ABSTRACT
The report presents a case of a patient with chronic neuropathic pain of the CRPS type II due to tibial neuroma that arose after failed tarsal release surgery. Despite the local and systemic pharmacological treatment, despite numerous sessions of manual therapy and physical therapy, the use of behavioral therapy and lignocaine infusions, topical botulinum toxin, hyaluronidase, and autologous serum, as well as multiple pulsed radiofrequencies, the patient developed a chronic pain syndrome that is only partially relieved by the use of a spinal cord stimulator and constant intake of anticonvulsants and opioids. The cryoanalgesia procedures performed several times turned out to be the only form of therapy that clearly relieves pain for a period of several months. The report describes the methodology of the indirect technique of cryoanalgesia under ultrasound control.
Keywords: Pain; Pain Therapy; Painful Neuromas; Cryoanalgesia
Introduction
Cryoanalgesia is a mini-invasive neurodestructive (ablative) procedure used to temporarily eliminate the pain signal conducted by the sensory branches through the action of low temperature. Due to the development of probes in the shape of long, rigid cannulas that allow the expansion of the gas (CO2, NO) circulating inside the probe, a temperature drop to -70 degrees C is achieved at the end of the active probe. This results in a mechanism called “heat theft” from the environment and the formation of the ice ball by freezing the water from the surrounding tissues. The mechanism of disruption of conduction is attributed to damage to Schwann cells while maintaining the continuity of the basement membrane and to micro embolisms in the nerve nutrient vessels. The method is used in the treatment of acute and chronic pain, and the denervation time is much longer than the duration of regional blocks, even with the use of continuous anesthesia catheters, and is even several months with minimal risk of infection and no risk of catheter removal [1,2]. The diameter of the ice ball formed at the top of the probe depends on the degree of tissue hydration, tissue blood supply, and freezing time, reaching even 1 cm in diameter [3]. Thanks to the coupling of the probe with the nerve stimulator and the use of ultrasound, X-ray, or CT imaging, very precise and safe percutaneous neuro destructive treatments have become possible. These treatments have been used for several decades in pain medicine and have found a very wide application, especially in the treatment of chronic pain in the spine of articular origin due to the possibility of precise identification and damage to the pain generator, i.e. medial branches departing from the dorsal spinal nerves without the risk of damaging the motor branches (nerve roots). Other applications include treatment of cervicogenic headaches, perineal pain, palliative denervation of peripheral joints in the course of severe osteoarthritis of the knee and hip (also preceding arthroplasty), and treatment of post-herpetic neuralgia or pain after rib fracture [4-12].
Cryoanalgesia, however, is also used wherever new pathological innervation arises, as in the case of a new network of periosteal sensory fibers in the sites of proliferative changes in osteoarthritis and musculotendinous attachments in tendinopathy and enthesopathy, or where, in the event of a change in the structure of the nerve, stump neuromas develop after amputations or in scars after unsuccessful surgeries to release the nerve entrapment syndromes [13-15]. The presented case relates precisely to such a situation where the chronic pain syndrome of the right foot fulfills the hallmarks of the failed tarsal release tunnel with the formation of a chronic pain syndrome of type II Complex Regional Pain Syndrome (CRPS) with typical allodynia, dysesthesia, and poor response for pharmacological treatment.
History of Complaints and Therapy
Female patient M.O. born in 2001 suffered a torsion right ankle injury in 2011. Treated conservatively, on an outpatient basis, locally and systemically anti-inflammatory with periodic plaster immobilization. The first available information sheet comes from 12/2014 when she was admitted to the children’s orthopedics department due to inflammation of the right ankle joint. In the epicrisis information on a three-year history of pain in the ankle joint previously treated with plaster casts, with CRP 16 mg / L, then 34 mg / L, was reported, and after antibiotic therapy and steroid therapy with temporary immobilization, the CRP was reduced to 1 mg / L. X-ray without evidence of bone destruction and without periosteal reactions, in scintigraphy, the hot area in the right ankle joint. Re-admitted to the same ward after 3 weeks with the diagnosis of Achilles tendonitis, and after local administration of steroid and plaster immobilization, she was discharged home with the recommendation of a follow-up in 2 weeks.
Another hospitalization on 02/2015 due to recurrent pain in the right ankle joint in the area of the medial ankle, laboratory tests showed low levels of Vit D (10 ng / ml), supplementation, and local physical treatment were included, and rheumatoid and neurological diseases were excluded. On 03/2015, a CT examination of the right ankle joint revealed an oval, hypodense lesion measuring 12 x 9 x 14 mm in the vicinity of the Flexor Halucis Longus tendon (FHL), but without specifying the diagnosis and possible relation to the symptoms. On 03/2015 stay in a rehabilitation center – for local physical treatment. On 04/2015, a recurrence of the pain - admission to the pediatric orthopedic department, treated with systemic and local anti-inflammatory drugs, and recommendation of orthopedic insoles - without making a specific diagnosis. On 10/2015, an MRI of the right ankle joint was performed with contrast, revealing an increased amount of fluid in the FHL tendon sheaths - otherwise, the image was normal.
Several visits to the emergency during an exacerbation of symptoms in 2016, but without elevated inflammatory markers, and treated with symptomatic anti-inflammatory treatment each time. On 05/2016, in the ultrasound examination, vascular disorders were excluded, but a hypoechoic lesion with a length of 8 mm was found along the tibial nerve, in the sinus of the steppe, with the conclusion of the description “possible scar after administration of the drug to the nerve”. The performed EMG (05/2016) revealed a slower conduction velocity in the medial plantar nerve. Another MRI scan found no damage to the tibial nerve. Nevertheless, in 06/2016 the patient was operated on and the indication for surgery was “tibial and medial plantar neuralgia and Baxter branch entrapment”. Relapse of the neuralgia occurred on 09/2016, Neurontin (Gabapentin) 3x300 mg was then recommended. The ultrasound examination on 10/2016 revealed extensive scarring involving the tibial nerve. Re-operated on 10/2016 - tibial nerve neurolysis and removal of scars complicated by postoperative wound dehiscence and prolonged skin healing. The prolonged postoperative pain was explained by the pressure of the tibial nerve entrapped into the scars, which was visualized in the next MRI examination in 2017. The attempt of botulinum injections and hyaluronidase in the area of the scar and pulsed radiofrequency of the tibial nerve in 2017 were unsuccessful. Due to the lack of improvement, for the third time the tibial neurolysis was performed on 11/2017, unfortunately without success.
In 2018, she was treated topically with the autologous conditioned serum (Orthokine) - 4 doses of ultrasound-guided infiltration of the tibial nerve surrounding without success. The only significant reduction in the intensity of neuropathic pain, sometimes to its complete abolition, although short-term, because only a few hours, was obtained after intravenous lignocaine infusions at a dose of 1.5 - 3 mg/kg / h calculated using the patient’s ideal body weight and given as an infusion over 20-30 min. Parallel to the lignocaine infusions, the mirror therapy used to desensitize the limb through behavioral patterns was also unsuccessful. The patient was qualified for spinal cord stimulator implantation with external stimulation, the procedure was performed on 06/2018 (with admission pain VAS 8-9, after its application pain reduction to VAS 4-5). Since then, the patient uses the pacemaker (Precision Montage™ MRI) at the time of exacerbation of symptoms, achieving a pain reduction by 2 points (with an average rating of 5-6), using a several-hour stimulation with the Burst program, but the pain has never been fully muted despite the constant intake of pregabalin (2x75 mg) and periodically Tramadol (50-100 mg).
Since 2019, the patient has undergone five cryoanalgesia treatments of tibial nerve neuroma in the area of postoperative scarring. According to the patient’s report, each of them reduces neuropathic pain with hyperalgesia by 3-4 points for several months (4-5 months on average), but it also does not completely eliminate it. For the patient, however, it is the only option so far to significantly reduce hyperalgesia and pain, without the risk of damage to the nerve trunk and loss of motor function of the foot.
Cryoanalgesia Technique
Depending on the type of pain generator, there are three main techniques for cryoanalgesia.
1) Classic technique - cryoablation of the sensory branch after effective sensory and motor stimulation - the most common model of the treatment in the area of the spine, where the goal is the medial branch innervating the intervertebral joints.
2) Periosteal technique - freezing of the pathological innervation around proliferative remodeling lesions on the tendon and ligament attachments or in the course of the osteoarthritis.
3) Indirect technique “in the lake” - in a situation that is impossible to identify with imaging techniques of a nerve branch due to its very small diameter (fascial or skin fine branches) or the edges of the neuroma in the postoperative scar, the technique of regional infiltration with lignocaine or saline and freezing in a block is used.
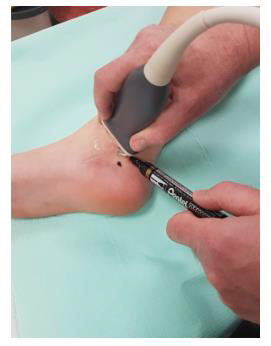
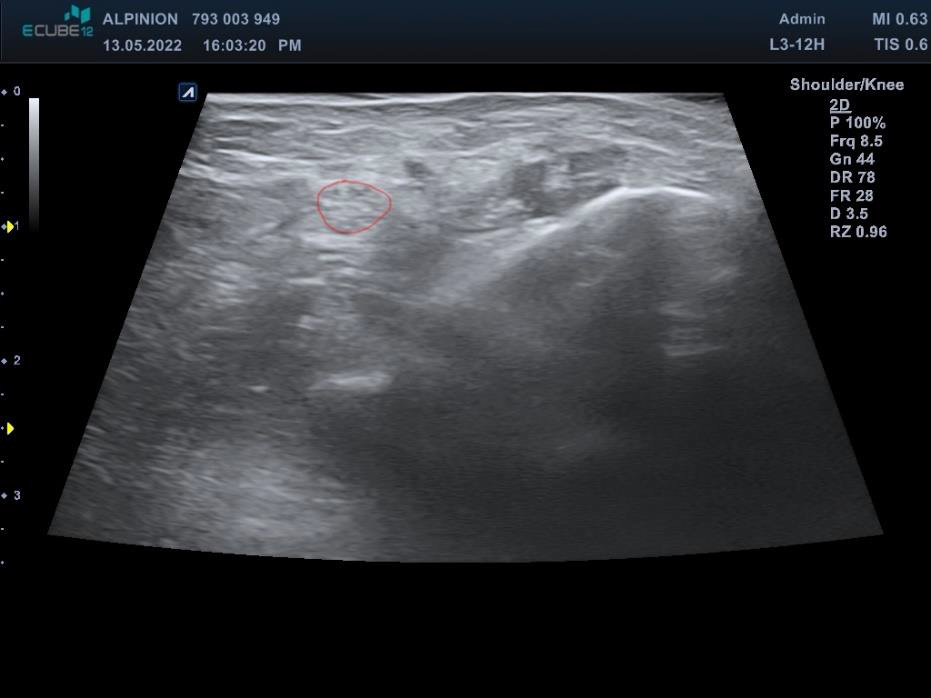
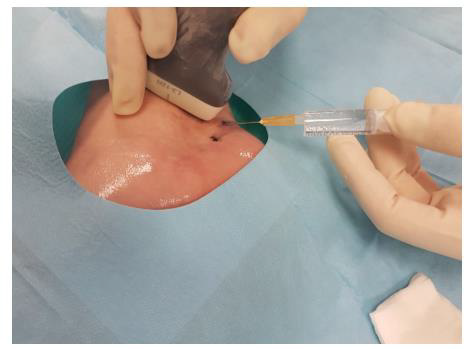
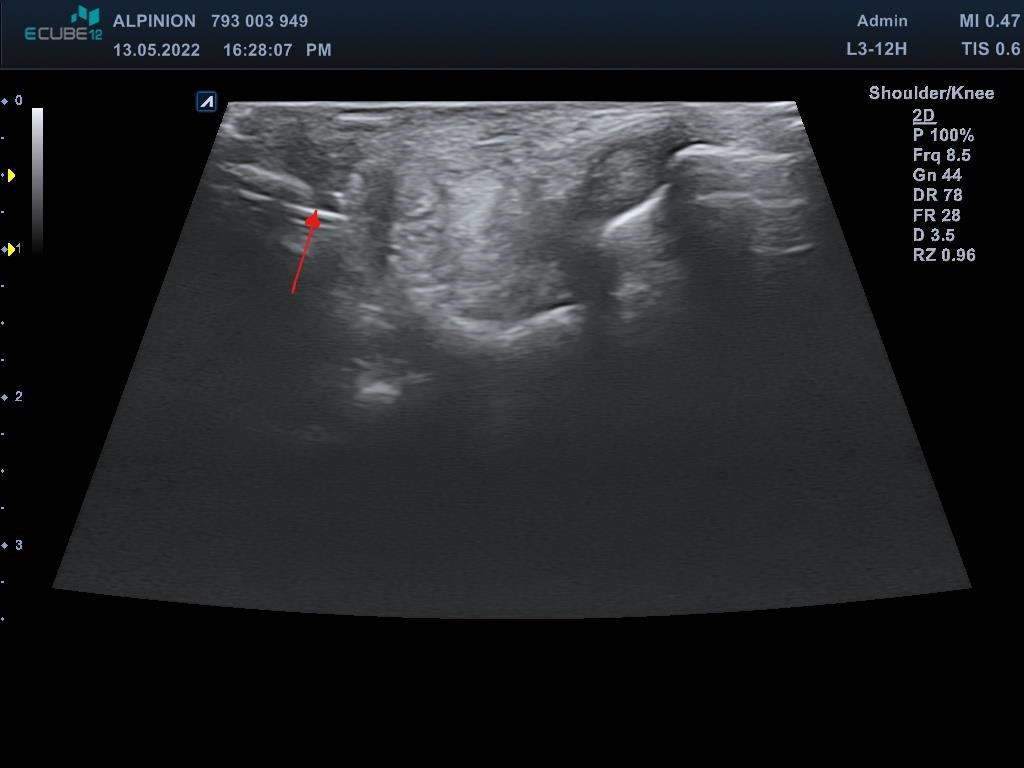
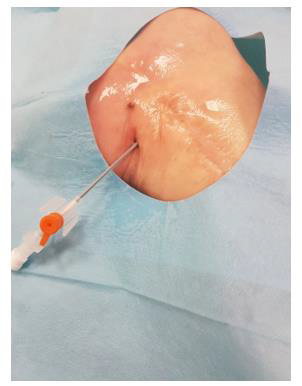
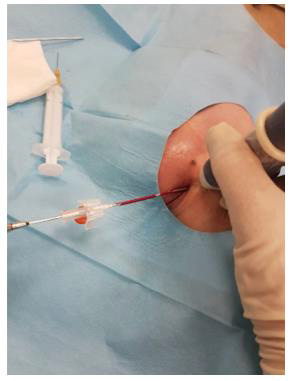
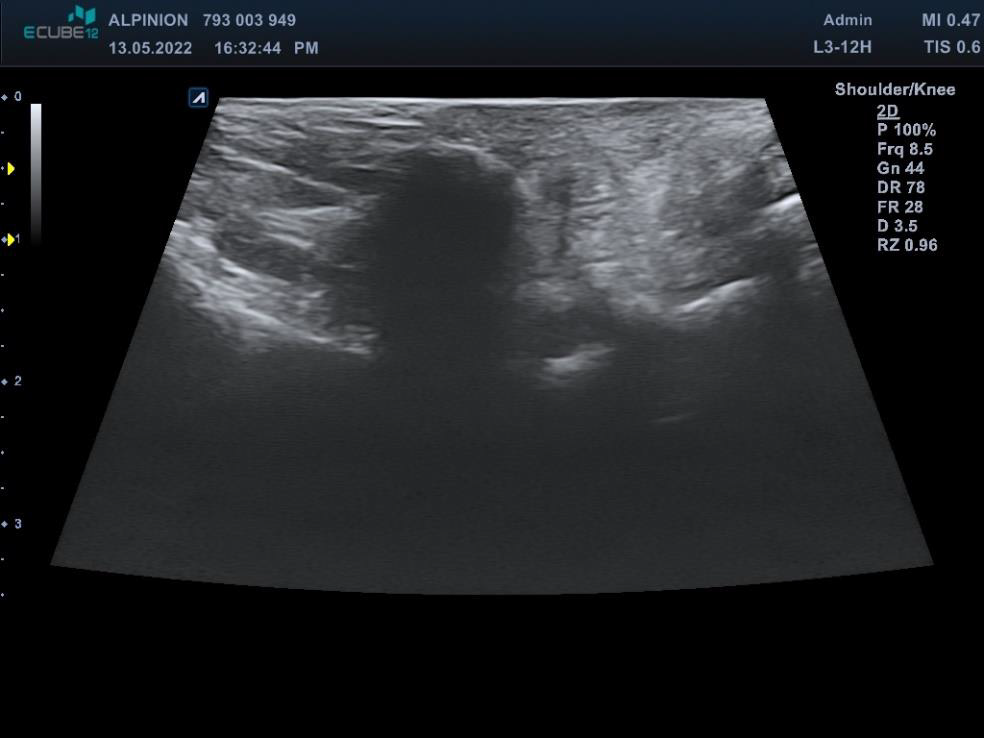
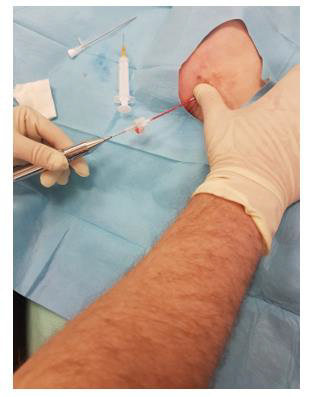
This third variant was used in the described case. In the first phase, the tibial nerve in the zone of its greatest tenderness was identified under ultrasound control, marking the boundaries of its course on the skin (Figures 1 & 2). Then, under ultrasound guidance, a thin needle was inserted towards the lateral wall of the neuroma, and lignocaine was infiltrated into the soft tissues forming scarring entrapment of the tibial nerve (Figures 3 & 4). A transcutaneous cannula was inserted through the path of the first needle and its position in the center of the fluid-soaked area was determined under ultrasound guidance (Figure 5). Then the cryoablation probe was inserted through the cannula and 2 cycles of 1.5 min freezing at minus 70 deg. C was performed with a 30-sec interval between the freezing cycles (Figure 6). During freezing, a growing ice ball was observed on the lateral wall of the tibial nerve in the area of the neuroma, and the skin was protected against frostbite by warming it with fingertips (Figures 7 & 8). After removing the probe, a pressure dressing was made.
Discussion
Cryoablation of neuromas is one of the widely recognized methods of treating chronic neuropathic pain caused by them [16,17]. In most cases, they result from the cutting of nerves in the amputated limb or crushing of the nerves during high-energy trauma and as such can be cryoanalgesia over an extended period of time until they are completely destroyed. A different situation is created by partial injuries of active large nerve trunks containing motor fibers, which form intraneural neuromas with the ectopic activity of the damaged nerve, and which should be differentiated from tumor-like neurogenic lesions [18,19]. The reason for the production of such neuromas is partial nerve damage - cuts, injection injuries, scar entrapment with chronic ischemia due to pressure. Then the cryoablation must be weak enough not to damage the motor fibers and effective enough to break spontaneous discharges and conductivity in the emphases. For this reason, the cryoanalgesia probe cannot directly adhere to the nerve trunk itself, and the lowered temperature zone transmits like an echo from adjacent infiltrated tissues sufficiently effectively to damage the thin unmyelinated newly formed pain-conducting fibers and not damage the myelin sheath and Schwann cells of the motor fibers. Thanks to ultrasound control, it is possible to infiltrate the area around the damaged nerve and to dose the freezing time in such a way (from 30 sec to 1.5 min) so that, by following the dimensions of the ice ball, it does not cause irreversible damage to the motor fibers. A similar technique can be successfully used in post-herpetic pain syndromes for cutaneous and fascial branches, without the need for cryoablation of the intercostal nerves, thanks to which there is no denervation of the intercostal muscles, the subpatellar branch of the saphenous nerve without the need to freeze the main nerve trunk or finally cryoanalgesia the suprascapular nerve without risk of the supraspinatus and infraspinatus denervation.
Conclusion
The indirect cryoablation technique can be used to treat chronic neuroblastoma pain syndromes even in the immediate vicinity of a large nerve trunk without the risk of damaging the motor fibers.
References
- Trescot AM (2003) Cryoanalgesia in interventional pain management. Pain Physician 6(3): 345-360.
- Finneran Iv JJ, Ilfeld BM (2021) Percutaneous cryoneurolysis for acute pain management: current status and future prospects. Expert Rev Med Devices 18(6): 533-543.
- Kastler A, Gruber H, Gizewski E, Loizides A (2018) Ultrasound Assessment of Ice-Ball Formation by Cryoneurolysis Device in an Ex Vivo Model 43(6): 631-633.
- Cunha E Sa M, Behari S (2011) Cryoneurolysis for the treatment of lumbar zygoapophyseal joint pain. Acta Neurochir (Wien) 153(5): 1021-1022.
- Staender M, Maerz U, Tonn JC, Steude U (2005) Computerized tomography-guided kryorhizotomy in 76 patients with lumbar facet joint syndrome. J Neurosurg Spine 3(6): 444-449.
- Matthews BG, Hurn SE, Harding MP, Henry RA, Ware RS (2019) The effectiveness of non-surgical interventions for common plantar digital compressive neuropathy (Morton's neuroma): A systematic review and meta-analysis. J Foot Ankle Res 12: 12.
- Bittman RW, Peters GL, Newsome JM, Eric B Friedberg, Jason W Mitchell, et al. (2018) Percutaneous Image-Guided Cryoneurolysis. AJR Am J Roentgenol 210(2): 454-465.
- Radnovich R, Scott D, Patel AT, R Olson, V Dasa, et al. (2017) Cryoneurolysis to treat the pain and symptoms of knee osteoarthritis: A multicenter, randomized, double-blind, sham-controlled trial. Osteoarthritis Cartilage 25(8): 1247-1256.
- Evans PJD, Lloyd JW, Jack TM (1981) Cryoanalgesia for intractable perineal pain. J R Soc Med 74(11): 804-809.
- Byas-Smith MG, Gulati A (2006) Ultrasound-guided intercostal nerve cryoablation. Anesth Analg 103(4): 1033-1035.
- Urban JA, Dolesh K, Martin E (2021) A Multimodal Pain Management Protocol Including Preoperative Cryoneurolysis for Total Knee Arthroplasty to Reduce Pain, Opioid Consumption, and Length of Stay. Arthroplast Today 10: 87-92.
- Finneran Iv JJ, Gabriel RA, Swisher MW, Berndtson AE, Godat LN, et al. (2020) Ultrasound-guided percutaneous intercostal nerve cryoneurolysis for analgesia following traumatic rib fracture -A case series. Korean J Anesthesiol 73(5): 455-459.
- Caporusso EF, Fallat LM, Savoy-Moore R (2002) Cryogenic neuroablation for the treatment of lower extremity neuromas. J Foot Ankle Surg 41(5): 286-290.
- Yoon JH, Grechushkin V, Chaudhry A, Bhattacharji P, Durkin B, et al. (2016) Cryoneurolysis in Patients with Refractory Chronic Peripheral Neuropathic Pain. J Vasc Interv Radiol 27(2): 239-243.
- Gould JS (2011) The failed tarsal tunnel release. Foot Ankle Clin 16(2): 287-293.
- Ramsook RR, Spinner D (2017) Ultrasound-Guided Cryoablation of a Traumatic Hip Disarticulation Neuroma. Pain Pract 17(7): 941-944.
- Von Falck C, Orgel M, Wacker F, Aschoff HH, Krettek C, et al. (2022) Icing the Pain-Ultrasound-Guided Cryoablation of Symptomatic Post-Amputation Stump Neuroma. Cardiovasc Intervent Radiol 45(2): 223-227.
- Finnerup NB, Kuner R, Jensen TS (2021) Neuropathic Pain: From Mechanisms to Treatment. Physiol Rev 101(1): 259-301.
- Abreu E, Aubert S, Wavreille G, Gheno R, Canella C, et al. (2013) Peripheral tumor and tumor-like neurogenic lesions. Eur J Radiol 82(1): 38-50.

 Case Report
Case Report