Impact Factor : 0.548
- NLM ID: 101723284
- OCoLC: 999826537
- LCCN: 2017202541
Seung Heon Shin*, Mi Kyung Ye, Mi Hyun Che and Dong Won Lee
Received: October 27, 2018; Published: November 15, 2018
*Corresponding author: Seung Heon Shin, Department of Otorhinolaryngology, South Korea
DOI: 10.26717/BJSTR.2018.11.002050
Objective: Nano-silver has antimicrobial and disinfectant properties with considerable cellular toxicity. The aim of this study was to investigate the effect of nano-silver on chronic rhinosinusitis (CRS) using an eosinophilic CRS mouse model.
Methods: The CRS mouse model was established with ovalbumin and Staphylococcus aureus enterotoxin B (SEB). Mice were treated with SEB for 16 weeks and nano-silver was intranasal applied three times a week from weeks 9 through 16. The level of inflammatory cell infiltration and chemical mediators in nasal lavage fluid were determined. Histologic changes were observed using hematoxylin and eosin staining and Periodic acid Schiff (PAS) staining.
Results: Intranasal administration of nano-silver resulted in a significant decrease in inflammatory cell infiltration and interferon-gamma levels in nasal lavage fluid. Nano-silver treated mice displayed significantly decreased inflammatory cell infiltration and PAS-positive cells in nasal mucosa. However, interleukin-4 level and epithelial thickness were not influenced.
Conclusion: Nano-silver effectively reduced Th1 inflammatory response in the eosinophilic CRS mouse model. Although, nano-silver had immunomodulatory effects, for the clinical usage further studies are needed to prove the pharmacokinetic and pharmacodynamic characteristics of nano-silver.
Keywords: Nano Silver; Chronic Rhinosinusitis; Mouse Model; Staphylococcus Aureus Enterotoxin B
Abbreviations: CRS: Chronic Rhinosinusitis; SEB: Staphylococcus Aureus Enterotoxin B; PAS: Periodic Acid Schiff; PBS: Phosphate Buffered Saline; NLF: Nasal Lavage Fluid
Silver has long been used as a disinfectant and antimicrobial agent. Metallic nanoparticles have been reported to have broad spectrum antibacterial properties [1]. The bioactivity and the properties of nano-sized metal particles are a very interesting area for researchers. Many metallic nanoparticles have been utilized as a special class of chemotherapy and are an emerging medical therapy. However, a high dose of metallic nanoparticles can be potently toxic and cytotoxic to organism they enter [2]. High concentration of nano-silver also has a cytotoxic effect associated with the modulation of chemical mediators in peripheral blood mononuclear cells at appropriate concentrations [3]. With the development of nanotechnology, nano-silver has been applied in various products from home appliances to medical devices [4,5]. Silver nanoparticle possess antibacterial and anti-inflammatory activities, and have been used for medical device to prevent infection [6]. The antibacterial activity of nano-silver is commonly studied and these nanoparticles have been demonstrated capable of restricting the survival of Escherichia coli, Staphylococcus aureus, and Streptococcus pyogenes [7,8]. Nano-silver can attenuate Staphylococcus aureus biofilm effect in chronic rhinosinusitis sheep model [9].
The anti-inflammatory effect of nano-silver was demonstrated in a murine model of allergic contact dermatitis and allergic rhinitis [10,11]. Nano-silver suppressed the expression of tumor necrosis factor-alpha (TNF-α) and interleukin (IL)-6 in dermatitis and suppresses IL-4 and IL-10 in allergic rhinitis. However, antimicrobial and anti-inflammatory mechanisms of nanoparticles are still under investigation and the specific immune characteristics of nanoparticles remain unclear. The properties of nanoparticles vary depending on the size uniformity, bioactivity, and purity of particles, and on the target surface molecules [12]. Nano-silver is widely applied in consumer products ranging from home appliances to medical devices [4]. The bioactivity, biological behavior, and potential toxicity of nano-silver are important areas to research to clarify potential medical applications. Because nanosilver has anti-inflammatory action, we presently used a mouse model of allergic chronic rhinosinusitis to evaluate the effect of nano-silver instillation on nasal mucosa inflammation.
Nano-silver colloidal solution was prepared by chemical reduction of silver ion with the aid of reducing agent and stabilizers. Silver nitrate was dissolved in distilled water and stabilizer. Then, the reducing agent dissolved in water was added drop-wise to silver ion-stabilizer solution while sonicating. Sodium hydroxide is another stabilizer. It neutralizes nano-silver. Sodium hydroxide was added; final products were sodium nitrate and silver. The particle size and ultraviolet-visible spectrum of nano-silver was determined by transmission electron microscopy using an ELS-8000 device (Otsuka electronics, Osaka, Japan). The average particle diameter was about 1.5nm. The size distributions were about 1-1.5nm in well dispersed colloidal solutions.
A chronic rhinosinusitis (CRS) mouse model was made as described previously with some modifications [13]. Female BALB/c mice free of murine specific pathogens were obtained from Hyosung Science Inc. (Daegu, South Korea). They were maintained under standard laboratory conditions in a pathogen-free cage. Food and water were freely available and all animal experiments in this study have been approved by the ethics committee and were conducted in accordance with the guidelines of the Institutional Review Board of Animal Experiments of Daegu Catholic University Medical Center. The model involved the use of ovalbumin (OVA, grade V; Sigma-Aldrich, St. Louis, MO, USA) and staphylococcal enterotoxin B (SEB; Sigma-Aldrich). Mice were sensitized by administration of an intraperitoneal injection of OVA at a concentration of 75μg in 200sμl phosphate buffered saline (PBS) containing 2mg of aluminum hydroxide (Sigma-Aldrich) on days 0, 7, 14, and 21, followed by a daily intranasal instillation of 500μg of OVA from days 22 to 25. Inflammation was maintained in the experimental mice by the subsequent nasal instillation of OVA three times a week for 4 consecutive weeks. To develop eosinophilic CRS, mice were challenged weekly with 5ng/ml or 500ng/ml of SEB from week 9 through 16 after OVA instillation.
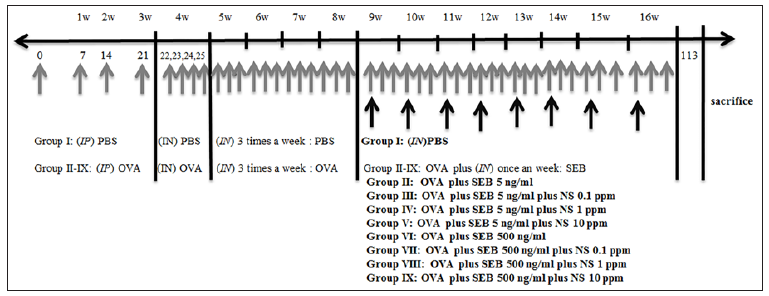
To determine the effect of nano-silver on the development of eosinophilic CRS, 0.1, 1, and 10ppm of nano-silver were intranasal applied three times a week 9 through 16. At day 113, mice were sacrificed for further study. There were nine study groups, with seven mice per group: PBS instillation only (group I), OVA with 5ng/ml instilled SEB (group II), OVA with 5ng/ml instilled SEB and treated with 0.1ppm NS (group III), OVA with 5ng/ml instilled SEB and treated with 1ppm NS (group IV), OVA with 5ng/ml instilled SEB and treated with 10ppm NS (group V), OVA with 500ng/ml instilled SEB (group VI), OVA with 500ng/ml instilled SEB and treated with 0.1ppm NS (group VII), OVA with 500ng/ml instilled SEB and treated with 1ppm NS (group VIII), and OVA with 500ng/ml instilled SEB and treated with 10ppm NS (group IX) (Figure 1).
Figure 1: Schematic diagram of ovalbumin (OVA) sensitized staphylococcal enterotoxin B (SEB) induction of allergic chronic rhinosinusitis mouse model. Nano-silver (NS) was intranasally (IN) applied three times a week from week 9 through 16 (gray arrow). IP: intraperitoneal, PBS: phosphate buffered saline; gray arrow: intranasal application of OVA or PBS; black arrow: intranasal application of SEB.
NLF was collected by an 18-gauge catheter through partial tracheal resection to the nasopharynx, just before sacrifice. The nasal cavity was gently washed with 1mL cold PBS and collected in a tube. The collected fluid was centrifuged to collect the supernatant and pellet. Supernatant was stored at -70 °C until being assayed for IL-4, IL-10, and interferon-gamma (INF-γ) using an ELISA quantification kit (R&D Systems, Minneapolis, MN, USA). The limit of detection was < 2pg/ml of each cytokine. To determine the inflammatory cell composition of NLF, the pellet was resuspended in PBS. Ten microliters of the cell suspension was stained with May- Grunwald-Giemsa stain and cells differentiated into eosinophils, neutrophils, lymphocytes, and other cells were determined as average number of cells in five high power field views.
Mice were painlessly sacrificed with a lethal dose of intraperitoneally administered pentobarbital sodium. Specimens were decalcified in ethylenediaminetetraacetic acid and embedded in paraffin. The tissue was cut in 5-μm-thick coronal sections. Three anatomically similar sections were chosen as in a previous study [11]. The first section, which was the most anterior, was at the level of the maxillary sinuses. The second section, which was more posterior, was at the end of the maxillary sinuses and the beginning of the complex ethmoid turbinals. The third section, which was the most posterior, contained the brain. Appearance of inflammatory cell infiltration and epithelial thickness were quantified in hematoxylin and eosin stained sections. The degree of submucosal inflammatory cell infiltration was quantified into four categories ranging from 0 to 3 (0: no, 1: mild, occasional scattered inflammatory cells, 2: moderate, 3: severe, diffuse infiltration of inflammatory cells). Goblet cell numbers were quantified by Periodic acid Schiff (PAS) staining at × 200 magnifications and the average number of goblet cells was counted using an eyepiece reticule. Epithelial thickness was directly measured at × 400 magnification through a video camera (Olympus Optical Co. Ltd, Tokyo, Japan) and analyzed with DP controller software (ver. 2.2.1.227). All tissue sections were examined blindly with respect to the source of the tissue and counts were determined at three different mucosal areas for each of the three sections per mouse.
All measured parameters are expressed as the mean standard error of mean for representative seven independent experiments for each group. The one-way analysis of variance followed by Tukey’s test for normally distributed data and the Kruskal-Wallis tests with post-hoc Bonferroni-Dunn test for nonnormally distributed data (SPSS ver. 21, IBM Corp., Armonk, NY, USA). A p value < 0.05 was considered statistically significant.
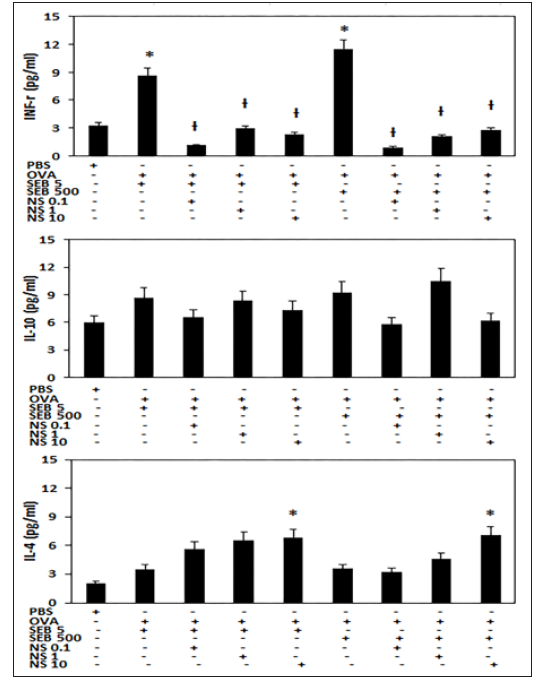
When the mice were treated with SEB, neutrophils and eosinophils were significantly increased in NLF (5ng/ml of SEB: neutrophils 6.3 ± 3.2, eosinophils 25.5 ± 13.6; 500 ng/ml of SEB: neutrophils 8.0 ±3.7, eosinophils 13.5 ± 2.7 with). Intranasal administration of nano-silver resulted in a significant decrease in inflammatory cell infiltration of NLF. However, the numbers of lymphocytes tended to increase with nano-silver instillation (Figure 2). When mice were treated with SEB, INF-γ levels in NLF was significantly increased in the eosinophilic CRS model (SEB 5ng/ml: 8.7 ± 3.6pg/ml, SEB 500ng/ml: 11.5 ± 6.2pg/ml), compared with control group (3.2 ± 2.5pg/ml). Increased level of INF-γ in NLF was significantly suppressed by 0.1ppm of nano-silver (SEB 5ng/ml: 1.2 ± 0.5pg/ml, SEB 500ng/ml: 0.9 ± 0.2pg/ml), 1ppm of nano-silver (SEB 5ng/ml: 3.0 ± 0.6pg/ml, SEB 500ng/ml: 2.1 ± 0.5pg/ml), and 10ppm of nano-silver (SEB 5ng/ml: 2.3 ± 0.5pg/ml, SEB 500ng/ml: 2.8 ± 0.8pg/ml). When the eosinophilic CRS mice were treated with nano-silver, the IL-4 level in NLF tended to increase in a nano-silver concentration dependent manner. However, nano-silver did not influence the IL-10 level in NLF (Figure 3).
Figure 2:Effects of nano-silver (NS) on inflammatory differentials in nasal lavage fluid of the staphylococcal enterotoxin B (SEB) allergic chronic rhinosinusitis mouse model. Neutrophil and eosinophil counts were significantly decreased with intranasal application of NS. NC: negative control, *: p<0.05 vs. PBS group, Ɨ: p<0.05 vs. SEB challenged group.
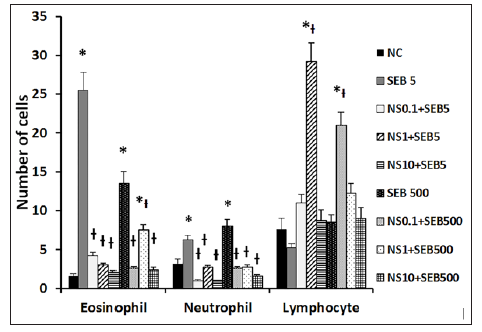
Figure 3: Effects of nano-silver (NS) on production of interferon (INF)-γ, interleukin (IL)-4, and IL-10 in nasal lavage fluid of the staphylococcal enterotoxin B (SEB) induced allergic chronic rhinosinusitis mouse model. SEB-induced INF-γ production was significantly inhibited by NS at concentrations of 0.1, 1.0, and 10 ppm. PBS: phosphate buffered saline, OVA: ovalbumin, *: p<0.05 vs. PBS group, Ɨ: p<0.05 vs. SEB challenged group.
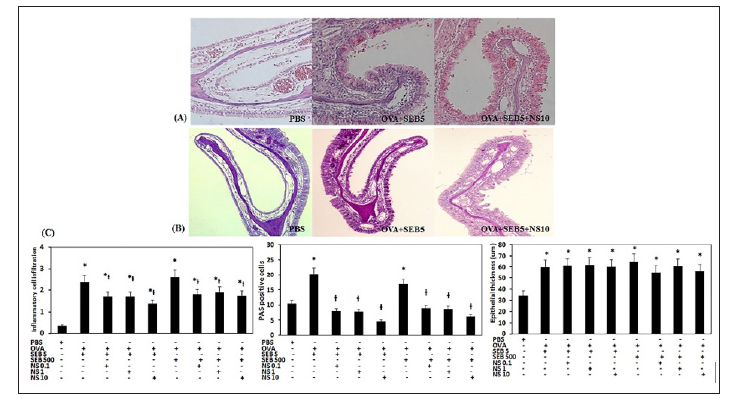
The eosinophilic CRS mouse model displayed increased inflammatory cell infiltration in the submucosal area of the nasal cavity (5ng/ml of SEB: 2.4 ± 0.5, 500ng/ml of SEB: 2.6 ± 0.3). When the nano-silver was intranasal distillated, inflammatory cell infiltration was significantly decreased; 0.1ppm of nano-silver (SEB 5ng/ml: 1.7 ± 1.0, SEB 500 ng/ml: 1.8 ±1.1), 1ppm of nano-silver (SEB 5ng/ml: 1.7 ± 0.8, SEB 500ng/ml: 1.9 ±1.2), and 10 ppm of nano-silver (SEB 5ng/ml: 1.4 ± 0.7, SEB 500ng/ml: 1.8 ±0.8). PAS-positive goblet cells in nasal mucosa showed a significantly increased in the eosinophilic CRS model (5ng/ml of SEB: 22.3 ± 4.3, 500ng/ml of SEB: 16.8 ± 3.7) compared to the negative control group (10.4 ± 2.7). This increased number of PAS-positive cells were subsequently significantly decreased with intranasal instillation of 0.1ppm of nano-silver (SEB 5ng/ml: 7.9 ± 2.4, SEB 500ng/ml: 9.0 ± 3.2), 1ppm of nano-silver (SEB 5ng/ml: 7.9 ± 2.1, SEB 500ng/ ml: 8.7 ± 1.8), and 10ppm of nano-silver (SEB 5ng/ml: 4.7 ± 1.1, SEB 500ng/ml: 6.2 ± 2.8). However, thickness of epithelial cells in nasal mucosa was not significantly influenced by the intranasal instillation of nano-silver (Figure 4).
Figure 4: Effects of nano-silver (NS) on the histologic change in the staphylococcal enterotoxin B (SEB) allergic chronic rhinosinusitis mouse model. Representative histological sections stained with hematoxylin and eosin staining (A) and periodic acid-Schiff (PAS) staining (B). Inflammatory cell infiltration and the number of PAS positive cells of nasal mucosa were significantly inhibited by NS at concentrations of 0.1, 1.0, and 10 ppm. Epithelial thickness was not influenced by intranasal instillation of NS (C). PBS: phosphate buffered saline, OVA: ovalbumin, *: p<0.05 vs. PBS group, Ɨ: p<0.05 vs. SEB challenged group.
The antibacterial, antifungal, anticancer, and application for drug delivery of nano-silver has been well documented [5,14,15]. However, the anti-inflammatory property of nano-silver is unclear. Nanocrystalline silver reduces wound inflammation and modulates fibrogenic cytokines, which could suppress inflammatory events in the early phase of wound healing [16]. CRS is a chronic inflammatory disease of the nasal and paranasal sinus mucosa with increased infiltration of inflammatory cells. SEB is a bacterial superantigen capable of inducing immune dysfunction of nasal mucosa. Although, nano-silver has shown anti-biofilm activity, it did not demonstrate meaningful subjective or objective improvements and is not superior to oral antibiotics in CRS patients [9,17,18]. In the present study, the appropriate concentration of nano-silver suppressed INF-γ but not IL-4 production and inflammatory cell infiltration in an eosinophilic CRS model. We used 0.1 to 10ppm of nano-silver due to the nasal epithelial cell survival was significantly decreased at nano-silver concentrations exceeding 10 ppm and higher concentration of nano-silver influenced the survival of mouse [11,19]. Nano-silver has both beneficial and harmful effects on human cells. Nanoparticles can undergo a series of process, such as binding and reaction with proteins, phagocytosis, deposition, clearance, and translocation.
These biologic activities are determined by the size, shape, morphology, particle composition, surface charge, particle reactivity in solution, efficiency of ion release, and the type of reducing agents used for the synthesis of nano-silver [20,21]. The physiochemical characteristics of each nanoparticle determines cellular uptake, biological distribution, and penetration into biological barriers [22]. Nano-silver used in this study was stabilized with a polymer capsule. When the nano-capsule comes in contact with cells, a polymer capsule can collapse and nanoparticles react with cells as a catalyst without intracellular accumulation. Polymer capsule can dissolve in water without aggregation. Intranasal application of polymerized nano-silver could have anti-inflammatory properties with cellular safety and without systemic absorption. The eosinophilic CRS mouse model displayed an increased number of eosinophils and neutrophil in NLF. The INF-γ level was significantly increased in NLF. Intranasal instillation of nano-silver suppressed INF-γ level in NLF. Nano-silver also suppressed inflammatory cell infiltration and the number of mucus producing cells in nasal mucosa. Nadworny et al. [23] suggested that the anti-inflammatory properties of nano-silver due to the increased apoptosis of inflammatory cells and decreased level of pro-inflammatory cytokines. This might be associated with the anti-inflammatory effect of nano-silver by suppressing the Th1 immune response and the production of mucus. 10ppm of nano-silver increased the IL-4 level in NLF of mouse model. These results indicate that nano-silver itself can induce adverse response at higher concentration and can irritate nasal mucosa and the interaction of SEB and nano-silver aggravated OVA-induced production of IL-4.
Nano-silver has anti-inflammatory effect in mouse model with eosinophilic CRS, decreasing the production of Th1 cytokine, mucus production, and inflammatory cell infiltration in nasal mucosa. However, intranasal application of nano-silver enhanced the production of Th2 cytokine. For the clinical use of nano-silver, further studies are needed to determine the pharmacokinetics and pharmacodynamics of nano-silver.

