Impact Factor : 0.548
- NLM ID: 101723284
- OCoLC: 999826537
- LCCN: 2017202541
Pavel F Zabrodskii*
Received: January 08, 2019; Published: January 22, 2019
*Corresponding author: Zabrodskii PF, Saratov Branch of the “REAVIZ” Samara Medical Institute, Saratov, Russia
DOI: 10.26717/BJSTR.2019.13.002399
It was established in experiments on random-bred albino mice that the reduction of mortality of mice from sepsis and decrease the blood concentration of proinflammatory cytokines (TNF-α, IL-1β, IL-6) after acute intoxication of organophosphorus compounds – OPC (DDVP, 0.5 LD50) due to the implementation of the cholinergic anti-inflammatory pathway: activation of m1-muscarinic acetylcholine receptors a of the brain and α7n-acetylcholine receptors of monocyte-macrophage system. The anti-inflammatory effect after acute intoxication of OPC is also associated with the excitation of the sympathetic nervous system, β2 adrenergic receptors of T lymphocytes, macrophages and monocytes and the subsequent reduction in blood of proinflammatory cytokines.
Keywords: Organophosphorus Compounds; M1-Muscarinic Acetylcholine Receptors; Α7n-Acetylcholine Receptors; β2 Adrenergic Receptors; Proinflammatory Cytokines
Abbreviations IOPC: Organophosphate Compounds; ELISA: Enzyme Immunosorbent Assay; DMSO: Dimethyl Sulfoxide
Organophosphate compounds (OPC) are widely used in agriculture, various industries and households, can cause environmental pollution, as well as acute and chronic intoxications [1-3]. Anticholinesterase drugs used in medicine have almost the same toxicodynamics as OPC [2-5]. More than 200 thousand people per year die from organophosphate insecticide intoxications in the world, mainly as a result of poisoning when used with safety violations [6]. The possibility of using OPC. in local armed conflicts for terrorist and criminal purposes is not excluded [2,6-8], and the occurrence of emergency situations at facilities engaged in the destruction of chemical weapons, in particular OPC, in accordance with international agreements [2,3]. In this case, possible group and mass acute intoxications of OPC [2,3]. Acute and chronic OPC intoxications reduce the humoral and cellular immune response [2,3], however, the study of the mechanisms of realization established by the 1987 “paradoxical effect” of OPC deserves attention-cholinergic stimulation significantly reduced the mortality of mice from sepsis [9]. Thus, we discovered the cholinergic anti-inflammatory mechanism (effect).
Subsequently, the expediency of using cholinomimetics for emergency activation of non-specific antimicrobial resistance of the organism was proved [10], and the identified phenomenon (mechanism) [9], began to be called since 2000 the “cholinergic anti-inflammatory pathway” [2,10,11-14]. Cholinergic anti-inflammatory pathway includes: acetylcholine (ACh) activation m1-acetylcholine receptors (m1AChRs) of the brain, modulating the immunoregulatory function of the vagus nerve [15-17], excitation of efferent fibers n. vagus; the effect of ACh on α7n-acetylcholine receptors (α7nAChRs) of monocyte-macrophage system cells (MMS); inhibition of proinflammatory cytokine production by these cells, TNF-α, IL-1β, IL-6 [2,16-21] and a decrease in mortality from sepsis [2,9,10,13,19,21-24]. When implementing the cholinergic anti-inflammatory pathway, in addition to the effects described, nAChRs of the adrenal medulla and sympathetic ganglia are activated, which leads to the production of epinephrine and norepinephrine (NE), which excite the β2-adrenoreceptor (β2ARs) of MMS cells [12,21,22] and reduce the production of inflammatory cells cytokines [12,13, 19,21,22].
n. vagus, releasing ACh in the celiac ganglia, causes excitation of the nerve of the spleen belonging to the sympathetic nervous system activating the α7nAChRs of the celiac ganglion [16,19,21,22]; NE is affected through its efferent fibers on β2ARs of T-lymphocytes [16,21,22], the production of ACh by these lymphocytes, which acts on α7nAChRs of MMS cells of the spleen; there is a reduction in the synthesis of proinflammatory cytokines [12,16,17,18,19]. Based on the foregoing, it is of interest to study the role of various links units of the cholinergic anti-inflammatory pathway during intoxication of OPC - m1AChRs of the brain, modulating the immunoregulatory function of the vagus nerve [16,17], as well as the associated sympathetic nervous system (the role of β2ARs of T lymphocytes and MMS cells) [12,13,18,21,22], activation of α7nAChRs of MMS cells in providing resistance to sepsis and inflammatory bowel diseases [2,10,18, 19,21].
The aim of the study was to determine the implementation mechanisms of the cholinergic anti-inflammatory pathway after acute intoxication with organophosphorus compounds by comparative assessment influences OPC and m1AChRs agonist, α7nAChRs agonist, epinephrine, norepinephrine, β2ARs agonist on mortality of mice after sepsis and concentration of proinflammatory cytokines in the blood
Experiments were performed on random-bred albino mice of both sexes weighing 18-22g. Control group of mice (control group 1, n = 8) received i.p. 2.0 ml isotonic sodium chloride solution (saline) 2 hours after subcutaneous administration of 0.5 ml saline. The second group of mice (control group 2, n = 55) was injected subcutaneously once with saline (0.5 ml), after 2 h mice received 2.5×109 CFUs in 2.0 ml of saline diurnal culture of E. coli O157:H7 (sepsis modeling) [2,9,17,18,21,24,25]. Mice of the third group (n = 30) were injected with OPC – DDVP (O, O-dimethyl-O-2,2- dichlorovinyl phosphate) (Sigma-Aldrich) intramuscularly once at a dose of 0.5 LD50 in 0.5 ml of a 0.25% solution of dimethyl sulfoxide (DMSO). DDVP was dissolved in DMSO, a 0.25% aqueous solution containing a toxicant was prepared. The LD50 DDVP was 52.5±3.3mg/kg. TBPB [1-(1’-2-methylbenzyl)-1,4’-bipiperidin-4- yl)-1H-benzo[d]imidazol-2(3H)-one] (Sigma-Aldrich) was used as a selective m1AChR agonist in the 4th group of animals (n=40), which was administered once subcutaneously at a dose of 60 mg/ kg [26]. The fifth group of mice (n = 35) received the α7nAChR agonist drug GTS-21 [3- (2,4-dimethoxybenzylidene)-anabaseine dihydrochloride] (Sigma-Aldrich) at a dose of 10 mg/kg in 0.5 ml saline (single subcutaneous injection) [27]. The sixth (n=35) and seventh groups of mice (n=33) received solutions of epinephrine hydrochloride and norepinephrine hydrotartrate (Sigma-Aldrich) in equitherapeutic doses subcutaneously, once in doses of 0.5 mg/kg and 0.9 mg/kg, respectively, in 0.5ml of physiological solution (activation of β2ARs of T-lymphocytes and MMS cells [12,16,18,21,22].
The eighth group of mice were injected single subcutaneous β2ARs selective agonist hexoprenaline sulfate (Nycomed) at a dose of 1.5mg/kg in 0.5ml of saline (group 3; n = 38). In groups 3, 4, 5, 6, 7 and 8, 2 h after drug administration was sepsis modeling. The mortality of mice (groups 2–8) after sepsis modeling was recorded after 4 and 24 h. The concentration of TNF-α, IL1β and IL-6 was studied in blood plasma of all groups of mice (groups 1-8) 4 and 24 h after the administration of E. coli (sepsis modeling) by enzyme immunosorbent assay (ELISA) using kits (ELISA Kits My Bio Soure) in accordance with the manufacturer’s instructions. Monoclonal antibodies MyBioSoure (TNF-α, IL1β, IL-6 - #MBS494184, #MBS494492, #MBS335516) were used to determine the concentration of proinflammatory cytokines. Blood for research was taken from the retroorbital venous sinus. The data obtained were processed statistically using the Student’s t-test. Differences between the parameters were considered reliable at p <0.05.
Acute intoxication of OPC (DDVP), the use of m1AChRs agonists (TBPB), an α7nAChRs agonist (GTS-21), epinephrine, norepinephrine and agonist β2ARs (hexoprenaline sulfate) 2 hours before the modeling of sepsis caused a significant decrease in mortality after 4 hours compared with the control group 2 (sepsis), respectively, in 3 64; 2.08; 2.55; 3.19; 2.39 and 1.98 times (p <0.05) (by 26.4; 18.9; 22.1; 25.0; 21.2 and 18.0%), and after 24 h - in 1.50; 1.57; 1.75; 1.97; 1.80 and 1.56 times (by 30.1; 32.6; 38.7; 44.4; 40.1 and 32.2%) (p <0.05), respectively (Table 1). The concentrations of cytokines TNF-α, IL-1β and IL-6 after sepsis modeling (control group 2) significantly increased in the blood of mice after 4 h compared to control group 1 (intact animals), respectively, at 17.8; 19.5 and 57.7 times (p <0.05), the concentrations of TNF-α, IL-1β and IL-6 after 24 h compared with their level after 4 h significantly decreased, exceeding the parameters of intact animals (group 1) in 1.5 (p>0.05), 4.5 and 8.2 times (p <0.05), respectively (Table 2).
Table 1: Effects of OPC (DDVP, 0.5 LD50), m1AChRs agonist TBPB (60 mg/kg), α7nAChRs agonist GTS-21 (10 mg/kg), epinephrine (0.5 mg/kg), norepinephrine (0.9 mg/kg) and agonist β2ARs hexoprenaline sulfate (1.5 mg/kg) on the mortality of mice from sepsis (М+m).
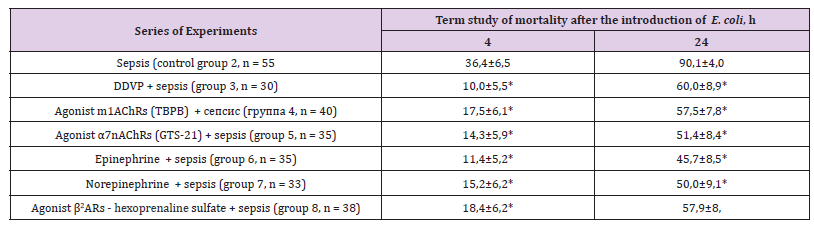
Note:*-p <0,05 as compared to control (group 2).
Table 2: Effects of OPC (DDVP, 0.5 LD50), m1AChRs agonist TBPB (60mg/kg), α7nAChRs agonist GTS-21 (10mg/kg), epinephrine (0.5mg/kg), norepinephrine (0.9mg/kg) and agonist β2ARs hexoprenaline sulfate (1.5mg/kg) on concentrations of proinflammatory cytokines in blood of mice with E. coli sepsis, pg/ml (M ± m; n = 6-8).
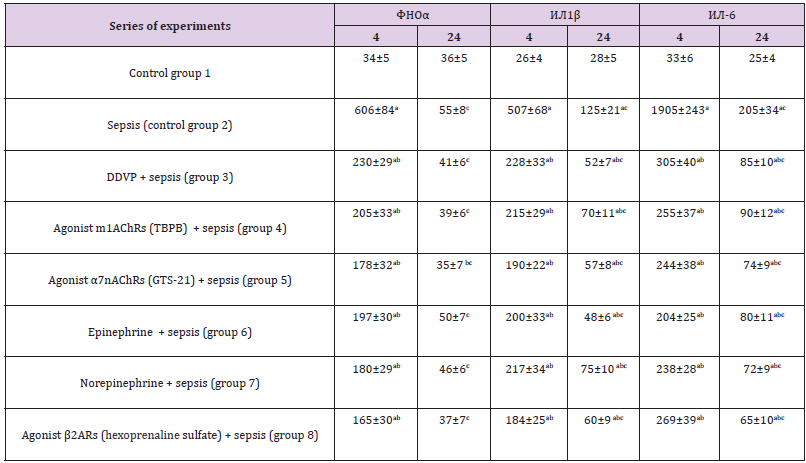
Note: 4 and 24 - after sepsis modeling, h; a-p <0,05 as compared to control (group 1); b-p <0,05 as compared to the corresponding parameter in sepsis (control group 2); с-p <0,05 as compared to the 4 h.
Acute intoxication of OPC (DDVP) decreased the TNF-α, IL-1β and IL-6 blood concentrations 4 h after sepsis modeling (group 3) compared to the control group 2 (sepsis without the use of drugs), respectively, by 2.6; 2.2 and 6.2 times (p <0.05). At the same time, the concentrations of proinflammatory cytokines in the blood significantly (p<0.05) exceeded the corresponding parameters of the control group 1. The concentrations of TNF-α, IL-1β and IL-6 24 h after sepsis modeling decreased in comparison with these parameters after 4 h, remaining below group 2 values in 1.3 (p>0.05), 2.4 and 2.4 times (p<0.05), respectively. The parameters of TNF-α, IL-1β and IL-6 after administration of m1AChRs agonist (TBPB) 4 h after modeling sepsis (group 4) decreased compared with the parameters of the control group 2, respectively, in 3.0; 2.4 and 8.0 times (p<0.05). A decrease in blood levels of proinflammatory cytokines was detected 24 h after sepsis modeling compared with the corresponding values after 4 h, while the concentrations of TNF-α, IL-1β and IL-6 remained below the values of group 2, respectively, in 1.4 (p> 0, 05); 1.8 and 2.3 times (p <0.05).
The values of TNF-α, IL-1β and IL-6, when using the α7nAChRs agonist (GTS-21), decreased 4 h after the modeling of sepsis (group 5) compared to the parameters of the control group 2, respectively, in 3.4; 2.7 and 7.8 times (p<0.05). A reduction of content of proinflammatory cytokines in the blood was established 24 h after the sepsis modeling compared with the corresponding values after 4 h, while the concentrations of TNF-α, IL-1β and IL-6 remained below the values of group 2, respectively, in 1.6 (p> 0,05); 2.2 and 2.8 times (p <0.05). Epinephrine reduced the TNF-α, IL-1β and IL-6 blood concentrations 4 h after the sepsis modeling (group 6) compared with the control group 2 (sepsis), respectively, in 3.1; 2.5 and 9.3 times (p <0.05). IL-1β and IL-6 values after 24 h decreased in 2.6 and 2.5 times (p <0.05), respectively, compared with these parameters in group 2, and the level of TNF-α in the blood did not significantly change. After administration NE the TNF-α, IL- 1β blood levels there was a reduction 4 h after sepsis modeling (group 7) compared with the control group 2 (sepsis), respectively, in 3.4; 1.7 and 8.0 times (p <0.05). The IL-1β and IL-6 blood concentrations were reduced after 24 h compared to the values after 4 h, respectively, 1.7 and 2.8 times (p<0.05), and the content of TNF-α remained almost unchanged.
The parameters of TNF-α, IL-1β and IL-6 after administration of β2ARs agonist (hexoprenaline sulfate) 4 h after sepsis modeling (group 8) decreased compared with the parameters of the control group 2, respectively, in 3.7; 2.8 and 7.1 times (p<0.05). A decrease in blood levels of proinflammatory cytokines was detected 24 h after sepsis modeling compared with the corresponding values after 4 h, while the concentrations of TNF-α, IL-1β and IL-6 remained below the values of group 2, respectively, in 1.5 (p>0, 05); 2.1 and 3.2 times (p <0.05). The IL-1β and IL-6 blood concentrations in groups 3, 4, 5, 6, 7 and 8 were significantly higher (p <0.05) than the corresponding values of the control group 1. The TNF-α, IL-1β and IL-6 blood concentrations after acute intoxication of OPC, the effects of the agonist m1AChRs (TBPB), α7nAChR agonist (GTS- 21), epinephrine, NE and β2ARs agonist (hexoprenaline sulfate) after modeling sepsis 2 h after administration of OPC (DDVP) and these drugs (groups 3, 4, 5, 6, 7, 8) decreased approximately equally compared with the reduction after the modeling sepsis (group 2; without the use of drugs).
After acute OPC intoxication, exposure to the m1AChRs agonist (TBPB), α7nAChRs agonist (GTS-21), epinephrine, NE and agonist β2ARs (hexoprenaline sulfate) 2 h after sepsis modeling (groups 3, 4, 5, 6, 7, 8) there was no significant difference in the mortality of mice . This suggests that the mechanism of action of OPC, which causes a decrease in animal mortality after sepsis, is associated with acetylcholine m1AChRs activation of the brain [16,17], α7nAChRs of MMS cells, nAChRs of adrenal medulla [2,13,16,18, 21,22], as well as activation of epinephrine, NE and agonist β2ARs (hexoprenaline sulfate) of β2ARs of MMS cells [21,22] due to the excitation of the ganglia of the sympathetic nervous system [2,18,21].
There is reason to conclude that the mechanism for reducing mortality in mice with acute intoxication of OPC from sepsis is due to activation in the brain m1AChRs (the first link of the cholinergic anti-inflammatory pathway) [2,16,17], α7nAChR MMS cells [12,17,19], as well as β2ARs macrophages and monocytes (due to the excitation of the sympathetic nervous system) and the action of norepinephrine on T lymphocytes of the spleen, [2,12,13,18]. The implementation of the reduction of pro-inflammatory cytokines TNF-α, IL-1β, IL-6, and others (the occurrence of anti-inflammatory effect) is provided by JAK2 kinase, STAT3 transcription factor, transcription factor NF-κB) [2,12, 17-21,22]. In addition, the decrease in mortality from sepsis after acute intoxication of OPC due to the suppression of the synthesis of proinflammatory cytokines is also associated with the effect of corticosteroids (activation of the hypothalamic-pituitary-adrenal system) [2,3,19,18].
Reduced mortality of mice from sepsis and the blood concentration of proinflammatory cytokines TNF-α, IL-1β, IL-6 after acute intoxication of organophosphorus compounds is due to the implementation of the cholinergic anti-inflammatory pathway: activation of m-acetylcholinergic receptors type 1 (m1AChRs) of the brain and α7n-acetylcholine receptors (α7nAChRs) of the monocyte-macrophage system, as well as stimulation of the sympathetic nervous system, β2-adrenoreceptors (β2ARs) T lymphocytes, macrophages and monocytes.


