Impact Factor : 0.548
- NLM ID: 101723284
- OCoLC: 999826537
- LCCN: 2017202541
Aleksandra Jukic*
Received: May 29, 2018; Published: June 12, 2018
*Corresponding author: Aleksandra Jukic, Department of Anesthesiology, resuscitation and intensive care, National cancer research center of Serbia, Belgrade, Serbia
DOI: 10.26717/BJSTR.2018.05.001202
Introduction: Almost all chemotherapy agents in use so far have a demonstrated cardiotoxic effect. New therapy protocols have significantly improved long term prognosis of patients with malignant tumors and helped increase their life span, but it also caused the increase of incidence of onset of cardiotoxic effects. The aim of this study was to examine whether preoperatively administered chemotherapy agents influence the onset of perioperative cardiovascular complications and to examine the influence of certain factors on the onset of cardiotoxic effects.
Materials and Methods: Retrospective cohort study was conducted at the National cancer research center of Serbia. Data were collected from medical case histories and anesthesiology documentation of patients without previous cardiovascular disease operated at this institution from January 2013-December 2017. The first group consisted of 1283 patients who were previously administered chemotherapy agents with demonstrated cardiotoxic effect with no regard to the type of tumor, stage of the disease and the period elapsed between the administration of chemotherapy and operation. The second group consisted of 1626 patients who were not administered chemotherapy. Perioperative presence or absence of cardiovascular effects like hypotension, hypertension, arrhythmias, and ischemic changes, signs of heart failure or thromboembolism were followed.
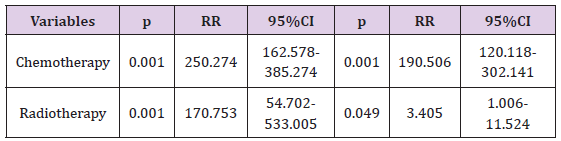
Results: The incidence of onset of cardiotoxic effects in patients who were previously administered chemotherapy found in this study was 98.21%. Univariate regression analysis demonstrated significant correlation of intraoperative onset of cardiotoxic effects and previous administration of chemotherapy (RR=250.274; 95% CI=162.578 – 385.274; p<0.001) as well as previously administered radiotherapy (RR=170.753; 95% CI=54.702 – 533.005; p<0.001). Multivariate regression analysis demonstrated significant correlation of onset of cardiotoxic effects after administration of chemotherapy and the administration of radiotherapy (RR=190.506; 95% CI=120.118 – 302.141; p<0.001).
Conclusion: Patients who were previously administered chemotherapy have increased risk of developing perioperative cardiovascular complications. This requires thorough cardiac examination of these patients before the operation in order to better diagnose and treat consequences of cardiotoxic effects of chemotherapy.
Keywords: Cardiotoxicity, Chemotherapy, Anesthesia, Cancer
Abbreviations: ASA: American Society of Anesthesiology, EFLV: Ejection Fraction of Left Ventricle, DM: Diabetes Mellitus, SD: Standard Deviation
Increase of incidence of malignant diseases which are among the leading causes of morbidity and mortality in the world, requires the development of new therapeutic possibilities and amelioration of the existing treatment protocols. Constant effort in this direction has led to the significant prolongation of life expectancy of oncologic patients but also increased incidence of adverse events of oncologic therapy. There are three main treatment possibilities for malignant diseases, surgery, chemotherapy and radiotherapy. As these treatment modalities are often combined, doctors treating patients who were previously treated due to malignant disease often see adverse events of the previously administered therapy. During the 1970s follow up of patients who previously received cytostatic anthracycline demonstrated increased incidence of development of heart failure in these patients. Various factors like the aging of world population in general, increase of patients with cardiovascular diseases, prolongation of life expectancy of oncologic patients, combination of several chemotherapy agents and/or radiotherapy caused the cardiotoxicity to become among the most serious and most common side-effects of chemotherapy [1].
A lot of patients who have survived the malignant disease after that have the same risk of development of cardiovascular disease and malignant recidivism [2] Although many studies examine cardiotoxicity of chemotherapy agents data are very variable due to the lack of uniformly accepted definition and criteria of cardiotoxicity [3] National cancer institute of United States developed one of the most thorough systems named the General criteria of toxicity which takes into account all important clinical and laboratory changes [4]. Cardiotoxicity of chemotherapy agents can be subacute, acute and chronic. Subacute and acute cardiotoxicity can develop from the moment of administration of chemotherapy agents up to two weeks after the end of the chemotherapy and it can manifest as the changes of ventricular repolarization, ECG changes of QT interval, supraventricular and ventricular arrhythmias, acute coronary syndrome and pericarditis or a syndrome similar to myocarditis.
Chronic cardiotoxicity can develop early, during the first year after the end of chemotherapy, or after this period. Chronic cardiotoxicity is characterized with asymptomatic systolic and/or diastolic dysfunction of the left ventricle which can cause serious congestive cardiomyopathy and even death [5] Cardiotoxicity is influenced by various factors which are in relation to the drug but also the patient. These factors are drug dose, cumulative dose, scheme and the manner of administration, drug formulation, combination of several drugs, age of the patient, the presence of cardiovascular disease, radiotherapy, metabolic abnormalities, hypersensitivity to certain drugs, other risk factors like smoking, obesity, diabetes mellitus [6] Toxicity can manifest itself even several years or even decades after the end of chemotherapy [7,8].
Retrospective cohort study was conducted at the National cancer research center of Serbia. Data were collected from medical histories and anesthesiology documents of patients who did not have previously diagnosed cardiovascular disease operated in this institution from January 1st, 2013-December 31st, 2017. Estimation of the cardiovascular condition of the patients was based upon the physical examination of the patient, data in the medical case history, blood pressure control, ECG finding, heart and lungs X-ray, CBC and the report of the Internal medicine specialist. Patients included in this study were American Society of Anesthesiology (ASA) class I and II. The first group consisted of 1283 patients who were previously administered any chemotherapy agent with proven cardiotoxic effect, no matter the type of the tumor, the stage of the disease and the time period between the administration of chemotherapy to the operational procedure. The second group included 1626 patients who were not previously administered any chemotherapy agents.
Patients having elective surgical procedures of skin, soft tissues, endocrine organs, breast, lungs, digestive and genitourinary system due to primary and metastatic tumors were included. Chemotherapy agents which were administered to the patients of the first group were: bleomycin, carboplatin, cisplatin, cyclophosphamide, dacarbazine, docetaxel, doxorubicin, etoposide, exemestane, fluorouracil, gemcitabine, letrozole, leucovorin, methotrexate, mitomycin, paclitaxel, vinblastine, vincristine, vinorelbine or combinations of these drugs, according to standard treatment regimens. During the operational procedure anesthesia was administered and cardiovascular condition of the patient was estimated by a specialist of anesthesia with at least 5 years of specialist experience. Anesthesia was administered according to the current anesthesia regimens. Instead of anesthetic propofol for induction of anesthesia etomidate was used and for maintenance of anesthesia inhalational anesthetic sevoflurane.
Propofol is known to cause hemodynamic instability so it was not used so that it would not interfere with the results of the study. Perioperative presence or absence of cardiovascular side effects like hypotension, hypertension, arrhythmias, ischemic changes, signs of heart failure or thromboembolism according to the National cancer institute common toxicity criteria, version 4 were followed through data in anesthesiology documents. Patients with clinical disorders class 3 and 4 were included (Table 1) these symptoms were followedduring operation and the first 24 hours after the end of operation. Other factors in relation with the patients were also followed like sex of the patients, age, diabetes mellitus, obesity, smoking, the number of administered chemotherapy cycles and previously administered radiotherapy. Institutional review board approved this study.
a) Patients with no previously diagnosed cardiovascular disease
b) Patients of ASA class I and II
c) Patients who had an elective surgical procedure
The existence of previous cardiovascular disease
a) Patients of ASA class III and IV
b) Patients who were administered cytostatic agents with not confirmed cardiotoxic effect
c) Patients who had an emergency surgical procedure.
Data were processed using X2 test, Student t-test for independent variables and the methods of univariate and multivariate regression analysis. Continual data were expressed as average value ± standard deviation (SD) while categorical variables were expressed in percentages(%).
Table 2: Demographic and other analyzed factors of the patients and presence or absence of cardiotoxic effects.
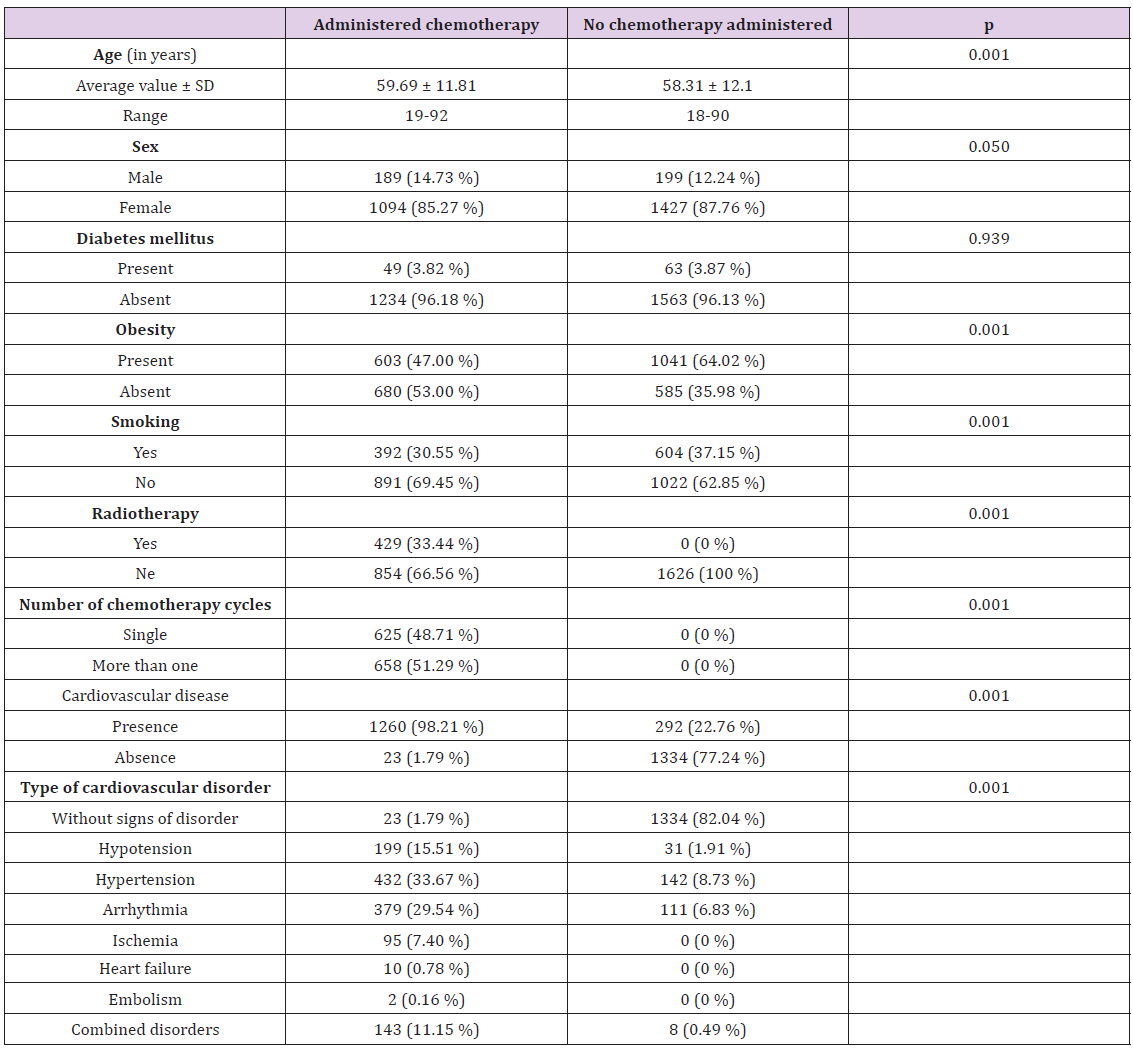
Demographic characteristics of patients (age and sex) and other factors (presence of diabetes mellitus, obesity, smoking, administered radiotherapy, number of cycles of chemotherapy and presence and the type of cardiovascular disorder) were analyzed (Table 2) Patients in both groups were of the similar average age (59. 69 years in the group which received chemotherapy and 58.31 years in the group which received no chemotherapy) There were more patients of female sex in both groups (85.27% in the chemotherapy group and 87.76% in the second group) due to the fact that there was a significant number of patients treated for breast cancer which occurs more frequently in female population. Also, patients with tumors of female genitourinary organs are treated in this institution, whereas patients with tumors of male genitourinary organs are not treated at the same institution. In both groups there was no difference between patients who suffered from diabetes mellitus (3.82% in the first, 3.87% in the second group).
There was a bigger percent of smokers among the patients who were not administered chemotherapy (37.15%) compared to the group of patients which have had chemotherapy administered (30.55%) due to the fact that more of patients with malignant disease who were already treated for some time were ready to give up the habit of smoking than the patients who were recently diagnosed and had a surgery as the first treatment option. The group who had chemotherapy administered had less obese patients than the second group (47.00% compared to 64.02%) due to the loss of weight as one of the principal features of malignant disease.
The incidence of onset of cardiotoxic effects in patients who were previously administered chemotherapy in this study is significantly higher compared to the patients who previously had no chemotherapy administered (98.21% compared to 22.76%, p<0.001). Patients who were previously administered chemotherapy have 250 times higher risk of onset of cardiotoxic effects than patients without previous chemotherapy (RR=250.274; 95% CI=162.578–385.274; p<0.001). Patients who received radiotherapy have 170 times higher risk for the onset of these effects than the patients who had no radiotherapy (RR=170.753; 95% CI=54.702-533.005; p<0.001). Patients who were administered both chemotherapy and radiotherapy have 190 times higher risk of development of cardiotoxic effects than patients who had neither of these treatments (RR=190.506; 95% CI=120.118-302.141; p<0,001).
In the group of patients who were administered chemotherapy the most frequent was hypertension (33.67%), arrhythmias (29.54%), hypotension (15.51%), combined disorders (11.15%), ischemia (7.40%) while the least frequent were signs of heart failure (0.78%) and pulmonary embolism (0.16%). Among the patients who received no chemotherapy the most frequent were hypertension (8.73%), arrhythmias (6.83%), hypotension (1.91%) and combined disorders (0.49%). There were no patients with signs of myocardial ischemia, heart failure and pulmonary embolism in the second group. High frequency of hypertension, hypotension and heart arrhythmias in both groups are also caused by surgical procedure, anesthesia and perioperative drugs.Univariate regression analysis presented significant relation of the occurrence of cardiotoxic effects and previous administration of chemotherapy as well as previous administration of radiotherapy. Multivariate regression analysis showed dependence of the occurrence of cardiotoxic effects after administration of chemotherapy and administration of radiotherapy (Table 3). Statistically significant correlation of other factors which were followed, age, sex, diabetes mellitus, obesity, smoking and the number of administered cycles of chemotherapy with increased incidence of onset of cardiotoxic effects after the administration of chemotherapy was not demonstrated (p˃0.05).
Table 3: Univariate and multivariate analysis showing correlation of examined factors with the onset of cardiotoxic effects.
Due to development and improvement of new treatment possibilities for cancer 5-year survival of patients with cancer diagnosed between 1975 and 1977 compared to the patients with cancer diagnosed between 1999 and 2005 in United States of America improved from 50% to 68% [9] However, those patients suffer complications of previously administered therapy. It is estimated that approximately 2 000 000 patients are in risk of developing late anthracycline toxicity [10] Almost every chemotherapy agent approved for use up to now can be considered cardiotoxic [11] According to the guidelines of the American College of Cardiology and American Heart Association patients who were administered chemotherapy are under increased risk of developing heart failure [12] The incidence of onset of cardiotoxic effects in this study is higher than in most of the studies of other authors. Tsibiribi et al. and Pai et al. [13-16] report that the risk of developing myocardial ischemia after administration of high doses of fluorouracil 5% varies from 1-68%. The incidence of chronic cardiotoxicity of 5-65% is reported in studies of Singal et al (1998), Jensen et al (2002), Lipshultz et al (1995) and Curigliano et al. There are several reasons for such a high incidence of cardiotoxic effects in this study. Most of other authors examined the cardiotoxicity of certain drugs which can be very variable.
In this study it was examined the onset of cardiovascular side-effects with no relation to the type of the administered drug, type and stage of malignant disease and the period from administration of chemotherapy to surgical procedure.However, the most important reason for such a high incidence of cardiotoxic effects in this study is the fact that although patients pass a thorough cardiology examination before they are administered chemotherapy, such a detailed examination is not compulsory for the patients having a surgical procedure who were previously administered chemotherapy [17- 18]. This examination does not include an echocardiographic examination or another manner of estimation of ejection fraction of left ventricle (EFLV) if the patient does not have a previous cardiovascular disease. If a patient does not have a previous cardiac disease and symptoms at the time of preparation for surgery such a thorough examination in our institution will not be done.
However, Meinardi et al. reports higher risk of cardiovascular events in long/term survivors of testicular cancer. Mitani et al. report that the studies which have defined a criterion for discontinuation of chemotherapy based upon the estimation of EFLV (reduction of EFLV˃10%, in relation to the absolute value of EFLV <50%) the risk for development of confirmed heart failure is reduced to less than 5% [13,19-22] Although this study has some limiting methodology factors, it can be safely concluded that in patients who previously had chemotherapy administered, several years or even decades before, there is a higher risk of onset of consequences of cardiotoxic effects. If such a patient has to have an operational procedure the stress from surgery and effects of anesthesia can potentiate this risk. That is why it is important to do a thorough cardiac examination of long/term cancer survivors who were treated with chemotherapy in order to be able to reduce this risk and treat these effects and reduce the risk of serious cardiovascular perioperative events.


