Impact Factor : 0.548
- NLM ID: 101723284
- OCoLC: 999826537
- LCCN: 2017202541
Saloua Khalfaoui*1 and Hafid Arabi2
Received: October 10, 2018; Published: November 14, 2018
*Corresponding author: Saloua Khalfaoui, Service de médecine physique et Réadaptation Fonctionnelle (MPR), Hôpital Militaire d’instructions Mohamed V, Rabat, Morocco
DOI: 10.26717/BJSTR.2018.11.002040
Etiopathogenesis of Heterotopic ossification or paraosteoarthropathy is unknown, all speculations remain open. Epilepsy and anti-epileptic drugs have never been mentioned as a risk factor. We report the case of an epileptic patient operated several times for fracture of the right humeral head. He was treated by anti-epileptic (Valproic Acid). He developed a stiffness of the right shoulder. Imaging showed a heterotopic ossification. Through this observation, we have tried to answer the question of the origin of the appearance of the heterotopic ossification; this genesis is multifactorial with a role of the anti-epileptic whose Valproic Acid.
Keywords: Heterotopic Ossification; Anti-Epileptic Drugs; Valproic Acid; Shoulder; Epilepsy
Abbreviations: HO: Heterotopic Ossification; AEDs: Anti-Epileptic Drugs; CT: Computed Tomography; BM: Bone Marrow; MSC: Mesenchymal Stromal Cells
Heterotopic ossification (HO) or paraosteoarthropathy is called neurogenic when it occurs as a result of central or peripheral nervous system involvement. A HO may also occur in a context of bone trauma or burns or in an orthopedic context [1]. Epilepsy and anti-epileptic drugs (AEDs) have never been mentioned as a risk factor.
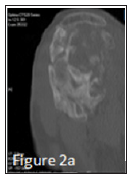
A 30 years old patient with no antecedent had a painful posterior dislocation of the right shoulder, due to an initial epileptic seizure (Figure 1a). Neurological examination and cerebral Computed tomography (CT) showed no abnormality; he was treated by an anti-epileptic (Valproic Acid). The reduction of the posterior dislocation with sedation caused a fracture of the head without neurological injury (Figure 1b). The patient was operated on three occasions: he was operated on day one for the initial fracture (anterior approach, Figure 1c). The postoperative was marked on day seven by a superficial cutaneous infection (C-reactive protein at 54 mg / L, normal complete blood count, and preservation of the general state). The sampling showed an enterobacter clouaca complex. He was treated by antibiotherapy based on gentamicin 160mg / L for five days and ciprofloxacin for two months, the evolution was remarkable after one month. He was operated at day 7 for insufficient reduction of the humeral head (removal of material and introduction of new material (Figure 1d, anterior and posterior approach). There was no sign of deep infection. The progress was marked by the absence of recurrence of epilepsy since the first crisis and stiffness. At day 30 of dislocation, there was an appearance of periarticular ossification compromising the function of the shoulder (Figures 1e & 1f). Computed tomography (CT) of the shoulder showed the localization of ossification (Figures 2a-2c). At day 45, Rehabilitation sessions were done but without improvement. He was operated at day 60 of the last act for the removal of the material without sign of infection (anterior approach). Currently, the patient always keeps stiffness on the activities of the daily life, the hand-mouth is possible but the limitation of the abduction and the external rotation makes difficult hand-neck. Scintigraphic evaluation of osteoblastic activity would be realized to decide on a possible arthrolysis of the shoulder.
Figure 2A: Heterotopic ossification on a sagittal cross section in computed tomography (CT) of the shoulder.
We distinguish metastatic calcification, dystrophic calcification, and ectopic ossification. In ectopic ossification, there is: progressive myositis (congenital disease), post-traumatic ossification myositis, sequelae of burns, complications of surgery, and neurological lesion. The etiopathogenesis of HO is not yet known exactly, although there are several models of HO induction in animals [1], like the technique of injecting bone marrow into a muscle [2]. Among the factors of the formation of the HO, some authors have evoked vascular changes related to a dysfunction of the autonomic nervous system [3]; the existence of a prolonged immobilization, it is responsible for a bone demineralization resulting in a release of calcium which can participate in the calcification of the neighboring soft tissues [4]; bone microtrauma, due to repeated passive kinesitherapic manipulations and going beyond the articular amplitudes, are responsibles for a local inflammatory state, favoring the release of humoral factors that stimulate osteogenesis [4]; the existence of sepsis, or a pressure ulcer leads to the appearance of a inflammatory syndrome, which also promotes the onset a HO [4]; spastic hypertonia would act by inducing bone micro-traumas linked to the stresses exerted by muscle contractures on the articulation [5].
In our case, we note several factors whose connection is unknown: the various surgical procedures, kinesitherapic manipulations, prolonged immobilization and a local inflammatory state such as sepsis. We eliminated the role of kinesitherapy that began after the onset of HO, we doubt about the role of infection because it was cutaneous and superficial; there was no damage to the central nervous system to incriminate spasticity. Could Epilepsy as a central neurological disorder promote HO with the intervention of other factors? We wonder what role the anti-epileptic drugs (AEDs), could play. Epilepsy is a common neurological disorder worldwide and AEDs are always the first choice for treatment, the treatment of our patient was Valproic Acid. However, more than 50% of patients with epilepsy who take AEDs have reported bone abnormalities [6]. In our observation, we believe that the multiple surgeries have favored HO by release of bone tissue and the intervention of anti-epileptic drugs as Valproic Acid. We wonder about the role of mesenchymal stromal cells in the genesis of OH after the fracture of the humerus. The mesenchymal stromal cells of adipose tissue were initially isolated from the bone marrow (BM-MSC) [7]. BM-MSC are multipotent, that is, they can differentiate into various types of cells derived from the mesoderm [8] (bone, cartilage, adipose tissue); these cells have great potential for cellular therapies as they can be directed to differentiate into certain lineages [9].
Recent studies have reported that Valproic Acid influences osteogenesis in vivo and in vitro [10,11], the study indicates that Valproic Acid increase the recruitment of Mesenchymal stromal cells (MSC) to sites of injury without compromising their ability to proliferate or differentiate [12]. Valproic Acid promoted osteoblastic differentiation [10]. Etiopathogenesis of HO is unknown, all speculations remain open, and the links that can exist between several clinical entities deserve clarification. In the case of a stiff shoulder post traumatic or postoperative consider making radiography and do not hastily think of a capsulate. Further investigation is warranted with regard to the predisposing a factor of HO.

