Impact Factor : 0.548
- NLM ID: 101723284
- OCoLC: 999826537
- LCCN: 2017202541
Hilla Shaviv1, Doron JD Rosen2 and Yossef Ezra*3
Received: January 19, 2018; Published: January 30, 2018
Corresponding author: Yossef Ezra, Department of Obstetrics and Gyencology, Hadassah Hebrew University Medical Center, Ein-Kerem, Jerusalem, Israel, POBox 12000, Zip 9112001
DOI: 10.26717/BJSTR.2018.02.000706
Objective: To evaluate a new treatment for dysmenorrhea by inducing menstrual fluid streaming mechanically.
Setting: A 4-month randomized double-blind, placebo-controlled, crossover study.
Participants: Study participants were women experiencing dysmenorrhea monthly (for the 6 preceding months) at a level >5 on the visual analog scale (0-10), who were otherwise healthy. The study participants also collected their menstrual fluid for 2 baseline months, while recording their pain levels (primary outcome) systematically. A correlation between menstrual fluid characteristics and menstrual pain was studied, too.
Intervention: A newly invented device was used to attenuate dysmenorrheic pain level.
Results: From September 2011 to December 2013, 28 study participants were enrolled; 3 withdrew and 3 were excluded for protocol non-adherence. Twenty-two study participants (132 menstrual cycles) who used the device reported an average reduction of 55±7.6% in pain levels vs. 22±12% reported with placebo (p = 0.008). Analysis of menstrual fluid collected from 19 study participants (38 menstrual cycles) showed high correlation between pain levels and menstrual fluid viscosity/tissue fragments, indicating that dysmenorrhea is closely related to the rheological characteristics of the menstrual fluid.
Conclusions: The new device is highly effective in relieving menstrual pain. Rheological characteristics of the menses are correlated with severity of dysmenorrhea.
Keywords: Dysmenorrhea; Menses; Randomized trial; Rheology; Vaginal device; Acti Lady
Abbreviations: PD: Primary Dysmenorrhea; NSAIDs: Nonsteroidal Anti-Inflammatory Drugs; AEs: Adverse Events; OC: Oral Contraceptives; IRB: Institutional Review Board
Approximately 50% of all menstruating women experience primary dysmenorrhea (PD) and 10-15% report severe symptoms, described as sharp, intermittent, or continuous pain, usually in the pelvis or lower abdomen within the first 48 hours of menstruation [1-6]. Although PD is prevalent, its pathophysiological origins are still unclear. PD may be associated with myometrial ischemia during menstruation. This ischemia is attributed to uterine contractions, induced by menstrual fluid prostaglandins [1]. The gold standard for treating PD is nonsteroidal anti-inflammatory drugs (NSAIDs), which inhibit prostaglandins production and limit their activity. Although effective, NSAID use may be associated with various adverse events (AEs) [7-14]. Oral contraceptives (OC) serve as the second therapy of choice is for PD, as they reduce menstrual fluid volume and decrease plasma vasopressin levels found in women with PD, leading to attenuation of the excessive uterine activity. However, OC use is also associated with AEs and medical limitations [15-17]. Another therapeutic approach, which is painful, temporary, invasive, and no longer in use, was cervical dilatation using mechanical instruments and medications [18,19].
The effectiveness of OC and cervical dilatation suggest that the pathophysiology of PD may be related to menstrual fluid physical characteristics such as corpuscular size and viscosity. Surprisingly, rheological characteristics of the menstrual fluid were never studied independently or in relation to PD. We hypothesize that the rheological properties of the menstrual fluid that flows though the uterine cervix [20,21], results in an impaired and even completely blocked flow. This impaired flow prolongs the exposure of the myometrium to the prostaglandins contained within the menstrual fluid, which in turn, increases the intensity of the uterine contractions [22], resulting in increased uterine pressure and ischemia, leading to menstrual pain [23,24]. In order to test this hypothesis we developed a vaginal device that promotes the flow of the menstrual fluid through the uterine cervix and as a result, would improve PD symptoms.
We intended to test the hypothesis of slow or obstructed flow through the cervical canal in 2 ways. First, to assess the correlation between menstrual fluid characteristics and pain levels by collecting the menstrual fluid and documenting pain levels systematically Then, to examine the efficacy of a novel vaginal device designed to promote better flow of the menstrual fluid through the cervical canal by creating oscillatory pressure gradient, leading to reduced menstrual fluid viscosity and accelerated passage.
The clinical trial was a prospective, randomized, double-blind, placebo-controlled, crossover study where for each participant we compared pain levels without the device (baseline), with the device, and with a placebo device. The protocol was approved by the institutional review board (IRB) at The Hadassah-Hebrew University Medical Center and all study participants signed an informed consent. Patients were used as their own controls, as PD typically exhibits similar pain levels every month [25]. The study was designed for 24 otherwise healthy women (as determined by physical examination and medical history). Key eligibility criteria included: 18-35 years of age; regular menstruation (28- day cycle ± 7 days); nonuse of OC in the 6 months preceding the study (and throughout the study); a history of PD (onset <1 year after menarche), which could be effectively treated with over-the- counter analgesics or OC; no history of pelvic pathology; and PD monthly at visual analog scale (VAS 0-10) pain level >5 in the 6 preceding months (for full inclusion and exclusion criteria see the Supplementary Materials).
Study participants were randomized (using a computergenerated randomization schedule prepared with R [R 3.1.1 R foundation]) into 4 groups of 6. The study spanned 6 menstrual cycles. For all groups, the first and last menstruations were baseline cycles (i.e., without using the device) and in between, each group had 3 cycles with the active device and 1 with placebo (for each group, the placebo was used in a different cycle). After the first baseline cycle, the device was inserted (not during menses) by a physician for a "safety test," activated for 5 minutes and then taken out. Any discomfort or pain from this procedure was documented. In subsequent cycles, the study participants inserted the device or the placebo by themselves. The study participants, monitors, and the medical team were blinded to the treatment received.
The study was carried out at the study participants' natural environment. The study participant documented their pain levels throughout the study (during the menstrual periods) every 4 hours if VAS pain levels were <5 and every 30 minutes if they were ≤5 and during the use of the device. Any significant changes in pain levels were also recorded. During sleep hours (7.5 hours every 24 hours) the study participants were exempt from documenting pain and using any devices. During the months with either the active or placebo device, each study participant was given 3 devices for her personal use.
Figure 1: Flow chart used to guide the study participants� use of the device.
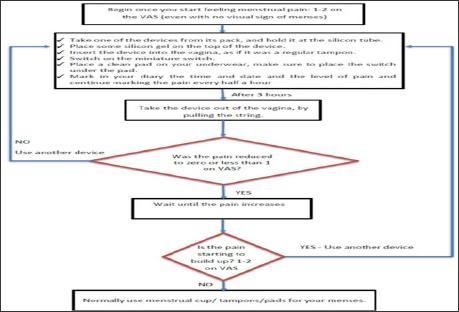
The study participants were instructed on how to use the device and where to locate it (as positioning it correctly adjacent to the cervical os is extremely important). If the device had no effect after 10 minutes, the study participants were instructed to slightly modify its positioning in the vagina. The devices were used according to the instruction depicted in Figure 1. Device use was to commence when VAS pain levels first reached 1 or 2 (even if bleeding signs were not yet apparent). Each use was 3 hours long. Each study participant determined for herself the number (at most 3) and timing of using the device, according to her pain level. The study participants were allowed to use their preferred analgesics as rescue medication, but were limited to one brand. Analgesics use was recorded (date/time/dose).
For the first part, during baseline months, study participants used a menstrual cup to collect the secreted menstrual fluid, emptying it every 4 hours into separate collecting test tubes while documenting the date/time of each collection. The test tubes were collected from each study participant once a day and kept refrigerated till examined. We used a simplistic approach to estimate menstrual fluid characteristics since the rheological properties of the menstrual fluid were never measured before. Furthermore, commonly used methods for studying viscous body fluids such as nasal fluid [26] are not applicable to menstrual fluid due to the limited amount of sample collected, the heterogeneity of the samples, and their opaque nature. The menstrual fluid was diluted with distilled water, particles (e.g., tissue fragments, blood clots) were isolated, visually inspected, their size was measured using a ruler, and tissue fragments >1 cm in diameter were counted.
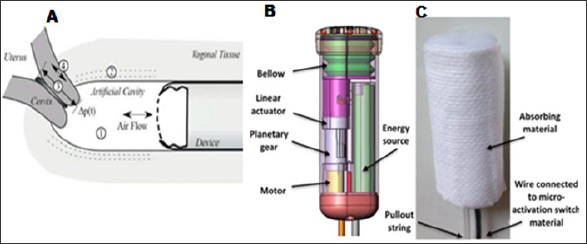
The development of the vaginal device was based on the hypothesis that blocked flow of menstrual fluid leads to PD. The menstrual fluid is a non-Newtonian fluid (i.e., a fluid whose viscosity is not linearly related to the shear rate) and its dynamic viscosity (resistance to flow) is reduced when subjected to oscillatory pressure gradient. This effect is known as "streaming” and is used in cystic fibrosis patients to facilitate the clearing of mucous from their lungs. The new device was designed to be positioned in the vagina, sized and shaped as a maximum absorbency tampon. The device creates a pressure oscillation in the closed artificial cavity formed next to the cervical os (Figure 2A). This action creates an oscillatory pressure gradient that is forced across the cervical canal, leading to reduced viscosity of the menstrual flow and accelerates its passage. The pressure oscillations are generated by a bellow connected to a rotating motor through a linear actuator (Figure 2B).
A micro activation switch is connected to a wire exiting the vaginal cavity (once activated, the micro switch is placed under a pad located on the study participant's underwear). The device is wrapped in a non-woven absorbent material, such that it can absorb the secreted menstrual fluid while in use (Figure 2C). The waveform is a 1 Hz slow rising and fast falling saw tooth shaped wave with an amplitude of 20 mmHg. The asymmetric waveform facilitates a unidirectional flow from the uterus to the vagina. The oscillations, which are not felt by the study participant, deliver a mechanical energy flux of approximately 2.4 mW/cm2 to the vaginal wall, over a typical duration of several hours. The placebo device is identical in appearance to the active device but the bellow was disabled.
Figure 2: The experimental setup and the vaginal device.
(A) A device is placed in the vagina, forming an artificial cavity (1) next to the cervical os. By periodically changing the volume of a bellow, the pressure in the cavity can be controlled through the finite compliance of the walls (2). A pressure gradient is therefore forced across the cervix (3). Any menstrual fluid present in the cervix is accelerated. The consequent shear stress reduces menstrual fluid viscosity, thereby facilitating their flow to the artificial cavity. Most of the acceleration is directly through air pressure variations. A minor acceleration is due to longitudinal acceleration of the cervix (4), whereas negligible acceleration is caused by vibrations of the vaginal tissue propagating to the cervix.
(B) Schematic representation of the vaginal device.
(C) Picture of the device.
Based on the results by Marsden et al. [27] on the effects of guaifenesin (a cervical dilator), we expected a reduction of at least 3 points on VAS (from a typical value of 6 to 3), with a scatter (variance) of ±3 points. Given that the placebo effect in analgesics is 1 point on VAS, 21 study participants were required to obtain on average a 3a result above the placebo effect (i.e., at least 99.8% confidence), with an 84% probability of obtaining at least a 2a result (i.e., at least 97.5% confidence). The raw VAS pain level and analgesic use data were reduced to simplified pain level indicators quantifying the amount of pain experienced in each menstrual cycle.
Four indicators were used for the VAS data and 1 for analgesics usage as follows:
i. Integrated Pain [P ] is the time integrated pain level over the menses duration (units: VAS•h), which provides an overall amount of pain;
ii. Maximal Pain [Pmax ] is the maximal recorded pain each month;
iii. Mean Post Treatment Pain [Ptreat] is the mean pain over 24 hours after the device is used for the first time;
iv. Mean Pain Following its Onset [Pthres] is the mean pain in the 24 hours from the time VAS pain levels reached 50% of the maximal pain on that month;
v. Average Number of Analgesics [Nanalg] is the number of analgesic pills taken over the menstrual period. Welch's t-test was used to compare unpaired quantitative pain indicators obtained with the placebo to those obtained with the active device. The test was 2 sided at a significance level of 0.05 and was performed using the R software (version 3.1.0).
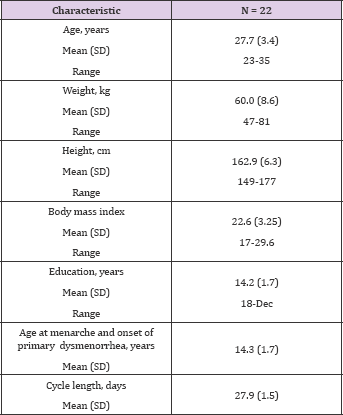
The study took place between September 2011 and December 2013. Twenty-eight participants were recruited and passed the screening tests; 3 withdrew (one became pregnant during her first month in the study, one was enlisted into the army, and one elected to withdraw), and 3 were excluded for protocol non-adherence. The current analysis includes the data collected from the remaining 22 study participants over 132 accumulated menstrual cycles (6 months per study participant). Participant demographics are presented in Table 1. Of the 22 participants who completed the study, 19 were able to use the menstrual cup and collect their menstrual fluid. For each of them, the temporal pain levels, as well as flux and appearance of clots/tissue fragments in the menstrual fluid during the baseline cycles, were evaluated.(6 months per study participant). Participant demographics are presented in Table 1. Of the 22 participants who completed the study, 19 were able to use the menstrual cup and collect their menstrual fluid. For each of them, the temporal pain levels, as well as flux and appearance of clots/tissue fragments in the menstrual fluid during the baseline cycles, were evaluated.
Table 1: Patients demographics.
We found that menstrual fluid could be grouped into 3 categories:
i. Bloody menstrual fluid containing tissue fragmvents larger than the average cervical diameter
ii. Very thick menstrual fluid (with egg yolk-like consistency), with or without such tissue fragments; and
iii. Bloody menstrual fluid with no tissue fragments (bloodlike consistency) (Figure 1).
Figure 3: The average pain and flux for all study participants (orange, flux; blue, pain). Time was measured from when the pain reached 50% of its maximal level; shaded areas denote one standard deviation
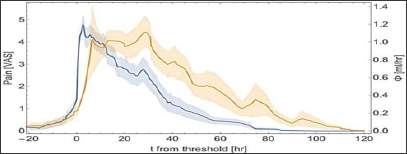
In all cases, the peak in the flux (flow divided by time interval) lagged the maximal pain level. This can be further quantified if mean time of the flux is defined as ∫tØ(t)dt/ ∫Ø(t)dt and mean time of the pain as∫tvas(t)dt/Ø(t)dt/ ∫tvas(t)dt. Doing so, yielded a lag of 11.2 ± 3.5 hours between the two means, and a signed a paired non-parametric test (comparing just the signs of the lags) ruled out the no-lag hypothesis at the 0.013% level. The average flux and pain values are presented in Figure 3. Moreover, particles or very thick menstrual fluid were generally observed only during the first 48 hours of menstruation, when pain was present. Subsequently, when no pain was reported, the menstrual fluid had a blood-like consistency. Notably, one study participant was atypically experiencing pain 72 hours after the onset of menstruation. Interestingly, in this study participant, particles were found only during that time, whereas during the rest of the menstruation the menstrual fluid had blood-like consistency.
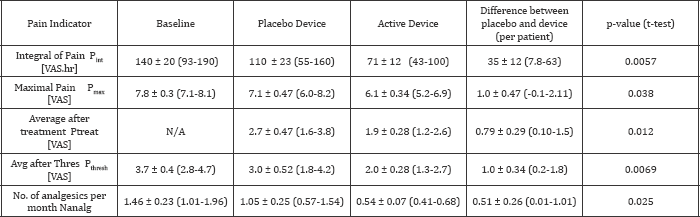
During the "safety test” no pain or discomfort were reported from using the device. Moreover, no pain or AE were reported throughout the study. No short- or long-term safety issues or irregularities were observed in routine gynecological examinations, nor were they reported by the study participants. Two women reported vaginal yeast infection. The reported pain was generally lower with the active than with the placebo device (Figure 4). There were cases that were characterized by low pain levels with the active as well with the placebo devices. The average pain indicators during the experimental cycles and the average number of analgesics taken to relieve pain are summarized in Table 2. Treatment with the active device was significantly more effective in reducing pain than treatment with the placebo device (regardless of the indicator used for the analysis).
Figure 4: Correlation between the average pain with an active device vs. placebo for Pint (A) and Ptreat (B). Each dot corresponds to an individual study participant. Each participant used up to three devices, according to need (see Fig. 1); each device was used for three hours and was then taken out. Dots in the greyed area represent individuals for who menstrual pain experienced with the active device was attenuated compared with the pain experienced with the placebo device.
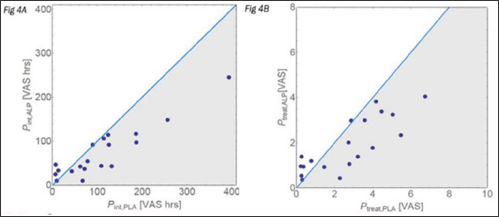
Table 2: Summary of the average pain indicators as measured during baseline, with an active device and a placebo device (N = 22).
Data are mean ± SD (95% CI).
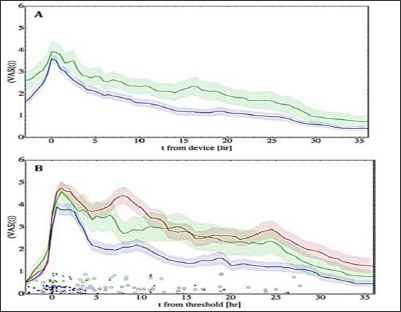
Compared with baseline, average pain reduction with the active device was 2.2 to 2.4 times greater than that observed with the placebo device (p <0.05 for all comparisons; t-test). In addition, we observed a significantly reduced analgesics intake in cycles where the active device was used compared to placebo (p<0.025; t-test). The mean time-dependent profiles of VAS pain (Figure 5) show that pain reduction with the active device was significantly greater than with the placebo device and that the effect could last over a day.
Figure 5: VAS pain levels over time.
(A) Mean VAS in study participants using an active (blue) or placebo device (green) over time (measured from the application of the device). Shaded area denotes the standard deviation.
(B) Same as panel A, except that time was measured from when the pain reached 50% of its maximal level. Red denotes baseline pain. Solid and empty blue dots denote the first and second use of the active device, respectively. Green dots denote use of the placebo device. Each study participant used up to three devices, according to need (see Fig. 1), each device was used for three hours and was then taken out.
In the present study we found that the newly invented vaginal device has been shown to be 2.3 times more effective in reducing menstrual pain than placebo. This reduction in pain supports our hypothesis on the role of blocked flow of menstrual fluid in PD. For comparison, aceclofenac and naproxen have been reported to be approximately 1.5 times more effective than placebo [7,28,29], and ibuprofen has been reported to be approximately 1.2 times more effective than placebo in reducing PD pain [29]. Notably, since rescue analgesics were allowed in our study, and their use was highest at baseline cycles and lowest when the active device was used, our observed VAS pain reductions represent the lower limit of the device efficacy. Furthermore, we speculate that some of the study participants might suffer from endometriosis and not from PD. Thus, if we could exclude women with proven endometriosis from this study, the actual efficacy of the device would have been higher.
PD is typically experienced during the first 24-36 hours after the onset of menstruation, after which the pain dissipates while the bleeding continues for a few more days [4,5]. In the current study, except for one case, PD followed this pattern and the pain was reported either with the first sign of menstruation or a few hours before bleeding was observed. In most cases, no pain was recorded after 48 hours. When the pain was at its highest level (i.e., when the uterine pressure peaks [30,31]), large particles consisting of tissue fragments or very thick menstrual fluid, were observed in the samples collected following the reported pain. Uterine contractions are excited by the secretion of prostaglandin F2a (PGF2a), found in excessive levels in women experiencing PD (compared with eumenorrheic women) [6,8,9,11-17]. Such uterine pressure cannot be generated unless the cervical lumen is blocked or significantly obstructed.
There is evidence suggesting that the elevated pressure results in additional PGF2a secretion, which leads to more contractions and pain. We found that the tissue fragments in the collected menstrual fluid were larger than the diameter of the rigid cervical canal, suggesting that they block the cervical lumen when exiting the uterus. Exceptionally, we noticed two study participants with no particles contained in the menstrual fluid and fluid that was not thick at any time. One possibility is that these participants actually suffered from undiagnosed endometriosis, although measures have been taken to reduce the probability of having such participants in our study (i.e., excluding participants whose medial history was suggestive of endometriosis such as those experiencing pain during sexual intercourse, bleeding between menses, etc).
Our study has a number of limitations. The sample size was relatively small and did not allow for statistical analysis of the rheological findings. Moreover, standard reproducible methods for evaluating rheological properties of menstrual fluid are currently lacking. Also, the study focused on women experiencing PD and non-PD controls were not included. Comparing PD and non-PD women with respect to rheological properties of the menstrual fluid and other parameters such as the size of the cervical opening could have provided further insights regarding the mechanism underlying PD. Also, the study was not designed to measure the potential impact of the device on the volume of the menstrual fluid or on the duration of the menses. Lastly, despite designing our inclusion/exclusion criteria to minimize the risk of including participants with secondary dysmenorrhea/endometriosis, we cannot rule out having such participants in our study.
In summary, our findings suggest that rheological properties of menstrual fluid might be the trigger for PD, and that induced streaming of menstrual fluid is highly effective in relieving pain. Future investigations with larger samples size (including non-PD controls) and longer follow up are warranted.
The study was funded by GalMedics Biotech Ltd. Hilla Shaviv is employed by GalMedics and is a shareholder. Doron Rosen is a shareholder in GalMedics. Yossef Ezra serves on the scientific board of GalMedics.
We thank Medicsense Ltd. (Israel) for helping with the design and submission of the clinical study as well as all the regulatory affairs concerning the development of the evaluated device, and for monitoring and examining the clinical site. We thank Prof. Nir Shaviv for performing the statistical analysis.


