ABSTRACT
Migraine is a disease of high prevalence, episodic, chronic and occupationally disproportionate for patients, presenting itself on a mundane level, meaning a public health problem, when in reality it is treated with hamburgers before migrating very diverse and comprehensive information with an arsenal of therapeutic options, preventive and bioconductive drugs, but it is also a multifactorial agent with clear points in treatment, which generates the need to investigate new therapies. The objective of this work is to keep up to date with the medications with which we are currently in the process of migration and to attend to the new treatment alternatives that have been emerging with the new studies that are being carried out.
Keywords: Migraine; New Alternatives; Treatment; Monoclonal Antibodies
Introduction
Migraine is a very prevalent pathology that is estimated to affect around 15% of adults and this represents the second cause of disabling neurological disease in the world. Migraine can be associated with morbidity and a negative impact on the quality of life of those who suffer from it. [1] As for epidemiological data, it is among the 10 main diseases that affect the general population, the estimated prevalence is 14.7% with predominance in women (17.7 vs 8%); according to estimates, it affects more than 44 million people in the United States and an estimated 170 million people in Europe. A study conducted in Latin America reported an overall prevalence of 8.5%, where Brazil had the highest figure with 12.6% and Argentina had the lowest rate with 5%, while in Mexico it was reported at 8%. [2,3] The frequency and intensity of pain episodes will vary depending on the factors of each individual, leading to consider appropriate the use of preventive medications to reduce the appearance of episodes, while, in other cases, this management will not be necessary [2].
The arsenal of drugs currently used as preventives will find calcium channel antagonists, antiepileptics, antidepressants, betablocker antihypertensives and botulinum toxin, among others, which, although they were developed for purposes other than migraine management, have currently been shown to be effective and safe in a group of patients with migraine without aura. [4] Thus, in recent years the efficacy and safety of monoclonal antibodies, which act in the peptide pathway related to the calcitonin gene (PRGC), one is specifically directed at the PRGC receptor (erenumab) and the other three of this family are directed at the peptide itself (fremanezumab, eptinezumab and galcanezumab). [5] These treatments have been shown to be effective and safe to date, which are an indication in the treatment of migraine in a given group of patients. Although studies on these treatments have been approved and their use has been incorporated into guidelines and consensus around the world, in many cases there is data from other regions that are alien to the reality of each region or country of the world in which it is located, as it happens in several countries of South America, that there are no different pharmacological groups. [6] However, it is very important to know the new treatment alternatives found in the literature to provide the most appropriate and updated treatment to patients.
Methodology
The study design is adapted to a systematic review of the evidence present in the scientific literature on screening for congenital heart disease. The literature search took place between 2000-2022 delving into various bibliographic databases in order to obtain information and review previous studies on the subject exposed. The keywords and Boolean operators used were “Migraine”, “treatment”, “alternative”, “monoclonal antibodies” described through DeCS (Descriptors in Health Sciences). To achieve a greater update on the subject, the articles published in the last 20 years were set as a temporary filter for the search.
Results
Based on the latest update of the International Headache Society of January 2018, where it defines migraine as the occurrence of headache crises of 4 to 72 hours duration, with its own characteristics (unilateral location, moderate or severe intensity, worsening with routine physical activity, pulsatile character,) associated with vomiting, nausea, phonophobia or photophobia [7]. Migraine may also be accompanied by aura. The diagnostic criteria for migraine with and without aura according to the latest update are described in Table 1. It is known that the diagnosis of migraine is clinical, through the anamnesis and welldetailed neurological physical examination, it can be made at diagnosis [8]. However, there is a possibility that other conditions generate similar symptoms, for this it is recommended at the time of diagnosis, that the patient with migraine without aura be evaluated by a neurological examination and a routine blood test, as well as by conducting neuroimaging studies [9]. All this in order to make a differential diagnosis on possible pathologies that may occur with similar symptoms and to be able to carry out an adequate diagnostic and therapeutic approach, ruling out structural anomalies.
Table 1: Diagnostic criteria for migraine without aura. Taken from international classification of headaches third edition.
Physiopathology is a disease in which many factors such as genetic, metabolic, environmental, hormonal, and exacerbation by means of some drugs intervene, in this way, mechanisms have been identified that cause alterations in the thalamuscortical circuits, hypothalamic and brain stem activation, brain connectivity, cortical depolarization and release of central pain modulating peptides, as is the (PRGC) calcitonin gene-related peptide, pituitary adenylate cyclase activating polypeptide. [10] It was established through studies that PRGC is a neuropeptide of 37 amino acids, is in the study phase since the 90s, is in the central and peripheral nervous system, forming part of a family that involves adrenomedulin, calcitonin and amylin, developing an important role in the neurotransmission of pain and in the pathophysiological mechanism of migraine acting through their respective receptors, thus activating when these bind to the PRGC molecule a cascade of information transduction that will end in the production and travel of the pain signal, which is why its study has allowed it to become a therapeutic target of new therapeutic behaviors. It has also been found increased in the intracranial circulation acute stage of migraine and its administration in experimental trials has been able to reproduce the symptoms in migraine patients, but not in those healthy volunteers. [11]
Conventional Migraine Treatments
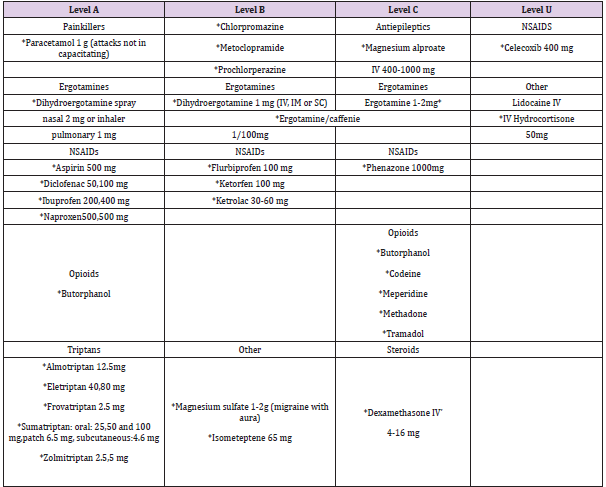
Table 2: Drugs used in the acute phase of migraine and their level of evidence Taken from Dominguez-Moreno R, et al. [21].
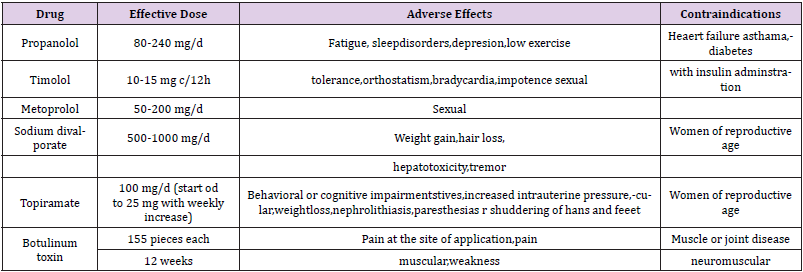
Table 3: Drugs used for migraine prevention level of evidence A Taken from Dominguez-Moreno R, et al. [21].
Conventional migraine treatment includes both preventive therapy, and its main goal is to reduce the frequency of occurrence of episodes and the intensity of attacks; Like acute treatment, which seeks to reduce the duration of crises, you should always warn of the possible adverse effects of medications and educate the patient to avoid the abuse of powerful analgesics. [12] Whose uses, indications, dosage are already well established in many guidelines and protocols, graphed in Tables 2 & 3.
Preventive Treatment with Monoclonal Antibodies:
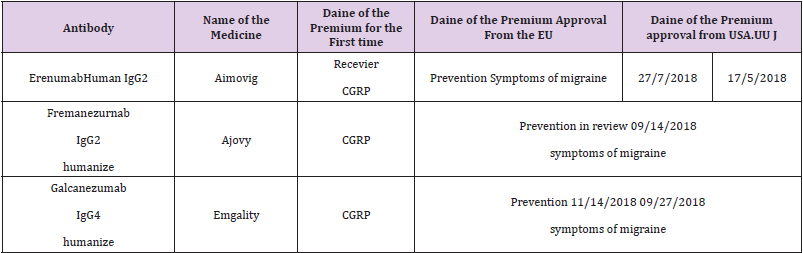
Currently in treatment more studied in recent years for its evidence revealed in the different studies carried out are monoclonal antibodies, in the preventive treatment of migraine, since 1985 the peptide related to the calcitonin gene (PRGC) was identified with an important role in the pathophysiology of migraine. This allowed a novel approach to be developed in the development of a treatment specifically targeting the pathophysiological mechanisms of migraine, giving rise to PRGC receptor antagonists and monoclonal antibodies against PRCG mechanisms. [13] Although studies were first done with PRGC receptor antagonists, which were shown to have efficacy for the treatment of acute migraine in several experimental trials, however, it was decided to discontinue them due to the toxicity at the hepatic level produced in long-term administration. This led to the development of monoclonal antibodies against PRGC or its receptor. [14] The new monoclonal antibodies have a long half-life which makes them ideal for treatment that requires chronic activity as occurs in migraine. Being macromolecules, they are not available orally, but they are available for intravenous or subcutaneous administration. Its properties give it a long duration of action that allows a dosage with a relatively low frequency, every month, or even quarterly, which improves patient compliance [15] In 2018 the first authorizations of antibody therapy for migraine were approved in the United States and in the European Union mentioned in Table 4.
Table 4: Mononuclear antibodies approved for the treatment of migraine Tomado de: Cury Summer, et al. [15].
At the international level, 4 antibodies have been approved for the preventive treatment of episodic and chronic migraine: erenumab, galcanezumab, fremanezumab and eptinezumab. Its mechanism of action focuses on the modulation of pain transmission induced by PRGC, the drugs Fremanezumab, galcanezumab and eptinemzumab, bind directly to PRGC, unlike erenumab which is the only one that acts by blocking the PRGC receptor causing in turn decreased central and peripheral sensitization. It should be noted that they have a limited ability to penetrate the blood-brain barrier, so their mechanism of action is not by direct action in the cerebral parenchyma but by way of the trigeminal ganglion and its distal terminals in dura and pia mater. [16] In recent years much has been studied about these drugs, multiple randomized, doubleblind works showed their effectiveness and excellent tolerance in a significant proportion of cases studied (40-62%) is achieved up to a reduction of 50% of migraine days per month with stopped with placebo (27-39%), at weeks 12 to 24 of treatment. There has even been a decrease in the non-painful symptoms of the migraine episode, such as vomiting, nausea, photophobia or phonophobia. [17]
Indication and Forms of Administration
Doses of erenumab 70 mg, fremanezumab 225 mg and galcanezumab 120 mg are available in self-injectable form to apply subcutaneously to the anterior side of the thighs, arm or abdomen. The first application is recommended to be performed by the doctor to instruct and educate the patient in its use and evaluate the initial tolerance. Eptinezumab requires intravenous infusion. [18] But those who are going to be candidates for this therapy, are considered candidates for treatment with monoclonal antibodies those people who meet the diagnostic criteria for migraine according to the latest International Classification of Headaches. Published work with mononuclear antibodies to the present, have included women and men between 18 and 70 years with episodic migraine, with chronic migraine, with at least 4 days of migraine per month, and at least 15 days per month of headache and 8 of them with migraine characteristics [19].
Discussion
It is necessary to make a good medical history and correlate it with a complete physical examination and thus evaluate being able to evaluate each patient and see whether or not he has the indication of treatment with mononuclear antibodies. It should be noted that these studies have not been carried out in pregnant women, leaving a gap in this population, so all women of reproductive age should be informed about this, and advise a contraceptive method during the time of treatment. [20] After the analysis of the current literature on migraine and its treatment alternatives, it was evident that there is still some uncertainty about the understanding as such of the disease and its pathophysiology. Monoclonal antibody therapy presents a hopeful expectation that supported by the results of multiple studies carried out that showed improvement under this treatment, thus, it produces an improvement of around days less migraine per month for patients, although a greater improvement was expected since it acts on the specific mechanisms of migraine. On the other hand, it should be noted that its subcutaneous administration is not the most ideal, but that it is compensated by having a long half-life, therefore, the frequency of administration of these treatments is lower and that is an advantage. Even so, these drugs presented the advantage of being based on a therapeutic target of the pathophysiology of migraine, which is a great advance with respect to the management and knowledge of it [15,20,21].
Conclusion
To conclude, to date there have been no comparative studies with currently conventional treatments, the data suggest that the efficacy rate could be similar, although it is expected to be significantly higher, however if the result were negative, the advantage that monoclonal antibodies would have would be argued in the ease of its administration either monthly or quarterly, and a lower rate of adverse effects and the absence of vasoconstrictor effect, with the disadvantage of economic costs and the limitation of access to these medicines by some countries, especially those that are in the process of development as is the case of South America, so it will be necessary to carefully assess the possible patients susceptible to receiving these therapies.
References
- (2016) GBD 2015 Disease and Injury Incidence and Prevalence Collaborators. Global, regional, and national incidence, prevalence, and years lived with disability for 310 diseases and injuries, 1990-2015: a systematic analysis for the Global Burden of Disease Study 2015. Lancet 388: 1545-602.
- Stovner LJ, Andree C, Eurolight Steering Committee (2008) Impact of headache in Europe: a review for the Eurolight project. J Headache Pain 9: 139-146.
- Charles A (2017) The pathophysiology of migraine: implications for clinical Management. Lancet Neurol 17(2): 174-182.
- Victor TW, Hu X, Campbell JC, Buse DC, Lipton RB (2010) Migraine prevalence by age and sex in the United States: a life-span study. Cephalalgia 30(9): 1065-1072.
- Sacco S, Bendtsen L, Ashina M, Reuter U, Terwindt G, et al. (2019) European Headache Federation guideline on the use of monoclonal antibodies acting on the calcitonin gene related peptide or its receptor for migraine prevention. J Headache Pain 20: 6.
- Mitsikostas DD, Reuter U (2017) Calcitonin gene-related peptide monoclonal antibodies for migraine prevention: comparisons across randomized controlled studies. Curr Opin Neurol 30(3): 272-280.
- (2018) Headache Classification Committee of the International Headache Society. The international classification of headache disorders (3rd)., Cephalalgia 38: 1-211.
- Worthington I, Pringsheim T, Gawel MJ, Gladstone J, Cooper P, et al. (2013) Canadian Headache Society Guideline: acute drug therapy for migraine headache. Can J Neurol Sci 40 (Suppl 3): S1-80.
- Lipton RB, Hamelsky SW, Kolodner KB, Steiner TJ, Stewart WF (2000) Migraine, quality of life, and depression: a population-based case-control study. Neurology 55(5): 629-635.
- Ho TW, Edvinsson L, Goadsby PJ (2010) CGRP and its receptors provide new insights into migraine pathophysiology. Nat Rev Neurol 6(10): 573-582.
- Edvisson L (2018) The CGRP pathway in migraine as a viable target for therapies. Headcache 58(1): 33-47.
- Antonci F, Ghiotto N, Wu S, Pucci E, Costa A (2016) Recent advances in migraine. Springer Plus 5: 637-650.
- Tso AR, Goadsby PJ (2017) Anti-CGRP Monoclonal Antibodies: the Next Era of Migraine Prevention? Curr Treat Options Neurol 19(8): 27-34.
- Schou WS, Ashina S, Amin FM, Goadsby PJ, Ashina M (2017) Calcitonin gene-related peptide and pain: a systematic review. J Headache Pain 18(1): 34-42.
- Cury Verano, Talia (2020) Anticuerpos comercializados en España para el tratamiento de la migrañ
- Dodick DW, Ashina M, Brandes JL, David Kudrow, Michel Lanteri Minet, et al. (2018) ARISE: a phase 3 randomized trial of erenumab for episodic migraine. Cephalalgia 38(6): 1026-1037.
- Silberstein SD, Dodick DW, Bigal ME, Paul P Yeung, Peter J Goadsby, et al. (2017) Fremanezumab for the preventive treatment of chronic migraine. N Engl J Med 377: 2113-2122.
- Doctorovich ED, Martín Bertuzzi F, Goicochea MT, Silvina Miranda, Maria de Lourdes Figuerola, et al. (2020) Consenso sobre el uso de anticuerpos monoclonales en la migraña en Argentina. Rev Neurol 70(4): 149-158.
- Rodríguez Almagro D, Achalandabaso A, Rus A, Obrero Gaitán E, Zagalaz Anula N, et al. (2020) Validation of the Spanish version of the migraine disability assess ment questionnaire (MIDAS) in university students with migraine. BMC Neurol 20: 67.
- Tiseo C, Ornello R, Pistoia F, Sacco S (2019) How to integrate monoclonal antibodies targeting the calcitonin gene-related peptide or its receptor in daily clinical practice. J Headache Pain 20: 49.
- Dominguez Moreno R, Vega Boada F, Mena Arceo RG (2019) Nuevos tratamientos contra la migrañ Med Int Méx 35(3): 397-405.

 Review Article
Review Article