Impact Factor : 0.548
- NLM ID: 101723284
- OCoLC: 999826537
- LCCN: 2017202541
Rüdiger Hardeland*
Received: August 23, 2017; Published: August 31, 2017
Corresponding author: Rüdiger Hardeland, Johann Friedrich Blumenbach Institute of Zoology and Anthropology, University of Goettingen, Buergerstr 50, D-37073 Göttingen, Germany
DOI: 10.26717/BJSTR.2017.01.000318
The melatonin metabolite N1-acetyl-5-methoxykynuramine (AMK) is a bioactive compound that acts as a potent inhibitor of cyclooxygenases, of neuronal NO synthase and myeloperoxidase, as a downregulator of cycloxygenase-2 and inducible NO synthase. Additionally, AMK is a potent scavenger of hydroxyl, peroxyl and carbonate radicals. It eliminates singlet oxygen more efficiently than histidine and related compounds. As a scavenger of all NO redox congeners, it forms a stable compound, 3-acetamidomethyl-6-methoxycinnolinone, that does not re-donate NO. Carbamoylating metabolites convert AMK into N-[2-(6-methoxyquinazolin-4-yl)-ethyl]-acetamide, another stable product. Upon oxidation, AMK forms an adduct-generating intermediate, which can attach to aromates, among them tyrosine and tryptophan. Protein AMKylation at tyrosines may cause inhibition of enzymes. With regard to receptor tyrosine kinases, this possibility may explain the observed growth inhibition in keratinocytes.
Keywords: AMK; Inflammation; NO synthases; Posttranslational modification; RNS; ROS
Abbreviations AMK: N1-acetyl-5-methoxykynuramine; CSF: Cerebrospinal Fluid; COX: Cyclooxygenase; iNOS : Inducible NO Synthase; MtiNOS: Mitochondrially Targeted Subform; NNOS: Neuronal NO Synthase; ROS: Reactive Oxygen Species
Melatonin (N-acetyl-5-methoxytryptamine; Figure 1), usually known as the nocturnally secreted hormone of the pineal gland, is, in fact, produced by numerous organs in quantities that exceed the amounts in the pineal and in the circulation by orders of magnitude [1]. However, in mammals, extrapineal melatonin is usually poorly released into the blood. The major catabolic pathway of circulating melatonin consists of monooxygenation to 6-hydroxymelatonin and subsequent conjugation, mostly to the easily excretable 6-sulfatoxymelatonin. Various other pathways exist as well. Among them, the kynuric pathway differs from the others by the degradation of the indole moiety [2]. In its initial step, the pyrrole ring of the indole is cleaved by dioxygenation in one or multiple steps. Remarkably many reactions of enzymatic, pseudoenzymatic, photocatalytic and free radical-mediated nature lead to the same product, N1-acetyl-N2-formyl-5-methoxykynuramine (AFMK; Figure 1) [2]. In the circulation, the amounts of AFMK mostly remain negligibly small. However, under conditions of oxidative stress, especially in inflammation, AFMK can attain remarkably high levels. In the cerebrospinal fluid (CSF) of patients with viral meningitis, AFMK concentrations were by orders of magnitude higher than the nocturnal maximum of melatonin [3]. This astonishing result may be explained by the fact that melatonin is readily oxidized under inflammatory conditions, whereas AFMK can accumulate because of much higher inertness [4,5], which only allows efficient interactions with the highly reactive hydroxyl radicals (•OH) [6], but not at comparable rates with oxidants of low reactivity.
AFMK can be also metabolized by enzymatic and nonenzymatic means. Besides several products of poorly understood or presumably absent biological activity [7,8], one metabolite exists that displays various biological effects of great interest, N1-acetyl- 5-methoxykynuramine (AMK; Figure 1). To date, three modes of formation from AFMK have been described, deformylation by arylamine formamidases [2] or by hemoperoxidases [9], and CO liberation by UV light of sufficient energy (UVB, UVC) [10]. Whether additional free radical-mediated pathways exist, would require more direct evidence. AMK displays several properties concerning inhibition or downregulation of biologically and medically important enzymes and, additionally, remarkably high reactivities towards reactive oxygen and nitrogen species, clearly exceeding those of AFMK and, sometimes, even those of melatonin.
The first report on biological activities of AMK dates back to 1984, when this compound was reported to be a cyclooxygenase (COX) inhibitor by above an order of magnitude more potent than acetylsalicylic acid (aspirin) [11]. At that time, no distinction was made between COX-1 and COX-2, but from the experimental settings, this effect should have concerned COX-1. In a later study [12], AMK was shown to downregulate COX-2 expression in macrophages. However, high pharmacological levels of 0.5 or 1 mM were required to demonstrate this effect. However, these elevated concentrations were necessary because the cells were challenged by bacterial lipopolysaccharide, a potent pro-inflammatory agent. No effects on COX-1 expression were observed [12], indicating that the earlier observed inhibition concerns actions at the enzyme protein rather than on gene suppression.
AMK was reported to possess various other anti-inflammatory properties. In neutrophils, it was shown to inhibit myeloperoxidase, thereby strongly reducing HOCl concentrations, an effect not shared with AFMK [13]. Another anti-inflammatory effect of substantial relevance concerns the downregulation of inducible NO synthase (iNOS), including its mitochondrially targeted subform (mt-iNOS) [14]. This effect, which was discovered in the context of mitochondrial oxidotoxicity, may be of relevance in terms of limiting inflammatory responses in microglia and pro-inflammatory communication between astrocytes and microglia [15]. Moreover, AMK was shown to inhibit, at remarkably low concentrations, the neuronal NO synthase (nNOS) [16,17]. Although half-inhibition was in the micromolar range, the IC20 value was as low as 10–11 M [17]. This discrepancy between half-inhibitory concentrations and measurable effects at extremely low levels has been detected in an entirely different function, too, as will be discussed and tried to explain in the section on “stickiness” of oxidatively formed AMK intermediates. Nevertheless, the increase of AFMK under inflammatory conditions may give rise to enzymatically produced AMK levels that may be biologically relevant. In such a way, AMK may contribute, in conjunction with its parent compound, melatonin, to the interruption of crosstalks between neurons, astrocytes and microglia and, therefore, antagonize microglia activation as caused by mechanisms of excitotoxicity [15,18]. Notably, the decreases in both iNOS and nNOS activities can be expected to jointly reduce both cytosolic and mitochondrial Ca2+ levels and, thus, support corresponding actions by melatonin that attenuate overexcitation and Ca2+-mediated mitochondrial dysfunction. Insofar, it is not surprising that mitochondrial protection and functional improvement have been demonstrated for AMK, too [14-19].
AMK has also been shown to be a potent scavenger of several reactive oxygen species (ROS) and other free radicals with oxidizing properties. In this regard and in all cases tested, AMK is much more effective than its precursor AFMK [2,4,5]. AMK was shown to efficiently scavenge hydroxyl radicals [4,5], peroxyl radicals [5], carbonate radicals [4,5], and the synthetic ABTS cation radicals [4,5,20,21] {ABTS = 2,2’-azino-bis-(3-ethylbenzthiazoline-6- sulfonic acid)} and to protect proteins from oxidative destruction [5]. Moreover, AMK turned out to be one of the most potent scavengers of a nonradical ROS, singlet oxygen [O2(1Δg)] [22], a property of interest to dermatologists. AMK was over 150-fold more effective than the frequently used synthetic scavenger DABCO {diazabicyclo-[2,2,2]-octane} and still much better than imidazole (16-fold), Nα-acetylhistidine (8-fold) and histidine (4-fold), the latter representing an experimentally often applied scavenger that is usually considered to be only surpassed by polymeric singlet oxygen quenchers. Among the low molecular weight compounds, only melatonin’s scavenging capacity proved to be more effective than histidine, but AMK was still about 1.6 times more potent [22]. Notably, AFMK remained practically inert towards singlet oxygen under same conditions.
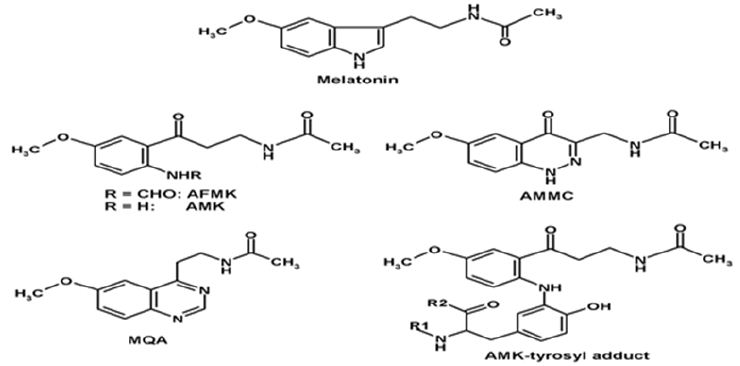
Figure 1: Structures of melatonin, AFMK, AMK, 3-acetamidomethyl-6-methoxycinnolinone (AMMC), N-[2-(6-methoxyquinazolin-4-yl)-ethyl]-acetamide (MQA), and the adduct of AMK with a tyrosyl residue in a peptide chain.
In addition to ROS, AMK interacted readily with reactive nitrogen species (RNS). This was first discovered with •NO, the radical form of nitric oxide as it is produced by the NO synthases. AMK was that much effective that it reacted already with traces of •NO in the air [2]. Previously, nitrosation had been studied in the case of melatonin, but the 1-nitrosomelatonin formed easily redonates •NO [23]. Therefore, melatonin is, in the balance, unable to efficiently eliminating •NO. AMK, being a substituted aniline, also contrasts with other anilines that form unstable nitrosamines which undergo secondary reactions [24]. Instead, AMK forms, by interaction with •NO, a second six-membered ring containing two nitrogen atoms, to give the, at that time, newly discovered compound 3-acetamidomethyl-6-methoxycinnolinone (AMMC; Figure 1) [25]. Through the formation of this new ring, AMMC is remarkably stable and does not re-donate •NO [24-26]. Since nitrosation is also possible by variant reactions of the other NO redox congeners, NO+ and HNO, i.e., the protonated form of NO–, AMK was also tested in presence of donors liberating these compounds. Again, the rapidly formed and stable product AMMC was generated [26].
A further product appeared by nitration of AMK, which took place in the presence of carbonate radicals and •NO2 that originate by decomposition of the peroxynitrite-CO2 adduct [25-27]. To date, no indication exists for a biological role of this metabolite, 3-nitro- AMK. Unexpectedly, AMK also formed another compound with a second 6-membered ring containing two nitrogen atoms. This metabolite, which did not result from interaction with •NOx, was identified as N-[2-(6-methoxyquinazolin-4-yl)-ethyl]-acetamide (MQA; (Figure 1) [25]. Initially, the chemical reaction by which this substance was generated remained somewhat enigmatic, but its formation was demonstrated upon incubation of yeast cells with AFMK [28].
In a further study, it turned out that MQA was neither formed by condensation of AFMK with ammonia, nor of AMK with formamide, but rather by carbamoylating reaction mixtures, especially by carbamoyl phosphate, a biologically occurring metabolite, in a reaction that was further enhanced by hydrogen peroxide and copper(II) [29]. This mode of formation notably differed from another well-known carbamoylation reaction by isocyanate. The enhancement by H2O2 and copper may be indicative for a reactivity of AMK towards the so-called crypto- or pseudo-hydroxyl radical, which may be a copper-hydroperoxo complex that undergoes •OHlike reactions, but is not inhibited by classic •OH scavengers such as mannitol [29]. Therefore, AMK may be classified as a scavenger of pseudo-hydroxyl radicals, too, a rather unusual property not known to date for melatonin or any other of its metabolites.
The high reactivity of AMK has led to the questions of further interaction partners and oxidation products. Oxidation studies with hydroxyl or carbonate radicals showed rapid destruction and formation of very short-lived products that have not yet been chemically characterized because of their instability and rapid decay [2]. Using the ABTS cation radical as a less reactive oxidant several products were identified and their structures described [20]. However, these compounds were oxidatively formed dimers and oligomers. Their occurrence may be physiologically irrelevant, but these products were indicative for a high reactivity of oxidatively formed AMK intermediates towards other aromates. This prompted us to investigate interactions of AMK with aromatic amino acids under oxidative conditions. In fact, AMKyl residues were found to become attached to tryptophan and tyrosine [2]. The mode of interaction was clarified by identifying the structure of the oxidatively formed adduct of AMK with the tyrosine side chain fragment, 4-ethylphenol [21].
AMK was shown to be attached with its anilinic nitrogen atom (N2) to the fragment in a position corresponding to ring atom 3 of tyrosine. Therefore, AMK may, under moderately oxidative conditions, posttranslationally modify proteins at superficially exposed tyrosine and, perhaps, tryptophan residues (Figure 1). AMKylation of proteins [21] was further discussed with regard to the possible interference with tyrosine phosphorylation by tyrosine kinases, especially tyrosine receptor kinases, in which multiple tyrosine residues in the cytosolic domains are rather easily accessible. For this scenario, interference with cell proliferation was predicted [2]. Recently, AMK was shown to be formed at substantial rates in the human epidermis [30]. In the same study, antiproliferative effects of AMK were observed in HaCaT keratinocytes and in the melanoma cell line SKMEL-188.
The half-effective doses (EC50) were found to be in the range between 10-12 and 10-10 M. However, the half-maximal inhibition [IC50] of [3H] thymidine incorporation and cell growth was not attained at these doses. This difference is reminiscent of the inhibition of nNOS [17], as mentioned above. Moreover, demonstrable effects at very low doses that contrast with the IC50 data indicate that AMK is not acting via a normal saturable receptor. In fact, no receptor or other binding site with association or dissociation constants in the physiological range have been described for AMK. However, these findings on incomplete inhibition would be compatible with the assumption of AMKylation. This possible type of protein modification needs further support or exclusion by direct experimental approaches.
AMK is a bioactive metabolite with several fascinating properties of biological and medical interest. The manifold actions in the fields of anti-inflammatory effects, protection of mitochondria and detoxification of various reactive intermediates would be worth further detailed investigations. The fact that AMK has been studied relatively rarely is mainly due to its poor availability from chemical providers. Another reason for why researchers may have been reluctant to work on AMK is presumably the assumed low concentration of the AMK, which may be regarded as physiologically unimportant. However, the absence of well-detectable amounts in the circulation cannot be taken as an argument for irrelevance. More important than blood levels are the concentrations in the tissues and in organelles.
The recent example of the skin [30] shows that substantial amounts of AMK can be present in a tissue and are measurable. Conditions under which the kynuric pathway of melatonin plays a larger role than normal have to be taken into consideration, too. This may be particularly the case in situations of high-grade inflammation, as exemplified by the strongly elevated AFMK levels in the CSF of meningitis patients. With regard to the relative inertness of AFMK, the subsequent formation of AMK may largely depend on the activities of the converting enzymes, arylamine formamidases and hemoperoxidases. Finally, it seems important to be aware that the concentration of AMK, which is readily consumed by various reactive compounds, is less important than the rate of AMK formation, which may be even close to the rate of consumption. Therefore, it might be promising to not limit a study on AMK levels alone, but rather to determine stable products that can have only been formed from AMK, in particular AMMC and MQA. Moreover, these compounds may also be used as indicators of processes leading to their formation. MQA may be used as an indicator of pseudo-hydroxyl radicals, which largely appear as rather elusive in biomedical studies.
Whether or not AMK may be suitable for pharmacological purposes, remains to be studied. One might think of antiinflammatory actions, reduction of copper toxicity, and antiproliferative effects. In experimental terms, the possibility of protein AMKylation at functionally important tyrosine residues could be of interest. However, in terms of eventual treatments, AMKylation may also be seen as a good reason for a caveat. If AMKylated proteins are formed or appear extracellularly, the possibility of immunogenicity [21] should not be left out of sight.


