Abstract
PC (pancreatic cancer) is that the fourth most common cause of death due to cancer worldwide. The incidence and mortality rates are increasing year by year worldwide, and this review has analyzed the foremost recent incidence and mortality data for pancreatic cancer occurrence in India. Several possible risk factors are discussed here, involving known established risk factors and novel possible risk factors. The development of this cancer may be a stepwise progression through intraepithelial neoplasia to carcinoma. Though early and accurate diagnosis is promising based on a combination of recent techniques including tumor markers and imaging modalities, lacking early clinical symptoms makes the diagnosis late. Pancreatic cancer is often difficult to diagnose. This is because there are no validated, specific screening tests that can easily and reliably find early-stage pancreatic cancer in people who do not show symptoms. Furthermore, people with pancreatic cancer often do not have clearly identified symptoms in the early stages of the disease. This means it is often not found until later stages when the cancer can no longer be removed with surgery and/or has spread from the pancreas to other parts of the body. Correct staging is critical because treatment is usually based on this parameter. Treatment options have improved throughout the last decades. However, surgical excision remains the first therapy and efficacy of conventional chemo-radiotherapy for PC is limited. Recently, some novel new therapies are developed and can be applied in clinics soon. This review will provide a summary of pancreatic cancer, including an understanding of the developments and controversies.
Abbreviations: nAChR: Nicotinic Acetylcholine Receptor; FAEEs: Fatty Acid Ethyl Esters; CCK: Cholecystokinin; VEGF-A: Vascular Endothelial Growth Factor-A; TAMs: Tumor-Associated Macrophages; IGFs: Insulin-Like Growth Factors; FPC: Familial Pancreatic Cancer; MEN: Multiple Endocrine Neoplasia Type 1; MCN: Mucinous Cystic Neoplasm; PDAC: Pancreatitis Ductal Adenocarcinoma; IPMN: Intra-Ductal Papillary Mucinous Neoplasia; MRI: Magnetic Resonance Imaging; EUS: Endoscopic Ultrasound; HPA: Hypothalamic-Pituitary-Adrenal; CRF: Corticotropin-Releasing Factor; CEA: Carcinoembryonic Antigen; OPN: Osteopontin; MIC-1: Macrophage Inhibitory Cytokine; PET: Positron Emission Tomography; ERCP: Endoscopic Retrograde Cholangiopancreatography
Epidemiology
Pancreatic cancer is the 12th most common cancer and the 4th leading cause of cancer-related deaths in the world. The agestandardized incidence rates of the cancer vary considerably in different parts of the world from as low as 0.6/100,000 persons per year in regions of Asia to as high as 12.6/100,000 in the West. The age-standardized incidence rates for pancreatic cancer on an average are 8.2 and 2.7/100,000 amongst males in the developed and developing countries, respectively and 5.4 and 2.1/100,000 amongst females in the developed and developing countries, respectively. Pancreatic cancer has a familial component in about 10% of cases. In India, the incidence rates of pancreatic cancer are low compared to western countries. In India, the incidence of pancreatic cancer is 0.5-2.4/100,000 persons per year in women - 0.2-1.8/100,000 persons per year in men. However, irrespective of the incidence of the disease, survival in patients with pancreatic cancer is generally low with the 1-year and 5-year relative survival rates for all stages being 29% and 7%, respectively.
The cause for such poor long-term outcomes is possibly related to the fact that the disease is largely asymptomatic in the early stages and by the time symptoms do develop, the disease is locally advanced or metastatic. Only 10-20% of patients have respectable pancreatic cancer at presentation.
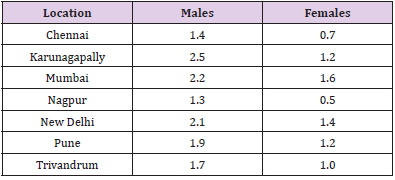
Table 1: Age-adjusted / age-standardized (ASR) pancreatic carcinoma incidence rates in India (expressed per 100,000 persons) (Data availability: ICMR Consensus Document for Management of Pancreatic Cancer).
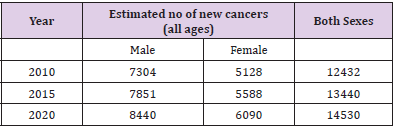
In the midst of all the dismal statistics for pancreatic cancer, there are some aspects that need to be appreciated, viz. the 5-year survival rates for patients with localized disease who are amenable to curative resection is 22% as compared to 2% for those with distant disease. Table 1 Provides the age-adjusted / agestandardized rates for pancreatic carcinoma from different parts of India. Pancreatic adenocarcinoma is more common in men as compared to women. Pancreatic cancer tends to occur later in life in the Western countries as compared to India where it probably occurs a decade earlier. Table 2 Provides an estimate of the number of pancreatic cancer cases in India over the next few years based on the data from the National Cancer Registry Programme (ICMR, Bangalore). The mortality figures from Indian registries suffer with problem of under-reporting because of problems in registration of death and in reporting of cause of death.
Table 2: Pancreatic Cancer estimates until 2020 in India. (Data availability: ICMR Consensus Document for Management of Pancreatic Cancer).
Risk Factors
Risk factors for pancreatic cancer can be divided into genetic and acquired. Pancreatic cancer has a familial component in about 10% of cases. The risk increases with the number of first degree relatives involved. The exact genetic basis of this inherited predisposition remains unknown in over 80% of cases. The most common inherited mutation in familial pancreatic cancer is probably in the BRCA2 gene. Other associated germline mutations include p16, ATM, STK11, PRSS1/PRSS2, SPINK1, PALB2,and DNA mismatch repair genes. The defined familial cancer syndromes associated with an increased risk for pancreatic cancer include hereditary pancreatitis, Peutz-Jeghers syndrome, hereditary breast and ovarian cancer syndrome, Familial Atypical Multiple Mole Melanoma syndrome, Lynch syndrome, and Li-Fraumeni syndrome. A detailed description of these syndromes is beyond the scope of this document. However, a thorough family history should be taken of additional relatives with pancreatic cancer, pancreatitis, melanoma, and cancers of the colon and rectum, breast, and ovaries. Acquired risk factors for pancreatic cancer include cigarette smoking, obesity, and diabetes (both type 1 and type 2). Recent onset after 50 years as well as long-term diabetes are considered as risk factors for pancreatic cancer. Other factors including alcohol use and dietary habits have less rigorous risk association. Perhaps the most important risk factor for pancreatic cancer in our country is chronic pancreatitis, which should be considered as a pre-malignant condition. However, because of its relatively low incidence, PC screening in the general population is less effective. As a result, it is urgent to explore the risk factors for PC and to identify the high-risk group. The possible risk factors for PC include gender, age, smoking, alcohol abuse, obesity, physical activities, diabetes, chronic pancreatitis, vitamin D, genetic alterations, dietary, and reproductive factors. Studies have consistently confirmed that smoking can increase the risk of PC and one-quarter of PC risk might be attributable to smoking. It is associated with 6-fold increase in the risk of PC.
There are greater than 60 chemicals identified as prospective carcinogens in cigarette smoke. Of these components swiftly absorbed in the upper aero digestive tract, nicotine is the major one, which predisposes to PC through causing genetic mutations in pancreatic cells. A recent study revealed that, through Src pathway, the ligation of nicotine and α7 nicotinic acetylcholine receptor (nAChR) stimulated metastasis and chemo resistance in PC [1]. Epidemiological evidence suggests that alcohol-abusing group have a higher PC incidence and mortality than nondrinkers. First, chronic pancreatitis, as a known risk factor for PC, is associated with heavy alcohol consumption. Acetaldehyde and fatty acid ethyl esters (FAEEs) are produced in the human body during the metabolism of alcohol and induce pancreatitis-like injury. Second, acetaldehyde has been proved as an organic chemical playing a significant role in carcinogenesis. But the exact mechanism linking alcohol consumption and PC has not been completely defined.
Dietary habits, particularly high-fat diets, resulted in a significant increase of cholecystokinin (CCK). High release of CCK was frequently associated with the development of intravascular tumor emboli, which was correlated with increased vascular endothelial growth factor-A (VEGF-A) [2]. Besides, people who consume a diet high in animal fat are at higher risk for diabetes. A link between diabetes and PC survival has also been suggested, but it remains inconsistent [3]. On one hand, it has been found that patients with long-term diabetes have a 1.5-fold to 2.0-fold increase in the risk of PC; on the other hand, the mean age of developing PC in these patients was significantly older than new-onset ones [4]. Diabetes may even be considered to be a consequence of PC. Chronic pancreatitis is a clearly identified and strong risk factor for PC which is up to 20 times greater than the general population [5]. Chronic pancreatitis is a long-term inflammation of pancreas. During the course of inflammation, a variety of pro- and antiinflammatory mediators (e.g., various cytokines, reactive oxygen species, and cyclooxygenase-2) released from the pancreas promote genomic damage and cellular proliferation and eventually lead to pancreatic malignancy. Tumor-associated macrophages (TAMs), a major inflammatory infiltrate, might link inflammation with cancer. In previous studies, high expression level of TAMs was detected and it might be associated with carcinogenesis, metastasis, and prognosis of PC.
Reproductive factors may be aetiologically associated with PC through estrogen exposure. Several studies, both in vivo and in vitro, have demonstrated that estrogen may lower women’s risk of PC. A 100-fold increase in circulating plasma level of estrogen is observed during pregnancy [6]. Women with the higher parity have longer term exposure to high estrogen. And high expression of steroid hormone receptors is frequently found in both benign and malignant neoplasm of pancreas. That is why long-term exposure to estrogen at high concentrations would inhibit the growth of PC, which has been shown in transplanted PC of rodent models. In addition, we know that insulin-like growth factors (IGFs) play a role in PC development, particularly in promoting cellular proliferation and inhibiting apoptosis. A research showed that circulating insulinlike growth factors (IGFs) concentrations in women who had given birth 4 or more times were significantly lower than nulliparous women. Recently, several genetic susceptibility loci of PC, which account for only 4% of all PC, have been frequently studied in relation to PC risk [7]. For example, BRCA2, PALB2, CDKN2a, and ATM germline mutations were carried by 10% to 15% families with familial pancreatic cancer (FPC). In the first stage of pancreatic adenocarcinoma (PDAC), the most common PC, KRAS mutations, are particularly frequent. Then, aberrations in P53, STAT3, SMAD4, and ARF/INK4 are involved in the development of PDAC.
Insulin-like growth factor-1 receptor (IGF-1R) is involved in cancer cell metabolism, proliferation, differentiation, apoptosis, and carcinogenesis and chemo-resistance [8]. Overexpression of IGF-1R in PC cells has been reported. In recent study, silencing IGF-1R could negatively regulate PC growth and metastasis via suppressing key signaling pathways such as PI3K/AKT, MAPK, JAK/ STAT, and EMT. Moreover, dysregulated genes involved in pathways, such as Sonic Hedgehog (Shh), Wnt, Notch, and transforming growth factor β (TGF-β) signaling, have revealed association with pancreatic tumor formation. Families with hereditary pancreatic cancer syndromes are considered at high risk. There are six certain hereditary conditions [9], such as multiple endocrine neoplasia type 1 (MEN1) syndrome, hereditary nonpolyposis colon cancer (Lynch syndrome), von Hippel-Lindau syndrome, Peutz-Jeghers syndrome, hereditary breast/ovarian cancer, and familial atypical multiple mole melanoma (FAMMM) syndrome.
Patho Physiology
The development of PC is a stepwise progression involving activation of oncogenes, inactivation of tumor suppressor genes, and deregulation of the cell cycle. There are three morphologic forms of noninvasive pancreatic neoplasia differing in biological and clinical behavior. These are
1) Intraductal papillary mucinous neoplasm (IPMN) which is composed of mucin-producing neoplastic cells growing in the main pancreatic duct or in one of its major branches,
2) Mucinous cystic neoplasm (MCN), as another mucinous cystic neoplasm, which does not connect to the native pancreatic ductal system and can be separated into three categories (benign, borderline, and malignant), and
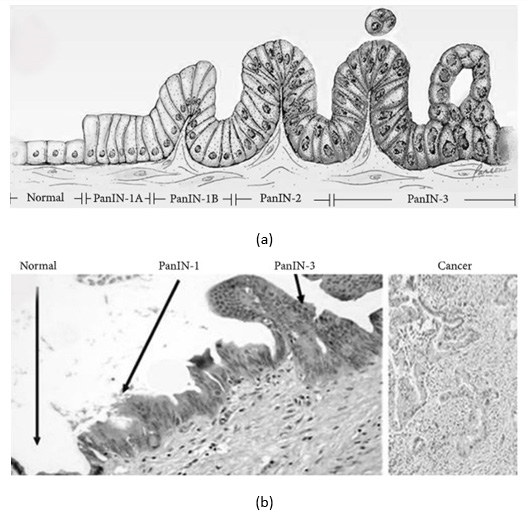
3) Pancreatic intraepithelial neoplasia (PanIN) which is the most common precursor to PC in human, proposed by Klimstra and Longnecker as a “gold standard” for describing the noninvasive lesions. PanINs are microscopic lesions initiating in small-caliber pancreatic ducts (<5 mm diameter) and may be classified into four consecutive stages accompanied by cumulative genetic alterations, as shown in Figures 1a & 1b [10]. Low-grade PanIN lesions (PanIN-1A/ PanIN-B) are flat or papillary epithelial lesions, which are characterized by epithelial cells with columnar shape and basally oriented uniform nuclei. As indicated above, activating KRAS mutations occur first (in PanIN-1 lesions).
Figure 1: Histological progression from normal pancreatic cells to pancreatic intraepithelial neoplasia.
a. Model for histological progression from normal pancreatic cells to pancreatic intraepithelial.
b. Micrograph of normal pancreas, pancreatic intraepithelial neoplasia (PanIN), and pancreatic cancer (PC) - Source [from Wikipedia]. Gradual transition from PanIN-1 to PanIN-3 was observed in a single duct. Haematoxylin and eosin stain.
As indicated above, TAMs might link inflammation with PC and play an important role in tumor growth and metastasis [11,12]. In the tumor micro environment, TAMs are mainly polarized towards M2 phenotype macrophages. In Japan, several studies have shown that high number of infiltrating M2-polarized macrophages in tumor tissue is related to a poor prognosis in PDAC patients [13- 15]. In our previous study, we also found that TAMs infiltration had a strong association with the incidence of lymph node metastasis [16]. In 2002, it was reported that TAMs expressed vascular endothelial growth factor- (VEGF-) C and impacted tumor lymphangiogenesis in the peritumoral inflammatory micro environment [17]. The results indicate that TAMs may have the ability to release cytokines and chemokines to affect tumor cell micro-environment, which enable lymph node metastasis. Additionally, 41 (58.6%) patients with PDAC in our previous study suffered from abdominal pain, and it was significantly associated with a higher level of infiltrating TAMs. These findings indicate that TAMs may involve the procedure of PC neural invasion. In the future, the clear molecule mechanism of TAMs in the PC tumor micro-environment requires further investigation.
The oncogenic KRASG12D is associated with invasive adenocarcinoma, through regulating division, differentiation, and apoptosis of pancreatic cells. The altered guanosine triphosphatase (GTPase) shows an increased activity of Ras-GTP, which stimulates downstream effector, namely, AKT. Activation of phosphatidylinositol 3 kinase (PI3K)/AKT pathway increases cell proliferation, survival, and protein synthesis in PC. In addition to its critical role in tumor initiation, KRAS is essential for the maintenance of PC. Compared to PanIN-1, PanIN-2 lesions are mostly papillary with higher nuclear atypia, including loss of nuclear polarity, nuclear crowding, enlarged nuclei, nuclear hyperchromasia, and nuclear pseudostratification. The inactivation of p16INK4A/CDKN2A gene occurs usually in PanIN-2. This tumor suppressor gene encodes protein p16, which binds to cyclin-dependent kinase 4/6 (Cdk4/6) and arrests cell cycle in G1 phase. Loss of the cycle-dependent kinase inhibitor, protein p16, arrests apoptosis. In PanIN-3 lesions, small clusters of epithelial cells with nuclear pleomorphism and high mitotic rate bud off into lumen. The lesion is a noninvasive form, known as “carcinoma in situ” of pancreatitis ductal adenocarcinoma (PDAC). In the progression from PanIN-3 to adenocarcinoma, accumulation of genetic alterations is detected, such as mutation in TP53, DPC4, and BRCA2. Maintenance of the G2/M arrest is dependent on a tumor suppressor, TP53. Another tumor suppressor gene, DPC4, was not found to be inactivated in PanIN-1/2. Inactivation of DPC4 induces disruption of TGF-β pathway, then leading to subsequent cell growth, differentiation, and oncogenesis. Compared to TP53 and DPC4, loss of BRCA2 occurs even later. BRCA2-mediated DNA repair is the most critical in the maintenance of genomic integrity. Mutations in BRCA2 cause an increased risk for PC. Though infiltrating adenocarcinomas are believed to develop from adjacent PanINs, the clinical significance of PanINs in the transection margin remains undefined.
Most pancreatic tumors are exocrine tumors, including ductal adenocarcinoma, acinar cell carcinoma, cystadenocarcinoma, adenosquamous carcinoma, signet ring cell carcinoma, hepatoid carcinoma, colloid carcinoma, undifferentiated carcinoma, Pancreatoblastoma, and pancreatic mucinous cystic neoplasm. And the most common form is ductal adenocarcinoma characterized by moderately to poorly differentiated glandular structures, comprising 80% to 90% of all pancreatic tumors. Pancreatoblastoma mostly occurs in childhood and it has a poor prognosis when it occurs in adult. Pancreatic mucinous cystic neoplasms range from totally benign to malignant, which can be diagnosed by EUS with cyst fluid analysis. By contrast, endocrine pancreatic tumors, the so-called neuro endocrine tumors of pancreas (PNET), are rare and they account for only 1-2% of all pancreatic tumors. Clinical manifestations of PNETs are varied based on the degree of differentiation and functionality. For many years, clinicians considered that these tumors displayed benign behavior with good prognosis. However, evidence has suggested that all PNETs larger than 0.5 cm were malignant.
Usually, PC is likely to metastasize early and rapidly, which is the primary cause of death. It first spreads to regional lymph nodes, followed by the liver and the peritoneal cavity. The prevalence of neural invasion is high, which is considered to be associated with abdominal pain. Metastasis to the lungs, bones, and brain is unusual. It is rare that PC metastasizes to the skin, which is called cutaneous metastasis, commonly to the umbilicus. But there have been a few number of cases of non-umbilical cutaneous metastases reported. In 2015, a case of 58-year-old PC patient with muscular metastasis was reported in France [18]. However, the mechanism of pancreatic tumor metastasis remains unknown. There are various studies on the mechanism of metastasis. As shown previously by Poomy P et al., high expression of amyloid precursor-like protein 2 (APLP2) is positive correlative to highly metastatic PC cells. Proliferator-activated receptor-γ (PPAP-γ), a nuclear receptor, is well accepted as a transcription factor in metastasis of PC. And the present studies aimed to investigate whether ligands of PPAP-γ, such as thiazolidinediones (TADs), inhibit metastasis of PC cells [19].
Pre-Malignant Lesions
Cystic neoplasia represent 10%–15% of cystic lesions of the pancreas. Intra-ductal papillary mucinous neoplasia (IPMN) and mucinous cystic neoplasia (MCN) are pre-malignant cystic lesions of the pancreas. The non-mucinous lesions have no malignant potential. With increasing use of abdominal imaging, incidental and often asymptomatic cystic pancreatic lesions are being detected with rising frequency. The initial evaluation of cystic pancreatic lesions should be contrast enhanced magnetic resonance imaging (MRI). Endoscopic ultrasound (EUS) is also indicated in most cases, unless the decision for surgery is evident. EUS-guided cyst fluid aspiration should only be performed when the results of cyst fluid analysis are expected to alter management. Treatment decisions in patients with IPMN are based on clinical and morphological criteria. All MCNs should be resected in medically fit patients with reasonable longevity. Recurrences are not seen after resection, and surveillance is not needed. After surgery for IPMN, surveillance of the remnant pancreas is indicated.
Diagnosis
Early and accurate diagnosis of PC, which often can be challenging, is important because it helps doctors choose the effective and timely treatment option for patients. It is usually based on a combination of imaging techniques such as computer tomography (CT) and endoscopic ultrasonography (EUS), tumor markers such as carbohydrate antigen 19-9 (CA19-9), clinical presentations, and the “gold standard” diagnosis-biopsy.
Clinical Presentation and Features
The initial symptoms of pancreatic cancer may be non-specific including weight loss, abdominal pain, nausea, and dyspepsia. Around 60%-70% of cancers arise in the head of pancreas, and these patients present with jaundice, pale stools, and itching. Tumour of the body and tail are often diagnosed at a more advanced stage. Some patients may have new onset diabetes, depression, or thrombophlebitis. Acute pancreatitis may be a manifestation of pancreatic cancer, especially when it occurs for the first time in an older adult without any obvious reason. Patients with chronic pancreatitis with super-added carcinoma may present with worsening pain, weight loss, and worsening diabetes control. In a longstanding diabetic patient sudden unexplained weight loss, or loss of blood sugar control may be features of pancreatic cancer. To date, lack of symptoms is the main cause of PC late diagnosis and therapy. The appearance of clinical presentations usually indicates an advanced stage and the most frequent presentations are progressive weight loss, anorexia, abdominal pain, and jaundice. These symptoms of PC are nonspecific and varied in different parts of pancreas. The tumor in the head of the pancreas (75%) produces symptoms such as weight loss, painless jaundice, nausea, and vomiting. The mass of pancreatic head causes blockage of the common bile duct, which results in jaundice, dark urine, light stool color, and itching.
Weight loss may be related to malabsorption of nutrients due to PC. Nausea, vomiting, and poor appetite, due to cancer-related gastric outlet (duodenum) obstruction, may also contribute to weight loss. If cancer is located at the body/tail of the pancreas, patients usually present with abdominal pain that radiates to the sides or the back. Previous reports showed that inflammatory and immune cells were associated with both the pain intensity and the extent of perineural invasion (PNI). And PNI is also involved in pain generation. Due to PC producing blood clotting chemicals, thrombus forms automatically in the portal blood vessels, the deep veins of the extremities, or the superficial veins on the body, which is known as Trousseau syndrome. In comparison to patients with other types of digestive cancer, patients in advanced stage of PC will experience more anxiety and depression. Earlier studies indicate that proinflammatory cytokines may be responsible for cancerrelated depression. And increased levels of several cytokines including interleukin-6 (IL-6), interleukin-8 (IL-18), and TNF-α in patients with PC have been found. These cytokines may correlate with the regulation of hypothalamic-pituitary-adrenal (HPA) axis and corticotropin-releasing factor (CRF). However, this hypothesis remains unclear. Other common symptoms include fatigue, diarrhea, and heartburn.
Tumor Markers
In the screening of asymptomatic patients with PC, the clinical role of serologic markers, which includes CA19-9, carcinoembryonic antigen (CEA), osteopontin (OPN), macrophage inhibitory cytokine 1 (MIC-1), and S100A6, has been limited. CA19-9 is an isolated Lewis antigen of the tumor-associated protein mucin 1 (MUC1). It can be helpful in the assessment of response to chemotherapy, in the early detection of tumor recurrence, and even in the predicting of the prognosis. The role of CA19-9 in PC diagnosis is inconclusive, though it is the most useful and routinely adopted, because highly elevated serum level of CA19-9 has been found in many other gastrointestinal tumors and ovarian cancer, as well as nonmalignant diseases. Chronic inflammation or acute injury may induce CA19-9 synthesis through pathologic fibrosis, which has been approved by immunohistochemical analysis for CA19-9 in hepatic inflammatory areas and bile ductule cells. That may be the reason why CA19-9 is elevated in chronic hepatitis and nonmalignant objective jaundice. Besides, CA19-9 level cannot be elevated in 10% of Caucasians even with large pancreatic tumor because they are Lewis-negative.
CEA, another biological marker for prognosis of PC, is a glycoprotein. A rising CEA level is associated with adenocarcinoma, including colon cancer, breast cancer, and stomach cancer. The sensitivity and specificity of CEA in PC were 83.78 and 69.44%, respectively. The level of CEA has significant correlation with tumor size, tumor differentiation, and lymphatic and liver metastasis. Serum OPN is one of the most recent biomarkers that have shown potential clinical applicability for PC. It is a highly phosphorylated sialoprotein discovered in 1986 in osteoblasts. Proinflammatory cytokines (e.g., TNF-α, IL-1β, and angiotensin II) upregulate the expression of OPN. Moreover, elevated levels of OPN were also found in a variety of cancers, including lung cancer, stomach cancer, and PC. It may promote cancer metastasis through the ligands-receptor interaction with the CD44 receptor family. A metaanalysis revealed that OPN was a serum diagnostic biomarker for the early-stage PC [20]. A previous study indicated that MIC-1 was a potential diagnostic biomarker in early diagnosis and postoperative monitoring for PC [21]. As a member of the TNF-β superfamily, MIC- 1 is weakly expressed under normal conditions, but it is markedly upregulated in inflammatory diseases as well as cancers. Compared to CA19-9, MIC-1 seems to have better sensitivity; however, it has lower specificity in differentiating pancreatitis from PC.
A number of proteins in the S100 family have been found to be related to PC progression and metastasis. S100A6 is a member of this family and PC patients with high level expression of S100A6 have poor outcome. It is significantly elevated in intraductal papillary mucinous neoplasms (IPMN), in pancreatic tumors, and even in PanIN lesions. S100A6 protein may influence the invasion of PC, but it is not yet clear what the precise mechanism is. In addition, several fecal markers have been studied, such as methylated bone morphogenetic protein 3 (mBMP3) and Adnab-9. In stools from PC patients, significantly higher mBMP3 was found when compared to stools from the controls. And BMP3 is recognized as a tumor suppressor. We therefore hypothesized that aberrant BMP3 promoter methylation PC led to the development of PC. The presence of Adnab-9 in stools has been shown to be associated with PC precursor lesions. As a fecal biomarker, Adnab-9 has a sensitivity of 80% and a specificity of 87% for the detection of PC [22]. In the future, effective tumor markers can be used to aid in the diagnosis of the presymptomatic PC, treatment assessment, and then monitoring for disease recurrence.
Imaging
Over the years, imaging techniques, such as trans abdominal ultrasound (US), CT, magnetic resonance imaging (MRI), positron emission tomography- (PET-) CT, endoscopic retrograde cholangiopancreatography (ERCP), and EUS, play a vital role in PC detection and staging. Although in small pancreatic lesions (less than 3cm) detection with fairly low sensitivity (67%) and specificity (40%) for PC, US is the most widely used image technique. Because it is inexpensive, safe, and painless, US is strongly recommended as the initial screening tool for PC. As one of the most convenient imaging tools, the new methods in the development of CT scans, including multi detectors, intravenous contrast, curved planner reformations, CT angiography, and some post-processing techniques, have shown promise in detection and staging of PC. Helical CT may detect masses larger than 2 cm with a sensitivity of 78–100%. CT imaging post-processing techniques have greatly enhanced its ability in preoperative TNM staging of PC. Planar reformatted images and curved reformatting are now being used to detect PC location and its relationship to adjacent structures, such as the pancreatic duct, common bile duct, and blood vessels. Maximum intensity projection (MIP) and volume rendering can identify narrowing or irregularity of vessels due to tumor encasement. In spite of some disadvantages of CT such as considerable radiation exposure and potential for contrast-induced nephropathy, it is a comprehensive primary imaging modality for PC diagnosis and staging.
MRI can help to clearly define pancreatic mass without abnormal CT findings. It is superior to CT in the detection of small pancreatic tumors, hypertrophied pancreatic head, iso attenuating pancreatic cancer, and focal fatty infiltration of parenchyma. Furthermore, magnetic resonance cholangiopancreatography (MRCP), which can delineate the pancreatic ductal system noninvasively, is currently used as an accurate diagnostic tool for patients with suspected biliopancreatic disease. PET-CT with fluorine-18 fluorodeoxyglucose (18F-FDG) is a combination of PET and high-end multi-detector-row CT, being widely used for diagnosis, staging, and monitoring cancer following treatment, such as PC. 18F-FDG PET/CT can detect the metabolic activity in PC and evaluate pancreatic tumor response to radiotherapy. Another important technique in the diagnosis of PC is ERCP, which combines upper gastrointestinal (GI) endoscopy and fluoroscopy. It provides direct visualization of pancreatic and bile duct system with morphologic alterations, like stenosis and dilation. The sensitivity of ERCP with respect to diagnosing pancreatic cancer was 70% and its specificity was 94% [23]. Moreover, during ERCP, we can collect the pancreatic juice and cells for pathological examination. But it is invasive and may cause some related complications, such as bleeding, perforation, and pancreatitis. The information of tumor size provided by ERCP is limited and metastasis cannot be assessed. Compared to ERCP-based brush cytology, the accuracy rates of EUSguided fine needle aspiration (FNA) of pancreatobiliary tumors are higher (over 80%) [24]. Furthermore, EUS is critical for preoperative staging of pancreatic head tumor by virtue of determining nearby blood vessels and lymph nodes involvement.
Management
Despite the improvement of PC diagnostic tests over these years, the rate of diagnosis at an early stage remains low, and so is the survival rate. These days, the efficacy of conventional chemoradiotherapy for PC is limited, and surgery is the best option for these patients.
Surgical Resection
Surgery remains the only possibility for curing of PC, though there are only 20% of patients with operable PC. The selection of an operative procedure for PC is based on factors such as the tumor location, tumor size, and tumor staging. The classic Whipple procedure (pancreatoduodenectomy), which involves removing the pancreatic head, as well as the curve of the duodenum, the gallbladder, and the common bile duct, is the most common operation for cancers of the head and/or neck of pancreas. In 1898, Alessandro Codivilla performed this procedure firstly on a patient with PC [25]. Unfortunately, this patient died of disseminated recurrence 24 days after surgery. Since an American doctor named Allen Oldfather Whipple devised the perfect version in 1935, it is called the Whipple procedure, which is performed on patients with pancreatic head cancer and periampullary cancer. When cancer involves the body and tail, distal/subtotal pancreatectomy is suggested. About 35% of the patients with body/tail PC were observed at the time of surgery, finding that the tumors have spread to surrounding tissues. In such cases, extended resection should be advisable. For patients with locally advanced pancreatic cancer (LAPC), multivesicular resection is technically feasible. Based on recent publications, perioperative mortality (3%) and morbidity (35%) did not differ between two groups of patients who underwent standard resection or multivesicular resection [26,27]. Although laparoscopy for PC treatment is controversial, laparoscopy in diagnosis and staging of PC is known to be critical, safe, and reliable. Refinements in surgical techniques will reduce perioperative morbidity and improve the outcomes.
Chemotherapy
For unresectable PC, chemotherapy is being extensively used, such as GEM/erlotinib, FOLFIRINOX, GEM/NAB-paclitaxel, GEM/ capecitabine, and capecitabine/oxaliplatin (XELOX). However, PC is characterized by a dense desmoplastic reaction which promotes resistance to chemotherapy [28]. As the key drug for chemotherapy of unresectable PC, gemcitabine (GEM) was first synthesized by Larry Hertel at Eli Lilly during the early 1980s. With the introduction of many new agents, such as 5-fluorouracil (5FU), cisplatin, epirubicin, oxaliplatin, leucovorin, and irinotecan, there have been multiple chemotherapy regimens for PC. In PC cells, epidermal growth factor receptor (EGFR) is overexpressed, and erlotinib is an EGFR-tyrosine kinase inhibitor. As a result, the level of EGFR expression may predict the efficacy of this combined chemotherapy in PC. And then, in 2011, Conroy et al. suggested that FOLFIRINOX, a combination regimen of oxaliplatin, 5FU, leucovorin, and irinotecan, should be used as first-line systemic chemotherapy in patients with advanced PC [29]. Because FOLFIRINOX toxicity is higher than GEM alone, this regimen is considered as an option for younger patients with a good performance status. More recently, the efficacy and safety of another combined chemotherapy of GEM plus NAB-paclitaxel (NAB-P) has been well evaluated in a clinical trial. NAB-P is an albumin nanoparticles, which is watersoluble formulation with less toxicity and a relatively higher local concentration in stromal-rich tumors. GEM sensitivity can be enhanced through inhibiting the primary GEM catabolic enzyme by NAB-P [30]. In addition, capecitabine (CAP) is widely used as an orally administered pro-drug that is enzymatically converted to 5FU by thymidine phosphorylase (dThdPase) preferentially located in tumors. So, CAP is much safer, more effective, and convenient than 5FU. Besides CAP, oxaliplatin is active as primary therapy for advanced PC. However, the combination of CAP plus oxaliplatin (XELOX) is just used as second-line chemotherapy because of limited experience. In spite of limited efficacy in metastasis PC, chemotherapy plays a central role in the adjuvant setting for patients with metastasis PC.
Radiotherapy
For unresectable PC, there is little evidence to support the efficacy of radiotherapy. However, radiotherapy can be used as a palliative treatment option for those unresectable locally advanced tumors. It can kill cancer cells and keep them from growth and recurrence. People will have side effects from radiotherapy, such as fatigue, gastrointestinal symptoms, skin rashes, and toxicity to the surrounding normal tissues. Fortunately, recent innovation in radiotherapy symbolized by intensity-modulated radiotherapy (IMRT) and image-guided radiotherapy (IGRT) provides alternative treatment which is much more effective and tolerable [31]. These technologies allow an increase of the target volume dose while minimizing the dose to the surrounding normal structures. As in a previous study, IGRT and IMRT after preradiation chemotherapy for longer than 9 months improved overall survival and progressionfree survival for these PC patients. In addition, induction of radio sensitization by injection of hydrogen peroxide and sodium hyaluronate into the unresectable pancreatic tumor would enhance the efficacy of radiotherapy, without serious complications [32]. Because of highly advanced technologies in radiotherapy, a new precisely targeted radiotherapy, named stereotactic body radiotherapy (SBRT), has been applied to treat PC, which has been successful in the treatment of thoracic tumors and early-stage non-small-cell lung cancer. It can deliver a high dose of radiation accurately while minimizing the dose to the surrounding normal tissues. Further investigation of radiotherapy is needed to improve its efficacy and safety in the treatment of local advanced PC.
Adjuvant Therapy
While the ESPAC-1 trial laid the foundation for the beneficial role of adjuvant chemotherapy in terms of a survival benefit, the ESPAC-3 trial confirmed the lack of a benefit of Gemcitabine over 5-FU. Despite this, single agent gemcitabine has been the preferred drug in the adjuvant setting. However, the results from the 30.5 month median follow up of the PRODIGE24 trial were recently presented at ASCO. For patients aged 18-79 years, 21-84 days after R0 or R1 resection, WHO Performance status, adequate hematologic and renal function, and no cardiac ischaemia, mFOLFIRINOX has not only been shown to be safe, but associated with a significantly better DFS and OS compared to Gemcitabine.
Other Treatment - Strategies
New promising therapies are urgently needed because only a few patients with PC can benefit from conventional treatments, like chemotherapy or radiotherapy. Gene therapy in PC is not yet applied in clinics, although it has become successful in vitro as well as in vivo. It includes gene replacement, gene modification, and gene blockade. PC gene therapy is mainly based on target genes, such as p16INK4A/CDKN2A, p21CIP1/WAF1, p14ARF, K-ras, LSM1/CaSm, HER-2/EerB-2, MDR1, BCRP, and VEGF. A bacterial cancer vaccine for PC, using a live attenuated Listeria strain as vector, is just beginning to reach early-phase clinical trial [33]. Recently, a novel new treatment has been developed, which is called high intensity focused ultrasound (HIFU). The first-in-human clinical trial of high intensity focused ultrasound (HIFU) in advanced PC was performed in China in 2000. Utilizing high intensity focused ultrasound energy, it causes all the targeted PC cells necrosis through heating. The effect of HIFU in ablation is a combination of direct and indirect effects. The direct effect includes thermal ablation on targeted cancer cells, mechanical effects such as acoustic pressure and shear stress. Indirect effect is associated with tumor blood vessel destruction. HIFU is a palliative treatment with less invasive and shorter recovery, which offers a suitable alternative. Another highly potent approach, which has been tested only in studies involving animals, is to enhance cancer cell death through an antiglycolytic agent called 3-bromopyruvate (3-BrPA) [34]. 3-BrPA inhibits enzyme activity of glyceraldehydes-3-phosphate dehydrogenase (GAPDH), one of the key glycolytic enzymes. Julius et al. developed the formulation of 3-BrPA, micro-encapsulated in a complex with β-cyclodextrin (β-CD), which limited exposure of 3-BrPA to normal cells. In the future, we wish to develop more and more novel therapeutic strategies, which could prove to be promising for PC patients [35-38].
Site of Tumour
Pancreaticoduodenectomy is performed for not only pancreatic cancer but also for tumors originating in the ampulla of Vater, periampullary duodenum and terminal end of common bile duct. Making a distinction between these 4 cancer types is essential as these are biologically distinct malignancies with different outcomes. This may be especially difficult to determine when the tumour is located in close proximity to, or is large and involving more than one of these adjoining sites. Therefore, during macroscopic evaluation it is vital to document the epicentre of the tumour (i.e. the site of tumour centre). This should be later corroborated with microscopic findings.
Tumour Size
Tumour size should be recorded in all three dimensions. This should be later confirmed or altered on the subsequent microscopy.
Histologic Type
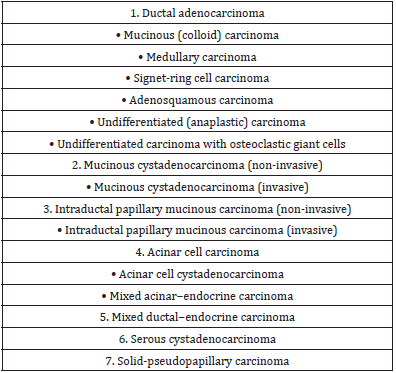
Histologically, PDAC is characterized by malignant glands infiltrating in a typical desmoplastic stroma. Many morphologic variants of ductal adenocarcinoma are recognized that differ in prognosis (Table 3). Colloid (mucinous) carcinoma and medullary carcinomas are associated with better outcome while other variants such as adenosquamous carcinoma and undifferentiated (anaplastic) carcinomas are associated with a poorer prognosis. Medullary carcinomas are characterized by solid syncytial growth pattern and a rich intratumorally lymphoid infiltrate. These variants are likely to display microsatellite instability and display a better prognosis. Acinar cell carcinomas are aggressive tumors however exhibit a slightly better prognosis than conventional PDAC. Adenosquamous carcinoma is a rare neoplasm characterized by the presence of variable proportions of glandular and squamous components. The squamous component should account for at least 30% of the tumour tissue.
Conflict of Interests
The author indicate that i have no significant financial relationship with the manufacturer of a commercial product or provider of a commercial service that may be discussed in this paper. And the authors have no conflict of interests.
Authors Contribution
Tshetiz Dahal carried out the conception and the design and participated in drafting the paper, with the analysis and the interpretation of data and gave the final approval of the version to be published.
References
- JG Treviño, S Pillai, S Kunigal, Sandeep Singh, William J Fulp, et al. (2012) Nicotine induces inhibitor of differentiation-1 in a Src-dependent pathway promoting metastasis and chemoresistance in pancreatic adenocarcinoma. Neoplasia 14(12): 1102-1114.
- GL Matters, TK Cooper, CO McGovern, Evan L Gilius, Jiangang Liao, et al. (2014) Cholecystokinin mediates progression and metastasis of pancreatic cancer associated with dietary fat. Digestive Diseases and Sciences 59(6): 1180-1191.
- AT Toriola, R Stolzenberg Solomon, L Dalidowitz, D Linehan, G Colditz (2014) Diabetes and pancreatic cancer survival: a prospective cohort-based study. British Journal of Cancer 111(1): 181-185.
- S Mizuno, Y Nakai, H Isayama, Shuhei Kawahata, Tomotaka Saito, et al. (2014) Smoking, family history of cancer, and diabetes mellitus are associated with the age of onset of pancreatic cancer in japanese patients. Pancreas 43(7): 1014-1017.
- AB Lowenfels, P Maisonneuve, G Cavallini, RW Ammann, PG Lankisch, et al. (1993) Pancreatitis and the risk of pancreatic cancer. International Pancreatitis Study Group. The New England Journal of Medicine 328(20): 1433-1437.
- JH Liu, RW Rebar (2014) Endocrinology of pregnancy in Maternal-Fetal Medicine: Principles and Practice. In: RK Creasy and R Resnik (Eds.)., Saunders, Philadelphia, Pa, USA, pp. 382-412.
- L Huang, C Wu, D Yu, Chengfeng Wang, Xu Che, et al. (2013) Identification of common variants in BRCA2 and MAP2K4 for susceptibility to sporadic pancreatic cancer. Carcinogenesis 34(5): 1001-1005.
- S T Guo, CC Jiang, GP Wang, YP Li, CY Wang, et al. (2013) MicroRNA-497 targets insulin-like growth factor 1 receptor and has a tumour suppressive role in human colorectal cancer. Oncogene 32(15): 1910-1920.
- AP Klein (2013) Identifying people at a high risk of developing pancreatic cancer. Nature Reviews Cancer 13(1): 66-74.
- T Muniraj, PA Jamidar, HR Aslanian (2013) Pancreatic cancer: a comprehensive review and update. Disease-a-Month 59(11): 368-402.
- P Allavena, A Sica, C Garlanda, A Mantovani (2008) The Yin-Yang of tumor-associated macrophages in neoplastic progression and immune surveillance. Immunological Reviews 222(1): 155-161.
- G Solinas, G Germano, A Mantovani, P Allavena (2009) Tumor-Associated Macrophages (TAM) as major players of the cancer-related inflammation. Journal of Leukocyte Biology 86(5): 1065-1073.
- H Kurahara, H Shinchi, Y Mataki, Kousei Maemura, Hidetoshi Noma, et al. (2011) Significance of M2-polarized tumor-associated macrophage in pancreatic cancer. Journal of Surgical Research 167(2): e211-e219.
- H Kurahara, S Takao, K Maemura, Yuko Mataki, Taisaku Kuwahata, et al. (2013) M2-Polarized tumor-associated macrophage infiltration of regional lymph nodes is associated with nodal lymphangiogenesis and occult nodal involvement in pn0 pancreatic cancer. Pancreas 42(1): 155-159.
- K Yoshikawa, S Mitsunaga, T Kinoshita, Masaru Konishi, Shinichiro Takahashi, et al. (2012) Impact of tumor-associated macrophages on invasive ductal carcinoma of the pancreas head. Cancer Science 103(11): 2012-2020.
- SJ Chen, QB Zhang, LJ Zeng, GD Lian MD, JJ Li, et al. (2015) Distribution and clinical significance of tumour-associated macrophages in pancreatic ductal adenocarcinoma: a retrospective analysis in China. Current Oncology 22(1): e11-e19.
- SF Schoppmann, P Birner, J Stöckl, Romana Kalt, Robert Ullrich, et al. (2002) Tumor-associated macrophages express lymphatic endothelial growth factors and are related to peritumoral lymphangiogenesis. The American Journal of Pathology 161(3): 947-956.
- R Belbarka, Z Fadoukheir, C Delafouchardiere, F Desseigne, H Errihani (2014) An unusual presentation of pancreatic cancer: muscular metastasis. Indian Journal of Cancer 51(3): 369-370.
- I Ninomiya, K Yamazaki, K Oyama, Hironori Hayashi, Hidehiro Tajima, et al. (2014) Pioglitazone inhibits the proliferation and metastasis of human pancreatic cancer cells. Oncology Letters 8(6): 2709-2714.
- JJ Li, HY Li, F Gu (2014) Diagnostic significance of serum osteopontin level for pancreatic cancer: a meta-analysis. Genetic Testing and Molecular Biomarkers 18(8): 580-586.
- X Wang, Y Li, H Tian, Jun Qi, Mo Li, et al. (2014) Macrophage inhibitory cytokine 1 (MIC-1/GDF15) as a novel diagnostic serum biomarker in pancreatic ductal adenocarcinoma. BMC Cancer 14(578).
- M Tobi, M Kim, DH Weinstein, Mary Ann Rambus, James Hatfield, et al. (2013) Prospective markers for early diagnosis and prognosis of sporadic pancreatic ductal adenocarcinoma. Digestive Diseases and Sciences 58(3): 744-750.
- HE Adamek, J Albert, H Breer, M Weitz, D Schilling, et al. (2000) Pancreatic cancer detection with magnetic resonance cholangiopancreatography and endoscopic retrograde cholangiopancreatography: a prospective controlled study. The Lancet 356(9225): 190-193.
- JB Klapman, R Logrono, CE Dye, I Waxman (2003) Clinical impact of on-site cytopathology interpretation on endoscopic ultrasound-guided fine needle aspiration. American Journal of Gastroenterology 98(6): 1289-1294.
- PJ Shukla, G Barreto, SV Shrikhande (2011) The evolution of pancreatoduodenectomy. Hepato-Gastroenterology 58(109): 1409-1412.
- W Hartwig, T Hackert, U Hinz, Matthias Hassenpflug, Oliver Strobel, et al. (2009) Multivisceral resection for pancreatic malignancies: risk-analysis and long-term outcome. Annals of Surgery 250(1): 81-87.
- AR Sasson, JP Hoffman, EA Ross, Steven A Kagan, James F Pingpank, et al. (2002) En bloc resection for locally advanced cancer of the pancreas: is it worthwhile? Journal of Gastrointestinal Surgery 6(2): 147-158.
- A Neesse, P Michl, KK Frese, Christine Feig, Natalie Cook, et al. (2011) Stromal biology and therapy in pancreatic cancer. Gut 60(6): 861-868.
- T Conroy, F Desseigne, M Ychou, Olivier Bouché, Rosine Guimbaud, et al. (2011) FOLFIRINOX versus gemcitabine for metastatic pancreatic cancer. The New England Journal of Medicine 364(19): 1817-1825.
- KK Frese, A Neesse, N Cook, Tashinga E Bapiro, Martijn P Lolkema, et al. (2012) Nab-paclitaxel potentiates gemcitabine activity by reducing cytidine deaminase levels in a mouse model of pancreatic cancer. Cancer Discovery 2(3): 260-269.
- SH Son, JH Song, BO Choi, Young Nam Kang, Myung Ah Lee, et al. (2012) The technical feasibility of an image-guided intensity-modulated radiotherapy (IG-IMRT) to perform a hypofractionated schedule in terms of toxicity and local control for patients with locally advanced or recurrent pancreatic cancer. Radiation Oncology 7: 203.
- A Nishioka, Y Ogawa, K Miyatake, Michiko Tadokoro, Munenobu Nogami, et al. (2014) Safety and efficacy of image-guided enzyme-targeting radiosensitization and intraoperative radiotherapy for locally advanced unresectable pancreatic cancer. Oncology Letters 8(1): 404-408.
- KC Soares, L Zheng, B Edil, EM Jaffee (2012) Vaccines for pancreatic cancer. Cancer Journal 18(6): 642-652.
- J Chapiro, S Sur, LJ Savic, Shanmugasundaram Ganapathy Kanniappan, Juvenal Reyes, et al. (2014) Systemic delivery of microencapsulated 3-bromopyruvate for the therapy of pancreatic cancer. Clinical Cancer Research 20(24): 6406-6417.
- Y Fang, Q Yao, Z Chen, Jianbin Xiang, Fisher E William, et al. (2013) Genetic and molecular alterations in pancreatic cancer: implications for personalized medicine. Medical Science Monitor 19: 916-926.
- (2012) IARC. GLOBOCAN.
- K Chen, Q Zhang, M Peng, Y Shen, P Wan, et al. (2014) Relationship between tea consumption and pancreatic cancer risk: a meta-analysis based on prospective cohort studies and case-control studies. European Journal of Cancer Prevention 23(5): 353-360.
- CL Wolfgang, JM Herman, DA Laheru, Alison P Klein, Michael A Erdek, et al. (2013) Recent progress in pancreatic cancer. CA: A Cancer Journal for Clinicians 63(5): 318-348.

 Review Article
Review Article