Impact Factor : 0.548
- NLM ID: 101723284
- OCoLC: 999826537
- LCCN: 2017202541
Orlando J Castejon*
Received: October 09, 2018; Published: October 31, 2018
*Corresponding author: Orlando J Castejon, Biological Research Institute, Venezuela
DOI: 10.26717/BJSTR.2018.10.001988
The alteration of nerve cell plasma membranes is reviewed in some neuropathological conditions. In moderate brain oedema a continuous plasma membrane is observed but the cytoplasmic membranes, such as smooth and rough endoplasmic reticulum membranes appear damaged. In severe oedema, fragmentation of plasma membrane, enlargement and focal necrosis of rough endoplasmic cisterns and nuclear envelope, detachment of membrane-bound ribosomes, and reduction of polysome are found. Shallow and deep invaginations of plasma membrane, and the formation of endocytic and clathrin-coated vesicles are seen. In astrocyte cells, areas of focal necrosis and fragmented limiting plasma membrane, overdistended rough endoplasmic reticulum cisterns with extense degranulated membrane domains, and vacuoles of smooth endoplasmic reticulum with necrotic limiting membrane are observed. Oligodendroglia cells show notably edematous changes featured by lacunar enlargement of rough endoplasmic reticulum and nuclear envelope, detachment of membrane bound ribosomes, and discontinuous plasma membrane. Plastic changes and damage of synaptic membranes are found.
Synaptic vesicle exocytosis and endocytosis at the non-specialized regions of presynaptic ending limiting membrane are frequently observed at activated synapses. In severe brain edema, synaptic disassembly occurs featured by wide separation of pre and postsynaptic membranes, and loss of peri synaptic astrocytic glial escheatment. Disruption, fusion and disassembly of interastrocytary gap junctions have also been observed. The endothelial cell luminal membrane of brain capillaries undergoes profound activity changes that characterize increased cerebrovascular permeability, such as increased formation of micro- and macropinocytotic vesicles, clathrin coated vesicles, and emission of pseudopods to form endothelial vacuoles. The alterations of nerve cell plasma membranes and cytomembranes are related with the anoxic-ischemic conditions of brain parenchyma. The role of free radical and lipid peroxidation, disturbed energy metabolism, altered metabolic cascades, glutamate excitotoxicity, haemoglobin toxicity, protein aggregation, and presence of extracellular oedema fluid are discussed in relation with the derangement of nerve cells membranes.
Abbreviations: Endoplasmic Reticulum (ER), Prion Protein (Prp), Parkinson's Disease (PD), Amyotrophic Lateral Sclerosis (ALS), Unfolded Protein Response (UPR), Mitochondria-Associated ER Membranes (Mams) Alzheimer's Disease (AD), Vesicle-Associated Membrane Protein-Associated Protein B (VAPB), Protein Tyrosine Phosphatase-Interacting Protein 51 (PTPIP51), Nitric Oxide Synthase (NOS), Mitochondria (M), Neural Cell Adhesion Molecules (NCAM)
Over the four past decades substantial progress have been made in elucidating the mechanisms by which nerve plasma membranes and cytomembranes are damaged. Nerve cell membrane alterations were earlier described by Bass [1] in Creutzfeldt- Jacob disease. Significant enrichment in cholesterol ester was found by [2] In nerve cell membranes in adrenoleukodystrophy. Disorganization of endoplasmic reticulum cisterns, formation of lamellar bodies, and hypertrophy of Golgi complex were described by Malunova and Samoilov [3] in cat cerebral cortex as early post-anoxic changes. Tissue surrounding hematomas, traumatic lesions, infective zones, and certain tumours undergo autocatalytic peroxidation [4], a lipidic disorder which greatly altered membrane functions. Demyelination-induced plasticity was found by Coria [5] in the nodal and internodal axolemma in rat lead-induced neuropathy. Nuclear membrane indentations were reported by Roos [6] in Huntington’s disease.
Biochemical and morphological changes in nerve cell membranes were reported by Salvati [7] in experimental allergic encephalomyelitis. Macromolecular structure of axonal membrane during acute experimental allergic encephalomyelitis, and myelin deficient rat optic nerve was described by Black [8] and Waxman [910] found a drop-in cholesterol content in neuronal plasmalemma after cerebral ischemia. [11] encountered continuities between the outer nuclear membrane and the rough endoplasmic reticulum in hyppocampal neurons during seizure-induced protein synthesis. Membrane damage during situations of acute or subacute cerebral aggression have been studied using experimental models [4,1216]. Glial membrane damage of glial axonal junction after diffusing axonal injury was reported by Maxwell.
Traumatic brain injuries produce damage to nodal axolemma [17] and a widespread derangement to the neuronal cytoskeleton [18,19] delayed phospholipids degradation [20], and calpain- mediated spectr in breakdown leading to ischemic neuronal death. Babu [21] reported several protein defects in the plasma membrane of neurons and astrocytes in chronic ethanol treated rats. Dux [22] described disaggregation of ribosomes as an early sign of histotoxic pathology heralding delayed neuronal death. Theriault described gap junction remodelling in the rat spinal cord after acute compression injury. Maxwell [23] found alterations in the axolemma and myelin sheath in guinea pig optic nerve after stretch injury. Praproknit [24] described plasma membrane fragility in dystrophic neuritis of senile plaques in Alzheimer's disease. Dabrowska-Bouta [25,26] showed alteration of myelin membranes after chronic lead intoxication in rats.
Torp [27] described association of fibrillar beta amyloid with neuronal membrane surface in aged dog brains. Similar findings were also reported by Yamaguchi [28] in hereditary cerebral haemorrhage with amyloidosis-Dutch type, Alzheimer disease, and non-demented aged subjects. Haik demonstrated that the putative transmembrane domain of prion protein induces neurotoxicity and destabilize nerve cell membranes.
Glutamate ecotoxicity-induced damage of plasma membrane occurs in transient global cerebral ischemia [29] and in traumatic brain injuries [30] Peroxidative damage to cell membranes occurs following cerebral ischemia [31-35]. Transient global cerebral ischemia triggers suppression of protein synthesis, a process controlled by endoplasmic reticulum function [36-38]. Protein aggregation examined by electron microscopy and laser-scanning confocal microscopy has been reported after focal brain ischemia. It has been suggested that nitric oxide may contribute to ischemia- induced cell injury acting upon endoplasmic reticulum, calcium homeostasis, protein synthesis, and energy metabolism [39].
Damaged of nerve cell plasma membranes, cytoskeleton, rough and smooth endoplasmic reticulum membranes, lysosomal limiting membrane, and outer and inner mitochondrial membranes have been reported by [41-43] in moderate and severe edema associated to congenital hydrocephalus, brain trauma and brain tumors. Recent studies have suggested that cholesterol, an important component of membranes that controls their physical properties and functions, plays a critical role in neurodegenerative diseases. Enrichment of neuronal plasma membrane with cholesterol protects cortical neurons from apoptosis induced by soluble oligomers of the Abeta (1-40) peptide. Conversely, cholesterol depletion renders cells more vulnerable to the cytotoxic effects of the Abeta soluble oligomers [44]. The binding of Abeta to membrane lipids facilitates Abeta fibrillation, which in turn disturbs the structure and function of membranes, such as membrane fluidity or the formation of ion channels [45].
Recent reports also indicate that dysfunction of endoplasmic reticulum, which not only mediates proteins processing, but also regulates intracellular calcium homeostasis and cell death signal activation, occurs at an early stage after ischemia, and might be the initial step of apoptotic cascades in neurons [46]. Singleton and Povlishock [47] reported plasma membrane disruption in diffuse brain injury. Luo and Shi [48] have found that acrolein, a by-product of oxidative stress and lipid peroxidation, inflicts severe axolemmal disruption. The membrane damage is likely mediated by reactive oxygen species and lipid peroxidation, which are elevated after acrolein exposure. Shi has also reported axolemmal disruption in guinea pig spinal cord following compression. Kurnellas [49] have described plasma membrane calcium ATPase deficiency in multiple sclerosis and spinal cord injury, as a potential mechanism of neurodegeneration. Farkas [50] demonstrated mechanoporation or disruption of neuronal plasma membrane induced by diffuse traumatic brain injury.
Thompson [51] demonstrated opening of neuronal gap junction hemichannels following ischemia after stroke. Yi have found an increase in complexing I and complexing II, considered respectively markers of inhibitory and excitatory synapses, after traumatic brain injury. Nitric oxide and its toxic metabolite, peroxynitrite, can inhibit components of the mitochondrial respiratory chain leading to cellular energy deficiency and, eventually, to cell death. Kynurenine metabolic pathway, its alterations and their potential association with cellular energy impair certain neurodegenerative diseases. During energy production, most of the O2 consumed by the mitochondria is reduced fully to water, but 1-2% of the O2 is reduced incompletely to give the superoxide anion O2- If the function of one or more respiratory chain complexes is impaired for any reason, the enhanced production of free radicals further worsens the mitochondrial function by causing oxidative damage to macromolecules, and by opening the mitochondrial permeability transition pores thereby inducing apoptosis [52]. Prion protein (PrP), normally a cell surface protein, has been detected in the cytosol of a subset of neurons. The appearance of PrP in the cytosol could result from either retro-translocation of misfolded PrP from the endoplasmic reticulum (ER) or impaired import of PrP into the ER [53].
Endoplasmic reticulum and mitochondria are in a close communication, establishing a dynamic ER-Ca2+-mitochondria interconnection that can play a prominent role in the neuronal cell death induction under stressful circumstances of Parkinson's disease pathology (PD). Also, endoplasmic reticulum (ER) stress in conjunction with abnormal protein degradation can contribute to the PD pathophysiology, [54]. Endoplasmic reticulum stress, initiated by the accumulation of unfolded or misfolded proteins, activates the unfolded protein response, which adapts cells to the stress. If this adaptive response is insufficient, the unfolded protein response activates an apoptotic program to eliminate the affected cells- Endoplasmic reticulum stress in myelinating cells is important in the pathogenesis of various disorders of myelin, including Charcot-Marie-Tooth disease, Pelizaeus-Merzbacher disease and Vanishing White Matter Disease, as well as in the most common myelin disorder, multiple sclerosis [55].
Amyotrophic lateral sclerosis (ALS) is a devastating neurodegenerative disease characterized by the misfolding and aggregation of distinct proteins in affected tissues, however, the pathogenic cause of disease remains unknown. Recent evidence indicates that endoplasmic reticulum (ER) stress plays a central role in ALS pathogenesis. ER stress activates the unfolded protein response (UPR), a homeostatic response to misfolded proteins. The UPR is initially protective by up-regulation of specific ER stress- regulated genes and inhibition of general protein translation. However, long-term ER stress leads to cell death via apoptotic signaling, thus providing a link to neurodegeneration [56]. a-synuclein pathology and its effects on diverse protein partners and specific cellular pathways in the membrane and/or cytosolic districts, such as endoplasmic reticulum/Golgi, axonal and synaptic compartments of dopaminergic neurons, may cause the onset of neuronal cell dysfunction and degeneration which are among the key pathological features of the Parkinson's disease (PD) brain. Besides, a-synuclein aggregation may induce dysfunction and degeneration of synapses via these multiple mechanisms [57].
According to Hedskog [58], it is well-established that sub compartments of endoplasmic reticulum (ER) are in physical contact with the mitochondria. These lipid raft-like regions of ER are referred to as mitochondria-associated ER membranes (MAMs), and they play an important role in, for example, lipid synthesis, calcium homeostasis, and apoptotic signaling. Perturbation of MAM function has previously been suggested in Alzheimer's disease (AD). Our data suggest an important role of ER-mitochondria contacts and cross-talk in AD pathology.
α-Synuclein is located in mitochondrial-associated endoplasmic reticulum membranes. PD-related mutated a-synuclein results in its reduced association with mitochondria-associated membranes, coincident with a lower degree of apposition of endoplasmic reticulum with mitochondria and an increase in mitochondrial fragmentation. Aging is associated to cognitive decline and susceptibility to neuron death, two processes related recently to subcellular Ca2+ homeostasis. Memory storage relies on mushroom spines stability that depends on store-operated Ca2+ entry (SOCE). In addition, Ca2+ transfer from endoplasmic reticulum (ER) to mitochondria sustains energy production but mitochondrial Ca2+ overload promotes apoptosis. We have addressed whether SOCE and ER-mitochondria Ca2+ transfer is influenced by culture time in long-term cultures of rat hippocampal neurons, a model of neuronal aging [59].
However, many neuronal functions damaged in Parkinson's disease are regulated by signaling between the endoplasmic reticulum (ER) and mitochondria. This signaling involves close physical associations between the two organelles that are mediated by binding of the integral ER protein vesicle-associated membrane protein-associated protein B (VAPB) to the outer mitochondrial membrane protein, protein tyrosine phosphatase-interacting protein 51 (PTPIP51). VAPB and PTPIP51 thus act as a scaffold to tether the two organelles. a-synuclein induced loosening of ER-mitochondria contacts is accompanied by disruption to Ca2+ exchange between the two organelles and mitochondrial ATP production. Such disruptions are likely to be particularly damaging to neurons that are heavily dependent on correct Ca2+ signaling and ATP [60].
Potentially toxic cytoplasmic calcium concentrations can also occur due to release from internal stores, either through physical damage to mitochondria and the endoplasmic reticulum, or a malfunction of receptors and channels present in their membranes. Such increases of cytoplasmic calcium concentrations can trigger a range of downstream neurotoxic cascades, including the uncoupling mitochondrial electron transfer from ATP synthesis, and the activation and overstimulation of enzymes such as calpains and other proteases, protein kinases, nitric oxide synthase (NOS), calcineurin and endonucleases [61].
Autophagy and apoptosis are basic physiologic processes contributing to the maintenance of cellular homeostasis. Autophagy encompasses pathways that target long-lived cytosolic proteins and damaged organelles. It involves a sequential set of events including double membrane formation, elongation, vesicle maturation and finally delivery of the targeted materials to the lysosome. Deregulation of autophagy plays a pivotal role in the etiology and/ or progress of many neurodegenerative disorders [62]. Neurons employ specialized mechanisms to modulate local gene expression in dendrites, via the dynamic regulation of micro RNA biogenesis factors at intracellular membranes of the endoplasmic reticulum, which in turn is crucial for neuronal dendrite complexity and therefore neuronal circuit formation and function.
In the present review we describe the ultrastructural alteration of nerve cell and glial plasma membrane, cytomembranes, synaptic membranes, and interastrocytary gap junctions in cortical biopsies of patients with vascular anomalies, congenital hydrocephalus, complicated brain traumatic injuries. The brain parenchyma of these patients exhibits moderate and severe oedema and sustained anoxic-ischemic conditions [63-66].
In vascular anomaly, the perifocal cerebral cortex exhibits a microstructure like that observed in normal animal brain cortex. The plasma membrane of non-pyramidal neurons shows in moderate oedema atly normal and continuous structure. The plasma membrane form endocytic and clathrin coated vesicles internalizing toward the cytoplasm. The limiting plasma membrane of rough endoplasmic reticulum cisterns exhibit fragmented areas and detachment of membrane associated ribosomes. The nuclear envelope appears irregularly dilated (Figures 1 & 2). Disaggregation of ribosomes was earlier demonstrated by Dux in primary cortical and hippocampal neuronal cultures after brief histotoxic hypoxia.
Figure 1: Anomaly of anterior cerebral artery. Right pa-rietal cortex. Non-pyramidal nerve cell showing an irreg-ularly dilated nuclear envelope (short arrows), enlarged endoplasmic reticulum cisterns (ER), detachment of mem-brane associated ribosomes (long arrows), and a contin-uous limiting plasma membrane (arrowheads). Note the prominent nucleus (N), the nucleolus (NL), the swollen mitochondria (M), and the non-dilated extracellular space in the neighboring neuropil that features moderate brain edema.
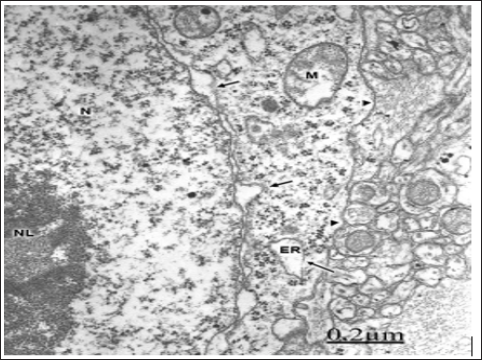
Figure 2: Anomaly of anterior cerebral artery. Right pari-etal cortex. Non-pyramidal neuron (NP) showing shallow invaginations of plasma membrane (long arrow) and for-mation of clathrin coated vesicle (arrowhead). Endocytic vesicles are also observed in sublemmal localization short arrows). Note the disrupted and degranulated rough en-doplasmic reticulum cisterns (ER).
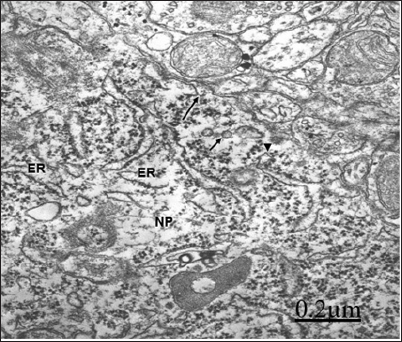
In relationship with congenital hydrocephalus in neonate patients, the presence of interstitial oedema fluid in the enlarged extracellular space of an immature neuropil induces fragmentation of plasma membrane, lacunar enlargement of rough endoplasmic reticulum, detachment of membrane associated ribosomes, irregularly dilated nuclear envelope, and nuclear pore disassembly [67] (Figure 3). Congenital hydrocephalus. Right frontal cortex. Non-pyramidal neuron exhibiting fragmented plasma membrane (long arrow), distended endoplasmic reticulum cisterns (ER), detachment of membrane associated ribosomes (short arrows), and disassembly of some nuclear pores (arrowheads). The mitochondria (M) appear swollen. Note the enlarged extracellular space in the neighbouring neuropil (asterisks). X 60.000.
Figure 3: Congenital hydrocephalus. Right frontal cortex. Non-pyramidal neuron exhibiting fragmented plasma membrane (long arrow), distended endoplasmic reticu-lum cisterns (ER), detachment of membrane associated ri-bosomes (short arrows), and disassembly of some nuclear pores (arrowheads). The mitochondria (M) appear swol-len. Note the enlarged extracellular space in the neigh-bouring neuropile (asterisks). X 60.000.
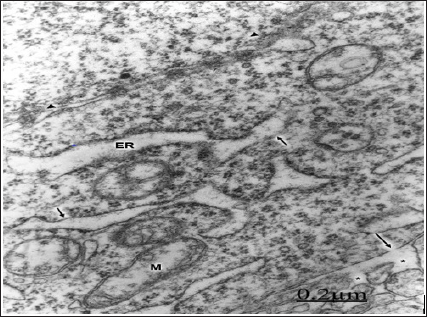
Figure 4: Congenital hydrocephalus. Right parietal cortex. The plasma membrane exhibits a deep invagination (long arrow) and formation of endocytic vesicles (short arrows) directed toward the swollen Golgi complex (GC). A lyso-some (L), moderately swollen and dark mitochondria (M), numerous Golgi vesicles and clathrin-coated vesicles (ar-rowheads) also are seen. X 60.000.
The plasma membrane shows deep invaginations and formation of numerous endocytic vesicles directed toward the endoplasmic reticulum and the Golgi apparatus [68]. The lysosomes show fragmented limiting membrane (Figure 4). In congenital hydrocephalus in neonate patients we are dealing with immature plasma membranes, characterized by changes in the integral membrane proteins, cholesterol domains, and in certain carbohydrates residues and anionic sites [69]. These different molecular compositions of immature plasma membranes explain its high sensitivity to injury factors. In hypertensive congenital hydrocephalus, the pressure exerted by the non-circulating cerebrospinal fluid induces plasma membrane fragmentation, and enlargement of intracellular nerve cell compartments. In addition, the presence of oedematous mitochondria evinces a disturbed energy metabolism.
Endocytosis is a fundamental mechanism by which neurons control intercellular signal, nutrient uptake, and synaptic transmission [70]. Clathrin-coated vesicles of different sizes have been isolated from rat brain, and related with the content of Na+, K (+)- ATPase, as a mechanism of Cl- uptake [71], as pathways for endocytosis of neural cell adhesion molecules (NCAM) [72], and for receptor-mediated vesicular transport [73], such as internalization of metabotropic glutamate receptors [74].
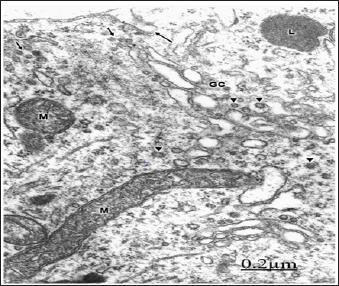
In patients with brain trauma and moderate oedema, some non-pyramidal neurons show a well preserved and continuous plasma membrane, swollen mitochondria [75,76] (Figure 5). In severe oedema of traumatic brain injuries complicated with subdural hematoma, the non-pyramidal neurons, astrocytes and oligodendrocytes show plasma membrane fragmentation, swollen mitochondria, enlargement of rough and smooth endoplasmic reticulum cisterns, and irregular dilation of nuclear envelope [77]. The rough endoplasmic reticulum displays extensive areas with detachment of membrane-bound associated ribosomes, and a marked reduction in the number of polysomes [78] (Figure 6).
Figure 5: Brain trauma. Left frontal hematoma. Non-py-ramidal neuron in an area of moderate perifocal oede-ma bearing a continuous plasma membrane (arrows), a non-dilated endoplasmic reticulum cistern (ER) and nucle-ar envelope (arrowhead), and swollen mitochondria (M). Note the well-preserved nucleolar substructures (NL).X 30.000.
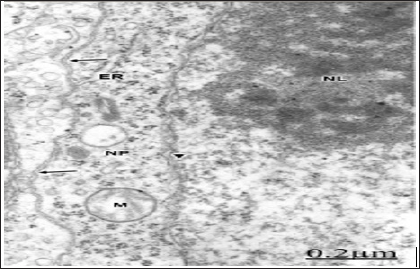
Figure 6: Brain trauma. Subdural hematoma. Left parietal cortex. Severe oedema. Non-pyramidal neuron (NP) dis-playing disrupted plasma membrane (long arrows), irreg-ularly dilated nuclear envelope (short arrows), and swol-len mitochondria (M). In the neighbouring neuropil, the asterisks label the enlarged extracellular space containing proteinaceous oedema fluid, which separates degenerat-ed synaptic endings (SE), and swollen astrocytic processes (A). Note the swollen nucleoplasm (N). X. 30.000.
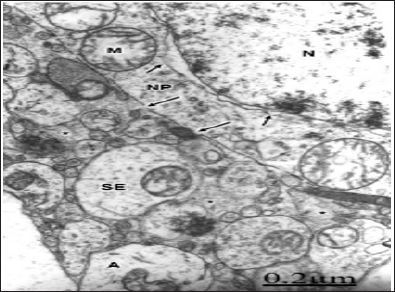
Singleton and Povlishock, Farkas demonstrated disruption of neuronal somatic plasmalemma in diffuse traumatic brain injury. Areas of focal necrosis of plasma membrane, cytomembranes, mainly outer nuclear membrane, smooth membranes of Golgi endoplasmic sacs, and lysosomes are observed [79] (Figure 7). In severe traumatic brain injuries with enlarged extracellular spaces containing proteinaceous oedema fluid, the plasma membrane of neurons appears. The degenerated myelinated axons show invaginations of the axolemmal membrane and formation of endocytic vesicles x. The myelin membranes appear separated forming large intra-period vacuoles. Traumatic damage of axolemmal membrane was reported by Castejon, Genarelli, Erb, Povlishock, and Maxswell.
Figure 7: Brain trauma. Subdural hematoma. Right pari-etal cortex. Non-pyramidal neuron showing focal necro-sis of nuclear envelope (long arrow) and of smooth Golgi complex (GC) membranes (short arrows). The lysosomes (L) show a discontinuous globular limiting membrane (ar-rowheads). X 75.000.
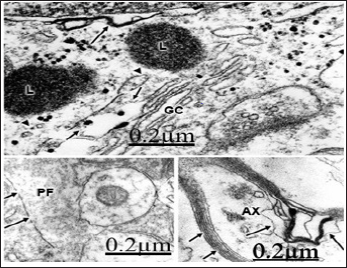
The swollen and clear astrocytes display marked degenerative changes induced by brain edema in comparison with those exhibited by neuronal cells. Areas of focal necrosis and fragmented limiting plasma membrane, over-distended rough endoplasmic reticulum cisterns with extended degranulated areas, and vacuoles of smooth endoplasmic reticulum with necrotic limiting membrane are found [80-82] (Figure 8). Oligodendroglial cells show also marked edematous changes featured by lacunar enlargement of rough endoplasmic reticulum and nuclear envelope, detachment of membrane bound ribosomes, swollen mitochondria associated to rough endoplasmic reticulum, and discontinuous plasma membrane [83,84]. (Figure 9). The endoplasmic reticulum is a subcellular compartment playing a fundamental role in the folding and processing of newly synthesized membranes and secretory processes, reactions which are strictly calcium/dependent. Severe disruption of neuronal Ca2+ homeostasis leading to a lethal Ca2+ overload as occurs in brain ischemia, can initiate a cascade of destructive processes which lead to the death of neurons during cerebral ischemia [85-86]. As stated above endoplasmic reticulum function is disturbed in many acute and chronic diseases of the brain, such as Parkinson's and Alzheimer's diseases [87]. Therefore, endoplasmic reticulum dysfunction is basically important to elucidate the pathogenetic mechanisms of neurodegeneration.
Figure 8: Brain trauma. Swollen and clear astrocyte cell (A) depicting fragmented plasma membrane (long ar-rows) and areas of focal necrosis (short arrow), degran-ulated and dilated rough endoplasmic reticulum cisterns (ER), glycogen granules (arrowheads), and a vacuole (V) limited by necrotic membrane. Note the degenerated and dense mitochondrion (M) associated with rough endo-plasmic reticulum X. 60.000.
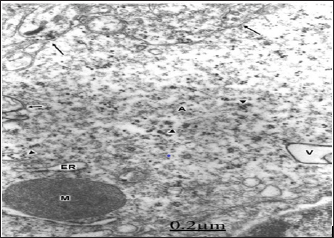
Figure 9: Brain trauma. Hydropic oligodendrocyte (OL) showing over-distended perinuclear (PN) and endoplasmic reticulum (ER) cisterns, disassembly of nuclear envelope (NE), and nuclear pores (long arrow). Note the swollen mitochondria (M) associated to rough endoplasmic reticulum. The neighboring neuropile exhibits degenerated axodendritic synapse (short arrow) and myelinated axon (AX). X 60.000.

Peroxidative stress has been implicated in mechanism leading to neuronal cell injury [88-91]. One source of free radicals in ischemic cells is arachidonic acid released by membrane phospholipids under the action of Ca2+ activated phospholipase A2 [92]. Several oxygen radical species besides superoxide radicals are produced following hypoxia. Superoxide radicals have been shown to change phospholipid and protein structure. Hydroxyl radicals are the most reactive and are known to initiate lipid peroxidation and protein oxidation [93,94].
In congenital hydrocephalus and in severe oedematous regions of complicated traumatic brain injuries, synaptic plasticity, synaptic degeneration and occur. Synaptic disassembly featured by wide separation of pre- and postsynaptic membranes and loss of peri synaptic astrocytic glial escheatment also is observed [95,96] (Figure 10).
Figure 10: Brain trauma. Subdural hematoma. Left parietal cortex. Severely edematous neuropil showing synaptic disassembly and disappearance of pre- and postsynap- tic densities (long arrow) of an axospinodendritic contact. The short arrow labels another spine synapse exhibiting partial vestiges of the postsynaptic density, and irregular profile of presynaptic membrane (arrowhead). Note the expansion of extracellular space (asterisks), and the detachment of peri synaptic astrocytic cytoplasm (A). X. 60.000.
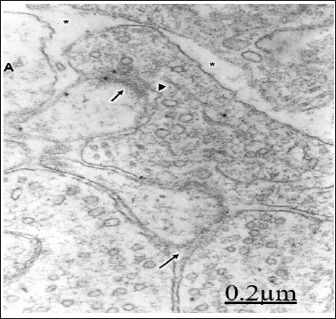
Numerous flat, curved and invaginated axodendritic and axospinous asymmetric synapses, and a less proportion of axodendritic and axosomatic symmetric synapses are found. Activated or sensitized synapses showed numerous frontline spheroid synaptic vesicles, prominent dense presynaptic dense projections and increased in length of synaptic membrane complex. Perforated synapses, multiple synapses and serial synapses are also found evincing synaptic splitting and formation of new synaptic connections (Figure 11).
The hydrocephalic or interstitial clear edema, the hematogenous edema fluid in traumatic brain injuries, and the proteinaceous edema fluid in brain tumors accumulated in the dilated extracellular space of cerebral cortex neuropil induce swelling and shrinkage of pre- and postsynaptic structures, increased amount of presynap- tic axoplasmic granular substance, and clumping, enlargement and depletion of synaptic vesicles, and filamentous hypertrophy of pre- synaptic endings (Figure 12). Disappearance of synaptic densities is evident in some cases. In severe brain edema, swollen and shrunken presynaptic endings with discontinuous limiting plasma appear separated from the postsynaptic structures and detached from the glial ensheathment expressing synaptic disassembly. Phagocytosis of isolated presynaptic endings, and of the entire synaptic contacts by astrocytes, microglial cells, and by non-nervous invading cells, such as monocytes and macrophages, are frequently observed [97].
Figure 11: Brain trauma. Epidural hematoma. Right temporal cortex. Moderate edematous region. An asymmetric ax spinous synapse (long arrow) appears showing a curved arrangement of the synaptic membrane complex. Another activated and flat symmetrical synapse (short arrows) is observed at the upper part of the figure. Note the peri synaptic glycogen-rich-astrocytic ensheathment (A) phagocyting a degenerated presynaptic ending (arrowheads).
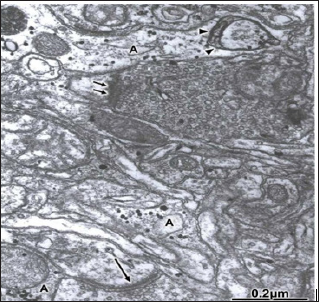
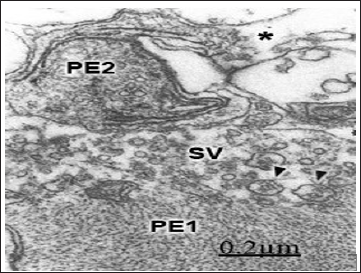
Figure 12: Brain trauma. Right parietal cortex. Subdural hematoma. Degenerated presynaptic ending (PE1) exhib-iting filamentous hypertrophy and reduced number of dis-placed synaptic vesicles (SV). Large vesicles (arrowheads) containing degenerated and small dense-cored nuclei are also observed. Another severely degenerated presynap-tic ending (PE2) shows necrotic osmiophilic membranes, dense axoplasmic substance and focal necrotic areas. The asterisk labels the enlarged extracellular space containing non-proteinaceous edema fluid.
Disruption and disassembly of interastrocytary gap junctions have been observed by us in human edematous cerebral cortex of patients with traumatic brain injuries and brain tumors [98] (Figure 13).
Figure 13: Anomaly of anterior cerebral artery. Left parietal cortex. Astrocytic end-feet (A) showing extensive and undulated interastrocytary gap junction and membrane domains with fusion of both confronted membranes (long arrows). Note the vacuolization (V) and swelling of perivascular astrocytic end feet, and the dense capillary basement membrane (BM). The pericyte cytoplasm (P), the endothelial cell (EC), and the capillary lumen (L) are also distinguished. X 36.000.
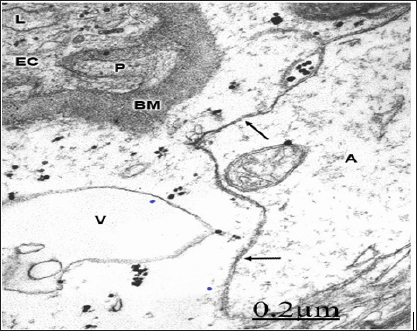
In severe traumatic brain injuries, gap junction disruption and disassembly are observed. Gap junction disassembly is characterized by wide separation of astrocytic end- feet confronted membranes [98] (Figure 14)
Figure 14: Brain trauma. Right epidural hematoma. Right temporal cortex. Capillary (C) showing the increased activity of endothelial cell luminal membrane, which exhibits the formation of pinocytotic vesicle (short arrow), deep invagination (long arrow), and formation of endothelial vacuoles (V). Note the swollen basement membrane (BM), and the degenerated myelinated axons (AX) in the neighboring neuropil. The arrow indicates the disrupted axo- lemmal membrane, and the circle the granular degeneration of neurofilaments. X 36.000.
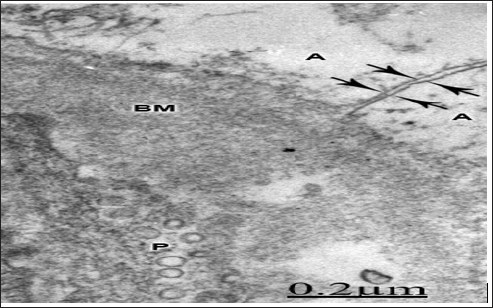
According to Hossain [99] rat astrocytes respond to ischemic insult by reorganizing their gap junction and increasing the junctional protein connexin43. Ochalski [100] reported extensive gap junctions, disruption and gap junction disassembly in kainic lesion sites of rat brain. Theriault described gap junction remodeling in rat spinal cord after acute compression injury. Li [101] found astrocytic gap junction internalization in rat brain after cerebral focal ischemia. Figure 15 Soroceanu [102] reported that high- grade brain tumors show reduced intercellular communication, and a decrease in connexin-43 protein levels. Aronica [103] found high expression of connexin proteins in low grade tumors, and in the peritumoral reactive astrocytes. Nakase [104-110] reported a protective role of astrocytic gap junctions in ischemic stroke reducing apoptosis and inflammation following ischemic insult. According to Perez- Velazquez [106,111-115], the actions of gap junctional coupling during injuries may be causally related to oxidative stress.
Figure 15: Brain trauma. Subdural parieto-temporal hematoma. Left parietal cortex. Disassembly of interas- trocytary gap junctions featured by wide separation of confronted astrocytic end feet membrane (A). Note the swollen and vacuolated basement membrane (BM), and the increased vesicular transport in the pericytal cytoplasm (P). X 90.000.
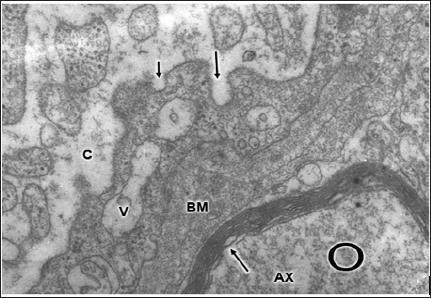
In severe and complicated traumatic brain injuries, the endothelial cell luminal membrane of brain capillaries undergoes profound activity changes that characterize the increased cerebrovascular permeability. Such changes are increased formation of micro- and macropinocytotic vesicles and clathrinid- coated vesicles, deep invaginations and formation of incomplete trans endothelia channels, and emission of pseudopods to form endothelial vacuoles [107,115-118].
In moderate brain oedema a continuous plasma membrane is observed in some neurons, but the cytoplasmic membranes, such as smooth and rough endoplasmic reticulum membranes appear damaged. In severe oedema, fragmentation of plasma membrane, enlargement and focal necrosis of rough endoplasmic cisterns and nuclear envelope, detachment of membrane-bound ribosomes, and reduction of polysome are found [118]. Shallow and deep invaginations of plasma membrane, and the formation of endocytic and clathrin-coated vesicles are seen. In astrocyte cells, areas of focal necrosis and fragmented limiting plasma membrane, overdistended rough endoplasmic reticulum cisterns with extense degranulated membrane domains, and vacuoles of smooth endoplasmic reticulum with necrotic limiting membrane are observed. Oligodendroglia cells show also notably edematous changes featured by lacunar enlargement of rough endoplasmic reticulum and nuclear envelope, detachment of membrane bound ribosomes, and discontinuous plasma membrane [119-121]. Plastic changes and damage of synaptic membranes are found. Synaptic vesicle exocytosis at the synaptic active zone, and endocytosis at the non-specialized regions of presynaptic ending limiting membrane are frequently observed at activated synapses.
In severe brain edema, synaptic disassembly occurs featured by wide separation of pre- and postsynaptic membranes and loss of peri synaptic astrocytic glial escheatment. Disruption, fusion and disassembly of interastrocytary gap junctions have also been observed. The endothelial cell luminal membrane of brain capillaries undergoes profound activity changes that characterize increased cerebrovascular permeability, such as increased formation of micro- and macropinocytotic vesicles, clathrin coated vesicles, and emission of pseudopods to form endothelial vacuoles, The alterations of nerve cell plasma membranes and cytomembranes are related with the anoxic-ischemic conditions of brain parenchyma [121-124]. The role of free radical and lipid peroxidation, disturbed energy metabolism, altered metabolic cascades, glutamate excitotoxicity, haemoglobin toxicity, protein aggregation, and presence of extracellular oedema fluid are discussed in relation with the derangement of nerve cells membranes.
This paper was carried out through the logistic support of Biological Research Institute. Faculty of Medicine. Zulia University and Castejon Foundation.


