ABSTRACT
The pandemic caused by the SARS COV2 coronavirus originated in the Asian country China, originated at the end of 2019 but it was not until March 2020 that it was cataloged by the World Health Organization (WHO) as a disease of international relevance and concern, however before it was ruled in this way it had already begun to study the implications on public health and especially in its clinical manifestations in order to know the natural history of this pathology of which nothing was known until that moment. After a little more than two years since this odyssey began, there have been multiple studies which have dimensioned and made known to the world the aggressiveness and organic complications generated by infection by this microorganism, one of the complications generated by this virus is given by the increase in thrombotic events in patients, these effects are influenced both by factors specific to the disease in question, and by the pathological predispositions of the patients. The disease itself produced by this virus has the ability to increase the procoagulant action generate an increase in fibrinolysis both intravascularly and extravascularly, but only in those patients with more aggressive or severe presentations of the infection, this increase in the fibrinolytic effect is reflected is an increase in various biomarkers as he D-dimer is for this reason that throughout the pandemic the association has been studied of elevated levels of this marker with many variables such as risk of ICU admission, the need to require invasive mechanical ventilation (IMV) and even the risk of mortality. That is, from different points of view the relationship between elevated levels of D-dimer and the severity of infection has been studied.
Keywords: D-Dimer; SARS-COV2 Infection; COVID19 and Hypercoagulability; Prothrombotic Status and COVID19
Abbreviations: IMV: Invasive Mechanical Ventilation ; ARDS: Acute Respiratory Distress Syndrome; CVD: Cerebrovascular Accident; PET: Pulmonary Thromboembolism; DVT: Deep Vein Thrombosis
Introduction
Infection by the SARS-COV2 virus is able to generate in the individual an aggressive pro-inflammatory response which is able to activate prothrombotic effects that further aggravate the clinical status of patients. Within the cascade of activation thrombin, factor XIIIa and fibrin there are many variables all of them measured by the inflammatory action, so that to understand this activation it is necessary to know initially the action of the virus on the organism which begins with its binding to receptors type ACE2 which are mostly at the pulmonary level but are also expressed in other organs such as the kidney or the heart. The binding of the virus to ACE2 generates a subsequent binding to lung cells especially pneumocytes type 2 triggering an injury at this level, with the migration of nuclear polymorphs produces a cell lysis. Cell damage is increased with the expression of PAMP and DAMP receptors which, in addition to occurring at the pulmonary level, have also been observed in the aforementioned organs. The accumulation of fibrin at the perilesional level in the alveolus and in the pulmonary venous capillary has as a response the activation of procoagulant factors which is clinically expressed with an increase in the time of PT, TPT and increase in serum levels of Dimero-D, in some cases with decreased platelets. The clinical manifestations secondary to the fibrinolitic effect of the infection are very variable generating from structural damage at the pulmonary level as microhematomas and parenchymal lesions lesions that in turn end up generating an increase in the pressure of pulmonary filling causing an alteration in ventricular filling pressures generating in the end a pump damage. At this time in patient has fallen into the worst was of the disease harassing all the known effects of hypotension, talking about a syndrome of multiple organ dysfunction.
Methodology
A narrative review was carried out in different platforms for the dissemination of scientific and academic information such as PubMed, ScienceDirect, Wiley, UpToDate, Elseiver among other indexed journals this using descriptors such as Dimero D and COVID19, hypercoagulability and COVID19, in order to obtain original and review articles with the aim of collecting articles of relevant information for our review, but whose year of publication was as recent as possible. We initially obtained 32 articles then applied our inclusion and exclusion criteria we were left with 15 of which we collected the most applicable and relevant information possible.
Results
At the end of 2019, a virus was identified for the first time in China that in a short time reached pandemic proportions. Over more than two years this microorganism has been studied by experts, for whom it has become a complete challenge to understand its diverse pathogenesis, which we do know so far is something about its involvement with the coagulability of patients who suffer from it [1]. Individuals who are affected by the disease often present various clinical manifestations, some with a very variable course of pathology ranging from an asymptomatic state or mild symptoms and others very unfortunate who suffer from severe states such as pneumonia that in some cases triggers acute respiratory distress syndrome (ARDS) with consequent refractory shock, multi-organ failure, and death [1,2]. Before it was declared a pandemic on March 11, 2020, scientists worldwide began to delve into the pathogenicity of covid19, studying multiple variables in order to establish or know what would be the clinical or laboratory parameters that would help medical personnel to have a better perception of the prognosis of patients in order to expand the therapeutic range and increase the life expectancy of affected patients. for this entity, one of the variables within the natural evolution of this disease that has been most talked about have been the procoagulant factors of which little or nothing was known at this time [3]. Today we know that one of the laboratory parameters that doctors have to determine the prothrombotic state of a patient is dimer D, this has been shown through various studies to be useful in the initial evaluation of the patient with COVID 19, but does the D-dimer serve as a predictor of severity in these patients?
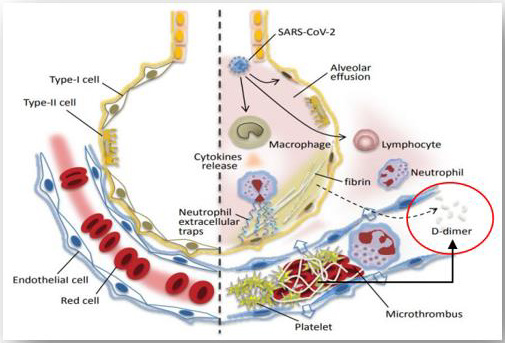
The increase of this biomarker can be observed in different conditions not only pathological but physiological scenarios, but its usefulness and development has been more studied in those cases in which a thrombotic pathology is presented as is the case for example of cerebrovascular accident (CVD), pathologies such as deep vein thrombosis (DVT), pulmonary thromboembolism (PET) and within the non-thrombotic events in which it can be seen increased are kidney disease chronic liver disease, chronic liver disease, congestive heart failure, burns, trauma, recent surgeries among others. Dimer-D is a biomarker generated from the degradation of fibrin by plasmin (Figure 1), it is considered an important marker of coagulation activation and fibrinolysis as it is the result of the sequential action of thrombin-factor XIIIa-plasmin. Its half-life reaches approximately 6 hours and its elimination is carried out almost entirely by renal form and another percentage through the reticulum-endothelial reticulum.
Illustration of the fibrinolytic mechanisms involved in dimer-D systensis both in the alveolar bed and at the systemic level. Available in; https://www.grupocaht.com/wp-content/uploads/2020/10/ DD-y-COVID-19-DMT-Set-2020.pdf The mechanisms by which SARS COV2 infection leads to increased D-dimer in patients is given by an increase in fibrinolysis of clots formed at the pulmonary level
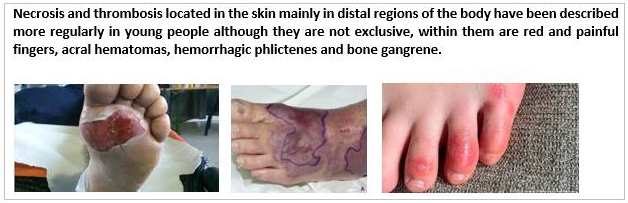
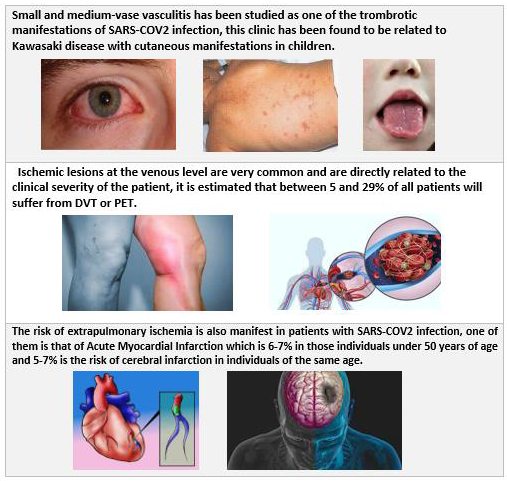
In this infection the virus in question enters our body through the respiratory tract initially binding to the angiotensin converting enzyme type 2 (ACE2) which in turn binds to type 2 pneumocytes located at the alveolar level and also in the pulmonary venous endothelium, but also have the ability to bind to other organs that express this type of proteins such as the kidney and the heart. Once here the microorganism has the ability to induce lysis of these cells through activation on the endothelium thus generating the accumulation of fibrin deposits and with this an activation of the procoagulant cascade [5]. damage and endothelitis end up generating intravascular lung injury or CIP, the same accumulation of fibrin deposits and the activation of plasmin causes the clinical increase in serum levels of Dimero-D, however the reasons why in the evolution of the disease despite not breaking down these fibrin deposits remain elevated procoagulant markers such as Dimero-D, PT and PTT and even mild platelet intake [6]. The CIP is considered the first systemic lesion generated by the virus reasons why it is the first to manifest clinically and does so generating own disorders associated with the ventilatory system large percentage reaching to cause massive pulmonary edema, respiratory acidosis, hypoxia forming the well-known syndrome of acute respiratory distress. However, since the receptors of the virus are at the cardiac level in said organs, clinical conditions can also be generated which differ in those of right origin this secondary to the initial increase in pressure on the pulmonary artery by lung injury which generate at the level of the right ventricle an increase at the end of systole of the same who ends in an increase in the pressure of atrial filling DER, the problems in the left heart are not foreign and develop as the disease progresses starting with a lower return of volume of venous origin with this a lower diastolic pressure is generated in the left atrium a consequent decrease in the systolic volume in the left ventricle which is reflected in a systemic hypotension[5,7]. It is clear that the infection presents a very extensive symptomatology associated with an immune response at the systemic level, within these the clinical manifestations product of the prothrombotic response induced by the virus could be described (Figures 2 & 3); Adapted from Oscar Sáenz, et al. [8] -- Images available at; * https://www.infosalus.com/salud-investigacion/ noticia-oms-estudia-relacion-sintomas-enfermedad-kawasakininos-covid-19-20200515183915.html, ** https://www.dw.com/ es/dedos-covid-ser%C3%ADan-un-efecto-secundario-de-la-luchadel- cuerpo-contra-el-coronavirus/a-59427904, *** https://www. tuotromedico.com/temas/infarto.htm The first studies carried out looking for the possible relationship between dimer D levels and the severity of the disease were carried out in the country where the virus was first isolated, this was carried out by Lippi and Favaloro in 2020 which was a meta-analysis carried out from 4 studies of which initially concluded the cut-off value for severity of the disease as 3.0 mg/L [9,10].
Serum dimer-D levels are increased with age so much that almost most individuals over the age of 80 find a biomarker result above normal limits could only be described as less than 5% of them presenting negative levels. In the review carried out by Sáenz et al. They describe as a positive predictive value a value of Dimer-D over 1.5 mg / L which would lead to patients presenting a higher risk of thrombotic events reaching a specificity and sensitivity between 85-88% [10,8].
The study carried out by Moreno G et al. in 2020 and in which the question was raised: Are D-dimer levels associated with the prognosis of patients with COVID19? was a systematic review with very complete parameters within which most articles of original content were obtained and from here they concluded that the elevated levels of this biomarker are significantly associated with a greater risk of disease severity, ARDS and death. In addition, they recommend using it as a tool in the evaluation of the prognosis and evaluation of the disease through monitors of its values at the time of admission and continue every 24 to 48 hours [11,12]. In Lima, Peru, a study was carried out by Lozano and Palacios in which they evaluated 76 patients in a clinic in the city in 2020 of whom they stand out had low frequency of comorbidities, despite being carried out retrospectively through their analysis they could conclude that not only the elevated levels of Dimer D, but the other variables studied by them such as DHL and PCR on the normal limits if they considerably increase the risk of admission to the ICU of the patients in question.
They affirm that the fact of presenting elevated levels of Dimer D is conditioned by a state of hypercoagulability explained by the aggressive pro-inflammatory response induced by the virus [13]. Also in 2020 it was carried out by Zhang et al. in an ambitious study they sought to determine the cut-off point of D-dimer levels from which the mortality risk of patients in the in-hospital setting could be predicted. Despite being a retrospective study, they were able to conclude that those patients whose serum D-dimer levels were greater than 2.0 mg/L had a higher probability of death when compared to those patients who have D-dimer values less than 2.0 mg/L [14,15]. From the onset of symptoms can be carried out an increase in this biomarker, but the question to which Zhou et al. answered through their study published in the Lancet. It was what was the behavior or evolution of the serum levels of Dimero D with the passing of the days and evolution of the patients. With a sample of 191 patients they managed to establish that once these levels rise, it continues in that sense in those patients who reach a severe stage of the disease and continues even in those who do not manage to survive the disease [16]. The same result in the study conducted with similar parameters by Tang et al. who in turn studied a total of 183 patients a sample similar to that of Zhou, et al. [17] (Table 1).
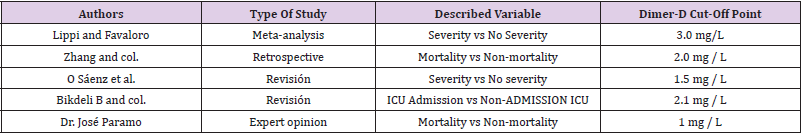
Table 1: Table of relationship between Dimero-D cut-off point and different variables under study. Adapted from references [5,8,9,14,20].
Discussion
The infection by this new coronavirus that causes COVID-19 has undoubtedly represented a great challenge for public health worldwide, even putting in check countries of the first world, some of them have been the most affected. However, this has generated that the studies carried out on the subject have been of greater magnitude something that conditions better medical recommendations, the topic described in our review has been widely studied given the relevance in clinical practice, the use of Dimer D has been abaado by multiple institutions as a predictor both severity of the disease as well as the risk of admission to ICU and even the probability of death, it was thus described by the consensus of experts in 2020 through the review of multiple studies conducted all conducted in China [18,19]. Now, the debate currently existing is about what should be the cut-off point to determine the probability that said patient will suffer any of the aforementioned events, within the review conducted by Bikdeli B, et al. [18]. published in the Journal of the American College of Cardiology in mid-2020 it is observed that all the events evaluated within the studies as they are severe vs non-severe cases, admission to ICU vs non-ICU and survivors vs non-survivors agree that patients with the most negative prognosis tend to have a Dimer-D value higher than 2.1 mg / L from then on the assessment is variable, this result coincides with what was presented in the study carried out by Zhang et al. which we cited above where it is observed that the cut-off point found by them that affects negatively the prognosis of patients is when the value of Dimero-D is greater than 2.0 mg /L. [14,18,19]. Despite the studies that demonstrate the cut-off value of Dimer-D as 2.0 mg /L. this result does not end up being the same for all scientists since in the meta-analysis carried out by Lippi and Favaloro which was a study of great relevance where patient evolution of patients from different Chinese medical institutions were studied, the result they show as a cut-off point of Dimero-D is 3.0 mg/L [10,18].
The studies practiced there are few that weigh on the other reasons why it is essential to take them into account together when bringing this to clinical practice salt moment with the revised theory it is valid to analyze and give a point of view on the serum levels of Dimero-D greater than 2.0 mg / L if they merit a medical intervention or stricter surveillance given that the evidence on the Greater risk of complications in these patients is conclusive, however in the medical profession there are in addition to massive studies personal opinions of experts as is the case of the one dictated by Dr. José A. Paramo Fernández president of the Spanish Society of Thrombosis and Hemostasis SETH, which varies from the cutoff point previously dictated when stating that even those patients with lower Dimer-D levels is said case of 1mg /L already have a risk of up to 18 times higher mortality when compared to those individuals with normal levels of the biomarker [20]. Dimero-D has undoubtedly proven to be useful to know the prognosis or scenarios that surround the patient and give a little information about what their clinical future could be, of course it is not even close to reaching an exact sensitivity or specificity for each of the scenarios described, the literature on the subject is wide but there is little information on studies carried out at the Latin American level with the specificities achieved. by Asian or European studies which does not make it clear if this biomarker will have equal utility in our region taking into account population variability.
References
- Wright FL, Vogler TO, Moore EE, Moore HB, Wohlauer MV, et al. (2020) Fibrinolysis Shutdown Correlation with Thromboembolic Events in Severe COVID-19 Infection. J Am Coll Surg 231(2):193-203.
- Mansory EM, Srigunapalan S, Lazo-Langner A (2021) Venous Thromboembolism in Hospitalized Critical and Noncritical COVID-19 Patients: A Systematic Review and Meta-analysis. TH Open 5(3): e286.
- Wu C, Liu Y, Cai X, Zhang W, Li Y, et al. (2021) Prevalencia de tromboembolismo venoso en pacientes críticamente enfermos con enfermedad por coronavirus 2019: un metaaná Front Med (Lausana) 8: 603558.
- Rosa CM (2020) DÍMERO D y COVID-19 Artículo de divulgació Setiembre 2020.
- Lippi G, Plebani M (2020) Laboratory abnormalities in patients with COVID-2019 infection. Clin Chem Lab Med 58(7): 1131-1134.
- Belen-Apak FB, Sarıalioglu F (2020) Pulmonary intravascular coagulation in COVID-19: possible pathogenesis and recommendations on anticoagulant/thrombolytic therapy. J Thromb Thrombolysis 50: 278-280.
- Thachil J (2020) The versatile heparin in COVID-19. J Thromb Haemost 18: 1020-1022.
- OA Sáenz Morales, AM Rubio, N Yomayusa, Nelson Gamba, Manuel Garay Fernández (2022) Coagulopatía en la infección por el virus SARS-CoV-2 (COVID-19): de los mecanismos fisiopatológicos al diagnóstico y tratamiento. Acta Colombiana de Cuidado Intensivo 22: 44-54.
- Lippi G, Favaloro E (2020) D-dimer is associated with severity of coronavirus disease 2019: a pooled analysis. Thrombosis and Haemostasis 120(5): 876-878.
- Moreno G, Carbonell R, Bodi M, Rodriguez A (2020) Revision sistematica sobre la utilidad pronostica del dimero-D, coagulacion intravascular diseminada y tratamiento anticoagulante en pacientes graves con COVID-19. Medicina intensiva 45: 42-55.
- Al Samkari H, Karp Leaf R, Dzik W, Carlson JCT, Fogerty AE, et al. (2020) COVID-19 and coagulation: bleeding and thrombotic manifestations of SARS-CoV-2 infection. Blood 136(4): 489-500.
- Ysabel Lozano, Enver V Palacios (2021) Factores asociados a la hospitalización de pacientes con COVID-19 en la Unidad de Cuidados Intensivos de una clínica en 2020. Universidad Científica del Sur. Lima, Perú. Horiz Med (Lima) 21(1): e1379.
- Zhang L, Yan X, Fan Q, Liu H, Liu X, et al. (2020) D-dimer levels on admission to predict in-hospital mortality in patients with COVID-19. J Thromb Haemost 18: 1324-1329.
- Berger JS, Kunichoff D, Adhikari S, Ahuja T, Amoroso N, et al. (2020) Prevalence and Outcomes of D-Dimer Elevation in Hospitalized Patients With COVID-19. Arterioscler Thromb Vasc Biol 40: 2539-2547.
- Zhou F, Yu T, Du R, Fan G, Liu Y, et al. (2020) Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet 395: 1054-1062.
- Tang N, Li D, Wang X, Sun Z (2020) Abnormal coagulation parameters are associated with poor prognosis in patients with novel coronavirus pneumonia. J Thromb Haemost 18: 844-847.
- Yury Lisseth González-Quiroz, María José Demera-Santos, María Luisa Freire-Pincay, Cindy Lisbeth Mecías-Zambrano (2021) Sensitivity and specificity of dimer D and ferritin as an indicator of severity in covid-19. Sitio Principal 7( 5): 213-232.
- Bikdeli B, Madhavan MV, Jimenez D, Taylor Chuich, Isaac Dreyfus, et al. (2020) COVID-19 and Thrombotic or Thromboembolic Disease: Implications for Prevention, Antithrombotic Therapy, and Follow-up. Journal of the American College of Cardiology 75(23): 2950-2973.
- Merad M, Martin JC (2020) Pathological inflammation in patients with COVID-19: a key role for monocytes and macrophages. Nat Rev Immunol 20: 355-362.
- Una reflexión de Jose A. Páramo Hernández, presidente de la Sociedad Española de Trombosis y Hemostasia (SETH).

 Review Article
Review Article