ABSTRACT
The generally accepted modus operandi for stimulating muscle strength adaptation is high-magnitude resistance training which is facilitated by protein and with the addition of augmented exogenous protein intake this outcome is potentiated. The aforementioned protocol has been observed in many studies over time periods (up to 12 weeks) however; very few longitudinal studies have examined the temporal dynamics of muscle strength adaptation throughout the entire period. These data could prove vital in constructing training and rehabilitation programs for both athletes and non-athletes. The objective of this review was to establish whether high-magnitude training with the addition of nutritional supplementation results in enhanced temporal adaptation dynamics and at what point these adaptations are significant. The searches were achieved utilising PubMed, Google Scholar, Medline, ScienceDirect, Wiley Online and SPORTS. Discuss databases. Of the inclusion criteria a total 362 participants from 11 studies were included in the review and concluded that the mean time point for muscle adaption was ~6.7-weeks. With the results indicating that adaptation to high-magnitude resistance training occurred at each time-point (up to and including 10, 12 and 24-weeks) for the training only groups of each study (placebo), this was equivalent to the supplement intervention group. While, both groups evidenced adaptation only ~27% showed significance between the placebo and intervention groups.
These findings are contrary to the known paradigm that supplements enhance muscle protein synthesis thus recovery and subsequent adaptation. However, there are considerations regarding the protocols used in the studies reviewed including dietary standardization that may have impacted the results. Notwithstanding, the results do indicate that supplements may enhance the effects of high-magnitude training and that this could have an impact on temporal adaptation dynamics leading to augmented muscle strength increases at an earlier stage in a training cycle than the current paradigm dictates. This could pose problems for other associated tissues such as tendinous tissue, as a differential in adaptation between muscles and tendons could cause non-uniformities within the muscle-tendon-unit and potentially lead to tendinopathy. Consequently, it may be prudent to investigate the temporal dynamics of muscle and tendon adaptation with a supplement intervention.
Keywords: Supplement; Protein; Whey; Leucine; Creatine; HMB; Hydroxy Methylbutyric Acid; Temporal Dynamics; Time-Related Adaptive Response; Muscle Adaptation; Strength Adaptation; Resistance Training; Nutritional Response; Muscle Strength
Introduction
Muscular adaptation is determined by parameters of mechanical loading, including intensity, load, and duration (Hickson, et al. [1,2]) and adequate nutritional support (Cermak, et al. [3]). Notwithstanding, the primary stimuli appear to be intensity and duration coupled with sufficient resistance (Schuenke, et al. [4]) and variants of these protocols are used throughout the world by professional and recreational athletes to promote muscle size and strength. Based on the classical overload principle a certain threshold needs to be exceeded in order to cause the optimum improvements in muscle strength, accordingly moderate to high mechanical loading has been proposed as the main stimuli to initiate muscle hypertrophy and increase strength adaptation (Hakkinen, et al. [5]). In general, it has been widely acknowledged that the initial phases of training that induce muscle strength improvements are predominantly redirected to the enhanced neuromuscular components (Carroll, et al. [2,6]), whereas morphological changes (hypertrophy) do not occur in muscle tissue until after six to eight weeks of high-magnitude resistance training (Verdijk, et al. [7]). The neurological adaptations that initiate and later augment muscular strength adaptation occurs as a resultant factor of mechanical resistance training and leads to significant increases in strength within the first two-weeks of training (Aagaard, et al. [8]). However, the specific mechanisms involved are still being debated and investigated. It has been postulated that the agonist skeletal muscle activation during high-magnitude mechanical loading could be as a result of augmented motor unit recruitment or firing frequency. While it has been shown that motor unit activation can be slightly changed with mechanical loading training protocols (Patten, et al. [9]) the study of motor units pre- and -post mechanical loading interventions appear to follow the size principle regardless of stimuli i.e., isometric or dynamic contractions [10].
Nevertheless, although the size principle appears to be conserved post training the absolute force that a specific motor unit is recruited depends on the augmented contractile force and contraction time of low-threshold units post training (Aagaard, et al. [11]). Another consideration that has been shown to impact neuromuscular adaptation is the application of a particular training protocol, explosive or steady state muscular contractions [9] evidenced that contraction type can determine neuromuscular response with progressive maximal contractions eliciting greater adaptations than rapid explosive contractions. The initial increase in neuromuscular adaptation enables larger loads and greater intensity to be achieved within the parameters of the training protocol therefore, the resultant factor will be further augmented adaptation leading to morphological changes (Duchateau, et al. [12]). Morphological changes are initiated to protect the skeletal muscular system from damage caused by external factors i.e., highmagnitude mechanical loading (to maintain homeostatic balance) (Haun, et al. [13,14]). The morphological change in response to high-magnitude mechanical loading results in enlargement of the muscle fibres, hypertrophy. The increase in the cross- sectional area (CSA) of skeletal muscle augments the strength of the muscle, as the CSA of a muscle is directly proportional to the force of the muscle. The morphological adaptation of muscle tissue is initially stimulated by mechanical stress which leads to metabolic stress (fatigue driven), initiating a plethora of other factors including hormonal changes; testosterone, insulin-like growth factor-1 (IGF- 1), insulin fluctuations; nutritional intake; protein, carbohydrates, electrolytes; recovery and timing of feeding to stimulate muscle adaptation, all these factors evoke adaptation [15].
High-magnitude mechanical loading causes mechanical stress during the concentric and specifically the eccentric phase of skeletal muscle contraction creating a greater hypertrophic response (Lixandrao, et al. [16]). Where eccentric contractions are challenging the sarcomere, the length is compromised and elongated beyond myofilament overlap, the tension augments and the actinmyosin cross-bridge integrity becomes compromised resulting in micro tears [17,18]. Continued contraction additionally damages the integrity of the sarcomere resulting in augmented calcium release and leads to damage to the sarcoplasmic reticulum leading to localised oedema and soreness as a result of inflammation. To elucidate further mechanical stress coupled with metabolic fatigue leads to localised metabolic stress resulting in increased activation of mechanosensitive calcium channels, intracellular enzymes and second messengers. IGF-1 is also secreted from the muscle cells triggering signalling cascades resulting in muscle protein synthesis (MPS) (Yang, et al. [19-23]). The increase in MPS stimulates additional molecular mechanisms that augment sarcomeres and myofibrils contractile elements size expanding the extracellular matrix to support growth/hypertrophy (Haun, et al. [13]). Regardless that this study is concentrating on the examination of skeletal muscle adaptation via high-magnitude mechanical loading (predominantly strength training protocols), heavy resistance lowvolume or light resistance and high-volume training effects strength adaptation to some degree, and a subsequent change in fibre type size (Campos, et al. [24-27,13]). There is evidence that resistance training elicits a greater effect on Type II muscle fibre with satellite cell proliferation and differentiation augmenting (Kadi, et al. [25,28]). Together with sarcoplasmic expansion and coordinated up-regulation of sarcoplasmic proteins involved in glycolysis and other metabolic processes related to ATP generation. This suggests that Type II muscle fibres (fast twitch fibres generating more force) can adapt significantly to resistance training by increasing their size (Verdijk, et al. [29]).
To facilitate the mechanisms of adaptation post mechanical stimuli adequate nutrition is essential, with specific attention to protein consumption (Esmarck, et al. [30-36]). A positive nitrogen balance provided by protein feeding is required to ensure remodelling of contractile machinery leading to hypertrophy (McGlory, et al. [37]). Nutrition is therefore a fundamental external element that can be manipulated to ensure a positive nitrogen balance is maintained post training to facilitate MPS, (Andrews, et al. [14]). The protocol for pre-training and post-training feeding is well documented (Berardi, et al. [33,38]) and there are a plethora of peer-reviewed papers and scientific publications providing robust paradigms for creating an environment for muscular adaptation (Hickson, et al. [1,30,39]). Many of these studies concentrate their attention on high-magnitude mechanical loading followed by protein feeding resulting in an augmentation of MPS and thus, over time, muscle hypertrophy (Cermak, et al. [3,39]). It has been established that to augment the adaptation of skeletal muscle tissue post resistance training an increase in protein consumption is required and in particular the amino acid (AA) leucine and supplements derived from leucine i.e., hydroxy methylbutyric acid (HMB) have been shown to be effective [40]. Leucine functions directly with mammalian target of rapamycin (mTOR), during resistance training mTOR activation is inhibited by AMP Activated protein kinase (APMK) as AA’s become available for energy metabolism. However, during recovery AMPK activation is decreased and mTOR reaches peak activity and as mTOR is known as an important signalling molecule involved in muscle hypertrophy it is important to potentiate its activation [41,42].
With the addition of protein to the habitual diet to enhance the anabolic environment post training other supplements have been determined to be effective. Although, there are a plethora of novel supplements available few supplements have been researched with more veracity than protein and creatine. These supplements have proven to have ergogenic characteristics and while the scientific evidence is esoteric, the general principles are known by the public therefore it would be prudent to ensure the validity of these claims and ensure that the public are utilising these supplements correctly. Creatine is unquestionably one of the most researched supplements of the past 30-years, the efficacy of creatine is well documented; it augments phosphocreatine (PCr) energy stores; augmenting PCr resynthesis; and reducing muscle damage through buffering the increases in lactate and hydrogen ions. However, there is no clear evidence that creatine directly enhances MPS and thus muscle strength adaptation however, it does impact performance positively by enabling greater force production. This creates an optimal environment to work/train with higher intensity over a short period of time, with this maximal intensity damage to the musculature is enhanced resulting in adaptation. The augmented response to training and the congruent adaptation results in hypertrophy and therefore an increase in the CSA of the muscle. (Wilder, et al. [43-50]).
With the addition of the traditional aforementioned supplements there has been a surge in research into additional supplements that will enhance the recovery and adaptation post mechanical loading protocols. Metabolites of leucine including Hydroxy methylbutyrate (HMB) and Hydroxyisocaproic Acid (HICA) are being increasingly used as supplements to augment the impact of leucine on the mTOR pathways to enhance MPS (Gallagher, et al. [51-54]). With HMB purporting to have anticatabolic actions acting antagonistically against the effects of acute bouts of resistance training where there is an increased breakdown of protein. HMB partly prevents this exercise induced proteolysis thus, causing assuaged muscle damage and therefore recovery becomes more expedient and gains in MPS follow (Phillips, et al. [19,20]). These effects have been observed in both young and elderly adults (Nissen, et al. [55]). There is substantial evidence that supplements augment skeletal muscle adaptation as a result of high-magnitude mechanical loading however, there is little evidence of the rate of adaptation. The accepted and established paradigm is that neurological changes occur within the first 2-3 weeks of training and morphological adaptation does not initiate transpire until after that period. Moreover, there is little known regarding the effects of the addition of supplements to in the equation, will there be a direct correlation with known studies evidencing an augmented response to supplement ingestion over a 12-week period when applied to a resistance training protocol or will this increase occur more expediently after 2-3 weeks rather than 6-8 weeks (Gabrie, et. [56,57]).
Aims of the Review
Research indicates that there are many parameters influencing adaptation time induced by mechanical loading, including intensity, load, duration and enhanced neural drive (which has an immediate response) (Hickson, et al. [1,2]). However, morphological changes do not occur in muscle tissue until after three to four weeks of high-magnitude resistance training. To elucidate further, neural adaptation increases the plasticity of the force generating capacity of the neuromuscular system which leads to enhanced muscle strength leading to greater adaptations (Fimland, et al. [2,58]). The inclusion of an enhanced nutritional regime with the addition of supplements, including but not limited to whey protein, leucine, HMB and creatine to augment the recovery process caused by mechanical stress (Vandenberghe, et al. [59,60,30,31,46,7,61,54]) could impact the temporal dynamics of strength adaptation. The rationale of the review therefore is to evaluate available peerreviewed studies for specific criteria relating to or including temporal dynamics (time-course changes) during strength training protocols over a period of at least 6-weeks (max. 12-weeks) with the addition of a nutritional/supplement intervention. With the aim of the review to elucidate current paradigms with special attention on potential gaps in current research. For the purpose of this study, the term ‘supplement’ will be used to refer to any oral product designed to augment the effects of resistance/strengthtraining exercises i.e., protein, leucine, HMB and creatine. There are cornucopia of studies relating to resistance training protocols to enhance muscle adaptation including studies that comprise a supplement element. However, these studies predominantly use pre- and post-intervention data to evidence adaptation, this review will use only data given from time-based adaptations to resistance training with a supplement intervention included (with a minimum of pre-, mid- and post-adaptation data).
Examining the temporal dynamics of muscular strength adaptation is vital as the information provided from researched based evidence could be used to provide a protocol for strength and recovery training programs. As discussed previously there is a convention regarding strength adaptation over a 10-week plus intervention, the evidence that adaptation does occur as a result of resistance training is overwhelming. There is also evidence that supplementation augments this adaptation however, at what time-point does the supplement augment the conventional muscle adaptation as this change could impact a training schedule. As supplements are utilised as ergogenic aids, with the paradigm that they elicit enhanced performance via maximising muscular adaptation as a result of training it is essential that the parameters are known. Paradoxically, using supplements to augment the recovery process thus potentiate muscle adaptation via resistance training could have adverse consequences. There could be imbalances in adaptation between muscle and tendon tissue thus causing disparities within the muscle-tendon-unit (MTU) (Mersmann, et al. [21]) this could potentially cause tendinopathy. Naturally, there are also implications for the development of training protocols to be considered for sports scientists, coaches etc., to use with their athletes. Regular adjustments will need to be assessed if the athlete has added supplements to their diet, ensuring that adequate stimuli is maintained to continue with training progression [55,62]).
Therefore, the objectives of this review are to find evidence of resistance training protocols that elicit strength adaptations over a time-course when a supplement intervention is added and to compare these results to a control group to evidence phases of adaptation. The resultant evidence may indicate a differential between the temporal dynamics of resistance training with and without a supplement intervention. Therefore, if there is found to be a correlation between supplement intervention and an increase in temporal dynamic adaptation the type of supplement and dosage will be exposed.
Materials and Method
The review was conducted in accordance with the search strategy guidelines using the criteria of the Preferred Reporting Items for Systemic Reviews and Meta-Analysis. The review was conducted via electronic databases search for peer-reviewed papers examining temporal dynamics of muscle strength adaptations resulting from supplement ingestion. With the term ‘supplement’ referring to oral consumption of a nutrient purported to augment the adaptation of muscle tissue as a result of mechanical stimuli [55]. The electronic searches were limited to English language citations published in PubMed database and Google Scholar from 1980 onward DISCOVERY resources at London South Bank University were also utilised for the initial citation and abstract searches. Additional refined searches were conducted to also include conference papers using EBSCO host which included the data bases Medline, ScienceDirect, Wiley Online and SPORTS Discuss. A combination of the words and phrases associated with the subject matter were used in the search; ‘muscle adaptation’, ‘strength training’, ‘time-course adaptations’, ‘temporal dynamics’, ‘resistance training’, ‘nutritional response’, ‘muscle strength’, ‘HMB Hydroxy Methylbutyrate Acid and adaptation’, ‘leucine and strength adaptation’, ‘protein and muscle adaptation’, Creatine and muscle strength’, ‘creatine and performance over time’, ‘supplements and muscle adaptation’. Further references were sourced through manual crosschecking of peer-reviewed papers that pertained to the relevant search criteria ensuring absolute saturation of related papers.
Primary citations and abstracts were searched for the following criteria and peer-reviewed papers were included therein:
a) Studies published in English
b) Randomised controlled or controlled trials
c) Studies containing a nutritional intervention related to augmenting strength increases
d) Studies pertaining to resistance training where the primary aim was to increase muscle strength via training frequencies of more than twice weekly
e) Studies which had a duration of no less than 6-weeks
f) Studies measuring muscle strength in terms of 1 maximal repetition (1RM) or maximal voluntary contraction (MVC) preintervention, at least once during intervention and at the end of the intervention
g) A placebo control administered
h) Peer-reviewed papers post 1980. The studies included in the final review contained all the search criteria idiosyncrasies.
Results
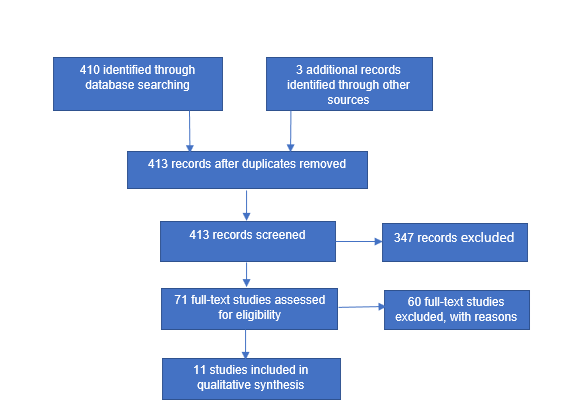
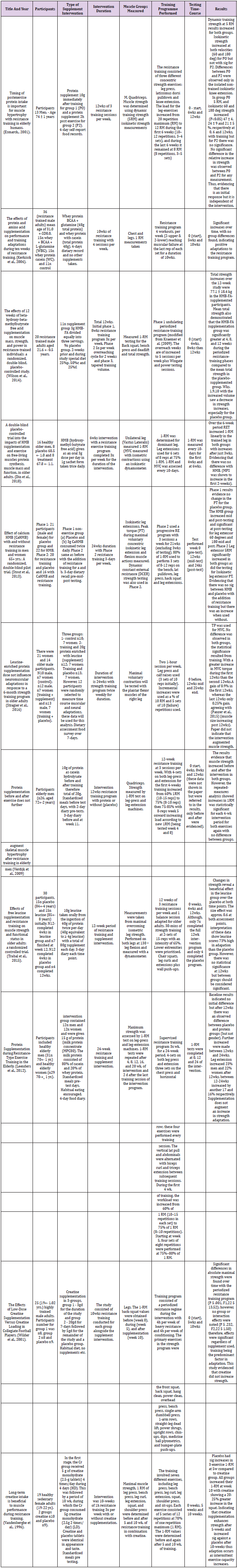
Initially the databased search process yielded 410 studies with three studies identified through other sources leading to 413 studies after duplicates were removed. Thereafter, abstracts were read to ensure the relevance to the review, this led to the exclusion of 347 studies. The remaining 51 studies were fully read and examined in miniscule detail, which led to identification of a further 20 papers leading to 71 studies. Of the 71 studies examined 60 were excluded while 11 matched the criteria genus. Many of the studies were excluded due to the frequency of measurements taken. The vast majority of the studies examined only the measurements before and after the intervention, heralding interesting results but not the results required for the review. Figure 1. represents evidence of the search process with the PRISMA search flow diagram. (Table 1). summaries each study. The details of the 11 peer-reviewed papers include in the review indicate that of the total 362 participants ~66% were male adults and ~34% were female adults with an age ranged between 18-93 years. Of the studies examined ~64% of the studies recruited exclusively male participants while ~27% utilised both genders with the remaining ~9% devoted to female participants. Regarding intervention duration, ~90% (n=10) were over 10-weeks with only one study lasting less that 10-weeks. However, the study lasting 6-weeks had measurements taken every 10-days for 40-days and then again at 6-weeks producing interesting data. The reviews characteristics pertained to a subject group that predominantly abstained from resistance training activities with ~73% of the studies including ~67% of the total participants. Only 27% of the studies reviewed used participants that used resistance training activities. A similar trend was established for the age of the participants with studies containing older adults comprising of ~64% of the total. With only one study recruiting sedentary women only between the ages of 19-22 years and three using trained individuals between 18-39 years.
The nutritional interventions used in the selected studies include supplements containing protein including protein enriched with leucine, HMB and creatine. Of these studies ~64% utilised a self-reported food diary/log, the studies used dietary records of between 3 and 7-days to analyse habitual diets of the participants. Only ~9% of the studies utilised meal standardisation for the duration of their studies (25% protein, 50% carbohydrate and 25% fat) however, a further ~27% did require a standardised meal scheduled to be used the day prior to testing/measurements. It is interesting the note that ~55% of the studies reviewed focused on protein (including protein enriched with leucine) with ~67% of these studies using a minimum of 20g as an addition to habitual diet. HMB supplementation consisted of ~27% of the studies reviewed with a standardised 3g taken per-day. The remaining ~18% (n=2) studied the effects of creatine both these studies used a ‘loading’ phase of 20g per-day split throughout the day followed by a maintenance phase, using 5g per-day for one study. The second study had a high (5g) and low (3g) loading phased approach for 4-days (total of 20g and 12g respectively) then maintenance of 5g and 3g respectively for the remaining duration. Regarding resistance strength, analysis of upper and lower limb muscle groups were tested and measured with ~73% of test completed on the lower limbs and ~27% on both upper and lower body tests. The tests used to measure the strength of the muscle included 1-repetition maximal lift (1RM) and/or dynamic training strength (5RM) and/or isokinetic strength measurement or a mixture of all three. Of these measures 1RM was the most prevalent measure used (with ~73% of studies using this method) with ~36% of the studies using isokinetic strength measures either independently or with other measures (other measurements e.g., free-fat mass was taken in each study; however, these measures were not directly pertinent to this review albeit very interesting).
The results of the reviewed studies are consistent with previous studies relating to the adaptation of skeletal muscle to resistance type training with specific regard to this review where studies were categorised to utilise high-magnitude training protocols >65% 1RM intensity. All of the reviewed studies evidenced adaptation to training regardless of supplement intervention or placebo given. However, it was interesting to note that Stout et al., 2013 evidenced that when HMB was taken as a supplement over a 24-week period without resistance training there were still significant mid- and post-testing adaptations. Interestingly, when resistance training was applied congruent to the previously mentioned paradigm there were significant changes at mid- and post-testing also however, there was not a significant difference between HMB and the placebo. Which contradicts Wilson et al. (2013) which demonstrated that HMB supplementation was significantly greater at 4, 8, and 12-weeks than the control group. The results of the protein supplement intervention confirmed that resistance training with high-magnitude led to increases in muscle strength adaptation at each time-point. All of the studies utilising protein as the supplement intervention used a control to evidence contrasts with the exception of (Esmarck, et al. [30]) which used pre-exercise and 2-hours post-exercise supplement interventions as comparisons rather than using a non-training placebo, again these showed mid-point (6-weeks) and post-trial (12-week) significant results, yet with no differential between interventions. The other protein interventions reviewed indicate there was no significance between intervention and control group regarding strength increase measurements. With the exception of (Trabal, et al. [63,64]) which evidenced that although there was no statistical significance at 12-weeks for isometric leg strength gains, betweengroup differences could be considered clinically significant at each time-point.
Creatine supplementation has been universally accepted to enhance performance with multiple short-term work bouts, improve recovery and long-term adaptation to training (Vandenberghe, et al. [59,60,43,65,49]). This review contained two studies that used creatine supplementation based on the criteria of reviewing temporal dynamics of strength adaptation and the differential between creatine consumption in the short-term and a control. The results of the two studies included in the review are not concomitant with each other, the results differ as (Wilder, et al. [45]) evidence significant time effects for 1RM strength increases, before and during, and during and after, and before and after supplementation. However, these data are similar to the control group which also showed increases in maximal strength thus showing that the addition of creatine (both low and high does) did create a significant differential (noting that the participants were trained athletes and their diets were not standardised). However, (Vandenberghe, et al. [59]) evidenced that creatine supplementation significantly increased 1RM strength after 5-weeks of resistance training with all six-exercise measured test showing improvements by +25 to +57% whereas, the placebo only increased in three measures tested (+15 to +40%). At 10-weeks the creatine group again showed greater difference in all strength tests evidencing that in lower leg measurements there was a 20- 25% greater strength adaptation than the placebo group. Thus, indicating that creatine supplementation can enhance strength over the short-term as well as the long-term.
Based on the information gathered from the review it is in congruent that the current strength training paradigm does evoke adaption of muscle tissue. However, it has also been established that a strategy of adequate nutrition in the form of additional protein potentiates the adaptation of muscle tissue as a result of resistance exercise (Cermak, et al. [3]). Moreover, of the studies reviewed that used protein as a supplement (with a control/ placebo) ~80% evidenced no statistically significant differential between supplement intervention and placebo at each time-point. Overall, this review does evidence that there was an increase in strength adaptation at the mid-point of each study for both intervention group and placebo (a mean time-point of 6.7-weeks). Additionally, further adaptation at the end of each 10-week and 12-weeks with additional increases thereafter in the three studies lasting 24-weeks. In Summation, there are a plethora of variables that could impact and skew the results of this review, as the search criteria has concise constraints the quantity of studies included in the review limits the data. These results suggest the predominant factor in strength adaptation is resistance training at magnitudes >65% 1RM regardless of supplement intervention, age and gender.
Discussion
This review was designed to extrapolate evidence from relevant peer-reviewed papers related to the effect of supplements coupled with the mechanical modality of high-magnitude resistance training on the temporal dynamics of muscular strength adaptation. Presently, to the knowledge of the authors the present review is thought to be the first review conducted specifically focused on the effects of supplementation on the temporal dynamics of muscle strength adaptation. The results of this review are congruent to the paradigm that resistance training containing intensities of >65% of 1RM induces muscular adaptation resulting in the augmentation of strength (Hickson et al., [1,2,25,16]). At each time-point across the total population of the review there was an increase in strength adaptation for both the training with intervention groups and the ‘control/placebo’ groups (with training but no supplement intervention). Indicating that the main protagonist in muscle strength adaptation is resistance training. These data were expected, as previously cited via adaptation mechanisms initially as a result of neural drive and the adaptation thereafter (Schuenke, et al. [4]). The review evidence that the initial adaptation occurs at a mean of the studies reviewed of ~6.7-weeks, the ‘mid’ period of the studies reviewed. The was one study which was not within same parameters as the other studies, this study measured strength every 10-days for 40-days and then subsequently on the final day, day-60. Again, there were incremental increases but this time it was over a shorter period of time, with each 10-day period evidencing early onset adaptation which continued until the end of the study (worth noting that the participants were untrained healthy older men).
The trend of the control groups which involved resistance training with no supplement intervention corresponded to the results of the supplement intervention groups examined in the review. All the studies showed an augmented response to resistance training in muscular strength when a supplement was included to the participants habitual diet. Although, many studies included food-diaries to monitor nutritional intake to eliminate potential outliers, only one study actually requested standardised meals to be used by their participants throughout the trial (other studies used standardised meals prior to testing days). This lack of conformity within the selected studies could have had an impact on the supplement intervention groups. Moreover, the lack of diet control may have skewed the results of the supplement intervention group for the individual studies. As the nature of taking a supplement is an exogenous addition to a nutritional regime if the aforementioned diet has for example adequate protein intake, and the intervention group has an extra 20g protein per day this ‘supplement’ may not have such an impact as 40g per day. However, again noting that the results of the review for both the control and supplement intervention groups indicating augmented adaptation to resistance training regardless, with only ~27% showing significant difference between the two measures. These data are not indicative to current knowledge regarding supplementation particularly protein which has numerous purported benefits in muscular adaptation (Volek, et al. [23,28,7,32,39,63]). Moreover, with protein requirement post-resistance training being axiomatic there must be other considerations to contemplate within group difference i.e., the type of protein consumed, the amount of protein consumed, and the timing of the protein consumed (protein-feeding). It has been shown that the muscles response to amino acids (AA) is transient and that the amount of protein absorbed is limited, subsequent to a single bolus of whey protein, after a latent period of 45-minutes MPS was augmented by approx. 200% for up to 90-minutes post consumption. This was prior to normal absorption rates, regardless of the availability of intramuscular and/or plasma leucine and essential AA (Atherton, et al. [36,42,25]). Thus, evidencing that there is a response to saturation of AA for a limited time of between 60-90 minutes where the rates of MPS (which results in strength adaptation) augments before returning to normal levels regardless of the amount of protein consumed or the availability of protein at that time, this has been referred to as the ‘muscle full theory’ (Atherton, et al. [36]).
The ‘muscle full theory’ indicates that there is a finite level of protein that is required to elicit MPS, and that leucine is the prominent AA that triggers the response. However, it is interesting to note that only two studies used >40g of protein per-day. One study used a total of 48g of protein (40g whey and 8g casein) this was ingested within 2-hours of exercise and on non-training days in the morning with a habitual diet. Although, this study did evidence an increase in fat-free mass during the study and an increase in the strength parameters set over time it did not indicate significance between groups at each time-point (over a 10-week period). However, (Trabal, et al. [64]) evidenced that the use of 40g of protein (to achieve a minimum of 5g leucine content) taken twice per-day did show differences between groups which were considered clinically significant. The other studies appeared to use the standard paradigm for protein intake, with the accepted textbook standard being 20- 25g of protein consumed post-exercise [15]. Notwithstanding, the amount of protein administered in the studies reviewed could have had an impact on the outcomes. Evidence from (MacNaughton et al. [17]) indicating that whole body resistance training responds more favourably to 40g of protein consumed post-training rather than the standard 20g of protein recommended post-resistance exercise. Although, this information seems contrary to one of the aforementioned studies in the review which administered >40g of protein (fat-free mass did increase which can be a marker for muscular adaptation), there is evidence that 40g of whey protein is the optimum amount of protein to consume to elicit maximum MPS (MacNaughton et al. [17]). Whereas the majority of the other studies in the review did not use the optimal amount of protein to elicit a response which may have led to limitations. Thus, utilising this methodology alongside Atherton’s ‘muscle full theory’ would create a perfect post-exercise protocol to ensure participants could achieve maximal MPS therefore, ensuring the best possible conditions for muscle strength adaptation.
This review also highlighted the effects of HMB supplementation with ~27% of the papers reviewed administering HMB to their participants. With all of the studies using the standard dosing of 3g per day, this protocol has been shown to be the optimum dose to achieve the purported anticatabolic actions (Gallagher, et al. [50- 53]). With HMB purporting to have anticatabolic actions as a result of acute bouts of resistance training where there is an increased breakdown of protein, HMB partly prevents this exercise induced proteolysis thus causing reduced muscle damage therefore, recovery becomes more expedient and gains in MPS follow. These effects have been observed in both young and elderly 70-plus adults (Nissen, et al. [55]). This is why HMB is being studied and why it may have benefits as a supplement for improving recovery and subsequent adaptation and muscular strength. Of the ~27% of studies administering HMB, ~33% of the studies reviewed evidenced significant difference between the control group and supplement intervention group. These data evidence that temporal dynamics is significant at each time-point however, the remaining ~67% of the studies evidence the contrary. Fascinatingly, the one study that used standardised meals ensuring that a minimum intake of 25% protein (thus perhaps changing the dynamics of their habitual diet) showed significant results in the difference between groups. Remarkably, one study (Stout, et al. [66]) showed that there was significance between a non-trained control group and a nontrained HMB supplement group, evidencing HMB can augment strength adaptations without resistance training.
The review also considered creatine as a supplement with ~18% of the studies reviewed using creatine as their supplement of choice. Understandingly, creatine was chosen as it is unquestionably one of the most researched supplements of the past 30-years, the efficacy of creatine is well documented; it augments phosphocreatine (PCr) energy stores; increasing PCr resynthesis; and reducing muscle damage through buffering the increases in lactate and hydrogen ions. However, there is no clear evidence that creatine directly enhances MPS and thus muscle strength adaptation however, it does impact performance positively by enabling greater force production. This creates an optimal environment to work/train with higher intensity over a short period of time, with this maximal intensity damage to the musculature is enhanced resulting in adaptation (Louis, et al.). Nevertheless, if you do not have rigorous training protocols with continued high-magnitude training but rather use low-magnitude training over a short period of time, there is little need for creatine as a supplement to buffer metabolites, or to implement direct MPS (Wilder, et al. [43-49]).
The two studies that applied creatine as their supplement intervention were not completely homogeneous, one study used highly trained individuals and the other study used sedentary adult females. They also used different loading and maintenance protocols with (Wilder, et al. [43]) using high and low doses (20g and 12g loading and 5g and 3g maintenance respectively) and (Vandenberghe, et al. [59]) using the standard 20g loading and 5g maintenance protocols. The results of the (Wilder, et al. [43]) study containing highly trained individuals evidenced an increase in both the control and intervention groups in strength adaptation. With hitherto, no within group significant differences at any time-point during the study for high or low dose regimes (the diets of the athletes were not standardised nor controlled also; the participants known supplement regimes may not have been adequately investigated). Whereas the results of (Vandenberghe, et al. [59]) containing sedentary participants evidenced significance at each time-point (5- and 10-weeks) between groups. These results indicate that creatine supplementation can impact muscle strength adaptation after only 5-weeks, whereas it is notionally accepted that repeated resistive exercises engaging the ATP-PC energy system evidences more expedient results than when 1RM measures are tested (Wilder, et al. [43]).
Notwithstanding, it would seem counterintuitive to use creatine as a standalone predominant supplement for strength adaptation as it is not a direct potentiate of MPS. Also, the level of intensity of the mechanical modulation needs to be high. To augment the adaptation of muscle tissue, protein and in particular the AA leucine and supplements derived from leucine i.e., HMB as previously noted would be the obvious supplement to administer in intervention protocol to increase muscle adaption [26]. Leucine functions directly with mammalian target of rapamycin (mTOR), during resistance training mTOR activation is inhibited by AMP Activated protein kinase (APMK) as AA become available for energy metabolism. However, during recovery AMPK activation is decreased and mTOR reaches peak activity and as mTOR is known as an important signalling molecule involved in muscle hypertrophy it is important to potentiate its activation [41,42]. To elucidate the relationship between mechanical loading and leucine, a group of young (24-years) and a group of elderly (63-66-years) participants received intravenously infused leucine post resistance-exercise and the results at the end of the 2-week trial evidenced that both groups increased their MPS (Yarasheski, et al. [67]).
This review does have its limitations, the number of peerreviewed papers relating to temporal dynamics of strength training adaptations with a supplement intervention are inadequate. Of the studies reviewed there were few comparisons that could be met; with each having different dosages of the same/ similar supplement, differences in supplement administered, age differences of participants, trained or untrained status, standardised meals or habitual diet etc. Nonetheless, there were enough similarities to draw trends, resistance high-magnitude training increased muscular strength adaptation regardless of age or activity levels, supplements provide additional adaptation in each study but only clinically significant in ~27% of studies reviewed. Most importantly temporal dynamics were evidenced, with changes in muscle strength adaptation at each time-point in each study. Notwithstanding, lessons can be learnt from the limitations of the review, with areas of improvement for future trials. Standardised nutritional meal plans must be adhered to alongside pre-intervention food-diaries to understand the habitual diets of all participants to ensure the gold-standard for nutritional interventions. This should be a fundamental requirement to ensure adequate data alongside the correct mechanical modulation techniques for muscular strength adaptations (dependant on age demographic and gender considerations) to standardise two clear elements.
It is interesting to reiterate that when standardised meals were incorporated in a study and also, when the supplement included >40g protein to be taken twice per day the results were positive at each time-point. This leads to the question that if strength adaptation in muscular tissue occurs earlier than expected (the majority of previous studies evidence before and after measures and are usually 8+ weeks in duration) how that impacts muscular synergistic tissues such as tendinous tissue. The discrepancy with temporal dynamics of muscle and tendon adaptation caused by the use of high-magnitude training could have implications on the mechanical loading of the complex, with the disparity between muscle strength and quality of the tendon stiffness to tolerate said mechanical loading. Again, this discrepancy could be exacerbated with the addition of nutritional supplementation.
As previously cited, supplementation of leucine post-exercise accelerates MPS and thus muscle tissue adaptation. However, there is also evidence in intervention studies that leucine also has an effect on collagen in tendinous tissue, with an augment in hydroxyproline content of the tension region, independently of training intervention and a greater increase in collagen synthesis when combined with exercise (Barbosa, et al. [68,69]). This would suggest that leucine has anabolic qualities for both muscular and tendinous tissue nonetheless, this adaptation may not be uniform. One might postulate that the impact of the supplementation intervention will exacerbate the increase in muscular strength without a corresponding increase in tendon stiffness. This may result in an imbalance within the muscle-tendon-unit, with the muscle producing more mechanical force this augmentation in mechanical demand on the tendon will lead to potential vicissitudes resulting in susceptibility to injury (Epro, et al. [70-93]).
Conclusion
This review suggests that muscular strength adaptation occurs as a result of high-magnitude strength training (>65% 1RM) regardless of age, gender and activity level after a short period of training with mean significant adaptations at ~6.7-weeks. These data correlate with both the training and placebo (control) groups in each trial and the supplement intervention groups with all groups experiencing augmented strength adaptation, and ~27% of the studies demonstrating significant differences between the placebo and the intervention groups. With the knowledge that initial adaptation of skeletal muscle when high-magnitude mechanical modalities are applied results in augmented muscular strength adaptation, the ingestion of supplements that could potentiate this effect to a greater extent need to be measured alongside tendinous tissue response. The potential differentiation in adaptation could create additional influence on the associated tendon tissue that may not respond as expediently as muscle tissue, resulting in potential tendinopathy. Moreover, an athletes’ coach/trainer must be made aware of the potential counterintuitive nature of supplementation and that high-magnitude resistance training may elicit strength gains faster than expected as a result of exogenous nutritional support. Therefore, a nutritional framework alongside training regimes needs to be applied synergistically with repeated testing to ensure potential injury risk is limited. Furthermore, future studies need to look directly at the temporal dynamics of muscular and tendon tissue strength adaptation in relation to highmagnitude mechanical modulation with supplement intervention to explore potential discrepancies in time adaptation as to prevent vicissitudes.
References
- Hickson R C, Hidaka K, Foster C, Falduto M T, Chatterton Jr R T, et al. (1994) Successive time courses of strength development and steroid hormone responses to heavy-resistance training. Journal of Applied Physiology 76 (2): 663-670.
- Fimland MS, Helgerud J, Solstad G M, Iversen V M, Leivseth G, et al. (2009) Neural adaptations underlying cross-education after unilateral strength training. European Journal of Applied Physiology 107(6): 723.
- Cermak N M, Res P T, de Groot L C, Saris W H, van Loon L J, et al. (2012) Protein supplementation augments the adaptive response of skeletal muscle to resistance-type exercise training: A meta-analysis. The American Journal of Clinical Nutrition 96(6): 1454-1464.
- Schuenke MD, Herman J R, Gliders R M, Hagerman F C, Hikida R S, et al. (2012) Early-phase muscular adaptations in response to slow-speed versus traditional resistance-training regimens. European Journal of Applied Physiology 112(10): 3585-3595.
- Hirayama K, Iwanuma S, Ikeda N, Yoshikawa A, Ema R, et al. (2017) Plyometric training favors optimizing muscle–tendon behavior during depth jumping. Frontiers in Physiology 8: 16.
- Häkkinen K, Kraemer W J, Newton R U, Alen M (2001a) Changes in electromyographic activity, muscle fibre and force production characteristics during heavy resistance/power strength training in middle‐aged and older men and women. Acta Physiologica Scandinavica 171(1): 51-62.
- Verdijk L B, Gleeson B G, Jonkers R A, Meijer K, Savelberg H H, et al. (2009) Skeletal muscle hypertrophy following resistance training is accompanied by a fiber type–specific increase in satellite cell content in elderly men. Journals of Gerontology Series A: Biomedical Sciences and Medical Sciences 64(3): 332-339.
- Aagaard P, Andersen JL, Dyhre‐Poulsen P, Leffers A, Wagner A, et al. (2001a) A mechanism for increased contractile strength of human pennate muscle in response to strength training: Changes in muscle architecture. The Journal of Physiology 534(2): 613-623.
- Patten C, Kamen G, Rowland D M (2001) Adaptations in maximal motor unit discharge rate to strength training in young and older adults. Muscle & Nerve: Official Journal of the American Association of Electrodiagnostic Medicine 24(4): 542-550.
- Tillin NA, Folland J P (2014) Maximal and explosive strength training elicit distinct neuromuscular adaptations, specific to the training stimulus. European Journal of Applied Physiology 114(2): 365-374.
- Aagaard P, Bojsen-Møller J, Lundbye-Jensen J (2020) Assessment of neuroplasticity with strength training. Exercise and Sport Sciences Reviews 48(4): 151-162.
- Duchateau J, Stragier S, Baudry S, Carpentier A (2021) Strength training: In search of optimal strategies to maximize neuromuscular performance. Exercise and Sport Sciences Reviews 49(1): 2-14.
- Haun C T, Vann C G, Osburn S C, Mumford P W, Roberson P A, et al. (2019) Muscle fiber hypertrophy in response to 6 weeks of high-volume resistance training in trained young men is largely attributed to sarcoplasmic hypertrophy. PLoS One 14(6): e0215267.
- Andrews RD, MacLean D A, Riechman S E (2006) Protein intake for skeletal muscle hypertrophy with resistance training in seniors. International Journal of Sport Nutrition and Exercise Metabolism 16(4): 362-372.
- Jeukendrup A, Gleeson M (2018) Sport nutrition. Human Kinetics.
- Lixandrao M E, Ugrinowitsch C, Berton R, Vechin FC, Conceição MS, et al. (2018) Magnitude of muscle strength and mass adaptations between high-load resistance training versus low-load resistance training associated with blood-flow restriction: A systematic review and meta-analysis, Sports Medicine 48(2): 361-378.
- Proske U, Morgan D L (2001) Muscle damage from eccentric exercise: Mechanism, mechanical signs, adaptation and clinical applications. The Journal of Physiology 537(2): 333-345.
- Yang Y, Breen L, Burd N A, Hector A J, Churchward-Venne TA, et al. (2012) Resistance exercise enhances myofibrillar protein synthesis with graded intakes of whey protein in older men. British Journal of Nutrition 108(10): 1780-1788.
- Mersmann F, Bohm S, Schroll A, Marzilger R, Arampatzis A, et al. (2016) Athletic training affects the uniformity of muscle and tendon adaptation during adolescence. Journal of Applied Physiology 121(4): 893-899.
- Campos G E, Luecke T J, Wendeln HK, Toma K, Hagerman F C, et al. (2002) Muscular adaptations in response to three different resistance-training regimens: Specificity of repetition maximum training zones. European Journal of Applied Physiology 88(1-2): 50-60.
- Kadi F, Schjerling P, Andersen L L, Charifi N, Madsen J L, et al. (2004) The effects of heavy resistance training and detraining on satellite cells in human skeletal muscles. The Journal of Physiology 558(3): 1005-1012.
- Drummond M J, Fry C S, Glynn E L, Timmerman K L, Dickinson J M, et al. (2011) Skeletal muscle amino acid transporter expression is increased in young and older adults following resistance exercise. Journal of Applied Physiology 111(1): 135-142.
- Mitchell C J, Churchward-Venne T A, West D W, Burd N A, Breen L, et al. (2012) Resistance exercise load does not determine training-mediated hypertrophic gains in young men. Journal of Applied Physiology 113(1): 71-77.
- Vierck J, O Reilly B, Hossner K, Antonio J, Byrne K, Bucci L, et al. (2000) Satellite cell regulation following myotrauma caused by resistance exercise. Cell Biology International 24(5): 263-272.
- Verdijk L B, Jonkers R A, Gleeson B G, Beelen M, Meijer K, et al. (2009b) Protein supplementation before and after exercise does not further augment skeletal muscle hypertrophy after resistance training in elderly men. The American Journal of Clinical Nutrition 89(2): 608-616.
- Esmarck B, Andersen JL, Olsen S, Richter E A, Mizuno M, et al. (2001a) Timing of postexercise protein intake is important for muscle hypertrophy with resistance training in elderly humans. The Journal of Physiology 535(1): 301-311.
- Esmarck B, Andersen J L, Olsen S, Richter E A, Mizuno M, et al. (2001b) Timing of postexercise protein intake is important for muscle hypertrophy with resistance training in elderly humans. The Journal of Physiology 535(1): 301-311.
- Volek J S (2003) Strength nutrition. Current Sports Medicine Reports 2(4): 189-193.
- Berardi J M, Price T B, Noreen EE, Lemon P W (2006) Postexercise muscle glycogen recovery enhanced with a carbohydrate-protein supplement. Medicine and Science in Sports and Exercise 38(6): 1106-1113.
- Verdijk L B, Gleeson B G, Jonkers R A, Meijer K, Savelberg H H, et al. (2009) Skeletal muscle hypertrophy following resistance training is accompanied by a fiber type–specific increase in satellite cell content in elderly men. Journals of Gerontology Series A: Biomedical Sciences and Medical Sciences 64(3): 332-339.
- Verdijk L B, Jonkers R A, Gleeson B G, Beelen M, Meijer K, et al. (2009a) Protein supplementation before and after exercise does not further augment skeletal muscle hypertrophy after resistance training in elderly men. The American Journal of Clinical Nutrition 89(2): 608-616.
- Atherton P J, Etheridge T, Watt PW, Wilkinson D, Selby A, et al. (2010) Muscle full effect after oral protein: Time-dependent concordance and discordance between human muscle protein synthesis and mTORC1 signaling, The American Journal of Clinical Nutrition 92(5): 1080-1088.
- McGlory C, Devries M C, Phillips S M (2017) Skeletal muscle and resistance exercise training; the role of protein synthesis in recovery and remodeling. Journal of Applied Physiology 122(3): 541-548.
- Baker L B, Heaton L E, Nuccio R P, Stein K W (2014) Dietitian-observed macronutrient intakes of young skill and team-sport athletes: Adequacy of pre, during, and postexercise nutrition. International Journal of Sport Nutrition and Exercise Metabolism 24(2): 166-176.
- Morton R W, McGlory C, Phillips S M (2015) Nutritional interventions to augment resistance training-induced skeletal muscle hypertrophy. Frontiers in Physiology 6: 245.
- Drummond M J, Rasmussen BB (2008) Leucine-enriched nutrients and the regulation of mTOR signalling and human skeletal muscle protein synthesis. Current Opinion in Clinical Nutrition and Metabolic Care 11(3): 222.
- Layman D K (2002) Role of leucine in protein metabolism during exercise and recovery. Canadian Journal of Applied Physiology 27(6): 646-662.
- Gil JH, Kim CK (2015) Effects of different doses of leucine ingestion following eight weeks of resistance exercise on protein synthesis and hypertrophy of skeletal muscle in rats. Journal of Exercise Nutrition & Biochemistry 19(1): 31.
- Wilder N, Deivert RG, Hagerman F, Gilders R (2001) The effects of low-dose creatine supplementation versus creatine loading in collegiate football players. Journal of Athletic Training 36(2): 124-129.
- Kreider R B (2003) Effects of creatine supplementation on performance and training adaptations, Molecular and Cellular Biochemistry 244(1-2): 89-94.
- Spillane M, Schoch R, Cooke M, Harvey T, Greenwood M, et al. (2009) The effects of creatine ethyl ester supplementation combined with heavy resistance training on body composition, muscle performance, and serum and muscle creatine levels. Journal of the International Society of Sports Nutrition 6(1): 6.
- Gallagher Pm, Carrithers Ja, Godard M P, Schulze K E, Trappe S W, et al. (2000) Β-hydroxy-β-methylbutyrate ingestion, part I: Effects on strength and fat free mass. Medicine & Science in Sports & Exercise 32 (12): 2109-2115.
- Flakoll P, Sharp R, Baier S, Levenhagen D, Carr C, et al. (2004) Effect of β-hydroxy-β-methylbutyrate, arginine, and lysine supplementation on strength, functionality, body composition, and protein metabolism in elderly women. Nutrition 20(5): 445-451.
- Thomson J S (2004) Β-hydroxy-β-methylbutyrate (HMB) supplementation of resistance trained men. Asia Pacific Journal of Clinical Nutrition 13.
- Silva V R, Belozo F L, Micheletti T O, Conrado M, Stout J R, et al. (2017) Β-hydroxy-β-methylbutyrate free acid supplementation may improve recovery and muscle adaptations after resistance training: A systematic review. Nutrition Research 45: 1-9.
- SALİMİ HH, RAHMİNİ-NİA F (2019) The effect of beta-hydroxy-beta-Methylbutyrate Supplementation on performance adaptations following Resistance Training in young males. Turkish Journal of Sport and Exercise 21(2): 349-354.
- Nissen S L, Sharp R L (2003) Effect of dietary supplements on lean mass and strength gains with resistance exercise: A meta-analysis. Journal of Applied Physiology 94(2): 651-659.
- Gabriel DA, Kamen G, Frost G (2006) Neural adaptations to resistive exercise. Sports Medicine 36(2): 133-149.
- Wernbom M, Augustsson J, Thomeé R (2007) The influence of frequency, intensity, volume and mode of strength training on whole muscle cross-sectional area in humans. Sports medicine 37(3): 225-264.
- Carroll T J, Selvanayagam V S, Riek S, Semmler J G (2011) Neural adaptations to strength training: Moving beyond transcranial magnetic stimulation and reflex studies. Acta Physiologica 202(2): 119-140.
- Vandenberghe K, Goris M, Van Hecke P, Van Leemputte M, et al. (1997) Long-term creatine intake is beneficial to muscle performance during resistance training. Journal of Applied Physiology 83(6): 2055-2063.
- Arazi H, Asadi A, Suzuki K (2018) The effects of beta-hydroxy-beta-methylbutyrate-free acid supplementation and resistance training on oxidative stress markers: A randomized, double-blind. placebo-controlled study. Antioxidants 7(6): 76.
- Tarnopolsky M A (2008) Building muscle: Nutrition to maximize bulk and strength adaptations to resistance exercise training. European Journal of Sport Science 8(2): 67-76.
- Trabal J, Forga M, Leyes P, Torres F, Rubio J, et al. (2015) Effects of free leucine supplementation and resistance training on muscle strength and functional status in older adults: A randomized controlled trial. Clinical Interventions in Aging 10: 713-723.
- Volek JS, Duncan ND, Mazzetti S A, Staron R S, Putukian M, et al. (1999) Performance and muscle fiber adaptations to creatine supplementation and heavy resistance training. Medicine and Science in Sports and Exercise 31(8): 1147-1156.
- Stout J R, Smith-Ryan AE, Fukuda D H, Kendall K L, Moon J R, et al. (2013) Effect of calcium β-hydroxy-β-methylbutyrate (CaHMB) with and without resistance training in men and women 65 yrs: A randomized, double-blind pilot trial, Experimental Gerontology 48(11): 1303-1310.
- Yarasheski K E, Zachwieja JJ, Bier D M (1993) Acute effects of resistance exercise on muscle protein synthesis rate in young and elderly men and women. American Journal of Physiology-Endocrinology and Metabolism 265(2): E210-E214.
- Barbosa A W, Benevides GP, Alferes L M T, Salomão E M, Gomes-Marcondes M C C, et al. (2012) A leucine-rich diet and exercise affect the biomechanical characteristics of the digital flexor tendon in rats after nutritional recovery. Amino Acids 42(1): 329-336.
- Farup J, Rahbek S K, Vendelbo MH, Matzon A, Hindhede J, et al. (2014) Whey protein hydrolysate augments tendon and muscle hypertrophy independent of resistance exercise contraction mode. Scandinavian Journal of Medicine & Science in Sports 24(5): 788-798.
- Epro G, Hunter S, König M, Schade F, Karamanidis K, et al. (2019) Evidence of a uniform muscle-tendon unit adaptation in healthy elite track and field jumpers: A cross sectional investigation. Frontiers in Physiology 10: 574.
- Bohm S, Mersmann F, Tettke M, Kraft M, Arampatzis A, et al. (2014) Human achilles tendon plasticity in response to cyclic strain: Effect of rate and duration. Journal of Experimental Biology 217(22): 4010-4017.
- Brose A, Parise G, Tarnopolsky M A (2003) Creatine supplementation enhances isometric strength and body composition improvements following strength exercise training in older adults. The Journals of Gerontology Series A: Biological Sciences and Medical Sciences 58(1): B11-B19.
- Brown N, Bubeck D, Haeufle D FB, Weickenmeier J, Kuhl E, et al. (2017) Weekly time course of neuro-muscular adaptation to intensive strength training.
- Din U, Brook M S, Selby A, Quinlan J, Boereboom C, et al. (2019) A double-blind placebo-controlled trial into the impacts of HMB supplementation and exercise on free-living muscle protein synthesis, muscle mass and function, in older adults. Clinical Nutrition 38(5): 2071-2078.
- Erskine R M, Fletcher G, Hanson B, Folland J P (2012) Whey protein does not enhance the adaptations to elbow flexor resistance training. Medicine and Science in Sports and Exercise 44(9): 1791-1800.
- Galloway MT, Lalley A L, Shearn J T (2013) The role of mechanical loading in tendon development, maintenance, injury, and repair. The Journal of Bone and Joint Surgery.American Volume 95(17): 1620-1628.
- Garazhian Y, Azimkhani A (2014) Combined effects of prolonged creatine supplementation and resistance training on muscular strength and body composition in collegiate athletes. Pamukkale Journal of Sport Sciences 5(2).
- Häkkinen K, Kraemer W J, Newton R U, Alen M (2001) Changes in electromyographic activity, muscle fibre and force production characteristics during heavy resistance/power strength training in middle‐aged and older men and women. Acta Physiologica Scandinavica 171(1): 51-62.
- Heinemeier K M, Schjerling P, Heinemeier J, Magnusson S P, Kjaer M, et al. (2013) Lack of tissue renewal in human adult achilles tendon is revealed by nuclear bomb 14C. The FASEB Journal 27(5): 2074-2079.
- Katsanos CS, Kobayashi H, Sheffield-Moore M, Aarsland A, Wolfe RR, et al. (2006) A high proportion of leucine is required for optimal stimulation of the rate of muscle protein synthesis by essential amino acids in the elderly. American Journal of Physiology-Endocrinology and Metabolism 291(2): E381-E387.
- keeler L K, Finkelstein L H, Miller W, Fernhall B O (2001) Early-phase adaptations of traditional-speed vs. superslow resistance training on strength and aerobic capacity in sedentary individuals. The Journal of Strength & Conditioning Research 15(3): 309-314.
- Kerksick C M, Rasmussen C J, Lancaster S L, Magu B, Smith P, et al. (2006) The effects of protein and amino acid supplementation on performance and training adaptations during ten weeks of resistance training. The Journal of Strength & Conditioning Research 20(3): 643-653.
- Kerksick C, Rasmussen C, Lancaster S, Magu B, Smith P, et al. (2006) The effects of protein and amino acid supplementation on performance and training adaptations during ten weeks of resistance training, Journal of Strength and Conditioning Research 20(3): 643-653.
- Kjær M (2004) Role of extracellular matrix in adaptation of tendon and skeletal muscle to mechanical loading. Physiological Reviews 84(2): 649-698.
- Kubo K, Ikebukuro T, Maki A, Yata H, Tsunoda N, et al. (2012) Time course of changes in the human achilles tendon properties and metabolism during training and detraining in vivo. European Journal of Applied Physiology 112(7): 2679-2691.
- Kubo K, Ikebukuro T, Yata H, Tsunoda N, Kanehisa H, et al. (2010) Time course of changes in muscle and tendon properties during strength training and detraining. The Journal of Strength & Conditioning Research 24(2): 322-331.
- Labata-Lezaun N, Llurda-Almuzara L, López-de-Celis C, Rodríguez-Sanz J, González-Rueda V, et al. (2020) Effectiveness of protein supplementation combined with resistance training on muscle strength and physical performance in elderly: A systematic review and meta-analysis. Nutrients 12(9): 2607.
- Leenders M, Verdijk L B, Van der Hoeven L, Van Kranenburg J, Nilwik R, et al. (2013) Protein supplementation during resistance-type exercise training in the elderly. Medicine and Science in Sports and Exercise 45(3): 542-552.
- Macnaughton L S, Wardle S L, Witard O C, McGlory C, Hamilton D L, et al. (2016) The response of muscle protein synthesis following whole‐body resistance exercise is greater following 40 g than 20 g of ingested whey protein. Physiological Reports 4(15): e12893.
- Mateos-Angulo A, Galán-Mercant A, Cuesta-Vargas A I (2020) Kinematic mobile drop jump analysis at different heights based on a smartphone inertial sensor. Journal of Human Kinetics 73(1): 57-65.
- Meyers M A, Chawla K K (2008) Mechanical behavior of materials. Cambridge university press.
- Miller B F, Hansen M, Olesen J L, Schwarz P, Babraj J A, et al. (2007) Tendon collagen synthesis at rest and after exercise in women. Journal of Applied Physiology 102(2): 541-546.
- Muramatsu T, Muraoka T, Takeshita D, Kawakami Y, Hirano Y, et al. (2001) Mechanical properties of tendon and aponeurosis of human gastrocnemius muscle in vivo. Journal of Applied Physiology 90(5): 1671-1678.
- Musiał K, Wiski M, Lipert A (2020) Protein intake among healthy adults undertaking regular muscle strength training. Medical Science Pulse 14(2): 23-29.
- Nabuco H C, Tomeleri C M, Fernandes R R, Junior P S, Cavalcante E F, et al. (2019) Effects of protein intake beyond habitual intakes associated with resistance training on metabolic syndrome-related parameters, isokinetic strength, and body composition in older women. Journal of Aging and Physical Activity 27(4): 545-552.
- Radaelli R, Botton CE, Wilhelm E N, Bottaro M, Brown LE, et al. (2014) Time course of low-and high-volume strength training on neuromuscular adaptations and muscle quality in older women Age 36(2): 881-892.
- Scaglioni G, Ferri A, Minetti A E, Martin A, Van Hoecke J, et al. (2002) Plantar flexor activation capacity and H reflex in older adults: Adaptations to strength training. Journal of Applied Physiology 92(6): 2292-2302.
- Stout J R, Smith-Ryan A E, Fukuda D H, Kendall K L, Moon J R, et al. (2013) Effect of calcium β-hydroxy-β-methylbutyrate (CaHMB) with and without resistance training in men and women 65 yrs: A randomized, double-blind pilot trial. Experimental Gerontology 48 (11): 1303-1310.
- Stragier S, Baudry S, Poortmans J, Duchateau J, Carpentier A, et al. (2016) Leucine-enriched protein supplementation does not influence neuromuscular adaptations in response to a 6-month strength training programme in older adults. Experimental Gerontology 82: 58-66.
- Tipton K D (2008) Protein for adaptations to exercise training, European Journal of Sport Science 8(2): 107-118.
- Villanueva M G, He J, Schroeder E T (2014) Periodized resistance training with and without supplementation improve body composition and performance in older men. European Journal of Applied Physiology 114(5): 891-905.
- Wilson JM, Lowery R P, Joy J M, Andersen J C, Wilson S M, et al. (2014) The effects of 12 weeks of beta-hydroxy-beta-methylbutyrate free acid supplementation on muscle mass, strength, and power in resistance-trained individuals: A randomized, double-blind, placebo-controlled study. European Journal of Applied Physiology 114(6): 1217-1227.
- Witard OC, Sarah R Jackman, Leigh Breen, Kenneth Smith, Anna Selby, et al. (2015) Myofibrillar muscle protein synthesis rates subsequent to a meal in response to increasing doses of whey protein at rest and after resistance exercise. American Journal of Clinical Nutrition 99(1): 86-95.

 Review Article
Review Article