Abstract
Dehydration, secondary to gastroenteritis, is one of the most common reasons for office visits and hospital admissions in developed countries. It results from a decrease in total body water content either due to less intake or more fluid loss. Common symptoms of dehydration are dry mouth/tongue, thirst, headache, and lethargy. A person can become dehydrated if they lose as little as 3% of their body weight from water depletion. However, little or no information is available on the impact of diet to the weight of such persons when dehydrated. In either of the two study centers, Benin or Nnewi, majority of the children who had diarrhea were those whose main diet is breast milk and solid food. For mild dehydration, we found that solid food is associated with weight gain in children (mean weight: 12.167, CI: 9.5151-14.818, p-Value: 0.00207; for Benin) and (mean weight: 12.11, CI: 10.597-13.623, p-Value: 0.00014; for Nnewi) using breast milk and water as a reference. In this study, we observed a weak correlation between age and weight of the children. The observed increase in weight among those with severe dehydration is found to be due to the kind of food they eat. It is observed that children who suffered severe dehydration were given more of solid food than other forms of diet. This will guide the quality of food given to children in order to increase their weight whenever they loose weight as a result of dehydration.
Keywords: Dehydration; Body Weight; Diet; Breast Milk and Water
Introduction
Dehydration, secondary to gastroenteritis, is one of the most common reasons for office visits and hospital admissions in developed countries [1-3]. Among European children, 3 years of age, incidence of diarrhea ranges from 0.5 to 1.9 episodes per child per year [4]. The management of acute diarrhea in children is largely based on clinical examination which allows assessment of hydration status. Underestimation of dehydration increases morbidity and mortality, while overestimation can result in inappropriate care and public expenditure. Dehydration is difficult to diagnose clinically [5]. Combinations of examination signs perform markedly better than any individual sign in predicting dehydration [6]. Many scores have been developed to estimate dehydration, but only one, the Clinical Dehydration Scale, has been validated to predict a longer length of Emergency Department (ED) stay and the need for intravenous fluid rehydration [7]. The development and validation of a dehydration scale requires the use of a gold standard. Weight loss is considered as the reference to diagnose dehydration in clinical practice and research [6]. In healthy adults with normal Body Mass Index (BMI), water accounts for 60% of body weight [8-12]. A person can become dehydrated if they lose as little as 3% of their body weight from water depletion [13].
The loss of body mass with no water intake is associated with poor memory and attention [14]. Studies have reported dehydration by 1–2%, impairs cognitive performance and impacts psychomotor and memory skills [15-18]. Fluid deficit of 4% decreases performance, causes headaches, irritability, sleepiness and increases respiratory rate with increase in temperature among children [19]. Dehydration also impairs muscle endurance and decreases muscle strength [20]. The fluid depletion of more than 8% can cause death [21]. Dehydration results from a decrease in total body water content either due to less intake or more fluid loss. Common symptoms of dehydration are dry mouth/tongue, thirst, headache, and lethargy [22]. The common symptoms of mild to moderate dehydration are dry mouth/tongue, thirst, headache, lethargy, fatigue, dry skin, muscle weakness, light-headedness, dizziness and a lack of focus. People with severe dehydration can present with sunken eyes, lack of tears, sunken fontanels (specifically among infants), hypotension, tachycardia and, in the worst-case scenario, unconsciousness [23]. However, there is little or no information about the impact of their diet to their weight irrespective of their dehydration status. This therefore becomes the aim of this study.
Methodology
Selection of Participants
A cross-sectional study was conducted among lactating mothers attending University of Benin Teaching Hospital and Nnamdi Azikiwe University Teaching Hospital in Benin and Nnewi respectively. All lactating mothers (and their children) attending the respective facilities were asked about their willingness to participate in the study. The study population refers to children 0-5 years of age presenting with clinically confirmed diarrhea. Among those who fulfilled the selection criteria, 327 children (on both study sites) were randomly recruited for this study. The research protocol was approved by the institutional ethics committee and all participants gave written informed consent.
Anthropometry and Socio-Demographic Information
Weight was measured in light clothes, without shoes, using an electronic digital scale to the nearest 0.1 kg. A structured and pre-tested questionnaire was administered to collect information on socio-demographic factors such as age, residence (rural or urban) and gender. Detailed history of lactation regarding whether baby was breastfed exclusively or partially [24], were recorded. Exclusive breastfeeding (EBF) was defined when the child received breast milk only and no other solids or liquids with the exception of vitamins, minerals, medicines or oral rehydration solution [24]; Partial breastfeeding (PBF) was defined when the infant received breast milk in addition to complementary foods; and complementary foods included milk, infant formula, gruel or semisolid foods given in addition to breast milk [24].
Statistical Analysis
We used unadjusted and multivariable linear regression models to assess the association of the weight of the child with the child’s diet and with the child’s dehydration status. We conducted data analyses using R based on R programming language.
Results and discussion
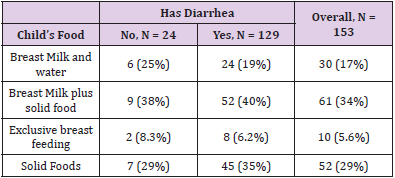
In Benin, Of the 129 children who had diarrhea, 24 (19%) fed mainly on breast milk and water, 52 (40%) fed on breast and solid food, 8 (6.2%) fed on exclusive breast milk and 45 (35%) fed only on solid food (Table 1).
Table 1: Bivariant analysis between child’s food and diarrhea status among children in Benin study site.
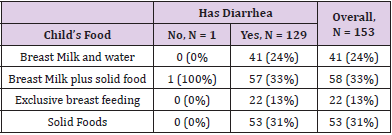
While in Nnewi, Of the 173 children who had diarrhea, 41 (24%) fed only on breast milk and water, 57 (33%) fed on breast and solid food, 22 (13%) fed on exclusive breast milk and 53 (31%) fed only on solid food (Table 2). A prolonged bout of diarrhea or vomiting can cause the body to lose more fluid than it can take in. The result is dehydration, which occurs when the body doesn’t have the fluid it needs to function properly. Severe dehydration can cause the kidneys to shut down [25]. In either of the centers, majority of the children who had diarrhea were those whose main diet is breast milk and solid food. Occasionally babies will react to something in their mother’s breastmilk, and this may sometimes cause diarrhea or constipation [26]. However, those who exclusively fed on breast milk had the least incidence of diarrhea. Exclusive breastfeeding, defined as feeding only human milk with no other liquids or foods, is known to offer maximum protection against diarrhea to infants younger than 6 months of age, whereas partial breastfeeding offers intermediate protection compared with no breastfeeding [27]. Previously, Lamberti, et al. analyzed the effect of suboptimal breastfeeding on diarrhea morbidity and mortality, based on review of 18 studies from developing countries published from 1980 to 2009 [28].
Table 2: Bivariant analysis between child’s food and diarrhea status among children in Nnewi study site.
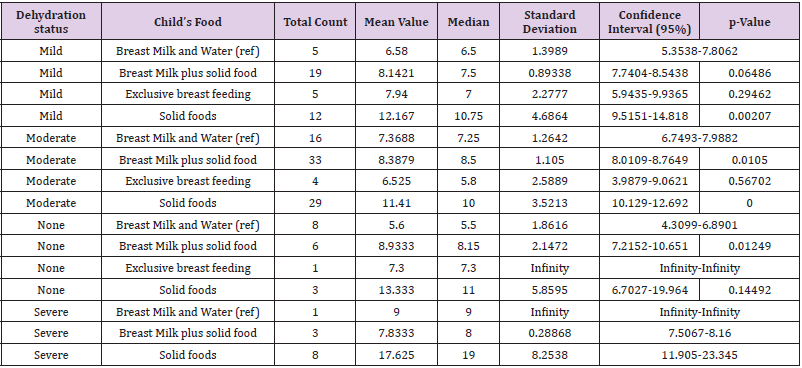
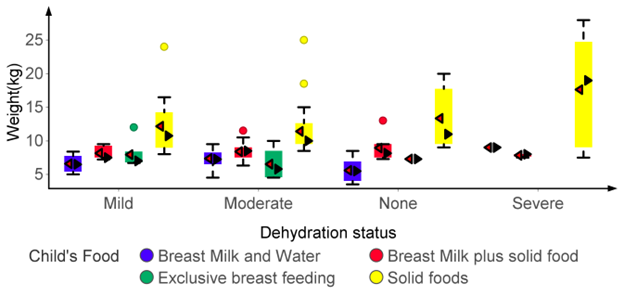
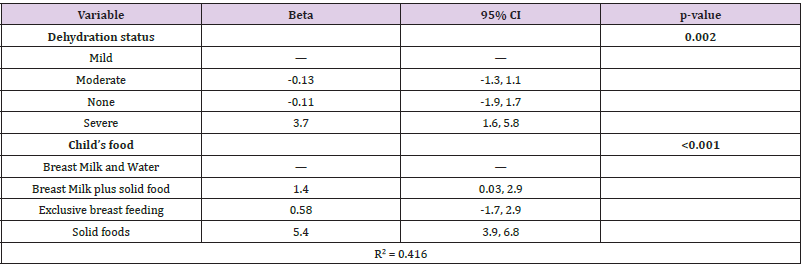
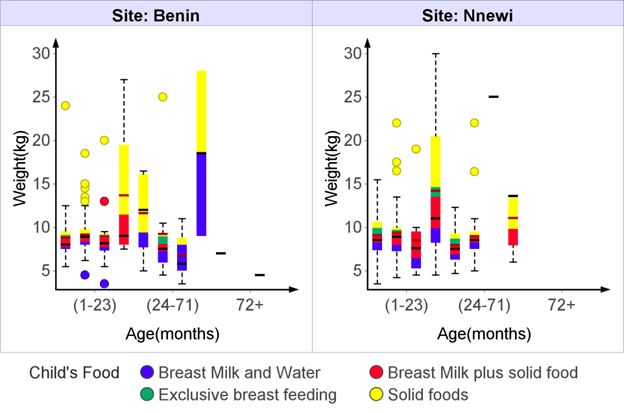
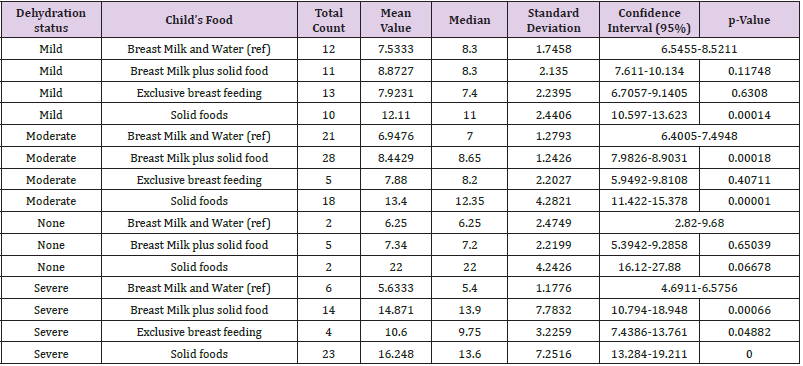
They found that not breast feeding was associated with a 165% (relative risk (RR) 2.65, 95% confidence interval (CI) 1.72–4.07) increase in diarrhea incidence in infants aged 0–5 months, a 32% (RR: 1.32, 95% CI 1.06–1.63) increase in those aged 6–11 month, and a 32% (RR: 1.32, 95% CI 1.06–1.63) increase in those aged 12–23 months. No breastfeeding was also associated with a 952% (RR: 10.52, 95% CI 2.79–49.6) increase in diarrhea mortality as compared to exclusive breastfeeding in infants aged 0–5 months of age, a 47% (RR: 1.47, 95% CI 0.67–3.25) increase as compared to any breastfeeding practice in those aged 6–11 months, and a 157% (RR: 2.57, 95% CI 1.10–6.01) increase in those aged 12–23 months. The indicator most commonly used to estimate dehydration status is acute weight loss [29]. For mild dehydration, we found that solid food is associated with weight gain in children (mean weight: 12.167, CI: 9.5151-14.818, p-Value: 0.00207; for Benin) (Figure 1 and Table 3) and (mean weight: 12.11, CI: 10.597-13.623, p-Value: 0.00014; for Nnewi) (Figure 1 and Table 3) using breast milk and water as a reference. The same was noticed in children with moderate dehydration, which is significant for those from Nnewi (mean weight: 13.4, CI: 11.422-15.378, p-Value: 0.00001) (Figure 1 and Table 3) at a level of significance tested at p<0.05. Data from a previous study suggest that the introduction of solid foods earlier than 4 months of age is associated with increased body fat or weight in childhood or with greater weight gain during infancy [30,31]. The combination of breast milk and solid food was observed to contribute significantly to the weight of the children with either moderate or severe dehydration Benin and Nnewi respectively (Figures 1 & 2).
Table 3: Statistical parameters based on the association between dehydration status and the child’s food (Benin site).
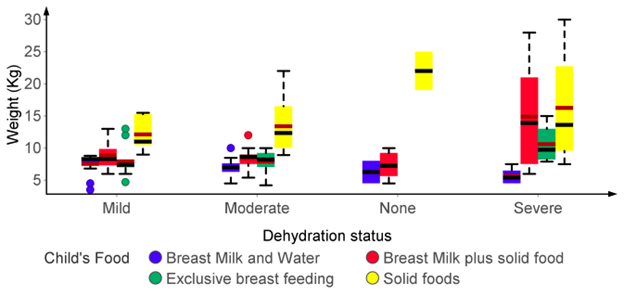
Figure 1: Variability of weight among children with different dehydration status based on their diet (Benin site).
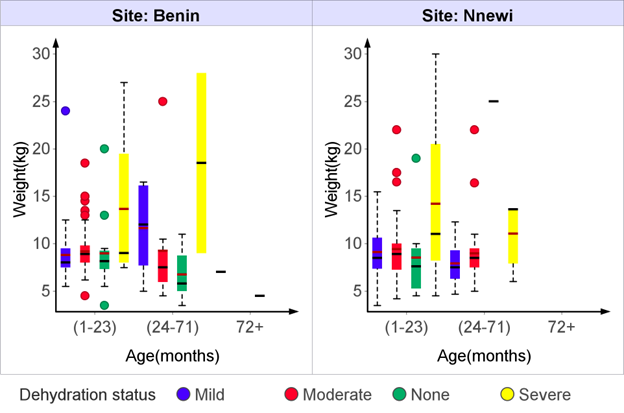
Figure 2: Variability of weight among children with different dehydration status based on their diet (Nnewi site).
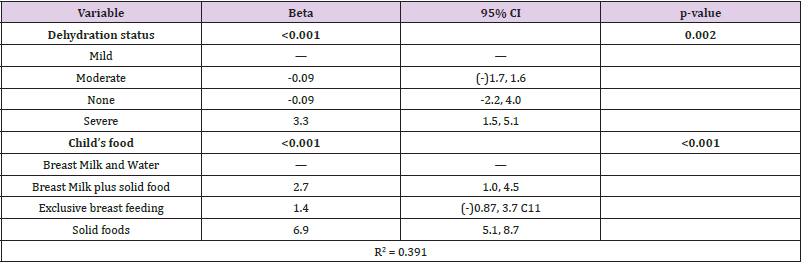
Other studies indicate that partial breastfeeding is associated with increased weight gain; this could be a starting point for obesity in children [8] irrespective of the dehydration status. In order to carry out a multivariant analysis, a linear regression model was developed based on the weight of the children as the dependent variable. When compared to the mild dehydration status of children in either study site, the results of our findings revealed that moderate dehydration status of children contribute negatively to the weight of the child i.e as the dehydration status progresses from mild to moderate, the weight of the child decreases drastically. Mild dehydration is the loss of no more than 5 to 6 percent loss of body weight. Loss of 7 to 10 percent is considered moderate dehydration [9]. However, as the condition worsen from mild to severe, we observed a multiple fold increase to the child’s weight irrespective of the study site (Tables 4 and 5). This is surprising as we expect a further decrease in the weight of the children with severe dehydration. Therefore, we considered other factors that may be responsible to a child’s weight despite their dehydration status and we selected the child’s age and sex. Body weight correlated more with age compared to gender [32]. In this study, we observed a weak correlation between age and weight of the children (Figure 3).
Figure 3: Variability of weight among children with different dehydration status based on their diet (Nnewi site).
This might be because age ranges within the adolescent and adulthood category were not considered in this study. The prevalence of each body weight category varied with age. Normal body weight was most prevalent (81.8%) at adolescence and least (27.5%) at middle adulthood. This implies that while adolescence is the healthiest age group, middle adult stage represents the most vulnerable segment of the population. This may be due to the fact that while young persons are physically active, the older ones are less active and tends to assume a sedentary lifestyle; at 40 years and above, most persons have passed the peak of their physical strength and biological functions [33-35]. However, those with severe dehydration experienced a dramatic rise in their weight despite their age category (Table 6). This increase in weight among those with severe dehydration is found to be due to the kind of food they eat. It is observed that children who suffered severe dehydration were given more of solid food than other forms of diet (Figure 4). This is highly commendable as we observed that diet compensate for weight loss in the presence of an infection that causes dehydration.
Figure 4: Variability of weight among children with different dehydration status based on their diet.
Table 6: Statistical parameters based on the association between dehydration status and the child’s food (Nnewi site).
Conclusion
The indicator most commonly used to estimate dehydration status is acute weight loss. The results of our findings revealed that moderate dehydration status of children contribute negatively to the weight of the child i.e as the dehydration status progresses from mild to moderate, the weight of the child decreases drastically. The observed increase in weight among those with severe dehydration is found to be due to the kind of food they eat. It is observed that children who suffered severe dehydration were given more of solid food than other forms of diet. This will guide the quality of food given to children in order to increase their weight whenever they lose weight as a result of dehydration.
Disclosure
The authors declare no conflict of interest.
References
- Parashar UD, Hummelman EG, Bresee JS, Miller MA, Glass RI (2003) Global illness and deaths caused by rotavirus disease in children. Emerg Infect Dis 9: 565-572.
- Soriano-Gabarro M, Mrukowicz J, Vesikari T, Verstraeten T (2006) Burden of rotavirus disease in European Union Countries. Pediatr Infect Dis J 25: S7-S11.
- Glass RI, Lew JF, Gangarosa RE, LeBaron CW, Ho MS (1991) Estimates of morbidity and mortality rates for diarrheal diseases in American children. J Pediatr 118: S27-33.
- Guarino A, Albano F, Ashkenazi S, Gendrel D, Hoekstra JH, et al. (2008) European Society for Paediatric Gastroenterology, Hepatology, and Nutrition/ European Society for Paediatric Infectious Diseases evidence-based guidelines for the management of acute gastroenteritis in children in Europe. J Pediatr Gastroenterol Nutr 46: S81-122.
- Steiner MJ, De Walt DA, Byerley JS (2004) Is this child dehydrated? JAMA 291: 2746-2754.
- Gorelick MH, Shaw KN, Murphy KO (1997) Validity and reliability of clinical signs in the diagnosis of dehydration in children. Pediatrics 99: 99-106.
- Friedman JN, Goldman RD, Srivastava R, Parkin PC (2004) Development of a clinical dehydration scale for use in children between 1 and 36 months of age. J Pediatr 145: 201-207.
- Kim J, Peterson KE, Scanlon KS, Garrett M Fitzmaurice, Aviva Must, et al. (2006)Trends in overweight from 1980 through 2001 among preschool-aged children enrolled in a health maintenance organization. Obesity (Silver Spring)14(7): 1107-1112 .
- Davis MM, Gance-Cleveland B, Hassink S, Johnson R, Paradis G, et al. (2007) Recommendations for prevention of childhood obesity. Pediatrics 120(suppl 4): S229-S253.
- Gillman MW (2008) The first months of life: a critical period for development of obesity. Am J Clin Nutr 87(6): 1587-1589.
- Wilson AC, Forsyth JS, Greene SA, Irvine L, Hau C, et al. (1998) Relation of infant diet to childhood health: seven year follow up of cohort of children in Dundee infant feeding study. BMJ 316(7124): 21-25.
- Baker JL, Michaelsen KF, Rasmussen KM, Sorensen TI. Maternal prepregnant body mass index, duration of.
- Kim J, Peterson KE (2008) Association of infant child care with infant feeding practices and weight gain among US infants. Arch Pediatr Adolesc Med 162(7): 627- 633
- Taveras EM, Rifas-Shiman SL, Belfort MB, Kleinman KP, Oken E, et al. (2009) Weight status in the first 6 months of life and obesity at 3 years of age. Pediatrics 123(4): 1177-1183
- Reilly JJ, Armstrong J, Dorosty AR, Emmett PM, Ness A, et al. (2005)Early life risk factors for obesity in childhood: cohort study. BMJ 330(7504): 1357.
- Burdette HL, Whitaker RC, Hall WC, Daniels SR (2006) Breastfeeding, introduction of complementary foods, and adiposity at 5 y of age. Am J Clin Nutr 83(3): 550 -558.
- Kramer MS (1981) Do breast-feeding and delayed introduction of solid foods protect against subsequent obesity? J Pediatr 98(6): 883- 887.
- Mehta KC, Specker BL, Bartholmey S, Giddens J, M L Ho (1998) Trial on timing of introduction to solids and food type on infant growth. Pediatrics 102(3 pt 1): 569-573.
- Agras WS, Kraemer HC, Berkowitz RI, Hammer LD (1990) Influence of early feeding style on adiposity at 6 years of age. J Pediatr 116(5): 805-809.
- Forsyth JS, Ogston SA, Clark A, Florey CD, Howie PW (1993) Relation between early introduction of solid food to infants and their weight and illnesses during the first two years of life. BMJ306(6892): 1572-1576.
- Gillman MW, Rifas-Shiman SL, Kleinman KP, Rich-Edwards JW, Lipshultz SE (2004) Maternal calcium intake and offspring blood pressure. Circulation 110(14): 1990-1995.
- Huh SY, Rifas-Shiman SL, Taveras EM, Oken E, Gillman MW (2011) Timing of solid food introduction and risk of obesity in preschool-aged children. Pediatrics 127(3): e544-e551.
- Heinig MJ, Nommsen LA, Peerson JM, Lonnerdal B, Dewey KG (1993) Intake and growth of breast-fed and formula-fed infants in relation to the timing of introduction of complementary foods: the DARLING study. Davis Area Research on Lactation, Infant Nutrition and Growth. Acta Paediatr 82(12): 999-1006.
- Mihrshahi S, Oddy WH, Peat JK, Iqbal Kabir (2008) Association between infant feeding patterns and diarrhoeal and respiratory illness: a cohort study in Chittagong, Bangladesh. Int Breastfeed J 3: 23-28.
- Ochoa TJ, Chea-Woo E (2019)Approach to patients with gastrointestinal tract infections and food poisoning. In: Cherry JD, Harrison GJ, Kaplan SL, Steinbach WJ, Hotez PJ (Eds.)., Feigin and Cherry's Textbook of Pediatric Infectious Diseases (8th)., Philadelphia, PA: Elsevier Saunders chap, p. 44.
- Morrow AL, Rangel JM (2004) Human milk protection against infectious diarrhea: implications for prevention and clinical care. Semin Pediatr Infect Dis 15: 221-228.
- Lamberti LM, Fischer Walker CL, Noiman A, Victoria C, Black RE (2011)Breastfeeding and the risk for diarrhea morbidity and mortality. BMC Public Health 11: S15.
- Hirschhorn N (1980) The treatment of acute diarrhea in children. An historical and physiological perspective. The American journal of clinical nutrition 33(3): 637-663.
- Pruvost I, Dubos F, Chazard E, Hue V, Duhamel A, et al. (2013) The value of body weight measurement to assess dehydration in children. PloS one 8(1): e55063.
- Baker JL, Michaelsen KF, Rasmussen KM, Sorensen TI (2004) Maternal prepregnant body mass index, duration of breastfeeding, and timing of complementary food introduction are associated with infant weight gain. Am J Clin Nutr 80(6): 1579-1588.
- Kim J, Peterson KE (2008) Association of infant child care with infant feeding practices and weight gain among US infants. Arch Pediatr Adolesc Med 162(7): 627-633.
- Kajale NA, Chiplonkar SA, Khadilkar V, Khadilkar AV (2016) Effect of breastfeeding practices and maternal nutrition on baby’s weight gain during first 6 months. The Journal of Obstetrics and Gynecology of India 66(1): 335-339.
- Steiner MJ, Darren A DeWalt, Julie S Byerley (2004) Is this child dehydrated? Journal of the American Medical Association291, no. 22: 2746-2754.
- Chinedu SN, Emiloju OC, Azuh DE, Ogunlana OO, Iheagwam FN (2017) Association between age, gender and body weight in educational institutions in Ota, Southwest Nigeria. Asian Journal of epidemiology 10(3): 144-149.
- Shephard RJ (1998) Aging and Exercise. In: Encyclopedia of Sports Medicine and Science, Fahey, T.D. (Eds.)., Internet Society for Sport Science, USA.

 Research Article
Research Article