Impact Factor : 0.548
- NLM ID: 101723284
- OCoLC: 999826537
- LCCN: 2017202541
Magdy K Khalil2, Mohamed H Afifi1, Essam A Ismael1, Nirmen A Fayed2 and Nahla K Gaballa2*
Received: June 17, 2017; Published: July 03, 2017
Corresponding author: Nahla Kamel Gaballa, Department of Anaesthiology, Menoufiya University, Shibin El Kom, Egypt
DOI: 10.26717/BJSTR.2017.01.000163
Objective: To study the perioperative effect of Dexmedetomidine on indirect calorimetry in recipients of adult living donor liver transplantation.
Background: Dexmedetomidine is newly used in liver transplant.
Material and Methods: forty patients were assigned randomly to get Dexmedetomidine (Dex) beginning at 0.5 μg/kg/h(0.2-0.7μg/ kg/h) or placebo (C) Anesthesia was guided by Enotropy (40-60) with Desflurane. Indirect calorimetry parameters, arterial blood gasses, hemodynamics were taken at base line, dissection, an hepatic, reperfusion and 3 hours after end of operation. Transoesophageal Doppler was used for fluid optimization.
Results: No significant difference was noticed in either group regarding demographic data, and operative data , Dexmedetomidine affects oxygen consumption(Vo2) and carbon dioxide production (Vco2) significantly at at reperfusion and 3hours postreperfusion; Mean ±SD for VO2 (273.1±36.1/227.1±82), (252.4 ±39/189.5±75) ml/min/m2 and P=0.019,0.005 for control /DEX groups at reperfusion and intensive care unit(I.C.U)respectively , VCO2(238.4±18/181.5±72), (210.5±27/159.0±63) ±78.77 ml/min/m2:p =0.002, 0.001 also for control /DEX respectively. Respiratory Quotient RQ and Energy expenditure EE were significant in C versus DEX at reperfusion and in intensive care, for RQ in C/DEX (0.7 9±0.08/0.73±0.08),(0.75±0.8/0.70±0.10) with p=0.014,0.001. EE for C/DEX (1358.5±152.4/1231.5±84.9), (1264.3±147.0/1068.1±122.9) and P=0.000, 0.001 sequentially. Haemodynamics were insignificantly decreased in DEX versus C .DEX decreased the utilization of desflurane in DEX/C (179±59/221±5 p=0.016) ml, fentanyl (988±202/1241±390p=0.041) μg, and insignificantly increased amounts of fluids needed and the in otropic support. pH and Paco2,HCO3 were insignificantly affected in both groups, except in reperfusion, where pH and HCO3 decreased and Paco2 increased significantly. The I.C.U. stay was comparable.
Conclusion: dexmedetomidine has a depressing effect on indirect calorimetry especially in reperfusion.
Keywords: Dexmedetomidine; Indirect Calorimetry; Liver Transplant
Abbreviations: Dex: Dexmedetomidine; ICU: Intensive Care Unit; TED: Transesophageal Doppler; CO: Cardiac Output; SVR: Systemic Vascular Resistance; ROTEM: Rotational Thromboelastography; BL: Baseline; DISS: During Dissection; ANH: Anhepatic; REP: Reperfusion Phases; SPSS: Statistical Package for Social Science; HR: Heart Rate; NLI: National Liver Institute; MAC: Minimal Alveolar Concentration.
As clinicians in operations with significant phases of fluctuations, we are normally concerned about hemodynamic solidity. While anesthesia and intubation are the principal components animating the neuronal and endocrinal responses [1-3]. Hepatectomy phase have significant blood loss, anhepatic phase is characterized by accumulation of acid metabolites and unclamping, is marked by vasodilatation of the splanchnic bed creating an insufficient preload and critical hemodynamic instability particularly in chirrotic patients with hyperdynamic circulation and fragile peripheral resistance [4].
Dexmedetomidine; a novel sedative profoundly selective α2 anesthetic agent may provide more hemodynamic stability [5,6] At low doses the dominant action of α2 -adrenoreceptor agonist activation is a reduction in sympathetic tone, The net effect of dexmedetomidine action is a significant reduction in circulating catecholamine with a slight decrease in blood pressure and a modest reduction in heart rate [7]. When dexmedetomidine is administered as a nonstop infusion, it is associated with an expected and stable hemodynamic reaction [8].
This study was approved by the institutional review board (00003413 FWA0000227– January 1st, 2013). pan African trial PACTR201402000776978. After providing written informed consent, 40 consecutive adult recipients (age 18-50 yr) , MELD (14- 20), scheduled for LDLT were enrolled, Exclusion criteria included recipients with evidence of cardiac decompensation ,severe hemodynamic instability, chronic obstructive pulmonary disease; pulmonary dysfunction (PaO2 less than 60 mmHg) and those on pre-operative beta blockers were excluded and increased oxygen requirements (FiO 2 >0.8) Patients were randomly allocated via a computer generated random number table into: control group( C n = 20) that received a placebo, and Dexmedetomidine group (DEX; n = 20) that received continuous infusion starting at 0.5 μg kg/h (0.2-0.7) (Precedex; Hospira, Inc., Lake Forest, IL 60045 USA). Dexmedetomidine 200 μg/ vial were diluted to a concentration of 4 μg /mL, and infusion started after the induction of anesthesia and continued till the end of surgery. The infusion in both groups had to be decreased or stopped with hemodynamic instability (heart rate <60 beats/min and/or mean blood pressure <60 mmHg), which did not respond to usual management including atropine or vasopressor drugs. All patients received noradrenaline support when indicated. The anesthesia team was blind to the cases that received Dexmedetomidine. After standard monitoring was in place, anesthesia was induced using propofol 2 mg/kg and rocuronium 1.2 mg/kg to facilitate endotracheal intubation. Anesthesia was maintained via desflurane in O2/air mixture (FIO2 = 0.4) and fentanyl as needed, anesthesia level was monitored by keeping spectral entropy (GE Healthcare, Helsinki, Finland) between( 40- 60). Rocuronium was given via neuromuscular blockade monitoring via accleromyography, NMT module, Dragger, USA), in intermittent boluses according to the clinical needs. Normothermia was achieved with a forced-air warming device. An arterial line was placed in the left radial artery, and a central line was deployed in the right internal jugular.
Settings were adjusted to keep Paco2 within normal, transesophageal Doppler (TED) Cardio QTM; Deltex Medical, Chichester, UK was used for fluids adjustment in otropic administration, and haemodynamic monitoring; cardiac output (CO), systemic vascular resistance (SVR), protocol of TED [9], base line data were collected continuously in patients operative sheet, blood products were given when clinically indicated guided by rotational thromboelastography (ROTEM) (Pentapharm GmbH, Munich, Germany). no patient received bicarbonate during the study. Patients in need for support received noradrenaline. M-COVX™ (Datex-Ohmeda S/5 Avance workstation™, GE Healthcare, Helsinki, Finland) Metabolic module was used in our operating room and ICU. It consists of a gas analyzer and a spirometer unit and displays oxygen consumption (VO2) and carbon dioxide production (VCO2). Partial pressures of O2 and CO2 are measured by the rapid paramagnetic analyzer and infrared analyzer, respectively and inspired tidal volume is measured using a pneumotachograph.
RQ can be calculated from the Halden-equation, Vco2 subsequently calculated from RQ as Vo2=Vco2/RQ, and EE =3.58xVo2+1.44xVco2-32.4. Reliability of displayed value of resting energy expenditure REE was assessed after considering the concomitant readings of RQ whose normal values range from 0.69 to 0.98).In each patient hemodynamics, a rterial blood gases(ABG,VO2, VCO2, RQ, and EE were measured at baseline (BL) ,during dissection(DISS), anhepatic (ANH) and reperfusion (REP) phases , and as a protocol all recipients were discharged to I.C.U on mechanical ventilation where the last measure were taken(I.C.U).
Data were collected and entered to the computer using (SPSS) Statistical Package for Social Science program for statistical analysis, version 21. Data were entered as numerical or categorical, as appropriate. In the present study α was set to 0.05, and maximum accepted = 20% with a minimum power of the study of 80%. A sample size of 20 per group would be required in each group to reveal a significant difference in the primary outcome of this RCT which is oxygen consumption (VO2) to detect a mean difference of 20 ml/kg/min and standard deviation of 19.2 ml/kg/min and 15.9 ml/kg/min in Dexmedetomedine and control groups respectively. Calculation of sample size was done using (IBM SPSS Sample power) software and was also confirmed using Length Java Applets for Power and Sample Size [Computer software].
Correction of p value for multiple testing was set p to 0.01 to detect significance (Bonforroni correction of multiple comparisons). So, in the present study an alpha level was set to 1% with a significance level of 99%, and a beta error accepted up to 20% with a power of study of 80%.
Two types of statistics were done: Descriptive statistics: Quantitative data were shown as mean, SD, and range. Qualitative data were expressed as frequency and percent at 95% confidence interval (95% CI). Analytical statistics: Chi-square test (X2) was used to measure association between qualitative variables. Student t- test and Mann Whitney U test were done to compare means and SD of 2 sets of quantitative data as appropriate. Paired sample t-test and Wilcoxon Signed Ranks Test were done to assess the follow up of quantitative data as appropriate. The results of comparing the correlation between two continuous variables were indicated by the correlation coefficient (r) using correlation analysis P (probability) value considered to be of statistical significance if it is less than 0.05.
Age, sex, body mass index (BMI), and MELD score were comparable (P>0.05) between DEX (n=20) and C (n=20) groups .Demographic, clinical characteristics and main indications for liver transplantation (mainly Hepatocellular carcinoma) are presented in (Table 1) that demonstrates no significant differences between the control and DEX groups. Operative data of the included patients as in (Table 1) demonstrating operative data in details, blood products and fluids transfusions in both groups.
Table 1: Demographic clinical characteristics and indications for liver transplantation with important intra operative data.
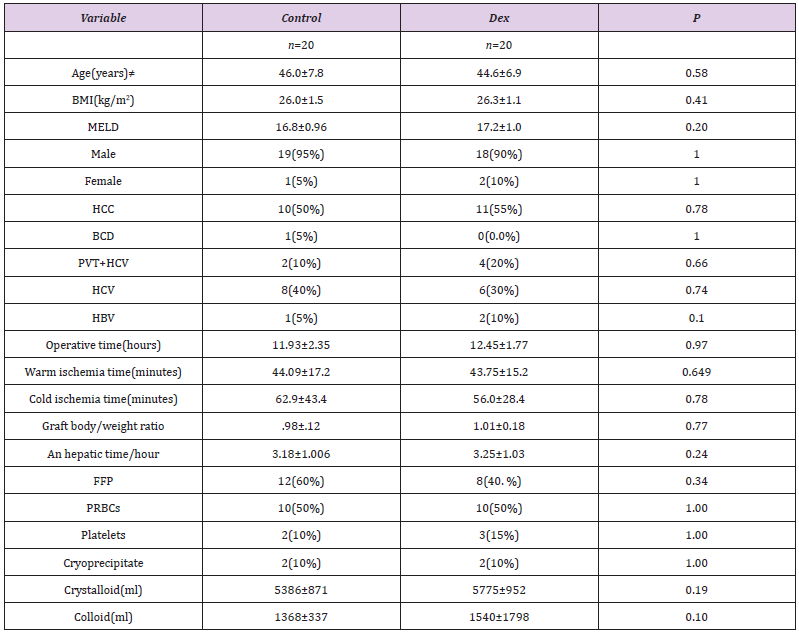
Mean ± SD fort-test ≠ for Mann Whitney test, data presented as percent age, tested by Cramer’s VP Value<0.05 statistically significant. BMI: Body Mass Index; MELD: Model for End Stage Liver Disease; HCV: Hepatitis C Virus; HCC: Hepato Cellular Carcinoma; BCS: Bud Chiarri Syndrome; HBV: Hepatitis B virus; PVT: Portal Vein Thrombosis.
Table 2: study the oxygen consumption VO2, carbon dioxide production VCO2, respiratory quitient RQ and energy expenditure EE values all over the follow up in Dexmedetomidine (DEX) and control groups.
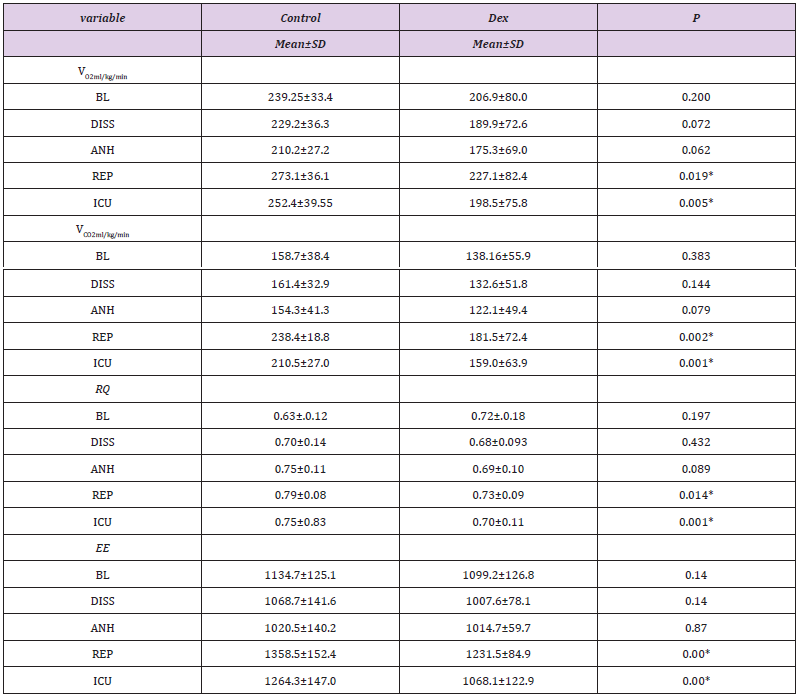
Mean ± SD fort-test ≠ for Mann Whitney test. P-value <0.05* statistically significant. BL: Baseline; DISS: Dissection; ANH: An hepatic; REP: Reperfusion; ICU: Intensive care.
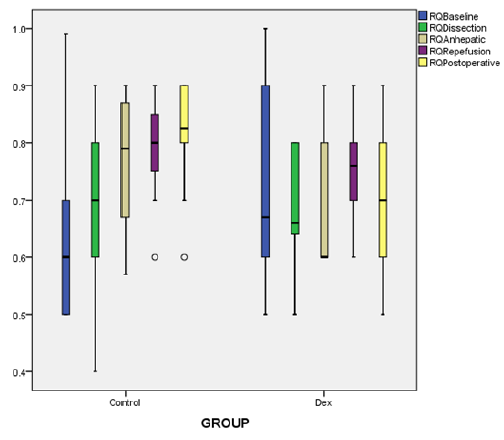
Figure 1: Box and whiskers graph of RQ;respiratort quotient showing; median values (line within the box) and inter-quartile range of RQ in the two studied groups; C group (control group) and DEX .p<0.005 at reperfusion and postoperatively.
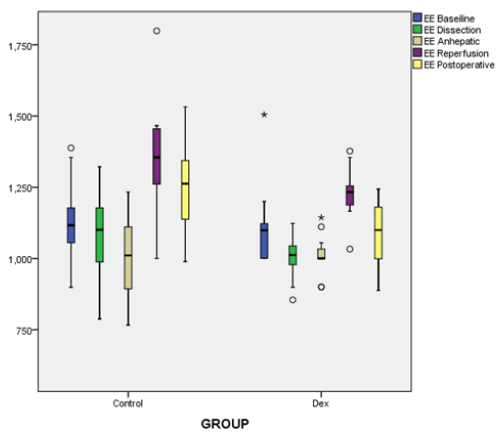
Figure 2: Box and whiskers graph of EE; energy expenditure showing; median values (line within the box) and inter-quartile range of EE in the two studied groups; C group (control group) and DEX .p<0.005 at reperfusion and postoperatively.
Continuous indirect calorimetry monitoring revealed insignificant changes in VO2 and VCO2 at ( BL, dissection, an hepatic phases) but significantly decreased at reperfusion and in I.C.U. in DEX vs C groups (Table 2), with a trend of decrease in DEX group EE and RQ showed the same fluctuations consistent with oxygen consumption and carbon dioxide production (Table 2), (Figures 1 & 2). As regarding hemodynamics, heart rate (HR) showed slight decrease in all phases in DEX group but in comparison to C significant changes were recorded at BL, anhepatic, I.C.U phases. Mean arterial pressure (MAP) systemic vascular resistance (SVR), corrected flow time (FTc) were maintained in both groups during different measuring points, and that was the same with cardiac output measurements (CO), (Table 3).
Table 3: Follow-up of hemodynamic parameters between DEX and Control groups.
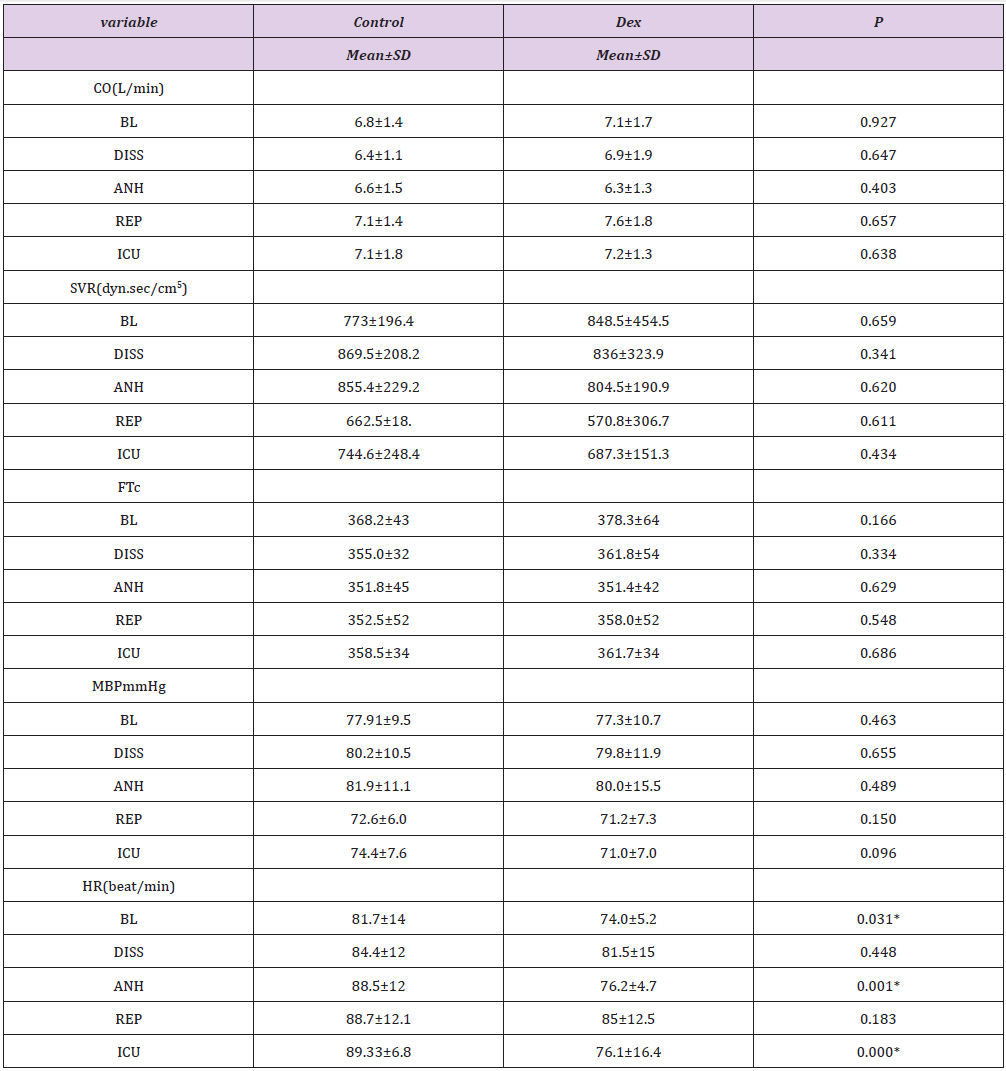
HR: Heart Rate; MAP: Mean Arterial Pressure; FTc: Corrected Flow time; CO: Cardiac Output SVR: Systemic Vascular Resistance; Data were presented as mean ± SD, tested by student t-test, P-value<0.05* statistically significant. BL: Baseline; DISS: Dissection; ANH: An hepatic: REP: Reperfusion; ICU: Intensive care.
Dex was delivered for 11.93±2.3hours with significant decrease in both Fentanyl (1241±390/988±202) μg p=0.041and Desflurane consumption (221±52/179± 59) ml p=0.016in C vs DEX respectively, and non significant Noradrenaline in C/DEX (9.4±1.5/10.3±1.0), P= 0.076. Regarding the ABG follow up during the operation there were no significant changes all over all times of measurements except at reperfusion where pH, Hco3 showed significant decrease in DEX than C and Paco2 showed increase with general tendency of ABG parameters towards acidosis especially in anhepatic and reperfusion stage (Table 4). ICU stay was comparable too, DEX/C (6.95 ±2.53/6.70 ±1.36), P =0.683.
Table 4: Followup of HCO3, PCO2 and pH parameters between DEX and Control groups.
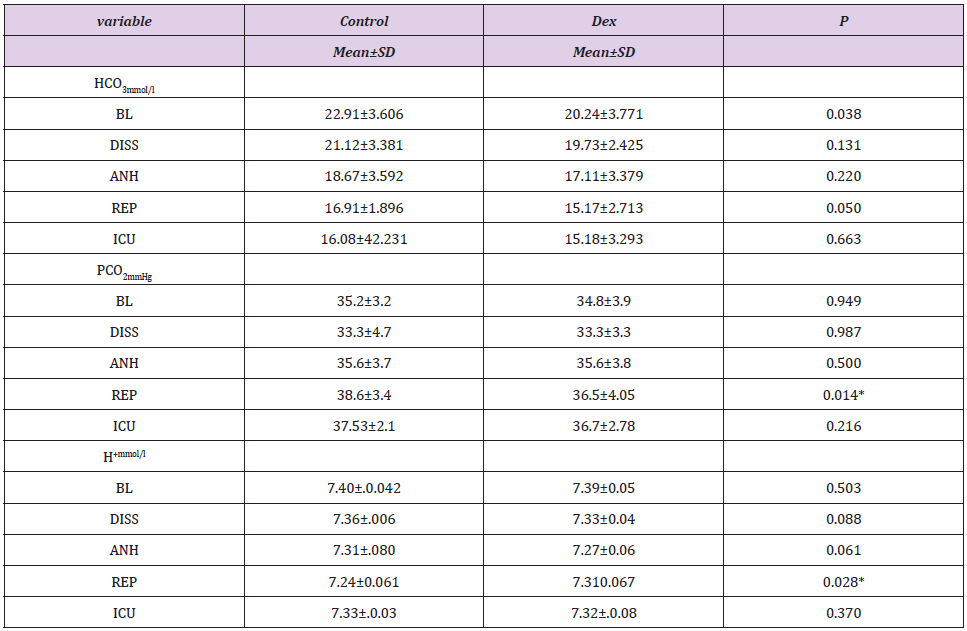
Data were presented as mean ± SD, tested by student t-test, P value<0.05 statistically significant. BL: Baseline; DISS: Dissection; ANH: Anhepatic; REP: Reperfusion, ICU: Intensive Care.
The vital finding in the review in National Liver Institute ( NLI) uncovered that dexmedetomidine diminished metabolic screen information ( Vo2, Vco2, RQ and EE) and the decline was critical in control versus dexmedetomidine group, and that happened clearly after reperfusion and couple of hours after later in intensive care, these progressions were steady with rebuilding of blood to the transplanted liver and were related with other settled metabolic changes incorporating the reduction in Hco3 and pH because of metabolites in the event of unclamping. This likewise was demonstrated by Walash et al. [10], who watched critical increment in VO2, VCO2 after reperfusion in uncomplicated transplant. The study done by Sayed E et al. [11] dexmedetomidine infusion during LDLT, there were significant decrease in VO2, VCO2 in DEX group than control in anhepatic, reperfusin and end of surgery in liver transplant.
In the study done by Tarrtonen et al [12] before operation dexmedetomidine and Clonidine caused greater decrease in VCO2, VO2 and EE than placebo and there were no difference in RQ between groups, the maximum decrease in VO2 of dexmedetomidine group was 7%. During the operation there were no differences in metabolic variables between groups, though VO2 and EE were lower in dexmedetomidine and clonidine groups than placebo. After operation VO2 was lower 17% in dexmedetomidine and Clonidine groups than placebo, also VCO2 and EE were lower (17%, 19%) respectively in patients.
Dexmedetomidine diminishes central sympathetic outflow and adjust intraoperative cardiovascular and endocrine reactions positively to surgical stimulus and laryngoscopy [13].The diminishment in tachycardia, hypertension, thoughtful movement, plasma catecholamine fixation and its impact on sedation results in diminished entire body metabolism subsequently adds the benefit up to body oxygen utilization [14,15] It has been exhibited that successful absence of pain in the postoperative period can diminish VO2 by up to 7-8% [16].
The heart rate was seen to be diminished at all measuring focuses with noteworthy decline in DEX versus C in baseline, a hepatic and I.C.U. stages where the impact of dexmedetomidine on HR was not meddled by the surgical incitement or blood loss. The baroceptor reflex is all around saved in patients who get dexmedetomidine, and the reflex heart rate reaction to a presser stimulus is enlarged other than the diminishing in noradrenaline discharge. Theseoutcomes outline that the cardiovascular reaction is evoked for the most part by declines in sympathetic outflow. Dexmedetomidine could bring about cardiovascular sorrow; bradycardia and hypotension [16] Despite what might be expected Unlugenc et al. [17] gave 1 mic/ kg dosage of dexmedetomidine inside 10 minutes of induction and they found a similar readings in HR inside 10 minutes, though HR and mean blood vessel pressure (MAP) were like qualities found in the placebo group. Alpha 2-agonists diminish the perioperative anesthetic and pain relieving prerequisites, however in the review by [18-20] there were no huge distinction between gatherings.
In our review, we utilized entropy to quantify the profundity of anesthesia. Enotropy showing reaction enotropy in charge of opioid need and state enotropy in charge of anesthesia required, permits keeping up anesthesia level between (40-60), to dodge the mistakes from relying upon hemodynamics to change inhalational anesthesia [21].
The requirement for more Fentanyl in control gathering of our review might be right due to the sympatholytic impact of dexmedetomidine intraoperatively and furthermore because of the time slack in ICU till extubation as a protocol in our organization thus the DEX group got the benefit of being sedated by dexmedetomidine. Aho et al. [22] indicated 25% diminishments of support focuses of isoflurane in patients who got dexmedetomidine. Another review [7] found 35% to 50% decreases of isoflurane prerequisites in patients treated with either low or high dosages of dexmedetomidine and isoflurane without premedication. The utilization of opioids (e.g.fentanyl) decreases minimal alveolar concentration (MAC) of sevoflurane [23] significantly, adding opiods to dexmedetomidine can potentiate its saving impact on inhalational utilization All are steady with our outcomes ,but on the other side a review [18] which uncovered insignificant distinction in opiods or i.v. analgesic necessities.
In a review by Basar et al. [24] watched that a solitary measurements of dexmedetomidine given before enlistment of anesthesia diminished thiopental prerequisites without genuine hemodynamic impacts or any impact on recuperation time. Correspondingly Khan et al. [25] watched additionally that dexmedetomidine diminished isoflurane necessities as an advantage of being calmed by the medication. The fluid volume required during the intraoperative period to maintain a strategic distance from hypotension was insignificantly higher in the dexmedetomidine group, a reaction that might be negative in hypovolumic patients with decreased left ventricular capacity [26]. This effect might be outweighed, however, by the diuretic effects of α2-adrenoceptor agonists, whose mechanisms may include attenuation of the secretion or effect of antidiuretic hormone, inhibition of renin, or release of natriuretic peptide [27].
Similarlyby Ralph, The fluid volume required during the intra operative period to maintain a strategic distance from hypotension was irrelevantly higher in the dexmedetomidine group [28]. In our review we recorded more colloids (1540±1798/1368±337) ml, crystalloids (5775±952/5386±871) ml and noradrenaline (10±1/9.4±1.5)mg in DEX/C groups non critical statistically, yet may add to stability of mean blood pressure weight as dexmedetomidine has a mild vasodilating impact on SVR and although we have fluctuations in hemodynamics ,those were less recognizable and statistically insignificant between groups.
Dexmedetomidine have a significant depressing effect on metabolim in the form of Vo2, Vo2, EE, which may be used as a method of protection from reperfusion injury (undergoing further study), and even in major hemodynamic fluctuations dexmedetomidine can be used with close monitoring, one of our draw backs that we could not assess the suitable dose for hepatic patients, we believe this will need further study in surgeries of less hemodynamic fluctuations.
Everyone who helped to accomplish the work perfectly, patients and nursing staff. We thank DR Haydizakari for support and nursing staff.
This research did not receive any specific grant from funding agencies in the public, commercial.
All relevant data are within the paper and its Supporting Information files.
Magdy kamal khalil, Tanta, Egypt, Tel: 01001466228; Email: magdykhalil20@yahoo.com. magdykhalil10@gmail.com.


