Impact Factor : 0.548
- NLM ID: 101723284
- OCoLC: 999826537
- LCCN: 2017202541
Wasana K Sumanasekera*1, William Nethery2, Loc Tran3 and Gopal Pillai4
Received: November 08, 2018; Published: November 19, 2018
*Corresponding author: Wasana K Sumanasekera, Department of Pharmaceutical Sciences, College of Pharmacy, Sullivan University, Louisville, KY. USA
DOI: 10.26717/BJSTR.2018.11.002064
Abbreviations: LMWH: Low Molecular Weight Heparins; DVT: Deep Vein Thromboembolism; VTE: Vein Thromboembolism; VEGF: Vascular Endothelial Growth Factor; UFH: Unfractionated Heparin; ULMWH: Ultra Low Molecular Weight Heparins; NCCN: National Comprehensive Cancer Network; TCM: Tumor Cell Conditioned Medium; VLA: Very Late Antigen; VCAM: Vascular Cell Adhesion Molecule; LAC: Low Anticoagulant Activity; AT: Antithrombin; NACH: Non Anti Coagulant Heparin; NALMWH: Non Anticoagulant Low Molecular Weight Heparin; TGFB: Transforming Growth Factor Beta
Cancer is a debilitating, life changing disease that not only affects patients but their families as well. Individuals who suffer through cancer constantly battle against the disease. There are several forms of cancer and many therapies are currently available. In addition to standard therapy, the anti-coagulant class of Low Molecular Weight Heparins (LMWH) can be used for supportive therapy in cancer patients [1]. The anti-cancer properties of LMWH were reviewed by a few groups [2,3]. Existing reviews on the subject are more generalized and do not provide the complete picture of the LMWH’s mechanistic role in attenuating cancer. Our review describes the current evidence supporting beneficial effects of LMWH in cancer patients and the mechanistic details of LMWH’s anti-tumor activities with special emphasis on breast and ovarian cancers.
Breast and ovarian cancers are prevalent in the United States. In addition, they are the leading causes of cancer related deaths among women worldwide [4,5]. In the United States, breast cancer has the highest estimated number of new cases each year and the third highest number of deaths of all cancers each year in women. Ovarian cancer ranks as the fifth highest in number of new cancer deaths (American cancer society, 2018). In 2018, the lifetime risk of developing ovarian cancer is 1.3% (American cancer society, 2018). There are many options to treat breast and ovarian cancers. Chemotherapy, hormonal therapy, surgery, and radiation are all valid options depending on certain factors. Factors such as the stage, node involvement, presence of specific receptors, and the patient’s performance status can help direct therapeutic choices. In addition, patients often require supportive therapy to combat the disease and to prevent adverse effects of their treatment regimen.
Heparin is a naturally occurring polysaccharide with an approximate molecular weight ranging from 5000-40000 Daltons and the mean molecular weight in the range of 12000-14000 Daltons [6]. It is a sulfated polyaminoglycan and therefore can bind to a wide range of molecules via electrostatic interactions. Heparin possesses numerous biological properties beyond their anticoagulant effects, including anticancer properties which have been a focus of interest for decades [7-11]. LMWH, on the other hand, consists of only short chains of polysaccharides with an average molecular weight of 4.5 KDa [12]. The LMWHs are obtained by various methods of fractionation and de- polymerization of polymeric heparin. Products prepared by different processes are dissimilar in physical, chemical and biological properties [6,13]. Due to these differences, several organizations, including the United States Food and Drug Administration, the European Medicines Agency, and the World Health Organization, regard LMWHs as individual products that should not be considered as clinically equivalent to one another [14,15]. The advantages of LMWHs compared to unfractionated heparin include lower risk of bleeding, osteoporosis and thrombocytopenia and they can be used subcutaneously rather than intravenously.
This has practical advantage in that they can be used in an outpatient setting, both for extended duration primary prophylaxis and long-term secondary prophylaxis without the need to monitor for anticoagulation in majority of patients. Patient compliance with LMWH is high in both cancer patients and non-cancer patients [16-18]. Overall, LMWHs offer significant advantages over warfarin in cancer patients in terms of practicality of use, safety profile, and high level of compliance as well as in efficacy. Heparin and the LMWHs are effective in the prevention and treatment of thromboembolic events in cancer patients. LMWH can be used in supportive anticoagulation therapy and recommended for Deep Vein Thromboembolism / venous thromboembolism (DVT / VTE) prophylaxis in cancer patients [1,19-22] because there is a strong association between cancer and thrombosis [2,23]. Cancer patients who are hospitalized or undergoing surgery require DVT prophylaxis [1, 21, 22]. LMWH is the standard recommended agent for both initial and continuing treatment [1, 21,22]. It has been reported that a high dose of Dalteparin (5000 IU) administered once daily for seven days for cancer patient during surgery served as a prophylactic measure [24]. The use of LMWH has shown to have beneficial anti-cancer effects if used in the treatment of patients in the earlier stages of the disease [25]. VTE is one of the leading causes of death in patients with cancer [1,21,22]. In a populationbased case-control study conducted in Olmsted, Minnesota, patients with a malignant neoplasm were 4.1 fold more likely to have a VTE and were 6.5 fold more likely to have a VTE if being treated with chemotherapy than the general population [26].
Clinically 15-20% of all patients with an acute VTE are associated with malignancy [24]. Older patients are also more at risk for developing VTE, which increases the risk of death or major hemorrhage [24]. Cancer patients who develop VTE have a lower chance of survival compared to patients who do not have a VTE [1,24,27]. Prophylactic LMWH has shown benefit in lung cancer patients by preventing VTE-related mortality and improving survival [28,29]. Post-surgical VTE prophylaxis is reported to increase survival rate of patients with advanced ovarian cancer [30]. VTE occurrence is less common in early stage breast cancer [29]; however, VTE occurrence increases during late stage metastatic breast cancer [31]. Patients who had surgery for breast or pelvic cancer demonstrated increased survival with Certoparin treatment, a LMWH, compared to unfractionated heparin [32]. The goal of using LMWH in cancer patient is to prevent complications and reduce mortality and morbidity associated with blood clots and embolism [1,8,21,22]. Use of LMWH improves cancer patients’ outcomes including DVT prevention and increased survival [23,33]. Data from randomized controlled trials suggest that LMWH applied a survival advantage for patients with breast cancer 650 days after treatment [32]. Patients with breast cancer who received LMWH in combination with their chemotherapy showed increased survival rate [1,3], which led many curious researchers to discover other advantageous properties of LMWH. Although LMWH has a primary indication as an anticoagulant, prolonged survival of cancer patients might be attributed to non-anticoagulant mechanisms [34].
Several studies have been done that observed clinical effects on patients’ cancer progression and demonstrate increased survival rates. In the FAMOUS study, advanced cancer patients received dalteparin or placebo and a subgroup analysis was performed. The study found that the sub group of patients who had a better prognosis had improved survival rates at year two and year three in patients receiving dalteparin for one year versus placebo. However, patients with advanced malignancy did not have improved survival rates at one year [35]. Another study also demonstrated similar results utilizing a 6week course of nadroparin versus placebo. An intention-to-treat analysis was performed and demonstrated that patients with advanced malignancy had significantly higher median survival times compared to placebo. A sub group analysis also demonstrated a significant difference in patients who were expected to survive greater than six months, while there was no difference in patients who were expected to survive less than six months [36]. Another study demonstrated a significant increase in progressionfree survival and median overall survival in patients with small cell lung cancer when dalteparin was administered in combination with chemotherapeutic agents versus placebo with chemotherapeutic agents [37]. A Cochrane review demonstrated that heparin based therapy improved survival in patient with limited disease [38]. Although there is evidence that shows improved survival, current guidelines through the National Comprehensive Cancer Network (NCCN) suggest that further study is needed before applying LMWH to antineoplastic regimens.
Heparin, was discovered in 1916 and was approved by FDA in 1930’s for the treatment of DVT and pulmonary embolism PE [39]. As a result of using LMWH for supportive therapy in cancer patients, researchers have identified anti-metastatic properties of LMWH [40,41]. It has been found that LMWH improves survival of cancer patients [40], may interfere with cancer-induced hyper coagulation [42], cancer cell proliferation [43], apoptosis [44], and angiogenesis [45].
Inhibition of Heparanase by LMWH: Heparanase, which is an endoglycosidase enzyme, plays a role in degradation and remodeling of the extracellular matrix via cleaving heparan sulfate (HS), a molecule located on adherent cells [46]. Heparanase is over expressed in human tumors and promote angiogenesis and cause tumor metastasis [46]. Heparan sulfate is a much larger molecule than heparin and, due to the proteoglycans attached to its surface [47], it functions as receptors for several growth factors that regulate processes such as angiogenesis and metastasis [48,49]. When the enzyme heparanase breaks down heparan, these ligands may be released, thereby increasing metastasis. The anti-metastatic effect of non-anti-coagulant LMWH, mimetic of heparan sulfate, can be attributed to their heparanase-inhibiting activity [46,50].
Role of LMWH on Cancer Cell Proliferation and Apoptosis: According to a study by Liang et al., modification of LMWH via acetylation caused decreased anticoagulant effects and increased antineoplastic effects [51]. Anticoagulant and antineoplastic effects of LMWH and its’ acetylated form were evaluated in mice and human breast cancer cells respectively. Both LMWH and acetylated LMWH inhibited proliferation of MDA-MB-231 and MCF-7 cells, while acetylated LMWH showed a greater effect [51]. Based on another study that examined heparin’s anti proliferative properties, LMWH significantly reduced adenocarcinoma in mice [52]. It is known that cancer cells bypass apoptosis, which is a safety mechanism that normal cells undergo when damaged. This allows cancer cells to proliferate unregulated by the normal cell cycle check point control mechanisms inherent to normal cells. Targeted apoptosis of cancer cells was investigated using LHT7-ApoPep-1, an apoptosis-homing peptide-conjugated variant of LHT7 [53]. LHT7 is a LMWH taurocholate conjugate, which exhibit tumorsuppressive activities such as anti-angiogenic characteristics and diminished anticoagulant properties [44].
In addition to apoptosis targeting, LHT7-ApoPep-1 also exhibited anti-angiogenic activity [53]. LHT7-ApoPep-1 has the unique characteristic of binding to apoptotic signaling regions in tumor cells and inducing apoptosis. More apoptotic cells were observed in tumor tissues from mice treated with LHT7-ApoPep-1 than in tumor tissues from mice treated with LHT7 alone. Tumor volume was shown to be decreased by 88.8% from control after 14 days in mice bearing MDA-MB-231 human breast cancer tumors (p< 0.05). Bay et al also described that the strong binding of LHT7-ApoPep-1 to apoptotic cells also appears to contribute to the antitumor activity of LHT7-ApoPep-1 through the “bystander effect”, which refers to the ability of cancer cells affected by anticancer agents to induce damage in neighboring cells [53].
Role of LMWH in Reducing Angiogenesis: Heparin possesses anti-angiogenic activity via binding and inhibiting angiogenic growth factors such as Vascular Endothelial Growth factor (VEGF) [53-55]. LMWH also interfere with fibrin formation induced by cancer cells resulting in inhibition of cancer metastasis [2,55]. Fibrin-coupled angiogenesis is an important determinant in the metastasis of many cancers [55]. Although unfractionated heparin (UFH) and first generation LMWH can be used as anti-angiogenic and anti-metastatic agents for chemotherapy, their anticoagulant activity makes them less suitable for cancer treatment due to the risk of hemorrhages [33]. Compared to unfractionated heparin and first generation LMWH, second generation LMWHs have a lower mean molecular weight, which may lead to a better efficacy and safety profile. Compared to heparin, dalteparin, which is a second generation LMWH, has a low binding affinity to endothelial cells [23], less able to inhibit thrombin and cause less platelet aggregation resulting in decreased risk of hemorrhages [56].
The compound LHT7, a LMWH –taurocholate conjugate, exhibited low anticoagulant activity [44]. LHT7 is reported to elicit anti – angiogenic properties via inhibiting VEGF leading to attenuated tumor growth in MDA-MB231 breast cancer cells along with the apoptotic effect [44]. Second generation LMWHs, such as Bemiparin and the Ultra-Low-MWH (ULMWH) RO-14, have demonstrated reduced endothelial angiogenic characteristics of breast cancer cells [23]. The researchers used breast and lung cancer tumor-cell-conditioned medium (TCM) to test the in vitro effects of Bemiparin and RO-14 on tumor-induced angiogenesis [23]. Bemiparin and s RO-14 significantly reduced TCM induced angiogenesis as measured by capillary-like tube formation assay [23]. Bemiparin and RO-14 are also possible candidates to be used as an adjuvant in chemotherapy.
Role of LMWH on Cancer Cell Adhesion, Invasion, and Metastasis: Heparin’s anti-metastatic property could be attributed to its antagonistic effect on cell-adhesion pathways, specifically P (platelet)-selectin-mediated binding and the VLA-4/VCAM-1 (very late antigen-4/vascular cell adhesion molecule-1) pathway [57]. Selectins are adhesion molecules expressed on leukocytes and endothelial cells which help the cells bind to one another. They are important in host defense mechanisms by facilitating the rolling mechanism of leukocytes along endothelium [58,59]. Tumor cell metastasis also relies heavily on cell adhesion pathways [34]. Therefore, heparin may be able to effectively combat the adhesion properties of tumor cells. In a study done on lung cancer, low anticoagulant activity (LAC) heparin prevented tumor cell attachment to the sub endothelial matrix of lung capillaries by inhibiting cell surface heparan sulfate functions [60]. It has also been theorized that the strong negative charge of heparins results in binding of cancer cells to heparin rather than the negatively charged endothelium, which would reduce the cancer’s ability to adhere to the endothelium [2]. A study by Sudha et al. concluded that second generation, non-anticoagulant heparin inhibits cell adhesion, invasion, and metastasis in mice with pancreatic cancer [61].
The modified heparin they used was sulfated non-anticoagulant heparin (S-NACH) which was devoid of antithrombin (AT) binding and inhibition of systemic AT-dependent coagulation factors such as factor Xa and II. S-NACH effectively inhibited P-selectin mediated pancreatic cancer cell adhesion and invasion similar to Tinzaparin [61]. This provided evidence that systemic anticoagulation is not a necessary component for heparin to inhibit cancer cell adhesion, invasion, and metastasis. This attribute of S-NACH would be desirable in patients at risk of bleeding and would not benefit from regular LMWH. Heparin’s ability to combat tumor cell metastasis seems to work in more than one way. LMWH have both anti-metastatic and anticoagulant properties [41], but also have side effects including hemorrhage, myocardial infarction, and even death. Therefore, designing a heparin derivative with all the appealing anti-metastatic properties is a challenge. To create a desired heparin molecule, one must create a heparin derivative that is a NA-LMWH (non-anticoagulant low molecular weight heparin) with anti-metastatic properties [41]. “Animal studies using nonanti- coagulant heparin (NAC heparin) suggest that it is possible to separate the anti-metastatic and anti-coagulant activities of heparin” [62]. These heparin derivatives could be given without implications in anti-coagulant activity.
Role of LMWH Induced Modulation of Signal Transduction Pathways Involved in Cell Proliferation and Apoptosis: Protein kinase C is involved in cell apoptosis, proliferation, invasion, apoptosis, drug resistance and angiogenesis. Therefore, there is potential for pharmacologic intervention that could help cancer patients (Kang JH, 2014). In a murine fibroblast model heparin inhibited proliferation of cells by blocking the expression of protooncogenes through alterations in the protein kinase C-dependent signal transduction pathway [2,63]. A protein kinase dependent reaction is necessary to induce two proto-oncogenes, c-fos and cmyc, that initiate DNA synthesis. When heparin was used in the fibroblast model, it suppressed fibroblast proliferation and it is possible that this mechanism could affect other cell types [63].
Heparin has also shown that it can inhibit proliferation of smooth muscle cells in vitro and in vivo murine models by inhibition of protein kinase C dependent mitogenic signals [64]. Several isozymes protein kinase C (α, β, δ, ε, ζ, η, and θ) are involved in breast cancer proliferation, differentiation, and apoptosis (Kang JH, 2014). There have not been any studies utilizing heparin or low molecular weight heparins in breast cancer while evaluating the inhibitory effects on protein kinase C. However, there have been studies utilizing small molecule protein kinase C inhibitors which demonstrated growth inhibition, cytotoxicity, and reduction of tumor sphere outgrowth of breast cancer cells in a mouse xenograft model [65]. Further study is needed to determine protein kinase C inhibitory activity of heparins in breast cancer cells.
Figure 1:Action of LMWH on CXCL12 pathway. SDF-1/CXCL12 binding to CXCR4 causes cascade of events such as increase in angiogenesis, cancer cell survival, cellular proliferation, motility, and invasion leading to breast cancer metastases. LMWH inhibits CXCL12 binding to CXCR4 resulting in anti-cancer properties. This figure is a modification from Xu et al., Drug Des Devel. Ther. 2015. Aug 28; 9: 4953-64. Abbreviations: LMWH= Low molecular weight heparin; CXCR4 = C-X-C chemokine receptor type 4; SDF-1 = Stromal derived factor 1. -| Inhibition / blocking. This figure is a modification from Xu et al., 2015.
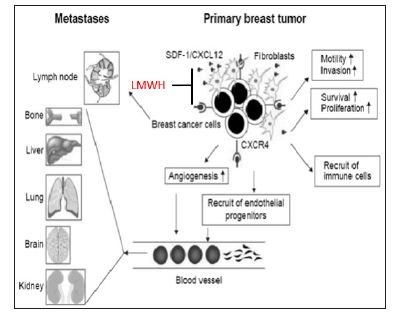
The majority of deaths that occur in patients with breast cancer are due to metastasis to other organs [69]. Favorable effects of LMWH on the CXCR4 mediated cancer metastasis were further investigated [68]. The anticoagulation properties of heparin may limit its usefulness in treating breast cancer patients [68]. Therefore, the heparin with the lowest chain that can inhibit CXCR4/ CXCL12-mediated cell migration with the least anticoagulation activity was investigated [68]. It has been found that dp12, a nonanticoagulant LMWH with an oligosaccharide of 12 sugar units, had significant inhibitory activity against CXCR4 and CXCL12 [68]. Results of this study indicate dp12’s beneficial roles in attenuating cancer metastasis leading to increase survival in patients with nonoperable tumors. Figure 1, modification from Xu et al., 2015 paper summarizes the involvement of CXCL12-CXCR4 pathway in cancer metastasis and the inhibitory actions of LMWH [70].
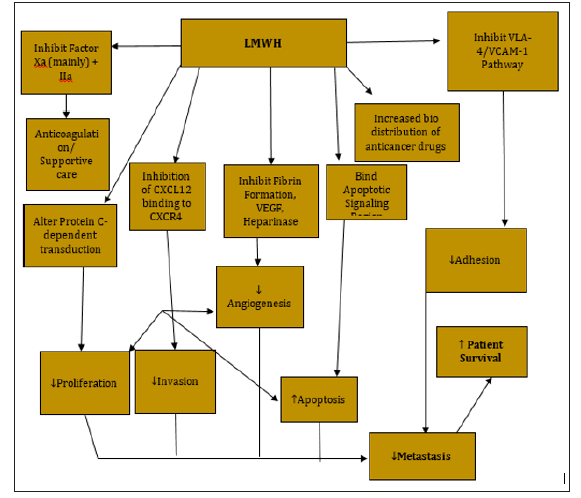
It was found that the group treated with a combination of tinzaparin and paclitaxel showed greater accumulation of paclitaxel in breast cancer tumors than the control groups. When Phillips et al used tinzaparin in combination with another conventional breast cancer chemotherapy agent (doxorubicin), a promising result was found. Tinzaparin was able to selectively increase doxorubicin uptake into breast tumor while reducing the uptake into the heart [3]. This is a unique and special property because doxorubicin is cardiotoxic. There was a synergistic effect of heparin and Doxorubicin. Potency of doxorubicin was increased with the use of heparin [71]. Concurrent use of heparin decreased the efflux of drugs from the cancer cells [71]. The concurrent use of LMWH and Adriamycin caused increased apoptosis and decreased metastasis of breast cancer cells [72]. With these results, the use of LMWH has potential to improve pharmacokinetic parameters of other chemotherapy agents. A model explaining therapeutic potential of LMWH and its mechanisms of action are illustrated in Figure 2.
Figure 2: A model explaining therapeutic potential of LMWH and its mechanism of action. Abbreviations: CXCR4, chemokine receptor type 4; CXCL12, chemokine ligand 12; LMWH, low molecular weight heparin; VLA-4/VCAM-1, very late antigen-4/ vascular cell adhesion molecule-1; VEGF, vascular endothelial growth factor.
Although the role of LMWH in DVT prophylaxis has been well established, there is much to be learned about their potential in anticancer therapy. Breast cancer, like many other types of cancer, possesses characteristics that can be targeted by therapeutic intervention. For example, breast cancer displays angiogenic characteristics, metastasis, or specific receptors located on tumor cells. These characteristics may well be the targets for the low molecular weight heparins which were originally designed to have efficient anticoagulant properties without the usual problems of using heparin. Results of recent investigations on the effects of LMWH on angiogenesis, apoptosis, chemokines and enhancement of pharmacokinetic properties of other established anticancer agents indicates a future role for these agents in cancer therapy. The problem of severe hemorrhage can now be overcome by selecting an LMWH that has very little anticoagulant property but retains the anticancer effect. The discovery of LMWH having anti-cancer properties is a glaring example of cancer research progression. Studies have shown that the usage of heparin can reduce the cancer-related mortality. Although there may be benefits to the use of LMWH, there are also drawbacks. While the adverse effects of LMWH are rare, they can be serious. However, benefits overweigh the risks.
We would like to acknowledge Drs. James Joseph, Nancy Nguyen, Diana Kuznetsova, and Robert Conzelman for their contributions in literature search.


