Impact Factor : 0.548
- NLM ID: 101723284
- OCoLC: 999826537
- LCCN: 2017202541
Eduardo Tena-Betancourt1,2*, Braulio Hernandez-Godinez3-6, Salvador A Solis-Chavez3,5,6, Yessica Heras-Romero2, Alberto Aranda-Fraustro7 and Alejandra Ibáñez-Contreras3,5,6
Received: August 22, 2017; Published: August 30, 2017
Corresponding author: Eduardo Tena-Betancourt, Coordinator, Animal Facility Services and Experimental Surgery, Facultad Mexicana de Medicina,Universidad La Salle A.C. CDMX 14000, Mexico
DOI: 10.26717/BJSTR.2017.01.000315
This paper describes a case of segmental testicular infarction (STI), a rarely described human condition resulting from partial ischemia of the testis and often resulting from complex, organic, traumatic or metabolic causes, although an idiopathic etiology has been also considered. Being an important human occurrence, STI has not been reported in non-human primates (NHP) and this work is aimed to characterize a fortuitous case of STI in a 20-year-old male rhesus macaque, demonstrated by clinical findings, ultrasonography and histopathology studies. Clinically, the subject presented a significant scrotal wall thickening and in duration of the right testis palpated at its lower pole, while ultrasonography revealed a hypoechoic irregular mass of poorly defined margins. Upon excising this organ, the histological findings demonstrated a well-defined hemorrhagic nodule measuring 1 cm in diameter consistent with segmental testicular infarction, confirmed by further detailed observations disclosing arteriolar wall thickening and a double lumen, indicating post-infarctrecanalization considered an analogous condition seen in human stroke.
Keywords: Macaca mulatta; Testicular Infarction; Ultrasonography; Histopathology
Abbreviations: STI: Segmental Testicular Infarction; NHP: Non-Human Primates; PI: Pulse-Inversion
Segmental testicular infarction (STI) is a rarity, and when diagnosed in humans it occurs as result of partial ischemia [1] or complex organic, traumatic [2] or other metabolic causes [3-6], although an idiopathic etiology has been also considered [7,8]. Other etiologic factors leading to STI have been reported, such as sickle cell anemia [9], vasculitis [10], acute epididymo-orchitis, epididymitis [8,11], torsion of testicular appendage, incarcerated hernia and trauma [1,12]or have included iatrogenic vascular injury [11,13], although sound reports suggests that it may also occur after spontaneous infection or surgery [2]. Another striking mechanism recently studied is cholesterol embolism-associated STI [3], which particularly affects males between their second and fourth decade of life [8]. In our description, a vast literature review showed that no previous reports of STI have been published in animal models such as non-human primates (NHP), and hence the purpose of this paper is to report the diagnosis and characterization of a fortuitous case of STI diagnosed in a 20-year-old male rhesus macaque, who´s lesion strongly resembled are human disease.
The STI case was observed in an aged rhesus macaque (Macaca mulatta) that according to our early breeding records was being regarded as non-productive. Upon palpation during an annual health monitoring, this male disclosed a minor decrease, in duration and hardening of the right testicle at the level of its lower pole without signs of pain. This animal was housed in adherence to Mexican regulations, [14] under harem conditions at the Primate Unit of the Centro de Investigación Proyecto CAMINA para Curar la Parálisis, A.C.located in México City (CDMX), a facility fully accredited by SEMARNAT(Secretaría del Medio Ambiente y Recursos Naturales) under Registry Number DGVSPIMVS-CR-IN-1014-D.F./08.
Under slight anesthesia (2-4 mg/kg IM. of Tiletaminezolazepam, Zoletil® LaboratoriosVirbac, Carros, France) [15], the monkey was studied by subjecting him to scrotal ultrasound at the Division of Radiology and Image of the Hospital Dr. Manuel Gea González, SSA, using an Aloka Ultrasound equipment (©Hitachi Medical Systems Europe),with a high frequency broadband linear transducer (7.5-10 MHz). The subject was positioned in a supine position with both legs slightly spread, while the scrotum placed over a towel to allow its full exposure; the relaxed penis was temporarily held towards the abdominal wall.
The scrotum and its contents were scanned along the longitudinal and transverse axis and both testes were compared. The affected testis was scanned from one extreme to another disclosing regular echo and texture, as well as one heterogeneous abnormality. Finally, under surgical anesthesia (4-6 mg/kg IM. of Tiletamine-zolazepam, Zoletil® Laboratories Virbac, Carros, France) an inguinal incision followed by orchiectomy was executed to biopsy the infarcted and healthy segments of the testis; the segments were further processed for histopathologic evaluation using the HE and Masson’s trichromatic method as described in [16]. The postoperative period was uneventful and supported by the use of antibiotics and analgesic drugs.

Figure 1: Ultrasound image showing an ancient round hypoechoic lesion in the upper pole of the right testis. The enhanced infarcted zone is outlined in a dotted yellow frame.

Figure 2: Right Histopathology micrograph showing normal testicular parenchyma. The lower portion of theImage shows the tunica albuginea. There are seminiferous tubules with active spermatogenesis. HE 4 X.
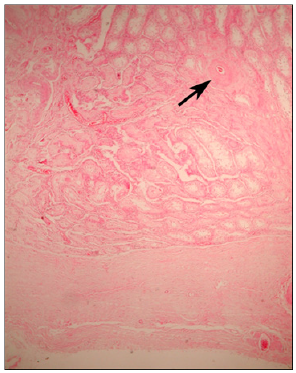
Figure 3: Left Histopathology micrograph showing an enlarged tunica albuginea (lower arrow) and atrophic Seminiferous tubules. A blood vessel (upper arrow) is seen coursing through a fibrous lesion consistent with an old infarct. HE 4 X.
The ultrasonography of the affected (right) testis revealed a markedly hypoechoic heterogeneous mass characterized by poorly irregular margins, (Figure 1) while the left testis was regarded as normal showing homogenous medium-level echoes; evidence of a healed old scar (not shown) was seen at the left inguinal canal probably associated to early fighting among cage mates. Macroscopically the excised testis presented a fibrous capsule, while the smooth tunicaalbuginea lacked any superficial nodules; during palpation a degree of in duration and thickening at the lower pole was also noted. The comparison with the contra lateral testis revealed a normal body.
The histopathologic examination of the indurate site (right testis) demonstrated a well-defined hemorrhagic nodule, resting closely adherent to a thickened tunica albuginea without erosion and showing extensive infarction, necrosis and atrophic seminiferous tubules (Figure 2). The non-affected tissue displayed a normal parenchyma and active spermatogenesis was observed at the seminiferous tubules (Figure 3). The rather distinct nodule was consistent with segmental testicular infarction showing arteriolar wall thickening and a double lumen, indicating postinfarctrecanalization (Figure 4). In addition, parenchymal replacement by proliferative and reparativecicatricial fibrous tissue was also present.

Figure 4: Histopathology micrograph showing arteriolar wall thickening and a double lumen, indicating post-infarct recanalization (arrow). Also, parenchymal replacement by proliferative-reparative cicatricial fibrous tissue is also observed. HE 40 X.
In this work, based on our initial clinical findings and the supporting ultrasonography and histopathologic studies, a rarity analogous to human STI was diagnosed in a mature rhesus acaque. This kind of pathology is seldom reported in young-adults, but when it is diagnosed in aged subjects is the result of very diverse etiology [1-3], and considering than more than 70% of patients are senile it may occur concomitantly to predisposing conditions such as sickle cell disease [9], epididymo-orchitis, polycythemia, angiitis and testicular damage following surgery or trauma [12]. STI can also be associated to hereditary conditions such as isolated protein S deficiency leading to vascular complications such as venous and arterial thrombosis [6].
Given its painful clinical impact and the potential effect on fertility, recent approaches have highlighted various qualitative/ quantitative methods for pulse-inversion (PI) imaging in a rabbit model of acute testicular ischemia [17], while murine models have been used to study the recanalization of arterial thrombosis [18] and to test the protective effect of erythropoietin in experimental torsion-detorsion providing further insight on this theme [19].
According to a previous report [20], although rarely observed STI is commonly associated to severe pain, while the ultrasound image sare largely related to the age and severity of the lesion. They had indicated also that during the early stages of STI, the lesions generally fail to demonstrate specific echogenic changes, but when advanced infarcted sections appear, they consist of well-defined rounded hypoechoic shapes in the first plane presenting welldefined and roughly triangular shaped forms in the perpendicular plane, tending to decrease in size when they calcified amplifying thus the echogenicity of the testis by the ensuing fibrosis.
In this case, it is considered that the fibroplasias of the tunica in tima without mineralization of the layer of the testicular artery was a significant anomaly, supported by human analogous findings, [11] while the absence of acute scrotum (pain and/or swelling), often mistaken for testicular torsion or detorsion, or epididymoorchitis was present probably associated to an old lesion, given that the monkey appeared healthy and without signs of any discomfort. This assumption is supported by the fact that the initial signs of this condition were determined by palpation of the testes when the routinely monitoring program was applied to the colony. Further characterization of STI could be supported also by the presence of a large-round ancient hypoechoic lesion seen at the inferior pole of the right testicle [7,11]; we assumed that either the level of echogenicity or discreteness of the lesion on sonography appears to relate to the old age of the infarct, given that the remaining tissue and the left testis showed normal echogenicity.
The microscopic findings depicted an enlarged tunica albuginea, scarce parenchyma and also atrophicseminiferous tubules with loss of active germ cells, while adistinct blood vessel was seen coursing through out the fibrotic lesion consistent with an old infarct. The infarcted area was shown focally amid theseminiferous tubules, and the parenchymal replacement was observed as proliferativereparativecicatricial fibrous tissue. Under higher magnification, arteriolar wall thickening and a double lumen was noted indicating post-infarct recanalization.
It is then postulated that the “organization/recanalization” of thrombus described by early pathologists [10] could led to their establishment of some degree of vascular flow in veins and arteries, and that according to previous work it could be modulated for therapeutic purposes [18]. In this case recanalization was the hallmark of these histologic findings; nevertheless, it is unclear when this event might took place explaining to a degree the lack of noticeable pain or discomfort in this animal. Being recanalization a complex event, showing up to reach 52.7% of rates after one week post-stroke, is that it has been described that the early recanalization within the first six hours have been positively correlated to a good clinical outcome, of patients that are suffering acuteischemic stroke [21]. Not having an objective explanation for this manifestation of this aged monkey, it can be safely assume that such process might have taken place earlier in his life and slowly evolved unnoticed.
On the other hand, it has been previously described how systemic degenerative vascular changes can be are seen in NHP, and occur analogously to human beings during advanced age [15]. Ischemic episodes have been associated to hyaline and fibrous degeneration of the arteries leading to blood flow change and thrombi formation [10]; consequently, the formation of clots may result in severe stroke insults to different organs [6]. However, in the majority of cases the testicular infarction affects testis globally and only in rare occasions a single segmental affectation is reported as seen in this case [5].
We estimate that lacking specific tumor markers consistent with additional studies, the reported condition of this monkey can be safely considered of idiopathic etiology. Abounding, it has been postulated also that disorders characterized by ischemia/ reperfusion (I/R), can be analogously compared to a series of events occurring in other organs under complex molecular mechanisms [4]; hence, we estimate that the progressive pathology seen in this monkey is responsible for the impending testicular atrophy, decreased blood flow and impaired spermatogenesis that occurred. No objective explanation exists for the apparent absence of pain in this monkey other than an early occurrence, since the cardinal signs of pain (vocalization, mutilation or lack of appetite) were absent and the animal displayed a normal social and grooming behavior.
It expected that the characterization of STI in advanced aged macaques should enhance the clinical awareness among scientists and veterinarians about the presence of unnoticed occasional conditions, providing a better understanding of the critical mechanisms associated with exceptional rarities mimicking human conditions. This report can also contribute to advice clinicians and researchers using NHP to execute comprehensive clinical assessments of their monkey colonies, assisted by non-invasive diagnostic to olsat different ontogenetic stages for the early detection of diseases.


