Impact Factor : 0.548
- NLM ID: 101723284
- OCoLC: 999826537
- LCCN: 2017202541
Bum Jun Kim1, Jung Hye Kwon*2, Do Yeun Kim3, Sun Kyung Baek4, Jung Hun Kang5, Myung Ah Lee6, Sung Yong Oh7 and In Gyu Hwang8
Received: January 04, 2018; Published: January 11, 2019
*Corresponding author: Kwon Jung Hye, Division of hemato-oncology, Kangdong Sacred Heart Hospital, 150, Sungan-ro, Gangdong-gu, Seoul, 134-701, Republic of Korea
DOI: 10.26717/BJSTR.2019.13.002354
Introduction: Breakthrough cancer pain (BTcP) has been reported to occur in more than half of cancer patients. However, despite the introduction of various treatment options, BTcP remains under-diagnosed and under-treated. Because physicians’ knowledge and attitudes are related to adequate BTcP management, it is important to explore physicians’ knowledge, attitude and clinical practice in managing BTcP.
Objectives: Purpose of this study is to evaluate medical oncologist’s knowledge, attitudes and clinical practice in BTcP management.
Method: We randomly contacted with a medical oncologist (MO) who has been experience more than five years in the field of oncology and conducted a survey.
Results: 30% (50/165) of contacted medical oncologists responded the survey. Median age was 46 years old (range, 35-65) and 70% (35/50) was male. The median knowledge score of BTcP was 7 (range, 2-10) and it was significantly lower than that of general cancer pain (median 9, range 4-10) (p = < 0.001). In the assessment of attitudes, 54% (27/50) of respondents replied to assess cancer pain only when the patient complained of pain and 44% (22/50) of respondents don’t evaluate the cancer pain in detail including breakthrough cancer pain. In the survey of drug preference, oxycodone was most frequently prescribed opioid for the background pain and BTcP. Considerable number of respondents (34%, 17/50) answered that they use transmucosal fentanyl in opioid dose titration.
Conclusion: The knowledge score of BTcP was significantly lower than that of general cancer pain in medical oncologists and a lack of understanding of BTcP led to inadequate management of BTcP.
Pain is one of the most common symptoms associated with cancer, and about 60% of cancer patients experience cancer pain during their illness [1]. Cancer pain is categorized by its nature and divided into two types: background pain and breakthrough pain. Breakthrough cancer pain (BTcP) was first identified in 1990 [2], and for decades, various definitions of BTcP have been proposed to differentiate BTcP from other types of pain. In recent years, the definition proposed by Davies et al. [3] has been widely accepted and is described as follows: “a transient exacerbation of pain that occurs either spontaneously or in relation to a specific predictable or unpredictable trigger, despite relatively stable and adequately controlled background pain.” To control BTcP, short acting opioids are usually recommended as rescue doses by guidelines [4-6]. Traditional short acting opioids, however, have limitations in their pharmacokinetic/pharmacodynamic profiles to control BTcP, which is characterized by short time to peak intensity (10 minutes), short duration (less than 60 mintues), and relatively high intensity [7].
Recently, fentanyl formulations with various delivery routes have been developed and are considered to be proper analgesics in the management of BTcP. Indeed, there are data showing that buccal fentanyl is effective in treatment of BTcP [8-10] and multiple randomized trials suggest that sublingual and nasal transmucosal formulations of fentanyl are also effective in managing BTcP [11,12]. With these results, rapid onset opioids are considered as optimal rescue medication for BTcP among experts. Despite the introduction of various treatment options, BTcP remains underdiagnosed and under-treated. Recent survey of 573 European cancer patients revealed that BTcP was occurred in 63% of cancer patients and only one third of these patients received adequate pain management [13]. One of the major barriers to optimal care for cancer pain is physicians’ misunderstanding and lack of knowledge of pain [14] and recent study has reported that there is a significant association between the level of physicians’ knowledge and proper pain management [15].
Unsurprisingly, uncontrolled BTcP affects patients’ activity, motivation, and overall quality of life [16]. Improperly relieved BTcP also increases the level of anxiety and makes patients be dissatisfied with their overall pain management [17]. Because physicians’ knowledge and attitudes are related to adequate BTcP management, it is important to explore physicians’ knowledge, attitude and clinical practice in managing BTcP.
In Korea, two thirds of physician who do palliative care in the cancer centers are medical oncology specialists [18]. Therefore, we carried out a survey among medical oncologists to explore physicians’ knowledge and attitudes, and clinical practice in BTcP management.
During the month of May 2016, medical oncologists who are registered in Korean Cancer Study Group (KCSG) were randomly contacted by telephone call with an introduction of a survey. Survey paper was distributed to only whom agreed the survey by interviewee. Eligibility includes more than 5 years of experience in the field of oncology and prescribe opioids for more than 20 patients per months. This study was approved by institutional review board.
The survey instrument was developed with referenced to previous research by three investigators (JH Kwon, SK Baek, and MA Lee) and review and feedback were performed among the investigators [15,19-21]. Finally, sixteen questionnaires and one vignette were selected and tested among investigators. Each questionnaire has its own specific objectives and can be grouped into broad categories: assessment of knowledge, attitudes and clinical practice of medical oncologists in the management of cancer pain. To evaluate medical oncologists’ knowledge of BTcP management, true or false questionnaires were used. Two kinds of true or false questionnaire was developed based on the NCCN adult cancer pain guidelines (ver.1.2016) and each included 10 questions. Two questionnaires asked participants about general cancer pain and BTcP. respectively. Scoring from 0 to 10 was used to assess medical oncologists’ knowledge of cancer pain management. To evaluate medical oncologists’ attitudes and their compliance to the cancer pain management guidelines, three questionnaires and one case vignette were used.
These questionnaires were focused on the patient selection for cancer pain assessment, the way how to assess cancer pain, follow up duration after initiation of opioid for cancer pain. A case vignette was presented at the last part of the survey which has two step clinical scenarios. First step is a clinical situation of uncontrolled background pain and second step is a situation for uncontrolled BTcP after controlling background pain. Proper pain management was asked for each step and the type of BTcP was asked for the second step. Additionally, we asked eleven questionnaires to identify their preferences in the management of BTcP in clinical practice. Participants are asked to choose the most preferred opioid for background cancer pain and BTcP. We also asked about individual treatment tendencies for BTcP and their thoughts for ideal characteristics of opioids adequate for BTcP management.
We used Mann-Whitney U test to compare median values and Chi-squared tests to compare percentages between different groups. All significance levels refer to two-sided tests. A P-value of less than 0.05 was considered significant. All statistical analyses were performed using the Statistical Package for the Social Sciences (IBM SPSS, Chicago, IL) version 21.0.
In the current study, we conducted the survey and evaluated medical oncologists’ knowledge, attitudes and clinical practice in the management of BTcP. This study showed that knowledge scores of BTcP was significantly lower than that of general cancer pain. We also identified that a lack of understanding of BTcP might lead to inadequate management of BTcP. In the BTcP knowledge assessment, respondents had difficulty with questions about the definition of BTcP and particularly lacked understanding of the background pain conditions to define BTcP. BTcP is a relatively new concept and it has been less than 30 years since the first effort to identify and define BTcP in 1990. Until now, there is no unanimous consensus on the clinical features to define BTcP despite an international effort to achieve consensus. Considering this situation, BTcP may still be an unfamiliar concept for physicians, even for oncology specialists in Korea. There are ongoing debates on criteria for severity or duration of BTcP and how to define ‘wellcontrolled background pain’ [7,22-25]. Consensus on these debates will lead to the improvement of knowledge for BTcP and until then, a comprehensive multidisciplinary approach is needed to diagnose and manage the BTcP appropriately.
Lack of understanding of BTcP has related to inadequate BTcP management. Half of respondents replied that they don’t ask patients about cancer pain in detail including BTcP and they only assess cancer pain when the patient complained of pain. Questionnaires and case vignette also revealed that considerable number of medical oncologist misuse transmucosal fentanyl during opioid titration. This is an unexpected outcome and may raise an important clinical issue related to the usage of transmucosal fentanyl in BTcP. Transmucosal fentanyl is considered the ideal analgesic for BTcP management and, in this survey, medical oncologists also responded that they prefer rapid onset opioids compared to parenteral or short acting opioids, significantly. However, since the equivalence dose of morphine to transmucosal fentanyl is not established, the usage of transmucosal fentanyl is limited to the management of BTcP and should not be used in the opioid dose titration for background pain. Indiscriminate use of transmucosal fentanyl in opioid dose titration can result in opioid abuse which can lead to aberrant behaviors with opioid and chemical coping.
In the retrospective analysis of 1,160 chronic pain patients receiving transmucosal fentanyl [26], the incidence of drug abuse events (1%) and aberrant drug-related behaviors (11%) were reported to be relatively low. However, opioid abuse events related to transmucosal fentanyl might have been underestimated because this report included patients from controlled clinical studies and over the past decade, the amount of transmucosal fentanyl prescription has increased significantly. There has no risk evaluation and mitigation strategies for opioids in Korea, therefore every physician has allow to prescribe any opioids including transmucosal fentanyl for cancer patients within the insurance guidance [27]. Given our worrying outcomes, thorough education for the diagnosis and management of BTcP is strongly recommended to prepare for the risk of opioid abuse in the future. 17 respondents (34%) replied that they use transmucosal fentanyl when titrating opioid in clinical practice and 14 respondents (28%) answered to the case vignette part 1 that they will use transmucosal fentanyl for opioid dose titration. However, the response to the knowledge questionnaire was not related to the choice for the case vignette in using transmucosal fentanyl for titration.
This might be a limitation of using case vignette to test the knowledge in the clinical practice. Current study has potential limitations. First, low contact number of oncologists and low response rate has some limitation to represent medical oncology societies of Korea. Second, Because the survey did not include the patients’ medical records regarding pain control the result might not reflect real knowledge of medical oncologist in Korea. We also could not find the relation between knowledge and clinical practice. In this survey, we found that some medical oncologists in Korea are unfamiliar with the definition of BTcP and they misuse transmucosal fentanyl in the opioid dose titration. Despite its advantages, transmucosal fentanyl is a double-edged sword which can cause opioid abuse and special warning and education is recommended. Further studies to find the factors related to inadequate knowledge of background pain among physicians and also need to conduct survey with large population reflecting knowledge, attitudes and clinical practice of BTcP management among physicians in Korea.
Table 1 demonstrates the characteristics of respondents. About 39% (165/425) of medical oncologists registered in KCSG were randomly contacted and 30% (50/165) of them were responded the survey. Median age was 46 years old (range, 35-65) and 70% (35/50) was male. Median duration of their career as a medical oncologist was 15 years (range, 6-30) and median number of patients who received opioid prescription during last 1 month before the survey per each medical oncologist was 100 (range, 25- 500).
Note: * n=47.
The 10 true or false questions and responses are listed in Table 2. Median knowledge score of general cancer pain management was 9 (range, 4-10). For most questions, correct answer rate was as high as > 80%. However, the correct answer rate to question about how to stop opioid in patients with resolved pain (Question I) was relatively low (74%). For questions related to BTcP, the median knowledge score was 7 (range, 2-10), and the score was significantly lower than that of questions about general cancer pain (median = 9, range, 4-10; p = < 0.001). Particularly, respondents had difficulty with questions about the definition of adequately controlled background pain to define BTcP (Question C, G) with 22-42% of correct answer rate. We compared knowledge scores between groups stratified by duration of career (>10 vs. ≤10 years) and number of patients receiving patients (>100 vs. ≤100 patients/ month) and there was no statistically significant difference between groups.
Note: * NRS=numeric rating scale.
We asked 3 questionnaires and 1 case vignette to assess medical oncologists’ attitudes and their compliance with cancer pain management guidelines. Twenty-seven respondents (54%) were assess cancer pain only when the patient complained of pain. Only 56% (28/50) of respondents evaluated the pain in detail including breakthrough cancer pain and 26% (13/50) of respondents answered that they only checked for the presence of pain (Table 3). Most of respondent followed up their patients in 2 weeks after initiating opioid analgesics for pain control (88%, 44/50) (Table 3).
Note: BTcP=breakthrough cancer pain.
Most characteristics of BTcP in the survey were considered important more than score 6 on a Likert scale (1 unimportant, 7 very important) (Table 4). More than 70% of respondents replied that they always ask location of pain, current prescribed opioids, and its efficacy when evaluating BTcP (Table 4). Respondents agreed that short action time less than 15 minutes, the maximum duration of action less than 2 hours, trans-mucosal route of administration are the ideal characteristics for BTcP medication (Table 5). Of the three different opioid formulations in the management of BTcP, respondents preferred rapid onset opioid to parenteral and short acting opioids and there was statistical significance (p = < 0.001) (Table 5). 88% (44/50) of respondent also replied that they titrate the dose of opioids according to the intensity of BTcP. They referred to the number of BTcP episodes/day as the most important factor when titrating the opioid dose (Table 5) and many of them responded that they would follow up patient in 3-7 days after the initiation of the first BTcP medication (36%, 18/50) (Table 5).
Table 4: Characteristics of BTcP, its importance and clinical practice among medical oncologist (n=50).
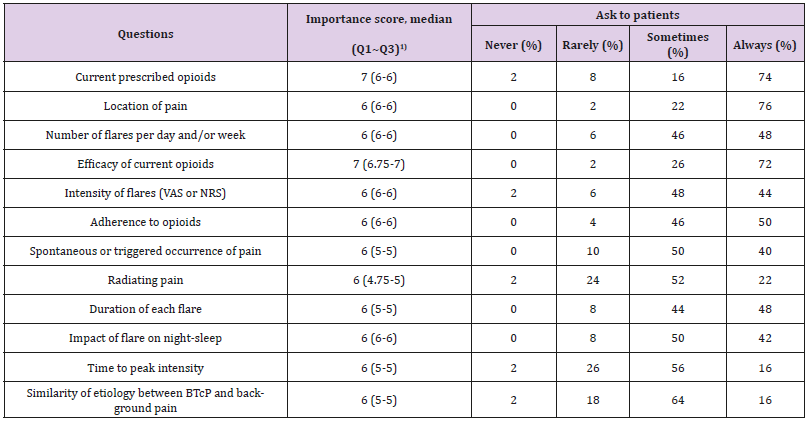
Note: 1) ranged from 1 (unimportant) to 7 (very important), BTcP, breakthrough cancer pain.
Table 5: Medical oncologists’ preference in the management of BTcP. Items were scored from 1 (unimportant or strongly disagree) to 7 (very important or strongly agree) {Citation}.
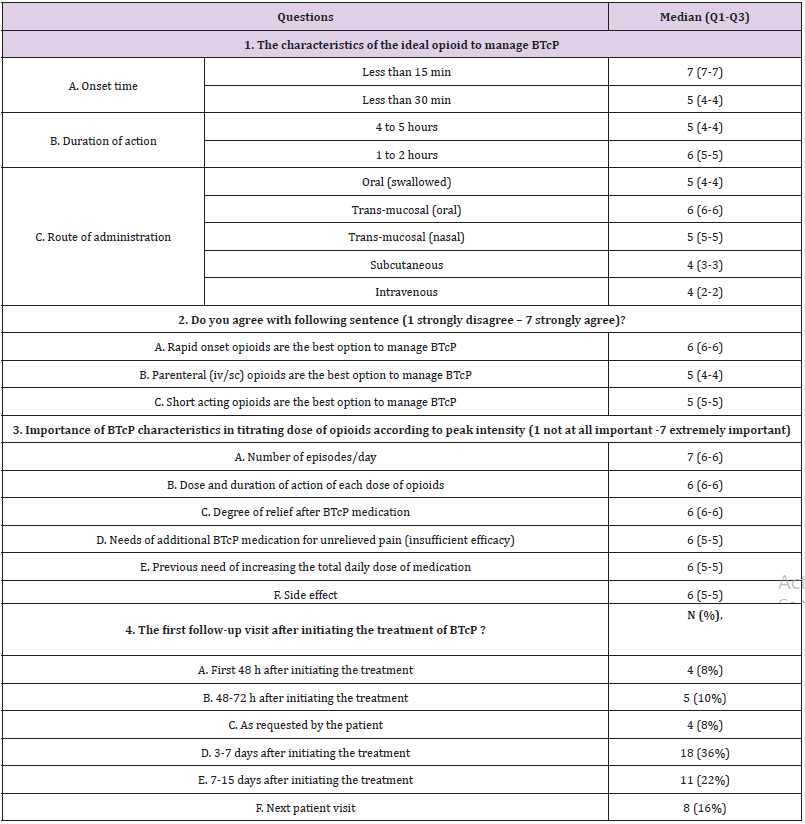
Note: BTcP=breakthough cancer pain.
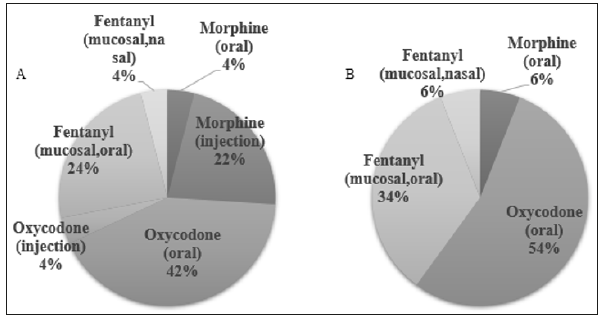
Oxycodone (50%) was most frequently prescribed long acting opioids for background pain followed by fentanyl patch (39%), hydromorphone (7%), and slow releasing morphine (2%) (Figure 1A). Short acting oxycodone was the first drug of choice during the titration of opioids and 34% of respondent replied that they use transmucosal fentanyl when titrating long acting opioids (Figure 1B). Preference for BTcP medication were slightly different between inpatient and outpatient clinic (Figure 2). Short acting oxycodone was most frequently selected medication in both situation (inpatient setting vs. outpatient setting, p = 0.171) followed by transmucosal fentanyl formula (inpatient setting vs. outpatient setting, p = 0.271) (Figure 2).
Figure 1: Long-acting opioids preferred in the management of background cancer pain (A). Short-acting opioids preferred in the opioid dose titration (B).
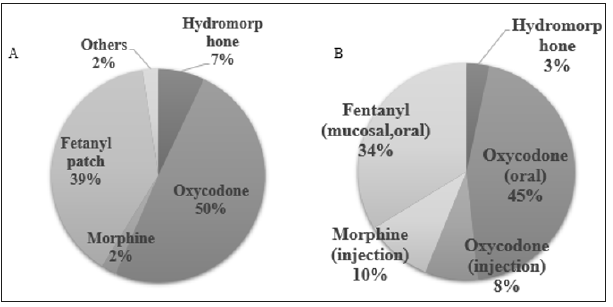
Figure 2: Rapid-acting opioids preferred in the management of breakthrough cancer pain in inpatient (A) and outpatient (B).
In the first case scenario, 38/50 of respondents chose to control background pain with opioid titration (answer D or answer A+E) and 6 of them answered they would like to add transmucosal fentanyl. Five were answered to use only transmucosal fentanyl for the case. Three were answered to increase the dose of long acting morphine and titrate with transmucosal fentanyl. Total 14 respondents replied that they will use transmucosal fentanyl in this case. Using transmucosal fentanyl in the case of uncontrolled background pain was not related to respondent’s age (p = 0.13), years of experience (p = 0.86), and number of patients with opioids (p = 0.66). For the part of BTcP, 49 diagnosed as idiopathic breakthrough pain or unpredictable incidental pain and only one answered as predictable idiopathic pain. 37 answered to use transmucosal fentanyl and 11 chose short acting opioids instead of transmucosal fentanyl. One chooses to increase the dose of long acting opioids and the other chose to increase the dose frequency of long acting opioids. Using transmucosal fentanyl was not related to respondent’s age (p= ?), years of experience (p = 0.67), and number of patients with opioids (p = 0.42).
This study was partly supported by a grant no.2017-06 from the Kangdong Sacred Heart Hospital Fund.


