Introduction
The hazards of tobacco consumption are clearly established. In France, where some 13 million people, 30% of the population, are tobacco users; there are 73 000 tobacco-related deaths a year [1-2]. One in two smokers who do not give up the habit die from a disease directly linked to tobacco use [3]. Women smokers, who account for 24% of the overall female population, are particularly at risk [4]. Smoking in combination with the taking of oral contraceptives increases the risk of cardiovascular events and in pregnant women can lead to complications such as preterm birth, delayed growth, and sudden infant death [5]. Against this background, are electronic cigarettes a viable alternative to traditional cigarettes or does the limited evidence make them an unknown risk? Following aggressive marketing campaigns, sales of e-cigarettes have increased exponentially [6,7]. Although additional data are becoming available, there are still areas of uncertainty. The French law n°2014-344 of 17 March 2014 prohibits the sale of e-cigarettes to minors [8,9]. This is a wise precautionary ruling since we do not know the long-term effects of vaping on health, which could well be greater for younger users. It is a measure that is also consistent with the decision not to let the e-cigarette become a “gateway” to smoking for those not allowed to buy tobacco. The French Ministry of Health also stated that it wished to restrict the use of e-cigarettes in public places [10]. In 2011, the ANSM (French Health Products Safety Agency) had already recommended against the use of e-cigarettes [11]. In 2013, the OFT (French Office for Smoking Prevention) and the DGS (Directorate General for Health) advised to regulate sale of the products rather than to ban them and to carry out assessment studies [3]. By a decision of 8 October 2013, the European Parliament ruled that the e-cigarette should not be considered as a medicine [12]. The HCSP (French Public Health Council), in response to a request by the MILDECA (Inter-ministerial Mission for the Fight against Drugs and Addictive Behaviour), performed a literature review to assess the risk-benefit ratio of e-cigarette use and its possible consequences for the general population [13]. The HCSP concluded that the risk-benefit analysis depends on the type of situation: “For smokers wishing to quit, using e-cigarettes is a means of reducing risk if it results in cessation; for tobacco smokers who also use e-cigarettes there are many unanswered questions and we need further studies before giving scientifically sound advice” [13]. The main cautionary notes expressed by the HCSP concern the risk of creating nicotine dependence in the young and subsequent resort to cigarette smoking, and the hazard of e-cigarette use by pregnant women [13]. In light of this, the HCSP issued the following recommendations: “Periodically monitor the extent and modes of e-cigarette consumption; explain and heighten consumers’ awareness of the risks involved; inform that the sale of e-cigarettes to minors is prohibited and that vaping constitutes a risk for pregnant women; periodically check the presence and concentration of nicotine in e-liquids; and introduce measures to counter attempts to normalize the use of nicotine, whether by smoking or inhaling”[13]. These recommendations are in line with those published by the WHO at the end of the summer 2014 [14].
The present work is based on the first three recommendations of the HCSP and on a survey of pregnant women and women taking combined oral contraceptives (COCs) conducted in a pharmacy setting. The survey was carried out by students of the Faculty of Pharmacy (Auvergne, France) during their work experience. The dual aim was to allow the students to apply the knowledge acquired during their initial training cycle and to collect data for a research project on e-cigarette use that would make them more aware of their future public health role. Assessment of the perception and knowledge of e-cigarettes and of their current and potential use by this targeted at-risk population was intended to provide information that would help in future cessation programs for those women wishing to quit.
Methods
Ethics Statement
a) This submission was reviewed by the independent medical ethics committee (Comitéde Protection des Personnes, CPP Sud Est 6, Clermont-Ferrand, France) which has determined that this submission does not constitute human research subjects as defined by French regulations (Article L.1221-1-1 and Article R. 1121-3) and has required IRB approval (IRB00008526).
b) This study was deemed as non-human subjects research, which is similar to an exemption. The study was an anonymous questionnaire-based survey. As a result, regulations for written consent are not applicable and an oral non opposition consent for participation was required from participants. Prior to the participation in the study, participants were asked to reply to an anonymous questionnaire of 68 items about perception and use of e-cigarette. Each participant had the opportunity to decline participation. When they were given oral consent to take part, the women were asked to reply to the anonymous questionnaire.
Recruitment and Sample
The recruitment was made by fourth year pharmacy students during a work placement from 9 to 13 December 2014, in the four departments (Puy de Dôme, Allier, Cantal and Haute Loire) of Auvergne, France. The target population comprised women of childbearing age who had come to the pharmacy to renew a prescription for COCs, and expectant mothers. Women who had poor command of written and/or spoken French were excluded from the study.
Survey Measures
After giving oral consent to take part, the women were asked to reply to an anonymous questionnaire of 68 items. The replies were divided into four main categories.
1. Main socio-demographic characteristics: department and commune of residence, population density of place of residence, level of education, socio economic category, and socioeconomic deprivation as assessed by the EPICES (Evaluation of the Deprivation and Inequalities of Health in Healthcare Centers) score [15]. Participants were divided into two groups according to their deprivation status (non-deprived: EPICES score<30.17; deprived: EPICES score ≥ 30.17) [15].
2. Smoking status (smoker or non-smoker, mini-Fagerström score).
3. Perception and knowledge of e-cigarettes.
4. Details of experimentation with and use of e-cigarettes.
Statistical Analysis
Statistical analysis was performed using Stata software, version 13(Stata Corp, College Station, TX, U.S.) and R software (cran.r-project.org). The tests were two-sided, with a type I error set at α=0.05. Characteristics were presented as the number of subjects and associated percentages for categorical parameters. The negative perception of e-cigarettes was assumed on the basis of positive replies to three questions concerning the awareness of related hazards, risk of dependence and passive vaping. This ordinal variable was considered as a quantitative parameter expressed as mean ± standard deviation. Assumption of normality was verified using Shapiro-Wilk test. Comparisons of the score between independent groups were performed by a nova or using Kruskal-Wallis test if conditions of a nova were not meet (homoscedasticity analysed by Bartlett’s test) followed when p<0.05, by an appropriate post-hoc test, i.e., Tukey-Kramer test or Dunn’s test, respectively. Qualitative data were compared between independent groups by Chi2 test or Fisher’s exact test, if necessary, followed when appropriate by Marascuillo procedure for multiple comparisons. Finally, a multidimensional approach completed these analyses using multiple correspondence analysis to establish relations between categorical variables in the light of previous results of univariate analysis and according to literature.
Results
Rate of Participation
Overall, 67 % (63/94) of the fourth-year students took part in the survey. Of the remaining 33% (31/94), 28 % (26/94) gave a reason for not handing in a completed questionnaire. Among these, four were repeaters and had already validated their work placement; four others were refused permission to take part in the survey by their placement supervisor (the sale of e-cigarettes is prohibited in pharmacies in France); and eighteen encountered no woman fulfilling the inclusion criteria during the period of the survey. Only 5 % (5/94) gave no reason for not carrying out the task. Of the 178 women who were invited to complete the questionnaire, 131 replied favorably, which represents a participation rate of 74 %. The main reason given for not taking part was a lack of time. Of the 131 questionnaires taken in, 7 were excluded from the survey because certain questions concerning socio-demographic characteristics and/or the EPICES score and/or experience of e-cigarettes had not been answered. A total number of 124 questionnaires were therefore included in the descriptive statistical analysis.
Socio-Demographic Characteristics of the Population Studied
The study population was made up of 18.5 % (23) pregnant women and 81.5 % (101) women taking COCs. Most were younger than 35 years old (78%, 96), with a mean age of 29.9 years (SD: 7.7). More than half were living with a partner (63%), in 42 % of cases (51) in an urban or semi-urban area, mainly in Puy de Dôme (59%). More than 62% (78) were in employment, including 12% in management (15), 26% in intermediate occupations (33), with a predominance of pharmaceutical assistants, and 26% salaried employees (32). 61% (76) had a higher education qualification. 15% (18) had an EPICES score ≥ 30.17, an indicator of material and social deprivation.
Use of Tobacco and/or E-Cigarettes
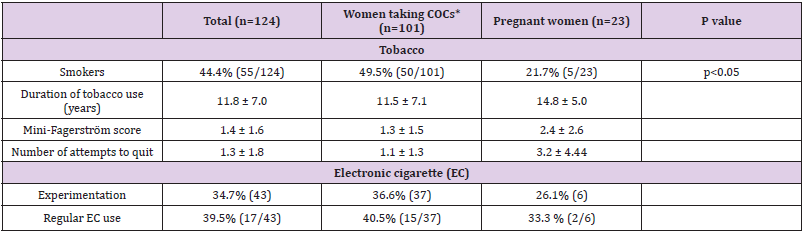
Use of tobacco of the women questioned, 44 % (55) were regular daily smokers, including 9% (11) who were trying to quit (Table 1). On average, they had been smoking 11 cigarettes a day for 12 years, with the first cigarette being smoked 2 hours after waking (Table 1). 22% of the women (12) had low to moderate nicotine dependence, as assessed by the Fagerström test(Score equal to or greater than 3). More than half (53 %, 29) had tried once or more to quit smoking, as against 47 % (26) who had never made the attempt. Of the 56% (69) who described themselves as non-smokers, 44 % (55) had never smoked regularly and 11% (14) were ex-smokers.
Table 1: Tobacco use and electronic-cigarette experimentation and regular use.
Note: COCs*: combined oral contraceptives
Experimentation with and/or Use of E-Cigarettes
All of the women in the survey knew about e-cigarettes, in most cases (72%, 89) through the media, otherwise by word of mouth (47%, 58), encounters in the street (42%, 52) and internet (17%, 21). Some had tried an e-cigarette and certain continue to use them.
E-cigarette Ever Use: One in three (43) of the women taking part had tried an e-cigarette, including 37% (37) of those who were taking COCs and 26% (6) of those who were pregnant (Table 1). Among the non-smokers, 14.5% (10) had also tried. A majority of the women (60%, 26) did not continue use. The reasons, in decreasing order of importance, were: “can’t replace an ordinary cigarette” (32%, 12), “just tried out of curiosity” (18%, 7), “don’t smoke” (16%, 6), “doesn’t feel like inhaling smoke” (13%, 5), “unpleasant taste” (11%, 5), “dangerous for your health” (5%, 2), “pregnancy” (3%, 1) and “price” (3%, 1).
E-Cigarette Regular Use: Of those who had tried e-cigarettes, 40% (17) continued to use them on an occasional or daily basis: 33% (2) of pregnant women and 41% (15) of women taking COCs (Table 1). Among the non-smokers, 30% (3) also continued e-cigarette use.
Perception of E-Cigarettes, User Profile and Characteristics
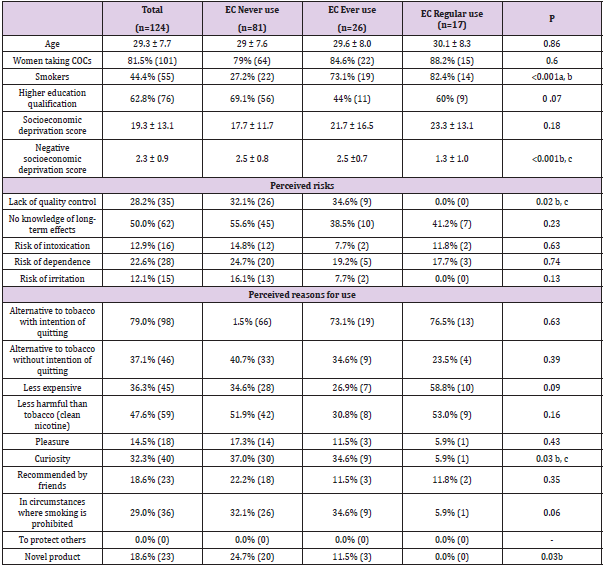
Table 2 shows the perception of e-cigarettes by regular users, those who had only experimented with them and never users. Regular users were more likely to be smokers and had a significantly less negative perception of e-cigarettes than the other participants (p<0.001, Table 2).
Table 2: Perception of e-cigarette.
Note: a EC Never use vs. EC Ever use.
b EC Never use vs. EC Regular use.
c EC Ever use vs. EC Regular use.
Perceived Risks Related to E-cigarette Use: The most commonly perceived risk of all the women responders was the lack of information concerning the potential long-term effects of e-cigarettes (50%), followed by the absence of quality control of the products (28%) (Table 2).It is noteworthy that, unlike experimental users and never users, regular e-cigarette smokers did not consider the latter factor to be an element of risk (p<0.02, Table 2). Only 23% of the women saw nicotine-dependence as being a risk of e-cigarette use (Table 2). The risk of intoxication in young children following ingestion of e-liquids was mentioned by only 13% of the participants, likewise the risk of irritation (Table 2). Interestingly, the risk of intoxication was cited by only 20% (3) of the pregnant women. In addition, two thirds of the women (81), including 70% (16) of pregnant women, did not know the answer or replied negatively to the question: “In your opinion, can e-liquids contain alcohol?”.
Perceived Reasons for Using E-cigarettes: The women in the survey considered that e-cigarettes were being taken up mainly as an alternative to tobacco, either in the hope of quitting smoking (79%) or without this intention (37%). Among e-cigarette users, only 24%thought the latter. The second main perceived reason was to use a product that was less harmful than tobacco (48%). More than a third considered that a lower cost than that of traditional cigarettes was the deciding factor. About 30% mentioned curiosity and the possibility of vaping in circumstances when smoking is forbidden as reasons. About 20% thought that people were attracted by the novelty aspect of the cigarettes or had been recommended to try them by friends. However, these last two reasons were not given by regular e-cigarette users (p=0.03). Finally, about 15% mentioned the notion of pleasure.
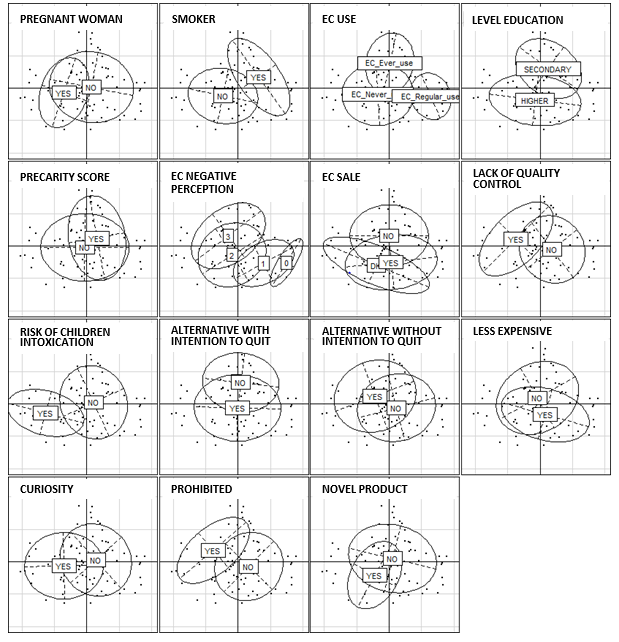
Profile of E-Cigarette Users: A multiple correspondence analysis was performed to produce a user profile (Figure 1). It showed that the e-cigarette users in our survey tended to be smokers without a strongly negative perception of the product. They were aware of the limited knowledge of the effects of e-cigarettes but not of the risk of intoxication they might pose for young children. They used e-cigarettes as an alternative to tobacco with the aim of quitting smoking and because they cost less than traditional cigarettes. In contrast, they were not drawn to e-cigarettes by curiosity, pleasure, or its novel character nor by the possibility of vaping in places where smoking is prohibited.
Patterns of Use: Most of our vapers preferred re-usable cigarettes (88%, 15) to the disposable variety (12%, 2). They bought the same brand of e-cigarette (41%, 14) and/or e-liquid (41%, 14) rather than different sorts of e-cigarette (9%, 3) and/or e-liquid (9%, 3). The two most common brands, cited by about a quarter of the users, were “Clopinettes” and “Alfa liquid”. 82% (14) of the women thought the liquid came from France or Europe as against 18% (3) who were unaware of its origin. More than three-quarters (68%, 13) reported buying their cigarettes in specialist shops and/ or at the tobacconist’s (16%, 3), or on internet (16%, 3). 71% (12) said they paid attention to the composition of their e-liquid.
More than half tried different flavors (53%, 9) rather than using the same one (47%, 8). The two most chosen flavors were tobacco (47%, 9) and fruit (37%, 7). More than half of the users (53%, 9) had been vaping for at least 3 months with a mean duration of 16 weeks (SD: 15). The mean volume of e-liquid used was 11.15 ml/ week (SD:12.09) initially and 10.54 ml/week (SD: 12.25) at the time of the survey, a decrease of 0.61ml/week (p=0.16). The mean dose of nicotine was 12.82 mg/ml (SD: 3.71) at the beginning of use as against 10.50 mg/ml (SD: 2.99) at the time of the survey, a significant reduction of 2.32 mg/ml (p=0.015). Since they had started vaping, 82% (14) of the women said they could breathe more easily. Two thirds of the users (11) reported no undesirable effects related to the e-cigarettes while 18% (3) had a burning or uncomfortable sensation in the mouth followed by an unpleasant taste (12%, 2) and then digestive problems such as nausea and vomiting (5%, 1), difficulty in breathing (5%, 1) and headache (5%, 1) None of the vapers had ever replaced their e-liquid by another substance. More than a quarter of the users said they had stopped smoking since using the e-cigarette (29%, 4); the others continued to smoke. Finally, 82% (14) of the vapers would be ready to recommend the use of e-cigarettes to relatives or friends who were smokers.
Perception of the Role of the Pharmacist with Regard to E-cigarettes
In France, the sale of e-cigarettes is prohibited in pharmacies. More than 70% (89) of the women questioned knew about this law, while 28% (35) were unaware or thought the opposite. The pharmacist, as a partner in public health, has a role to play about e- cigarettes but this role was perceived by only 33% (42) of the survey participants. The women considered the pharmacist’s role was to inform, and advise on prevention (65% of replies), help with smoking cessation by targeting potential users (22%) and to guarantee the safety of the products sold (13%). More than half of the women (6) who said they were trying to quit smoking were not receiving any follow-up, 27% (3) had consulted their pharmacist and 9% (1) a specialist in tobacco addiction. Likewise, more than half used e-cigarettes as an aid to quitting smoking (6), 41% (5) used nicotine substitutes, particularly transdermal patches (43% of replies), followed by gum (29%), inhalers (14%) and sublingual tablets (14%). One woman sought no help in quitting (9%). Those who were trying to quit had generally made attempts to stop before (82%, 9).
Discussion
These results point up the current limited knowledge about e-cigarettes and the difficulties encountered in quitting smoking and underline the importance of the advisory role of the pharmacist, particularly for at-risk individuals. The main limitation of our study is the small survey sample, in particular the number of e-cigarette users (17/124). The other potential source of bias lies in the socioeconomic category of the target population, which is overrepresented by pharmaceutical assistants among the intermediate occupations. Nevertheless, our survey shows that among the users of e- cigarettes there are also non-smokers and at-risk subjects such as pregnant women. These findings are consistent with those of the French ETINCEL telephone survey carried out in November 2013 at the request of the OFDT (Monitoring Centre for Drugs and Drug Addiction) [6]. This survey showed that 88 % of French people were familiar with e-cigarettes [6]. The proportion rose to 99% in the 2014 French “National Health Barometer”, a large telephone survey of health behaviors in a representative random sample of the population aged 15-75 years living in France [7]. According with this result, the present study shows that in 2014, all the subjects interviewed were aware of the product. About one in five (18 %) of those questioned in the ETINCEL survey had already experimented with the electronic cigarette [6]. The proportion rose to 26% in the 2014 French “National Health Barometer”, 3% use a cigarette on a daily basis and the majority of users tend to be young [7]. The women in our study were of childbearing age, and hence relatively young, which might explain why many of them had tried e-cigarettes.
In a literature review of e-cigarettes 2006-2013, Pepper et al. reported that awareness of the product among the public increased from 16% to 58% between 2009 and 2011 along with its use, which rose from 1% to 6% [16]. Most users were smokers or ex-smokers; some smoked both electronic and traditional cigarettes [16].
E- cigarettes were used as an aid to quit smoking, as a healthier alternative to tobacco or to replace a traditional cigarette in areas where smoking is not allowed [16]. These findings are in line with those of our study except for the last one, probably because new legislation is being introduced in France to prohibit vaping in public places [10]. Certain concerns related to e-cigarette use have been expressed by the HCSP and reiterated by the WHO [14]. It is feared that the e-cigarette could be the gateway to nicotine consumption among young people and make cigarette smoking seem normal [14]. These concerns find an echo in our results since among the non-smoking women in our survey who had tried e-cigarettes certain continued to use them thereafter. The WHO report makes mention of irritation of the eyes and respiratory tract as a possible adverse side effect of e-cigarettes, as the result of exposure to propylene glycol [14]. These side effects also figured among those referred to by participants in our survey.
Although meta-analyses showed that e-cigarettes were, in the short term, less harmful than the traditional cigarette and seemed to be of help in smoking cessation [17,18], they nevertheless involve the inhalation of nicotine, which is addictive and toxic at high concentrations. The main health risk resulting from nicotine exposure via routes other than inhalation is intoxication by ingestion or contact with the skin [19]. This risk is all the greater in young children since intoxication can be fatal [19]. There was low awareness of this hazard in our survey, which is worrying and suggests that efforts should be made to fully inform the public of the potential danger. Most countries have no monitoring assessments of these incidents and hence information is scarce. However, reports issued by the United States and the United Kingdom indicate that the number of cases of nicotine poisoning have risen considerably as the use of electronic inhalers has become more widespread [20]. The participants in our survey were unaware of the contents of e-liquids and did not know whether they contained alcohol. The works of Mark et al. revealed that misconceptions about electronic cigarettes are common among pregnant women [21]. In their report published in 2014, the WHO emphasized the potential danger of e-liquids for the unborn baby and recommended that their use be prohibited in pregnant women [14]. The report called for caution, stating that while e-cigarettes held out promise in the fight against tobacco use, they also constituted a potential threat [14]. It was also reminded that scientific studies were needed on the effects of e-cigarettes on health even if their use is probably less harmful than that of tobacco [14].
Conclusion
In conclusion, although a good proportion of the women in our survey had experimented with, or were using, e-cigarettes they were largely unaware of their possible dangers. These findings suggest that the pharmacist has a role to play as a partner in public health not only in providing preventive information but also in helping smoking cessation. However, because the sale of e-cigarettes is prohibited in pharmacies, this advisory role is not generally appreciated. Over- the-counter conversations could help change perceptions and warrant official encouragement as part of the new tasks assigned to pharmacists by the Hospitals Patients, Health, and Territories law of 2009 [22].
Competing Interests
The authors have declared that no competing interest exist.
References
- European Commission. Attitude of Europeans toward tobacco (2012) Special Eurobarometer 385: 167.
- Lermenier-Jeannet A. Le tabac en France: un bilan des années 2004-2014 (2014) Office Français de Prévention du Tabagisme [in French]. Tendances 92: 1-6.
- Office Français de Prévention du Tabagisme (2013) Rapport et avis d’experts sur la cigarette électronique [in French], pp. 212.
- Guignard M, Neck F, Richard JB, Lermenier A, Wilquin JL, et al. (2015) La consommation de tabac en France en 2014: caractéristiques et évolutions ré [in French] Evolutions 31: 1-6.
- Rogers JM (2008) Tobacco and pregnancy: overview of exposures and effects. Birth Defects Res C Embryo Today 84(1): 1-15.
- Observatoire Français de Drogues et des Toxicomanies (2014) Résultats de l’enquête ETINCEL-OFDT sur la cigarette électronique [in French]1: 1-15.
- Institut National de Prévention et d’éducation pour la santé (2014) Baromètre santé Premiers résultats tabac et e-cigarette. Caractéristiques et évolutions récentes [in French].
- Law n°2014-344 dated 17th march 2014 relative to the consummation (2014). Official Journal n°65 of 18th March 2014 [in French], p. 75.
- Cahn Z (2013) France acts on electronic cigarettes. J Public Health Policy 34(4): 560-564.
- (2014) Ministère des Affaires Sociales de la Santé et des Droits des Femmes (2014) Programme National de réduction du Tabagisme 2014-2019 [in French], p. 55.
- (2011) Agence Nationale de Sécurité du Mé Cigarette électronique: Point d'information.
- (2013) European Parliament. Amendments adopted by the European Parliament on 8October 2013 on the proposal for a directive of the European Parliament and of the Council on the approximation of the laws, regulations and administrative provisions of the Member States concerning the manufacture, presentation and sale of tobacco and related products.
- (2014) High Council for Public Health. Opinion concerning the risks and benefits in the use of electronic cigarettes by the general population, p. 10.
- (2014) WHO Framework Convention on Tobacco Control. Electronic nicotine delivery systems. Report, p. 13.
- Labbe E, Blanquet M, Gerbaud L, Poirier G, Sass C, et al. (2015) A new reliable index to measure individual deprivation: the EPICES score. Eur J Public Health 25(4): 604-609.
- Pepper JK, Brewer NT (2014) Electronic nicotine delivery system (electronic cigarette) awareness, use, reactions and beliefs: a systematic review (2014). Tob Control 23(5): 375-384.
- Hajek P, Etter JF, Benowitz N, Eissenberg T, McRobbie H (2014) Electronic cigarettes: review of use, content, safety, effects on smokers and potential for harm and benefit. Addiction 109(11): 1801-1810.
- McRobbie H, Bullen C, Hartmann-Boyce J, Hajek P (2014) Electronic cigarettes for smoking cessation and reduction. Cochrane Database Syst Rev 12: CD010216.
- Normandin PA, Benotti SA (2015) Pediatric Emergency Update: Lethality of Liquid Nicotine in E-Cigarettes. J Emerg Nurs 41(4): 357-390.
- Durmowicz EL (2014) The impact of electronic cigarettes on the paediatric population. Tob Control Suppl 2(Suppl 2): ii 41-46.
- Mark KS, Farquhar B, Chisolm MS, Coleman-Cowger VH, Terplan M (2015) Knowledge, Attitudes, and Practice of Electronic Cigarette Use Among Pregnant Women. J Addict Med 13 “in press”.
- Law n°2009-879 dated 21th july 2009 relative to Hospitals Patients, Health and Territories [in French].

 Research Article
Research Article