Impact Factor : 0.548
- NLM ID: 101723284
- OCoLC: 999826537
- LCCN: 2017202541
Ibraim Rudhani, Naim Morina*, Marigonë Zubaku and Ahmet Avdullahu
Received: June 20, 2018; Published: July 03, 2018
*Corresponding author: Naim Morina, Specialist of Internal Medicine, Clinic for Neophrology and Hemodialysis UCCK Kosovo, Hospital Circle, Kosovo
DOI: 10.26717/BJSTR.2018.06.001331
Introduction: Renal Osteodistrophy is a skeletal change most commonly caused by chronic renal resulting diseases. It is associated with hyperphosphatemia, hypocalcemia, increased secretion of parathyroid hormone, metabolic acidosis and decreased vitamin D activity, which results in skeletal changes.
Purpose of the Research: notification of pathologies of renal osteodystrophy and analysis of collected data.
Materials and Methods: The research was retrospective, conducted at the Hemodialysis Department of Nephrology Clinic, QKUK Prishtina. Part of this study was 89 patients selected in regular hemodialysis sessions. The age of patients ranges from 25 to 80 years, with an average of 57 years. The study was conducted through the collection of data from laboratory documentation of patients who suffered from terminal renal insufficiency.
Results: Out of 89 patients, 42 (47.19%) were women, 47 (52.81%) were males. The most vulnerable age group was 60-69 years old (47.19%), 44 patients with adult phosphorus, 4 patients with decreased phosphorus, 5 were with calcium, 11 with decreased calcium, 5 patients with phosphorus and adult calcium, 1 patient with phosphorus and decreased calcium and 14 patients with increased phosphorus and decreased calcium.
Conclusion: Renal osteodystrophy affects more about 60-69 years of age, while the most affected gender is female and has changes in calcium and phosphorus laboratory values. The most manifested manifestations of both sexes are accompanied by changes in bone remodeling.
Keywords: Renal Osteodystrophy; Chronic Renal Insufficiency
Renal osteodystrophy refers to skeletal changes that result from chronic renal disease and are caused by disorders in calcium and phosphorus metabolism, abnormal vitamin D metabolism and increased thyroid gland activity. In early stages of renal insufficiency, intestinal calcium absorption is reduced because the kidneys are unable to convert vitamin D to its active form 1,25 dihydroxyclaliferol [1-3]. Thus, with the breakdown of the active form of vitamin D, calcium metabolism is regulated, which is the most biologically active form in the absorption of calcium from the digestive tract. Damaged absorbation of calcium occur due to impaired kidney function and the corresponding phosphatase retention due to lower serum calcium percentage. This hypokalcemia is associated with parathyroid gland compensator hyperactivity that increases phosphate excretion in urine, decreases calcium excretion in the urine, and regulates and promotes the release of calcium from the bone. The most frequent changes observed in association with compensatory hyperparathyroidism are those that affect and include the skeletal system. Bone changes are associated with alteration of bone remodeling, osteomalation, osteocytic cystic fibrosis (osteotomy associated with fibrotic degeneration and cystic regions resulting from parathyroid gland hyperfunction) and osteosclerosis. Bone lesions usually appear on the fingers, the clavicle and the acromioclavicular articulation. Other lesions that can be observed are found in the skull, distal clavicle erosion and pubic symphysis margins, rib fractures, and femoral head necrosis [4-8]. In children as predominant lesion is osteomalacia which is associated with bone mitigation, leading to rib and pelvic deformities. Early renal osteodystrophic stages can be detected histologically or biochemically without the presence of distinct radiographic changes because radiographic data of bone disease appear only if 30% of the bone elements are lost [9,10].
The purpose of this paper is to inform about the most common pathologies that lead to the appearance of renal osteodystrophy. Especially the analysis of the data collected by gender, age in patients with renal osteodistrophy and research of calcium and phosphorus values in these patients. Short description or brief description of the aforementioned pathologies (etiology, clinical features, diagnostics, treatment) [11-15].
The research method is retrospective study. Part of this research was 89 patients selected at regular hemodialysis session. The age of patients ranges from 25 to 80 years, with an average of 57 years. The site of the research was the Clinic of Nephrology-Hemodialysis Department, QKUK Prishtina. The study was conducted between collecting data from laboratory records of patients who suffered from terminal renal failure and who were in regular Hemodialysis sessions from 02.01.2017 to 30 June 2017 (retrospective study) [15-18].
For this research, a permission from the director of the Nephrology Clinic was initially granted. The data collection was made from 02 January 2014 to 30 June 2017. The data is processed in Excel and are presented through tables and graphs [19-24].
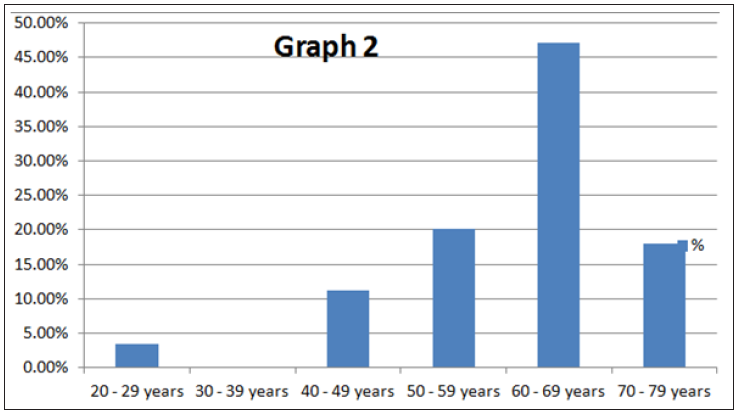
Presentation of the results begins with the data of the patients grouped according to age, gender, calcium and phosphorus values and the frequency of occurrence of diseases in the hemodialysis unit at QKUK (Table 1). In the diagram we see that the age most affected in the hemodialysis ward at UCCK from the total number of samples was 60-69 years old and the more attacked were the males (Graph 1) and (Table 2). From the diagram we find that the most frequent age in patients with renal osteodistrophia in the hemodialysis ward at QKUK from the total number of samples is 60-69 years (47.19%) (Graph 2) and (Table 3). From the diagram we can see that the most affected gender of the patient with renal osteodystrophy in the dialysis ward at UCCK from the total number of samples was female by 57% (Graph 3) and (Table 4). From the diagram we can see that the phosphorus values have been the highest and the most stressed was the male gender, while the values of decreased calcium and increased phosphorus were the most affected by female gender (Graph 4).
Graph 2: Graphic presentation of patients with renal osteodistrophia - terminal renal insufficiency.
Graph 3: Graphic presentation of patients with renal osteodistrophia - terminal renal insufficiency.

Graph 4: Graphic presentation of patients with renal osteodistrophia - terminal renal insufficiency.

During the period January-June 2017, 89 patients were screened for sample In the total number of patients the age and sex frequency was investigated, the most affected age was 60-69 years, and the most common male gender. Over age-related research in patients with renal osteodystrophy, the most affected age was 60- 69 years of age [25-27]. Based on the appearance of gender in renal osteodystoma patients, the most affected gender was female. If we compare with the state of Germany, then we see that the most common pathology of renal osteodystrophy is chronic renal failure. According to the statistics of 2016, we understand that from the diagnosed cases the most common age was 70-79 years old and the most affected gender was female [28-30]. (https://www.aerzteblatt. de/int/archive/article/174773)
The research carried out has shown that the pathology of renal osteodystrophy is terminal chronic insufficiency, with regard to age, the most affected age was around 60-69 years, the most commonly invasive gender among patients with renal osteodystrophy was female gender. The research also includes age-related incidence based on changes in phosphorus and calcium levels. As evidenced by statistics, care of early diagnosis of terminal renal disease, proper treatment and prevention, preclude the appearance of renal osteodystrophy.


