Impact Factor : 0.548
- NLM ID: 101723284
- OCoLC: 999826537
- LCCN: 2017202541
Pawar RR1, Suryavanshi VJ1, Patil MM1, Patil SS2 and Mulik GN*1
Received: October 10, 2018; Published: October 29, 2018
*Corresponding author: Mulik GN, P.G. Department of Chemistry, Balwant College, Vita-415 311, India
DOI: 10.26717/BJSTR.2018.10.001965
The extraction of Zinc(II) from acid media by N-n-heptylaniline in xylene has been studied for metal distribution measurements. Various physicochemical parameters like acid concentration, reagent concentration, equilibrium time, effect of diluents, aqueous to organic phase ratio, stripping agents and loading capacity were investigated for the quantitative extraction. (The extraction was carried out from 3 M hydrochloric acid and 0.5 M N-n-heptylaniline concentration). Zinc(II) was selectively extracted and separated from many metal ions and synthetic mixtures. The nature of the extracted species was determined. Zinc(II) was analyzed from pharmaceutical samples and nycil talk powder.
The uses of Zinc(II) are in the production of die-casting alloys, in galvanizing industry, pharmaceutical samples, in the manufacture of brass products, rolled Zinc(II) products of various types, light metal alloys, in desilvering lead and in wet-batteries. It is starting material for the production of Zinc(II) oxide. Zinc(II) is an essential trace element in plant and animal life. The Zinc(II) content in humans is 2 - 4g [1]. Zinc(II) is also essential constituent of several enzymes necessary for metabolism. It is necessary to develop the simple, rapid and selective method of separation for Zinc(II) from different elements. For extraction and separation of Zinc(II) solvent extraction is useful method. Solvent extraction of zinc(II), indium(III), thallium(III) and bismuth(III) with n-octylaniline from hydrochloric acid media and their separation carried out [2].
The distribution equilibrium of Zinc(II) between synergistic mixture of N-n-octylaniline and trioctylamine in xylene and thiocyanate media has been investigated [3]. The solvent extraction of Zinc(II) from thiocyanate and sulphuric acid media using N-n- hexylaniline in xylene is described [4]. Solvent extraction of zinc from strong hydrochloric acid solution with alamine 336 has been carried out [5]. Liquid-liquid extraction of Zinc(II) by 3-methyl- quinoxaline-2-thione from nitrate medium investigated [6]. Separation of iron and Zinc(II) from manganese nodule leach liquor using TBP as extractant is studied [7]. Solvent extraction utilized for the selective separation of Zinc(II) from other elements in hydro metallurgical processing of resources [8]. Zinc(II) chloride and hydrochloric acid extraction from solutions of high Zinc(II) concentration by tri-n-butyl phosphate diluted in ShellSol2046 (an aliphatic solvent) has been studied with a combination of experiments and mathematical Modeling [9]. From chloride solutions solvent extraction of Zinc(II) carried out [10].
The mean centering of ratio kinetic profiles method was used for the simultaneous determination of binary mixtures of Ni(II) and Zn(II) in water samples, without prior separation steps [11]. Synergistic extraction of Zinc(II) by mixtures of primary amine N1923 and cyanex 272 investigated [12]. The solvent extraction of zinc(II), cadmium(II) and chromium(III) from phosphoric acid solutions by tri-n-butyl phosphate in kerosene as diluent was investigated [13].The extraction and separation of zinc(II), manganese, cobalt and nickel from nickel laterite bacteria leach liquor were carried out using sodium salts of TOPS-99 and Cyanex 272 in kerosene [14]. The selective removal of Zinc(II) over iron (II) by liquid-liquid extraction from spent hydrochloric acid pickling effluents produced by the Zinc(II) hot-dip galvanizing industry was studied at room temperature [15]. Extraction and separation of cobalt and Zinc(II) was studied from a sulphate solution using NaD2EHPA, NaPC88A and NaCyanex 271 of 0.04M concentration [16]. The liquid-liquid extraction of Zinc(II) using D2EHPA as extractant has been investigated in order to recover zinc(II) sulphate from an industrial effluent produced by Votorantim Co. which contains several metallic species such as cadmium, cobalt, iron, lead, calcium, magnesium, manganese and nickel [17].
The extraction of Zinc(II) and cadmium(II) with mixtures of neutral organophosphorus extractants and amine extractants has been investigated [18]. Synergistic solvent extraction and transport of Zn(II) and Cu(II) across polymer inclusion membranes with a mixture of TOPO and aliquat 336 carried out [19]. Extraction of Zn(II) from aqueous hydrochloric acid solutions into alamine-336 m-xylene systems was studied [20]. 3-hydroxybenzylaminobenzoic acid synthesized as a reagent for the determination of Zinc(II) in various water samples [21]. Recovery of both sulphuric acid and Zinc(II) bleed stream generated during the electrowinning of Zinc(II) in the Zinc(II) refineries using tris-(2-ethylhexyl) amine dissolved in kerosene has been investigated in detail [22]. The extraction of Zn(II), Fe(II), Fe(III) and Cd(II) with TMbutylphosphate (TBP) and commercial trioctyl phosphine oxide in kerosene from chloride medium has been studied [23]. Solvent extraction behavior of zinc(II), cadmium(II), mercury(II) and bismuth(III) with n-octylaniline in different organic solvents from various aqueous acid solutions has been investigated [24].
An accurate, inexpensive and less laborious liquid-liquid extractive spectrophotometric procedure for the determination of Zinc(II) in aqueous media has been developed [25]. Previously we have reported number of solvent extraction methods for the quantitative extraction of platinum group metals with amines [2639]. In present research paper a systematic study of extraction of Zinc(II) with N-n-heptylaniline in xylene has been carried out. The extracted complex species in the organic phase Zinc(II) was back extracted with 0.5M ammonia solution (2X10mL) and was determined complexometrically. Various parameters such as acid-reagent concentration, equilibrium period, effect of various diluents, enrichment study, loading capacity and diverse ions were also studied. The study was also extended for analysis of synthetic mixtures and pharmaceutical sample.
Mettler toledo model-ML204 electronic balance with accuracy 0.0001g was used for weighing. For pH measurement Elico digital pH meter model LI-120 with combined glass electrode was used. All glass wares were cleaned by acidified solution of potassium dichromate and finally rinsed with water
Standard Zinc(II) Sulphate Solution (1 mg/mL)
The stock solution of Zinc(II) was prepared by dissolving 5.49g of Zinc(II) sulphate (ZnSO4.7H2O) in 250mL of distilled water containing 0.5mL of concentrated sulphuric acid. The solution was standardized and diluted as required for working purpose.
Ethylene Diaminetetraacetic Acid Solution (0.001m)
A solution of ethylene diaminetetraacetic acid (0.01 M) was prepared by dissolving 3.722g EDTA in distilled water and diluting to 1000mL and standardized complexometrically. Working solution of EDTA (0.001M) was prepared by proper dilution.
Buffer Solution (pH 10)
A pH 10 buffer solution was prepared by adding 142mL concentrated ammonia solution (sp.gr. 0.88-0.90) to 17.5g ammonium chloride and dilute it to 250mL with distilled water
N-N-Heptylaniline Solution (0.5M)
N-n-heptylaniline was synthesized by the method of Z. G. Gardlund [40] and its solution (%, v/v) was prepared by using xylene as the diluent.
To an aliquot of solution containing 1mg of zinc(II), add sufficient quantity of hydrochloric acid to make the concentration of 3M in a total volume of 10mL. Transfer the solution into a 125mL separating funnel and shake the solution for 1 minute with 10mL of 0.5 M N-n-heptylaniline. The swirl the solution and allow to separate the two phases. After phase separation, strip the Zinc(II) from organic phase with two 20mL portions of 0.5 M ammonia solution. Collect and combine the aqueous extract and estimate Zinc(II) complexometrically [41].
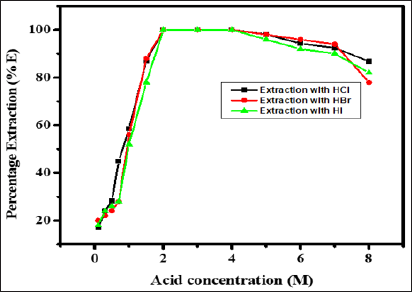
The extraction of 1mg of Zinc(II) was studied from hydrochloric, hydrobromic, hydroiodic, sulphuric, nitric and perchloric acid media in the range of 0.1 to 8.0 M with 0.5 M N-n-heptylaniline in xylene keeping the aqueous to organic volume ratio 1:1. The extraction of Zinc(II) increases with increase in acid concentration and becomes quantitative in 2.0 to 4.0M hydrochloric, hydrobromic and hydroiodic acid. While Zinc(II) was not extracted with remaining acids. Upon further increase in hydrochloric, hydrobromic and hydroiodic acid concentration the extraction of Zinc(II) decreases (Figure 1), 3.0 M concentration of hydrochloric acid was used throughout the work, as it has a wide range of applications as compared with hydrobromic and hydroiodic acid.
Figure 1: Extraction behaviour of Zn(II) as a function of hydrochloric, hydrobromic and hydroiodic acid concentration, Conditions: Zn(II) = 1mg, N-n-heptylaniline = 0.5M in xylene, aq.: org. ratio = 1:1, shaking time = 1min, strippant = ammonia buffer (2 *10mL).
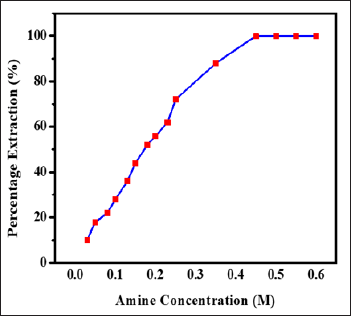
The effect of extractant concentration was scanned for the range of 0.03 M to 0.6M of N-n-heptylaniline on the 1mg Zinc(II) from 3M hydrochloric acid. Increase in N-n-heptylaniline concentration was found to be increase the extraction of zinc(II). The excess of reagent concentration had no adverse effect on magnitude of extraction (Figure 2). It was found that, 10mL of 0.5 M N-n-heptylaniline was sufficient for the quantitative extraction of 1.0mg of Zinc(II) from 3.0 M hydrochloric acid. Therefore, in the recommended procedure 0.5 M N-n-heptylaniline in xylene has to be used to ensure complete extraction of zinc(II).
Figure 2: Influence of amine concentration on extraction of Zinc(II), Conditions: Zn(II) = lmg, HCl = 3.0M, aq.: org. ratio = 1:1, shaking time = 1min, strippant = ammonia buffer (2 *10mL).
The effect of phase contact time on the distribution ratio for the extraction of Zinc(II) from 3 M hydrochloric acid using 0.5M N-n-heptylaniline in xylene has been studied (Table 1). Variation of shaking time from 10 s to 20min showed that a minimum of 30s shaking is needed for the complete extraction of zinc(II). The prolonged shaking had no adverse effect on the extraction, hence 1min shaking is recommended in the general procedure to ensure the quantitative extraction of zinc(II).
Note: *Recommended for general extraction procedure
Conditions: Zn(II) = 1mg, N-n-heptylaniline = 0.5M in xylene, HCl = 3.0M, aq.: organic ratio = 1:1, strippant = ammonia buffer (2 x10mL).
According to Sole appropriate hydrocarbon mixture used as diluents, having flash and boiling point above 600 C and density of 0.8g / cm-3 to aid phase separation. The extra ctability and selectivity for the extraction of metal ions by organic extractant are greatly affected by the nature of solvent. A number of different diluents were tested for the extraction of zinc(II). To discern the effect of nature of various aromatic and aliphatic diluents on the extraction of Zinc(II) the organic diluents namely xylene, toluene, benzene, nitrobenzene, carbon tetrachloride and chloroform (Table 2). Out of these diluents, Zinc(II) was quantitatively extracted in the xylene, toluene, benzene and nitrobenzene while the remaining solvents give incomplete extraction of zinc(II). Among this xylene was preferred as a diluent for further studies since it provide better phase separation, relatively lower aqueous solubility, ready availability and relative low cost.
Note: *Recommended for general extraction procedure
Conditions: Zn(II) = 1mg, N-n-heptylaniline = 0.5M in xylene, HCl = 3.0M, Shaking time = 1min, strippant = ammonia buffer (2 x10mL).
To find out whether large aqueous: organic volume ratio could be tolerated for the successfully extraction of Zinc(II) by N-n- heptylaniline under optimum condition. Zinc(II) was extracted from aqueous (10 to 150mL) of 3.0M HCl with 10mL of 0.5M N-n- heptylaniline in xylene. Zinc(II) was stripped and determined as described in the general procedure. It was found that extraction of Zinc(II) was quantitative when aqueous to organic volume ratio was only 1:1 and it decreased beyond it this may be attribute to the less stability of ion pair formed under conditions (Table 3).
Note: *Recommended for general extraction procedure
Conditions: Zn(II) = 1mg, N-n-heptylaniline = 0.5M in xylene, HCl = 3.0M, Shaking time = 1min, strippant = ammonia buffer (2 x10mL).
The loading capacity of N-n-heptylaniline in xylene was studied by equilibrating Zinc(II) in 3M hydrochloric acid at fixed aqueous to organic ratio (1:1) for 1min. The two phases were separated, and the same organic phase was again used for the extraction of fresh zinc(II). The extraction of Zinc(II) by same N-n-heptylaniline was repeated till no further extraction of Zinc(II) was observed in the organic phase. In the saturated organic phase of N-n-heptylaniline the amount of Zinc(II) was found to be 5mg.
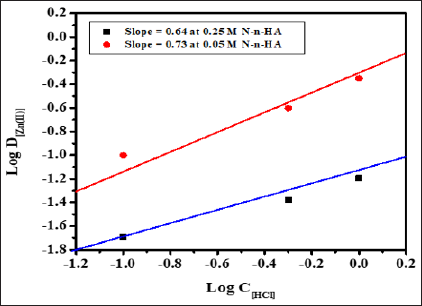
Stoichiometry of the extracted species was determined by plotting Log D[Zn(II)] versus Log C[HCl] at 0.05 and 0.25M N-n-heptylaniline concentration having slopes 0.73 and 0.64 respectively (Figure 3). Similarly, the graph of Log D[Zn(II)] versus Log C[N-n-heptylaniline] at 2.0M and 6.0M HCl concentration having the slopes of 2.03 and 1.69 respectively (Figure 4). The stoichiometry of the extracted species is calculated to be 1:1:2 (metal: acid: extractant).
Figure 3: Log-Log plot of distribution ratio versus N-n- heptylaniline concentration at 2.0M and 6.0M hydrochloric acid, Conditions: Zn(II) = 1mg, N-n-heptylaniline = 0.5M in xylene, aq.: org ratio : 1:1, shaking time = 1min, strippant = ammonia buffer (2 x10mL).
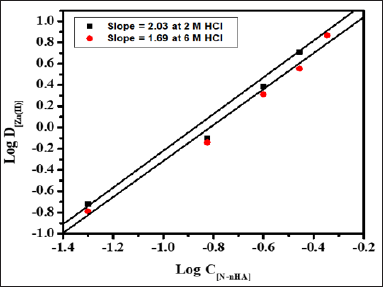
Figure 4: Log-Log plot of distribution ratio Log D[Zn(II)] versus Log C[HCl] at 0.05 and 0.25M N-n-heptylaniline, Conditions: Zn(II) = 1mg, aq.: org. ratio = 1:1, shaking time = 1min, strippant = ammonia buffer (2 x10mL).
Hence the probable extracted species is: [(RR' NH2+)2 ZnCl42-] and the extraction mechanism appears to be,
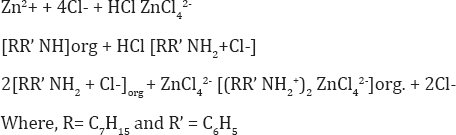
A number of ions were tested for the interference in the extraction of Zinc(II) and complexometric determination. Varying amounts of foreign ions were added to 1mg of Zinc(II) (in 10mL of solution) and recommended extraction procedure was followed. There was no interference from (as shown by less than 1.0 % deviation of Zinc(II) recovery) from 5mg each of manganese(II), silver(I), gold(III) and thallium(I), aluminium(III); 2.5mg each of calcium(II), beryllium(II), magnesium(II), chromium(VI); 100mg each of fluoride, acetate, thiocyanate; 60mg each of thiourea, ascorbate, 40mg each of thiosulphate, 10mg each of phosphate and malonate. Copper(II), nickel(II), cobalt(II), mercury(II), bismuth(III) and lead co-extract. Of which bismuth and lead were masked with thiourea. The results are tabulated in Table 4.
Note: a = Masked with thiourea.
In order to establish the validity of the proposed extraction procedure, the method has been applied for the separation of Zinc(II) from binary mixture. The binary separation of each metal ion was achieved by selective extraction of Zinc(II) leaving behind other metals in raffinate. The method was applied for the separation of Al(III), In(III), Th(IV), Tl(I), Bi(III) and lead(II) Table 5. The extracted Zinc(II) from loaded N-n-heptylaniline was stripped by washing the organic phase using 0.5M ammonia.
Note:a = Masked with thiourea.
Synthetic mixtures containing chromium(VI), gold(III), silver(I) in addition to Zinc(II) are analyzed by the proposed method. Zinc(II) from organic phase is back stripped with 0.5M ammonia and is determined complexometrically. The results given in Table 6 show that the separation of Zinc(II) is possible from synthetic multicomponent mixtures.
To 0.5g of sample placed in a flask add 2mL of conc. nitric acid and 10mL 3% KMnO4. The mixture is heated on water-bath for 30min at 70-800C. Excess permanganate was removed by adding solid oxalic acid. The solution was diluted to 100mL and extract Zinc(II) as described in extraction procedure. The results of analysis are reported in Table 7.
Note: *Average of six determinations.
A tablet was dissolved in concentrated perchloric acid and solution was evaporated to near dryness. The residue was taken up in the minimum amount of perchloric acid and solution was evaporated to dryness again. The residue was then leached with water and diluted to 100mL with water. An aliquot was taken for the extraction and estimation of Zinc(II) by the recommended procedure. The mean of six results is reported in Table 8.
Note: *Average of six determinations
Conditions: Zn(II) = 1mg, N-n-heptylaniline = 0.5M, HCl = 3.0M, aq.: org. ratio = 1:1, Shaking time = 1min, strippant = ammonia buffer (2 x10mL).
The present investigation highlights that N-n-heptylaniline is very effective ion-pair forming extractant. The method can be applied for simultaneous determination of Zinc(II) in synthetic mixtures, commercial sample and pharmaceutical sample. The stoichiometry of the proposed method was found to be 1:1:2 (metal: extractant: acid).

