Abstract
What Is the Meaning of Fetal Heart Rate Patterns According to Physiopathology?
The analysis and interpretation of the Fetal Heart Rate in the parturition watch have shown their limits in the identification of the fetuses risking neonatal asphyxia, which consequences are death or Cerebral Palsy. The weak specificity of the continuous electronic heart rate monitoring generates an unjustified C-section excess, associated to an increasing of the immediate and ulterior materno-fetal morbi-mortality. Presently, the visual analysis of the Fetal Heart Rate exclusively lays on chronomorphological patterns. However, there is another way that calls on the knowledge of the physiological processes of the fetus’ cardiovascular control:
a. In situation of normoxemia, the peripheral baroreceptors of the near term fetus are responsible for the control of the arterial pressure with both systems in action: the permanent accelerating sympathetic system and the intermittent braking parasympathetic system, which causes short-term variability.
b. In situation of acute hypoxemia, the peripheral chemoreceptors are stimulated and induce a rapid vagal mediated fall in heart rate (i.e. deceleration), and a peripheral vasoconstriction mediated by sympathetic nervous system.
In situation of acidosis, the central chemoreceptor is activated. It then stimulates the sympathetic system generating a fetal tachycardia, and inhibits the functioning of the peripheral baroreceptors, producing minimal to absent variability. From that comes out that the variability and the heart baseline are the only two patterns that reflect the fetal prognosis.
Keywords: Fetal Heart Rate; Physiopathological Approach; Baroreceptors; Neonatal Asphyxia; Caesarean Section
Abbreviations: SNS: Sympathetic Nervous System; BPM: Beats Per Minute; FHR: Fetal Heart Rate
Introduction
The development of continuous electronic heart rate monitoring during labour in the 1960s led to expectations that have not yet been satisfied. Also, several prospective studies have established the responsibility of continuous electronic monitoring in higher instrumental vaginal and abdominal delivery ratios without significant modifications of the neonatal prognosis [1,2]. The reasons of this relative failure are currently well known: First and foremost, electronic monitoring of the fetal heart rate (FHR) is a very sensitive technique but has a weak positive predictive value [3]. Furthermore, deficiencies exist in the interpretations and decisions regarding pathological heart rates [4-7] although these deficiencies can be improved through professional training [8,9]. Learned societies have produced remarkable efforts to standardize the terminology and simplify the FHR abnormality classification [3,10,11] along with encouraging the use of second line methods. These efforts, although restructured multiple times, have not really changed major perinatal indicators. Among the difficulties encountered, confusion is often seen in the examination and understanding of a pathological FHR monitoring. To this day, the visual analysis of the FHR exclusively relies on descriptive morphological and chronological criteria, and it is having been shown that this method produces substantial individual interpretation gaps [12-14]. However, there exists another approach based on knowledge of the physiological processes of the fetus’ cardiovascular control and which is advocated by some teams [15-25]. We propose a review of this method and intend to show that this kind of analysis leads to a better understanding of the fetus’ condition and therefore a simplified obstetrical decision.
Current State of Knowledge
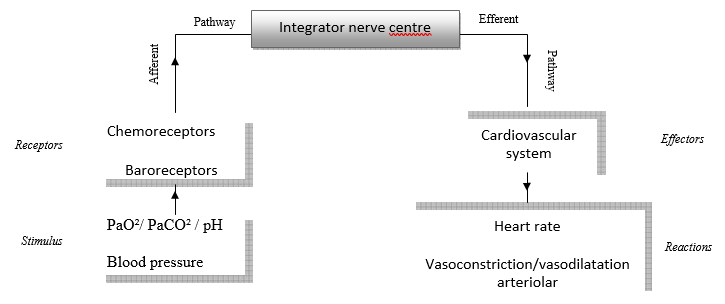
The cardiovascular control of a near-term fetus is like that of adults in certain aspects. The animal model generally used in experimentation is a near-term fetal sheep because of its many similarities with the human fetus [26]. The autonomous nervous system provides continuous adaptation of cardiac function to the fetus’ needs and to energetic availability (oxygen and glucose). This cardiovascular control relies on peripheral receptors (Figure 1): There are 2 types of peripheral fetal receptors (baroreceptors and chemoreceptors) located in the major elastic arteries of the neck and thorax – the aortic arch and the carotid bifurcations – and a central chemoreceptor located in the ventro-lateral surface of the medulla [16,27,28].
Peripheral Receptors
Baroreceptor: The baroreceptor responds to arterial pressure changes in the lumen of the aortic arch and the carotid bifurcations [29,30]. High blood pressure produces a fall in heart rate by the activation of the parasympathetic brake, whereas hypotension induces cardiac acceleration by a return of the basal sympathetic tone. This phenomenon is known as the baroreflex and is the most important component for the immediate control of arterial blood pressure [29].
Baroreflex Control of Blood Pressure: In the near-term fetus, baroreflex control seems to be exclusively achieved by the carotid baroreceptors [31,32]. The arterial baroreceptor’s stimulation by an elevation of the intraluminal pressure produces a spike train in the afferent nerve, and the response of the bulbar centre, mediated by the vague nerve, results in a brake effect on the sinus node. The delay between two cardiac systoles is consequently increased, resulting in a diminished heart rate and a decrease in blood pressure.
The Baroreflex and The Genesis of Short-Term Variability: During the 1960s and 1970s, Edward Hon and Jelte de Haan, quoted by Chester B. Martin, [33] noticed the preservation of the short term variability (STV) of the FHR in anencephalic fetuses, when the brainstem’s rostral structures – location of the sympathetic nuclei – are absent and when the parasympathetic nuclei and the hypothalamus are functional. In the physiological state, the peripheral baroreceptor is responsible by itself for maintaining the FHR, which is directly related to changes of intraluminal pressure in the aortic arch and carotid bifurcations. The baroreceptors are thus constantly submitted to an alternation of high and low pressures in relation with the cardiac cycle: each cardiac systole results in a blood surge which momentarily stretches the baroreceptors and provokes a deceleration in return. The opposite phenomenon occurs for the diastole. This deceleration - acceleration alternating causes constant heart rate variations, which produces the STV [33]. In other words, STV depicts the constant conflict between the parasympathetic brake and the sympathetic acceleration. STV is also an indicator of cardiac control by the peripheral baroreceptor. Its alteration reflects the overtaking of the cardiac function by the central chemoreceptor which we will see later. Long-term variability (LTV) shares the same origin as STV, yet also includes accelerating impulses of a sympathetic origin. This sympathetic overprint essentially comes from the fetal breathing activity [33] as well as from active movements, and more generally from variations of the fetal brain’s cortical activity [32].
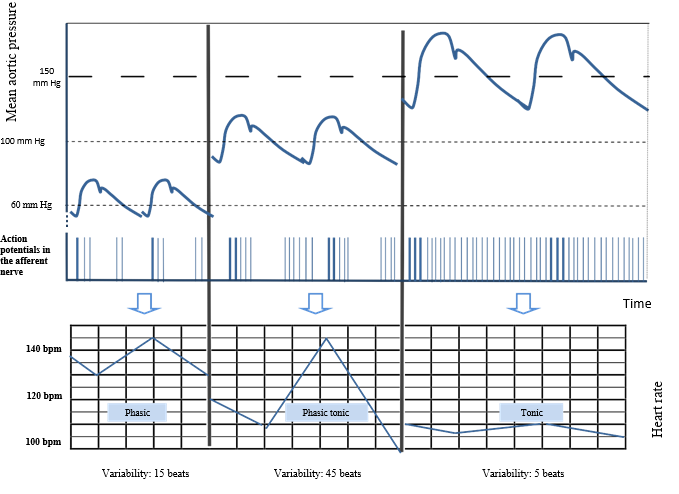
The Baroreceptors Characteristics are Those of Sensory Receptors [34-37] with Some Distinctive Features: In a situation of normoxemia, the baroreceptor operates on a phasic mode: it responds to the stimulus installation as well as its interruption by a spike train. It does not code for the duration of the stimulus but functions with an “on-off” effect. It is a fast adapting receptor. In the event of hypoxemia, the behaviour of the baroreceptor changes. Indeed, the sympathetic system, stimulated by the chemoreceptors, responds with peripheral vasoconstriction and vascular redistribution in order to preserve the oxygen supply to major organs such as the heart, brain and adrenal glands. The increase in blood pressure generated by this peripheral vasoconstriction changes the behaviour of the baroreceptor, causing it to go successively from a phasic mode to a phasic/tonic mode, ending with a tonic state. This is illustrated in (Figure 2) On the left side, with a normal mean aortic pressure, the variability of the FHR, characterized by alternating sympathetic acceleration and parasympathetic braking is estimated at 20 beats per minute (BPM). As the aortic pressure rises, parasympathetic braking is more intense, resulting in a decrease of the FHR. Consequently, sympathetic accelerations produce a greater contrast. This leads to a higher variability, estimated here at 45 BPM. In extreme cases, a severe elevation in blood pressure causes the baroreceptor to operate on a tonic mode: it fires action potentials on a permanent basis. The disappearance of the sympathetic activity results in a decrease of STV and even more of the LTV, which depends substantially on sympathetic tonus. This last stage, characterized by a succession of persistent action potentials, shows that a tonic receptor therefore codes for the duration of the hypertensive stimulation, and displays slow adaptation.
Figure 2: The baroreceptor’s behavior characterized by the train of action potentials recorded at the afferent nerve related to the level of the pressure in the aorta.
The Peripheral Chemoreceptor Detects a Decrease in PaO2 and Generates a Chemoreflex: The key efferent components of the peripheral chemoreflex are rapid vagal mediated falls in heart rate, i.e. decelerations in labour, and sympathetic nervous system (SNS) mediated peripheral vasoconstriction [25]. The recent works of Lear et al. have given us a better understanding of the origin of decelerations observed with FHR monitoring during labour. All atypical variable decelerations, whether prolonged or early, are signs of acute hypoxemia generated by a chemoreflex [25]. It is plausible that late decelerations are also signs of acute hypoxemia. They are directly associated with placental insufficiency [30]. The central chemoreceptor is influenced by interstitial fluid and cerebrospinal fluid pH, which reflect hydrogen ion levels of the plasma (metabolic acidosis) and by the level of the PaCO2 (respiratory acidosis) [38]. When activated, the chemoreceptor stimulates the SNS through the nucleus of the solitary tract, resulting in fetal tachycardia, inhibits the baroreceptors causing minimal or even absent FHR variability, and modulates the responses of the peripheral chemoreceptors [25].
Physiopathological Proposals
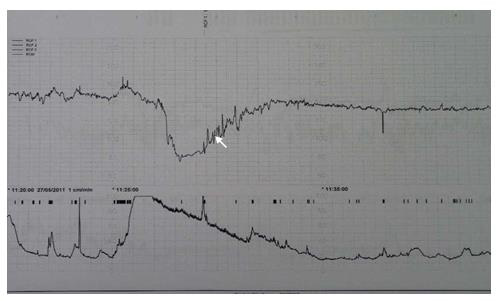
The Marked Variability in the Rising Phase of a Deceleration (Figures 2 & 3): This transient accentuation of the variability in relation to the post-acute phase of hypoxemia [32] is believed to be secondary to adrenalin secretion [39]. It is however probable that this marked variability is associated with the baroreceptor going into a phasic/tonic mode. The physiopathology of this event being that acute hypoxemia generates a chemoreflex that elevates blood pressure [40].
The FHR not returning to the baseline after a deceleration, along with a decrease of the variability reveals the tonic behaviour of the baroreceptor. This indicates sub-acute or persistent hypoxemia (Figure 3). The oxygen deprivation experienced by the fetus after a deceleration with hypoxic implications induces a quiet sleep state, allowing it to spare oxygen while restoring its reserves [40]. Quiet sleep state is primarily under parasympathetic control. It is easily identified on the FHR monitoring and consists of minimal variability without significant accelerations and a slightly decreased FHR baseline.
Figure 3: Transient increased variability during the rising phase and the lower level of the heart rate after the deceleration in relation to persistent hypoxemia.
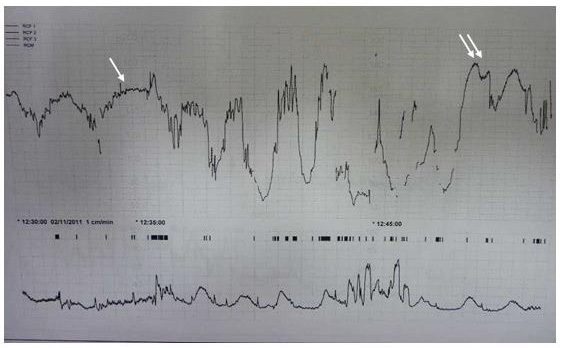
An Overshoot is a Prolonged Temporary Acceleration that Follows a Variable Deceleration: Its magnitude and duration are higher than the shoulder that ends a typical variable deceleration. Also, the FHR will show decreased variability. It is perceived as a warning signal. To this day, there is no consensus on the physiopathology of the overshoot. The hypotheses based on animal experiments and clinical observations rely mainly on the significant role of acidosis following a deceleration with myocardial hypoxia and adrenalin secretion [17,41]. As early as 1974, Dawes et al. showed that hypercapnia, which leads to respiratory acidosis, caused an increase in heart rate for the near-term fetal sheep [42]. There are therefore several concurring arguments that allow us to propose the following explanation: The overshoot reflects the transient hypercapnia that follows CO2 release by haemoglobin saturating in O2 after a phase of acute hypoxemia is over and oxygen levels go up again. This is known as the Haldane effect: the affinity of haemoglobin for CO2 decreases when the blood’s partial pressure of oxygen rises. Consequently, a brutal discharge of CO2 generating fetal respiratory acidemia stimulates the central chemoreceptor which, by a direct adrenergic effect (sympathetic b1 or b2) or indirect (adrenalin), causes a short acceleration that disappears as soon as the PaCO2 becomes normal. The overshoot prognosis is appreciated in several ways throughout publications, because there are two morphological types of overshoots, each with different implications. The first kind of overshoot is the one in which variability is preserved and that follows a deceleration with hypoxemia. It indicates a transient respiratory acidemia and has a reasonably good prognosis [(Figure 4) single arrow]. On the other hand, an overshoot with diminished variability following a deceleration with myocardial hypoxia is rather an indicator of mixed acidosis. In this situation, respiratory acidosis is manifested by the overshoot and metabolic acidosis by the disappearance of the STV [(Figure 4) double arrow].
Practical Proposals
A Wise Use of Oxytocin
Oxytocin must not be used in case of decelerations because it increases the hypoxic aggression [43].
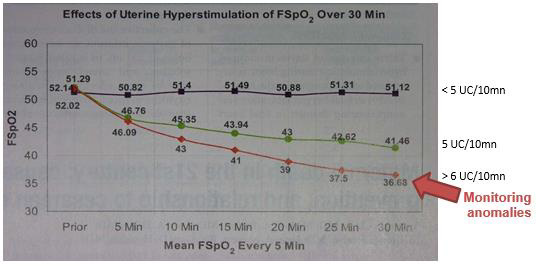
The objective is an average of four uterine contractions every ten minutes, threshold beyond which the fetal SaO2 decreases, even without decelerations (Figure 5) [43]. The interval between two contractions- the time necessary for reperfusion- is enough for a healthy fetus to restore full oxygenation and glycogenic reserves [25].
In case of pathologies with supposed placental insufficiencies (retarded fetal growth, diabetes, prolonged pregnancies…) a careful attitude is required because fetal recovery capacities are all severely altered [44,45].
The most important parameters when evaluating the FHR are the baseline and variability whom, when normal, show that the peripheral baroreceptor is functional and indicate the absence of metabolic acidosis.
The chrono-morphological and sometimes worrying aspects of decelerations do not enable the identification of acidosis [46,47], however they give an insight of the momentary state of the fetus adapting to a hostile environment. Decelerations can indicate acute hypoxemia (all decelerations except the typical variable, a marked variability), persistent hypoxemia (no restitution of the FHR baseline with a decrease in variability at the end of the deceleration), tissue hypoxia (decelerations combined with myocardial hypoxia), respiratory acidosis (overshoot with stable variability) or metabolic acidosis (fetal tachycardia and minimal or absent variability). Therefore, the physician must consider each type of deceleration as a sign of aggression against the fetus, but not as an immediate danger requiring extraction. Indeed, there are two steps of the fetal response to an acute hypoxemia:
Figure 5: Effects of uterine hyper stimulation on fetal oxygen status and FHR pattern.
Note: Rice Simpson K, James DC. Effects of oxytocin-induced uterine hyperstimulation during labor on fetal oxygen status and fetal heart rate patterns. Am J Obstet Gynecol 2008; 199: 34.e1-34.e5.
a. First a deceleration whose morphology and chronology depend on the nature and the mechanism of the stimulus, and whose magnitude depends on the intensity of the stimulus (i.e. the hypoxemia): the fetus “signals” an acute hypoxemia.
b. Secondly, after the deceleration, a change in variability (sometimes during the increase phase of the deceleration) and of the FHR baseline.
c. These modifications which occur immediately after the deceleration are observed on the interval between two decelerations. It is essential for the physician to take them into account, as they will determine the fetal prognosis according to three criteria: the amplitude range of the variability, the decrease or increase of the FHR baseline, and most significantly, the duration of these two anomalies. This analysis allows a dynamic interpretation combining the evolution of fetal data and ante and per-natal circumstances.
Examples:
Recurrent deceleration without any changes of neither the baseline FHR nor the baseline FHR variability: the fetus is healthy and has an unchanged acid/base balance [25,40]. An obstetrical intervention is unnecessary, except stopping oxytocin delivery if any and encouraging patient mobility.
Recurrent deceleration with a gradual increase of the baseline FHR and a gradual decrease of the baseline FHR variability: the fetus alters its acid/base balance [17]. In this case a fetal blood sampling allows an acidosis quantification, enabling either performing an obstetrical intervention or continuing monitoring, based on the context of evolution of fetal data and ante and per-natal circumstances. Let’s remember that the rate of Cerebral Palsy among new-borns with acidosis is 0.2% [48,49].
Prospects
The surveillance of the fetal well-being is still based on continuous electronic heart rate monitoring. Its weak specificity is responsible for an increase of caesarean rates with no improvements in the number of cerebral palsies or neonatal deaths. No alternative method (fetal pulse oxymetry, fetal blood samplings, STAN) has improved this specificity [50]. However, a physiopathological approach can increase this specificity because it allows a better understanding of the fetal acid-base state in real-time [51]. Hopefully, this point of view will spread and allow better indications for fetal blood sampling, reduce the risk of underestimating a danger for the fetus and limit caesareans during labour for fetal asphyxia suspicion.
Recommendations to Reach these Goals:
a. Conform to the international guidelines with an emphasis on a physiopathological analysis and dynamic interpretation.
b. A university degree of fetal heart rythmology should be created, with neurophysiologists, obstetricians, mid-wives, paediatricians and anaesthetists working together in order to train the future instructors.
c. Perinatal care networks as well as experimentation in the Aquitaine area should develop local FHR interpretation training sessions. It will be important to underline the benefits of a precise description of adverse outcomes in order to improve the data of perinatal indicators at a national level.
Conflict of Interests
None
Acknowledgement
Acknowledgements to Guillaume Doubre for his great help in this translation.
References
- Thacker SB, Stroup D, Chang MH (2006) Continuous electronic heart rate monitoring for fetal assessment during labour. Cochrane Database of Systematic Reviews (3).
- Alfirevic Z, Devane D, Gyte GML, Cuthbert A (2017) Continuous cardiotocography (CTG) as a form of electronic fetal monitoring (EFM) for fetal assessment during labour. Cochrane Database of Systematic Reviews (3).
- (2009) Intrapartum fetal heart rate monitoring: nomenclature, interpretation, and general management principles. ACOG Practice Bulletin No. 106, Obstet Gynecol 114(1): 192-202.
- Berglund S, Grunewald C, Pettersson H, Cnattingius S (2008) Severe asphyxia due to delivery- related malpractice in Sweden 1990–2005. BJOG 115(3): 316-323.
- Jonsson M, Nordén-Lindeberg S, Östlund I, Hanson U (2009) Metabolic acidosis at birth and suboptimal care-illustration of the gap between knowledge and clinical practice- BJOG 116(11): 1453-1460.
- Shaw RW (2000) Confidential Enquiry into Stillbirths and Deaths in Infancy. report 7th Annual. Maternal and Child Health Research Consortium London, UK, p. 53-64.
- (2007) Confidential Enquiry into Maternal and Child Health. Perinatal Mortality 2005: England Wales and Northern Ireland. London CEMACH, UK.
- Young P, Hamilton R, Hodgett S, Moss M, Rigby C, et al. (2001) Reducing risk by improving standards of intrapartum fetal care. J R Soc Med 94(5): 226-231.
- Pehrson C, Sorensen JL, Amer Wåhlin (2011) I Evaluation and impact of cardiotocography training programmes: a systematic review. BJOG 118(8): 926-935.
- (2008) Collège National des Gynécologues-Obstétriciens Franç Recommandations pour la Pratique Cinique: modalités de surveillance fœtale pendant le travail. J Gynecol Obstet Biol Reprod 37(1): 1-107.
- (2001) The use of electronic fetal monitoring: the use and interpretation of cardiotocography in intrapartum fetal surveillance. NICE Inherited Clinical Guidelines C. London: National Institute for Clinical Excellence.
- Rhöse S, Heinis AMF, Vandenbussche F, Van Drongelen J, Van Dillen J, et al. (2014) Inter- and intra- observer agreement of non-reassuring cardiotocography analysis and subsequent clinical management. Acta Obstet Gynecol Scand 93(6): 596-602.
- Beaulieu MD, Fabia J, Leduc B, Brisson J, Bastide A, et al. (1982) The reproducibility of intrapartum cardiotocogram assessments. Can Med Assoc J 127(3): 214-613.
- Palomäki O, Luukkaala T, Riikka L, Tuimala R (2006) Intrapartum cardiotocography –the dilemna of interpretational variation. J Perinat Med 34(4): 298-302.
- Giussiani DA, Spencer J, Hanson M (1994) Fetal cardiovascular reflex responses to hypoxemia. Fetal and maternal medicine Review 6(1): 17-37
- Westgate J, Wibbens B, Bennet L, Wassink G, Parer JT, et al. (2007) The intrapartum deceleration in center stage: a physiologic approach to the interpretation of fetal heart rate changes in labor. Am J Obstet Gynecol 197(3): 236.e1-236.e11.
- Bennet L, Gunn AJ (2009) The Fetal Heart Rate Response to hypoxia: Insights from Animal Models. Clin Perinatol 36(3): 655-672.
- Cibils LA (2009) On intrapartum fetal monitoring. Am J Obstet Gynecol 4: 1382-1389.
- Chandraharan E (2010) Rational approach to fetal monitoring during labour in “all” resource settings. Sri Lanka Journal of Obstetrics and gynaecology 32(4): 77-84.
- Doret M, Constans A, Gaucherand P (2010) Physiologic basis for fetal heart rate analysis during labour. Journal de Gynécologie Obstétrique et Biologie de la Reproduction 39: 276-283.
- Secourgeon JF (2012) Hypoxemia, peripheral chemoreceptors and fetal heart rate. Journal de Gynécologie Obstétrique et Biologie de la Reproduction 41(1): 26-40.
- Ugwumadu A (2013) Understanding cardiotocographic patterns associated with intrapartum fetal hypoxia and neurologic injury. Bpobgyn 27(4): 509-536.
- Ugwumadu A (2014) Are we (mis)guided by current guidelines on intrapartum fetal heart rate monitoring? Case for a more physiological approach to interpretation. BJOG 121(9): 1063-1070.
- Pinas A, Chandraharan E (2016) Continuous cardiotocography during labour: Analysis, classification and management. Bpobgyn 30: 33-47.
- Lear CA, Galinsky R, Wassink G, Yamaguchi K, Davidson JO, et al. (2016) The myths and physiology surrounding intrapartum decelerations: the critical role of the peripheral chemoreflex. J Physiol 594(17): 4711-4725.
- Garabedian C, De Jonckheere J, Butruille L, Deruelle P, Storme L, et al. (2017) Understanding fetal physiology and second line monitoring during labor. J Gynecol Obstet Hum Reprod 46(2): 6113-6117.
- O Regan RG, Majcherczyk S (1982) Role of peripheral chemoreceptors and central chemosensitivity in the regulation of respiration and circulation. J Exp Biol 100: 23-40.
- Dobbins EG, Feldman JL (1994) Brainstem network controlling descending drive to phrenic motoneurons in rat. J Comp Neurol 347(1): 64-86.
- Benarroch E (2008) The arterial baroreflex. Functional organization and involvement in neurologic disease. Neurology 71(21): 1733-1738.
- Itskovitz J, LaGamma EF, Rudolph AM (1983) Heart rate and blood pressure responses to umbilical cord compression in fetal lamb with special reference to the mechanism of variable deceleration. Am J Obstet Gynecol 147(4): 451-457.
- De Haan J, Martin CB, Evers JLH, Jongsma HW (1981) Pathophysiology of variable and late decelerations. J Perinatal Medicine 9(1): 7-16.
- Afors K, Chandraharan E (2011) Use of continuous electronic fetal monitoring in a preterm fetus: clinical dilemna and recommendations for practice. J Pregnancy 2011: 1-7.
- Martin CB (1982) Physiology and clinical use of fetal heart rate variability. Clinics in perinatology 9(2): 339-351.
- Blanco CE, Dawes GS, Hanson MA, McCooke HB (1988) Carotid Baroreceptors in Fetal and Newborn Sheep. Pediatric Research 24(3): 342-346.
- Berne RM, Levy MN (1993) The peripheral circulation and its control In Physiology (3rd ): R. Farrel, St Louis, (Eds.). pp. 438.
- Constant I (2006) Le système nerveux autonome revisité, Conférences d'actualisation, p. 51-72.
- Sherwood L (2016) The blood vessels In Human physiology (9th Edn). Boston Cengage learning, p. 366-367.
- Darnall RA (2010) The role of CO2 and central chemoreception in the control of breathing in the fetus and the neonate. Respiratory Physiology & Neurobiology 173(3): 201-212.
- Giussani DA, Spencer JA, Moore PJ, Bennet L, Hanson MA, et al. (1993) Afferent and efferent components of the cardiovascular reflex responses to acute hypoxia in term fetal sheep. J Physiol 461: 431-449.
- Lear CA, Westgate JA, Ugwumadu A, Nijhuis JG, Stone PR, et al. (2018) Understanding Fetal Heart Rate Patterns That May Predict Antenatal and Intrapartum Neural Injury. Semin Pediatr Neurol 28: 3-16.
- Westgate JA, Bennet L, De Haan HH, Gunn AJ (2001) Fetal heart rate overshoots during repeated umbilical cord occlusion in sheep. Obstet Gynecol 97(3): 454-459.
- Boddy K, Dawes GS, Fischer R, Pinter S, Robinson JS, et al. (1974) Foetal respiratory movements, electrocortical and cardiovascular responses to hypoxaemia and hypercapnia in sheep. J Physiol 243(3): 599-618.
- Rice Simpson K, James DC (2008) Effects of oxytocin-induced uterine hyperstimulation during labour on fetal oxygen status and fetal heart rate patterns. Am J Obstet Gynecol 199(34): 1-5.
- Westgate JA, Wassink G, Bennet L, Gunn AJ (2005) Spontaneous hypoxia in multiple pregnancies is associated with early fetal decompensation and greater T-wave elevation during brief repeated cord occlusion in near-term fetal sheep. Am J Obstet Gynecol 193(4): 1526-1533.
- Wassink G, Bennet L, Davidson JO, Westgate JA, Gunn AJ, et al. (2013) Pre-existing hypoxia is associated with greater EEG suppression and early onset of evolving seizure activity during brief repeated asphyxia in near-term fetal sheep 8(8): e73895.
- Parer JT, Ikeda T (2007) A framework for standardized management of intrapartum fetal heart rate patterns. Am J Obstet Gynecol 197(26): 1-6.
- Cahill AG, Roehl KA, Odibo AO, Macones GA (2012) Association and prediction of neonatal acidemia. Am J Obstet Gynecol 207(3): 206.e1-8.
- Nelson KB, Dambrosia JM, Ting TY, Grether JK (1996) Uncertain value of electronic fetal monitoring in predicting cerebral palsy. N Engl J Med 334(10): 613-618.
- Parer JT, King T, Flanders S, Fox M, Kilpatrick SJ, et al. (2006) Fetal acidemia and electronic fetal heart rate patterns: Is there evidence of an association? J Mat Fetal Med 19(5): 289-294.
- Audibert F (2008) Alternative techniques of fetal assessment during labor: Future or past? Journal de Gynécologie Obstétrique et Biologie de la Reproduction 37(7): 633.
- Haddad S, Secourgeon JF, Pueyo S (2016) Implication of a Perinatal Care Network in the Regional Harmonization of the Evaluation of Fetal State during Parturition. Rev Med Perinat 8: 226-231.

 Research Article
Research Article