metabolism, they are known to disrupt cellular homeostasis [18].
Chronic stress shifts the balance of hormones to low levels of anabolic hormones and high levels of catabolic hormones [19]. From hormonal point of view, knowing that testosterone is anabolic, and cortisol is catabolic, and that testosterone/cortisol ratio is a great marker for stress [19], it therefore suggest a relationship between the testis and adrenal gland which released these hormones. In fact, steroidogenic cells in the adrenal cortex and testis are reported to arise from a common pool of progenitors in the adrenogonadal primordium, a specialized group of coelomic epithelial cells in the urogenital ridge [20]. According to Heikinheimo et al. [21], the common developmental origin of the adrenal cortex and testis is reflected in overlapping functional profiles for these organs. Thus, the organs may suffer or enjoy together in a condition of exposure to substances or agents. Considering that the adrenal gland and testis are easily exposed to substances ingested by man and animals, and are from common developmental origin couple with the assertion that they may suffer or enjoy together, this study was therefore undertaken to investigate the effect of G. kola on the histology of adrenal and testicular tissue as well as the oxidative and antioxidant activities in normal Wistar rats.
Materials and Methods
Adult male Wistar rats (N=32) of comparable weight (180 to 200g) were obtained from the Department of Physiology animal holding unit of the Ambrose Alli University, Ekpoma, Nigeria and were transferred to the site of the experiments. They were housed in plastic cages (measuring 50 x 35 x 25) under standard environmental condition of 12/12hr light/dark cycle and were allowed to acclimatize for two weeks to the laboratory conditions before the experiment and fed ad libitum with rat chow and tap water. Garcinia kola (G. kola) Heckel Seeds were purchased commercially from a local market in Ekpoma in Edo State, Nigeria. The plant material was authenticated in the herbarium unit of Forestry Research Institute, Ibadan, Nigeria. Thereafter the testas were peeled off the seeds, cut into small sizes and sun dried. This was blended into powder and packed in clean and well covered containers pending when it was used. Oral acute toxicity (LD50) was performed on the grounded seed by Lorke [22] method using 12 rats.
Based on the results of the oral acute toxicity, the remaining 20 rats were divided into 4 groups as followed; group 1 served as untreated control while groups 2, 3 and 4 served as the test groups treated on 1000mg/kg, 1200mg/kg and 1400mg/kg dose of the prepared G. kola respectively. These doses of G. kola chosen are comparable with previous study by Ahumibe and Braide [23]. Administration of the prepared G. kola was as describe by Nwaopara et al. [24]. Briefly, the required grams of crude G. kola powder (1000mg/kg, and 1200mg/kg and 1,400mg/kg) were mixed with 20% of normal daily feed (5.0g rat chow) to form paste that was then given to each group of animal. After the rat finished the formed paste, a measured quantity of normal rat chow (80% of 150g) was given to the animal for that day. At the end 7 days treatment the animals were euthanized following cervical decapitation. The testis and adrenal glands were dissected out, devoid of accessory fats and tissues, weighted (using electronic weighing scale) and each dropped in containers containing phosphate buffer (1:10w/v) and 10% formal saline for tissue oxidative stress analysis and histological evaluation respectively.
The tissues (testis and adrenal gland) in phosphate buffer were homogenized and centrifuged at 4000 rpm for 10 minutes to obtain the supernatant which was used for the determination oxidative stress and antioxidant marker. The protein concentration was determined by means of the Biuret method [25] while lipid peroxidation was determined by measuring the thiobarbituric acid reactive substances (TBARS) produced during lipid peroxidation following the method by Varshney and Kale [26]. The level of SOD and Catalase activities were determined by the methods of Misra and Fridovich [27] and Sinha [28] respectively. Following standard histological processing procedures, the tissues in the container containing 10% formal saline were processed and then evaluated under the microscope. The result was analyzed with appropriate statistical analysis (ANOVA) using SPSS 20 and p<0.05 (LSD) was considered statistically significant and then presented as means ± SEM in charts, tables and microscopic slides.
Results
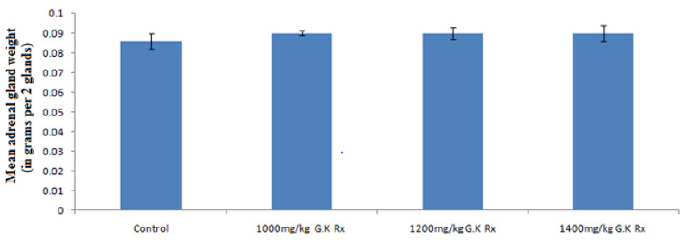
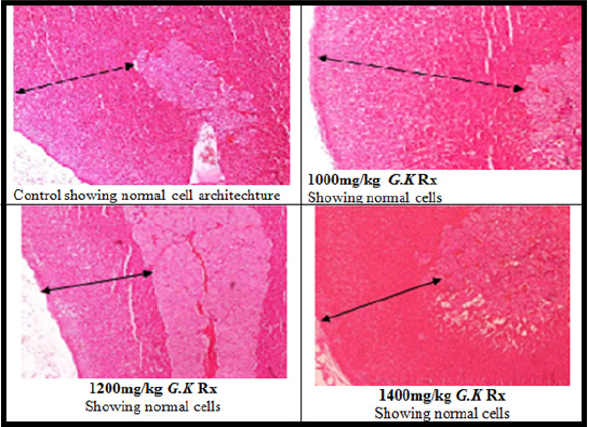
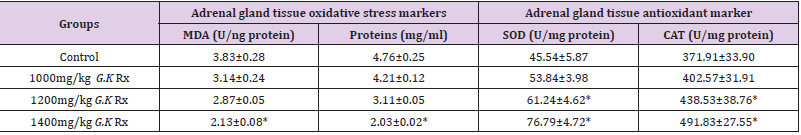
Table 1 shows the findings of oral acute toxicity study of G. kola. Orally, G. kola seed was found to be safe at dose as high as 5000 mg/ kg body weight. Figure 1 presents the effect of G. kola treatments on adrenal gland weight. Although G. kola causes a slight increase in adrenal weight, the difference was not significant compared to the control. Table 2 presents the effect of G. kola treatments on adrenal gland oxidative and antioxidant markers. There were significant reductions (p<0.05) in adrenal gland tissue MDA and protein levels with 1400mg/kg G. kola treatment compared with the control. However, 1200mg/kg and 1400mg/kg G. kola treatment resulted in significant increase (p<0.05) in adrenal gland tissue SOD and CAT levels compared to the control. Histological presentations showed (plate 1) normal pattern of cortex zonation showing stainable zona glomerulosa cytoplasm, clear zona fasciculata cytoplasm and zona reticularis acidophilic cytoplasm with normal medulla layers in the 1000, 1200 and 1400mg/kg G. kola treated and in the control.
Figure 1: Effect of G. kola treatments on adrenal gland weight.
Note: Mean ± SEM of 5 male rats per group.
Plate 1: Effect of G. kola treatments on adrenal gland tissue histology (Haematoxylin and Eosin stained x100).
Table 2: Effect of G. kola treatments on adrenal gland oxidative and antioxidant markers.
Note: Mean ± SEM of 5 male rats per group, * = p<0.05 compared to control.
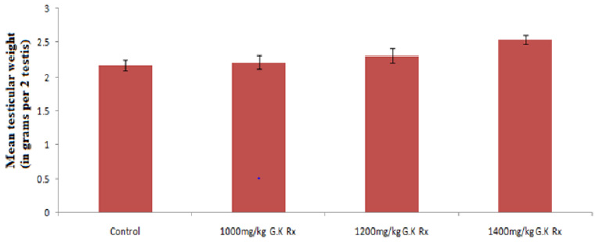
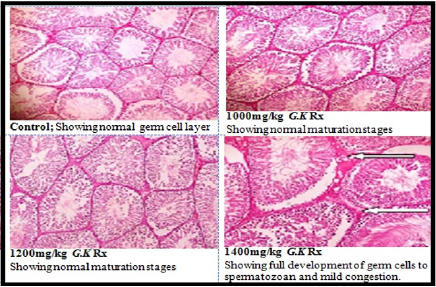
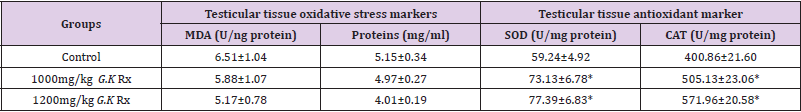
Figure 2 represents the effect of G. kola treatments on testicular weight. There was a non-significantly dose depended increase (p>0.05) in testicular weight with graded dose of G. kola treatments compared to the control. Table 3 presents the effect of G. kola treatments on testicular tissue oxidative and antioxidant marker activities. There was dose dependent decrease in testicular tissue MDA and protein levels that were significant (p<0.05) in the 1400mg/kg G. kola treated group compared to the control. Testicular tissue SOD and CAT activities were significantly increased (p<0.05) in a dose dependent fashion compared to the control. Histological findings on testicular tissue (Plate 2) showed comparable normal spermatogonia cells in the seminiferous tubules epithelium, normal spermatocytes, spermatids and interstitial leydig cells in the G. kola treated groups with the control. However, there was a dose dependent germ cells development within the lumen in the G. kola treated groups compared to the control with mild congestion (white arrow) in the 1400mg/kg treated group.
Figure 2: Effect of G. kola treatments on testicular weight.
Note: Mean ± SEM of 5 male rats per group.
Plate 2: Effect of G. kola treatments on testicular tissue histology (Haematoxylin and Eosin stained x100).
Table 3: Effect of G. kola treatments on testicular tissue oxidative and antioxidant markers.
Note: Mean ± SEM of 5 male rats per group, * = p<0.05 compared to control.
Discussion
Reactive oxygen species are considered to be unavoidable byproducts of normal aerobic metabolism, however, when not balance, they can disrupt cellular homeostasis [18]. In the present study the ingestion of G. kola in normal adult male Wistar rat stimulated adrenal gland and testicular weights although not significantly different from the control. In line with this observation, Mosunmola et al. [5] has also reported no significant differences in the testicular weight between control and G. kola extract treated rats not exposed to arsenate. Also observed in the present study, the ingestion of G. kola in normal adult male Wistar rat weakens the oxidative stress activities and improved antioxidants status of the adrenal gland and testicular tissues in a dose dependent manner. Abarikwu et al. [29] have reported increase MRNA expression of SOD in culture interstitial leydig cells in the present of kolaviron.
The antioxidant and scavenging activity of Garcinia biflavonoid complex has been investigated in a range of established in vitro assays involving reactive oxygen species and studies showed that kolaviron elicited significant reducing power and a dose-dependent inhibition of oxidation of linoleic acid [30], reduced damage to proteins and lipids induced by Fe3+/EDTA/ascorbate mixtures ex vivo [31] as well as caused reduced background levels of protein oxidation marker in plasma and liver and γ-glutamyl semialdehyde (GGS) and malondialdehyde in liver [32]. The inhibitory activity of kolaviron in deoxyribose assay was suggested to directly relate to prevention of the propagation of the process of lipid peroxidation and modulation of other biomarkers of oxidative stress in animal model [33,34]. Thus, the observed decreased in adrenal and testicular oxidative stress markers in this study is in line.
Histological, the graded doses of G. kola have no effect on the adrenal gland but 1400mg/kg resulted in a full germ cells development and mild congestion on the testis. In comparison with the findings of this study on the histology of the adrenal gland, Mosunmola et al. [5] has reported comparable number of basophils with the control group in groups not exposed to arsenate. Also in line with the findings on the testis, the study by Mosunmola et al. [5] has reported prominent and healthy spermatogonia in rats unexposed to arsenat but treated with G. kola extract and was concluded that the extract improved initiation of spermatogenesis. The observed non significant effect of G. kola on the histology of the adrenal gland indicates that the extract is safe. On the other hand, the improved development on the germ cells of the testis by the ingestion of G. kola indicates that the extract stimulates spermatogenesis probably via enhancing hormonal interplay that promote germ cells development through its phytochemical and antioxidant properties.
It is known that plants use a diverse array of anti-oxidative enzymes as well as low molecular weight antioxidants to scavenge different types of reactive oxygen intermediates, thereby protecting cells against injury and potential damage [18]. Thus, the observed effects of G. kola on the adrenal gland and testis may be due to its phytochemical and antioxidants components. In support of this assertion, Okoko [35] has previously reported G. kola to be a powerful antioxidant. Also, the cytoprotective activity of flavonoids; a phytochemical in G. kola, has been reported to be via direct antioxidant effect [36,37]. In fact, flavonoids are documented to possess indirect antioxidant activity through inducing the upregulation of glutathione, γ-glutamylcystein ligase, glutathione s-transferase and NAD (P) H: quinine oxidoreductase in different cell systems and inhibit cell death by suppressing oxidative stress [38]. Thus, the observed boosting of the adrenal and testicular tissue antioxidant status and weakness of oxidative markers in this study are in accordance and is owned to the antioxidants and phytochemical properties of G. kola.
References
- Uko O J, Usman A, Ataja AM (2001) Some biological activities of Garcinia kola in growing rats. Veterinarski Arhiv 71(5): 287-297.
- Iwu MM (1993) Handbook of African medical plants. Bola Raton: CRC Press Inc pp. 223-224.
- Iwu MM, Igboko OA, Okunji CO, Tempesta MS (1990) Anti-diabetic and aldose reductase activities of biflavanones of Garcinia kola. J. Pharmacy and Pharmacology 42: 2903-2922.
- Olayinka OA, Saburi AA, Thomas OI, Oluwakemi CO, Oyesiku AO, et al. (2008) Clinical effects of Garcinia kola in knee osteoarthritis. J Orthop Surg 3: 34.
- Mosunmola BO, Isaac OA, Efere MO, Rufus OA, Olugbenga AA (2017) Assessment of the Effects of Graded Doses of Polyphenolic-Rich Fraction of Garcinia kola Seeds on Pituitary-Testicular Axis of Male Wistar Rats. Dose Response 15(4): 1-8.
- Tcheghebe TO, Signe M, Seukep JM, Tatong FN (2016) Review on traditional uses, phytochemical and pharmacological profiles of Garcinia kola Merit Research Journal of Medicine and Medical Sciences 4(11): 480-489.
- Esimone CO, Adikwu MU, Nworu CS, Okoye FBC, Odimegwu DC (2007) Adaptogenic potentials of Camellia sinensis leaves, Garcinia kola and Kola nitida seeds. Science Research Essays 2: 232-237.
- Seanego CT, Ndip RN (2012) Identification and antibacterial evaluation of bioactive compounds from Garcinia kola (Heckel) seeds. Molecules 17(6): 6569-6584.
- Antia BS, Pansanit A, Ekpa OD, Ekpe UJ, Mahidol C, et al. (2010) Alpha-glucosidase inhibitory, aromatase inhibitory and antiplasmodial activities of a biflavonoid GB1 from Garcinia kola stem bark. Planta Medica 76(3): 276-277.
- Adaramoye OA, Farombi EO, Adeyemi EO, Emerole GO (2005) Inhibition of human low-density lipoprotein oxidation by flavonoids of Garcinia kola Pakistan J Med Sci 21(3): 331-339.
- Lacmata ST, Kuete V, Dzoyem JP, Tankeo SB, Teke GN, et al. (2012) Antibacterial activities of selected cameroonian plants and their synergistic effects with antibiotics against bacteria expressing MDR phenotypes. Evidence Based Complementary and Alternative Medicine pp. 11.
- Obi AU, Nwoha PU (2014) Effects of Kolaviron, the Major Constituent of Garciniakola, on the Histology of the Hypothalamus, Pituitary, and Testes Using Adult Male Wistar Rats as a Model Organism. Forensic Medicine and Anatomy Research 2: 80-87.
- Mazi EA, Okoronkwo KA, Ibe UK (2013) Physico-Chemical and Nutritive Properties of Bitter Kola (Garcinia kola). J. Nutrition and Food Sciences 3: 218-224.
- Odebunmi EO, Oluwanili OO, Awolola GV, Adediji OD (2009) Proximate and nutritional composition of Kola nut (cola nitida), bitter kola (Garcinia kola), and Alligator pepper (Afromomum melegueta). African Journal of Biotechnology 8(2): 308-310.
- Steinmetz KA, Potter JD (1996) Vegetables, fruit, and cancer prevention: a review. J Am Diet Assoc 96(10): 1027-1039.
- Block G (1992) The data support a role for antioxidants in reducing cancer risk. Nutr Rev 50(7): 207-213.
- Hertog MG, Feskens EJ, Hollman PC, Katan MB, Kromhout D (1993) Dietary antioxidant flavonoids and risk of coronary heart disease: the Zutphen Elderly Study. Lancet 342(8878): 1007-10011.
- Mittler R (2002) Oxidative stress, antioxidants and stress tolerance. Trends Plant Sci 7(9): 405-410.
- Vasunilashorn S, Cohen AA (2014) Stress Responsive Biochemical Anabolic/Catabolic Ratio and Telomere Length in Older Adults. Biodemography Soc Biol 60(2): 174-184.
- Hatano O, Takakusu A, Nomura M, Morohashi K (1996). Identical origin of adrenal cortex and gonad revealed by expression profiles of Ad4BP/SF‐1. Genes Cells 1: 663-671.
- Heikinheimo M, Pihlajoki M, Schrade A, Kyrönlahti A, Wilson BD (2015) Testicular steroidogenic cells to the rescue. Endocrinology 156(5): 1616-1619.
- Lorke D (1983) A new approach to practical acute toxicity testing. Archives of Toxicology 54: 275-287.
- Ahumibe AA, Braide BV (2009) Effect of gavage treatment with pulverised garcinia kolaseeds on erythrocyte membrane integrity and selected haematological indices in male albino Wistar rats. Nig J Physiological Sciences 24 (1): 47-52.
- Nwaopara AO, Anibeze CIP, Akpuaka FC (2010) Histological signs of neurodegeneration in the cerebrum of rats fed with diet containing Yaji: The complex Nigerian suya meat sauce. Asian J Med Sci 2(1): 16-21.
- Gornall AG, Bardawill CJ, David MM (1949) Determination of serum proteins by means of the biureto reaction. J Biol Chem 177(2): 751-766.
- Varshney R, Kale RK (1990) Effects of Calmodulin Antagonists on Radiation-induced Lipid Peroxidation in Microsomes. Int. J Rad Biol 58: 733-743.
- Misra HP, Fridovich I (1972) The role of superoxide anion in the autoxidation of epinephrine and a simple assay for superoxide dismutase. J Biol Chem 247: 3170-3175.
- Sinha KA (1972) Colorimetric Assay of Catalase. Analytical Biochemistry 47(2): 389-394.
- Abarikwu SO, Farombi EO, Pant AB (2012) Kolaviron biflavanoids of Garcinia Kola seeds protect atrazine-induced cytotoxicity in primary cultures of rat leydig cells. Int J Toxicol 31(4): 407-415.
- Farombi EO, Akanni OO, Emerole GO (2002) Antioxidative and scavenging activities of kolaviron in vitro. Pharm Biol 40: 107-116.
- Farombi EO, Møller P, Dragsted LO (2004) Ex-vivo and in vitro protective effects of kolaviron against oxygen-derived radical-induced DNA damage and oxidative stress in human lymphocytes and rat liver cells. Cell Biol Toxicol 20(2): 71-82.
- Farombi EO, Hansen M, Ravn Haren G, Møller P, et al. (2004) Commonly consumed and naturally occurring dietary substances affect biomarkers of oxidative stress and DNA damage in healthy rats. Food Chem Toxicol 42(8): 1315-1322.
- Farombi EO, Tahnteng JG, Agboola AO, Nwankwo JO, Emerole GO (2000) Chemoprevention of 2-acetylaminofluorene-induced hepatotoxicity and lipid peroxidation in rats by kolaviron a Garcinia kola seed extract. Food Chem Toxicol 38(6): 535-541.
- Farombi EO (2000) Mechanisms for the hepatoprotective action of kolaviron: studies on hepatic enzymes, microsomal lipids and lipid peroxidation in carbontetrachloride-treated rats. Pharmacol Res 42(1): 75-80.
- Okoko T (2009) In vitro antioxidant and free radical scavenging activities of Garcinia kola Food Chem. Toxicol 47(10): 2620-2623.
- Chen TJ, Jeng JY, Lin CW, Wu CY, Chen YC (2006) Quercetin inhibition of ROSdependent and-independent apoptosis in rat glioma C6 cells. Toxicol 223(2): 113-126.
- Chow JM, Shen SC, Huan SK, Lin HY, Chen YC (2005) Quercetin, but not rutin and quercitrin, prevention of H2O2-induced apoptosis via antioxidant activity and heme oxygenase 1 gene expression in macrophages. Biochem Pharmacol 69: 1839-1851.
- Ayepola OR, Cerf ME, Brooks NL, Oguntibeju OO (2014) Kolaviron, a biflavonoid complex of Garcinia kola seeds modulates apoptosis by suppressing oxidative stress and inflammation in diabetes-induced nephrotoxic rats. Phytomed 21: 1785-1793.

 Review Article
Review Article