Abstract
Background: The robotic lobectomy is performed by either a three- or a fourarm technique. We hypothesized that the robotic-assisted four-arm lobectomy might be faster and may allow dissection of a higher number and mass of lymph nodes in patients with lung cancer.
Methods: A total of 50 lung cancer patients (32 males and 18 females) who underwent a robotic-assisted lobectomy from November 2018 to February 2020 by a single surgeon were retrospectively analyzed. Operative time, dissected lymph nodes and post-operative outcome were compared following a three- and four-arm robotic lobectomy.
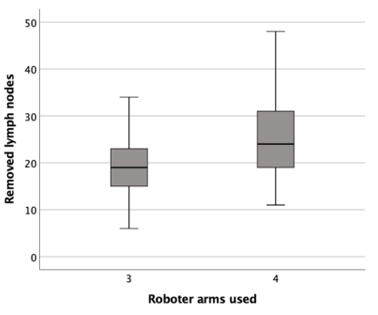
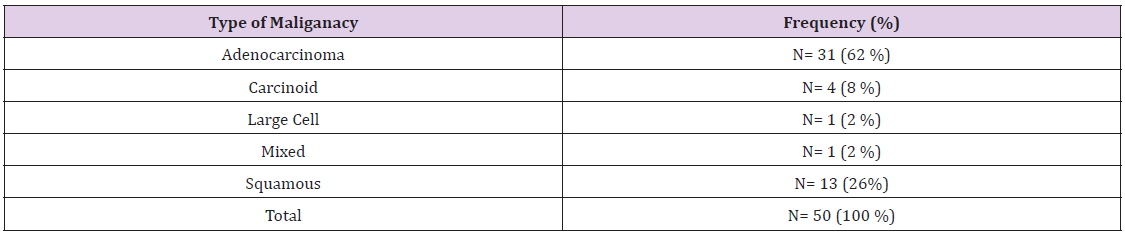
Results: There was no significant difference in operating time between the four-arm and three-arm groups (178.29 versus 164.29 minutes; p= 0.26). The mean operation duration was determined to be 172 minutes. Of the 50 patients reviewed, 31(62%) presented histologically an adenocarcinoma. The operating time was significantly longer for left upper lobectomy as compared to right lower lobectomy (p=0.04). On average, 23 lymph nodes were harvested. The four-arm technique was associated with increased lymph node harvest as compared to the three-arm technique (mean, 25.5 versus 19.1; p=0.012). There was no significant difference in complication rates by both techniques, and the 90-day mortality was zero.
Conclusion: The robotic-assisted lobectomy using the four-arm technique is safe and harvests yields potentially more lymph nodes than when using the three-arm technique, confirming our hypothesis. This preliminary result advocates for further four-arm robotic lobectomy in patients with lung cancer.
Keywords: Robotic; Lobectomy; Fourarm Robot; Three-arm Robot; Outcomes; Lymphadenectomy; Lung cancer
Abbreviations: FDG-PET: Fluoro-2- Deoxyglucose; MRI: Magnetic Resonance Imaging; PFS: Progression-Free; OS: Overall Survival; RATS: Robotic-Assisted Thoracoscopic Surgery; MIT: Minimally Invasive Techniques
Introduction
Robotic-Assisted Thoracoscopic Surgery (RATS) is a recent development in minimally invasive techniques (MIT). It has been used in gynecology, urology as well as in the thoracic surgery. This development has led to an increase in the number of minimally invasive procedures in the thoracic surgery. The reasons for this observation are the endo-wrist function with the seven grades of freedom, their ability to mimic human hand movement intrathoracically, and magnified 3D imaging, which offers a useful view, as small vessels can easily be identified and managed in case of any bleeding by early coagulation [1]. Furthermore, the exposition is optimized, and it makes precise maneuvers around critical structures easy and accurate. Moreover, the camera is stable and controlled by the surgeon at the console [2]. Many trials have demonstrated the safety and feasibility of robotic lobectomy [3,4]. The technique of RATS lobectomy can be learned in a center of excellence and by receiving help from a proctor, particularly in the early phase of the program [5]. Many assessments and an operating phase designed to develop a methodical approach to the RATS have been described, and a stepwise increase in the case complexity during the initial learning curve has been recommended [5,6]. Several studies have explored oncological aspects of the robotic lobectomy for lung cancer. The robotic lobectomy has proven to be safe with oncologic efficacy similar to lobectomy via a thoracotomy or video-assisted thoracoscopic surgery (VATS) [7]. A particular strength of the robotic platform is the improvement of a lymph node dissection. Studies have shown that lymphadenectomy with RATS can lead to increased lymph node harvest as compared with VATS and has been associated with a high rates lymph node upstaging [8].
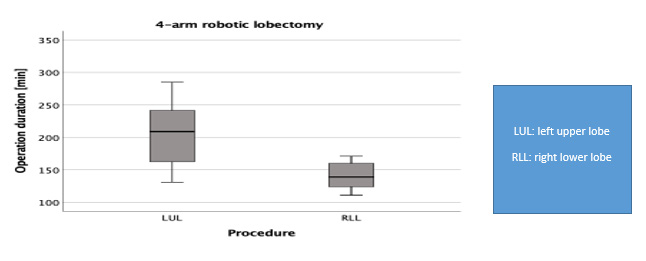
Conversely, some authors have found no differences in the number of resected lymph nodes between the two techniques for a lobectomy for lung cancer [9,10] (Figure 1). The operative techniques for the robotic-assisted lobectomy have not been standardized at yet. Robotic approaches for a lobectomy may differ in terms of port placement, as the number 3 vs. 4 and the localization of the ports is variable. Two common techniques have been reported using either a three-arm or a four-arm robotic technique. Dylewski, et al. reported on the three-port approach as known in the VATS technique for robotic anatomic resection [11]. Other groups, including Cerfolio, et al. and Melfi, et al. favored the four-port approach for anatomical resection, demonstrating good results [1]. Up to now a comparison of both techniques with regards to operative metrics and outcomes is difficult, as few surgeons are experienced in both technics, and one been typically favored over the other in most centers. The purpose of this study was to report on our clinical experience with both the three- and four-arm technique. In particular, we compared the operative times and lymph node yield between the two techniques, as these were anecdotally different (Figure 2).
Methods
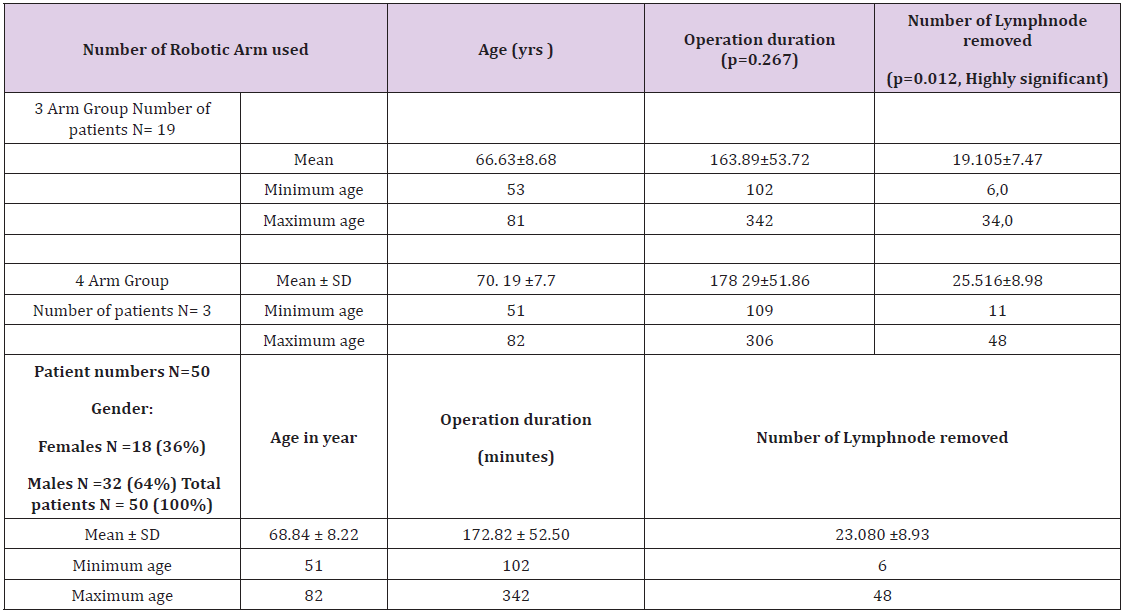
We retrospectively studied a cohort of 50 patients with nonsmall cell lung cancer (32 males and 18 females; mean age 68.84 years) who underwent a robotic lobectomy by one surgeon at a single institution because of lung cancer consecutive between November 2018 and February 2020.Given its retrospective nature, the local ethics committee of the University of Würzburg waived the requirement for additional approval. Informed written consent was obtained from all patients prior to robotic lobectomy. Well-informed consent was taken from all patients and the study was approved by the ethics committee of the University of Würzburg. Patients were routinely staged by positron emission tomography with 2-[18F] Fluoro-2-deoxyglucose (FDG-PET) and brain magnetic resonance imaging (MRI). Follow-up data for calculation of progression-free (PFS) and overall survival (OS) were available for XY± YZ months from the date of robotic lobectomy. Criteria for patients’ selection for robotic lobectomy were clinical stage 8th Edition UICC (Union International Contra le Cancer) I and II. Patients who underwent robotic lobectomy for a benign disease as well as patients with stage III disease who had received neo-adjuvant treatment were excluded.
Robotic Technique
The first series of nineteen patients were consecutively operated with the three-arm technique as previously described [3]. For the next 31 patients, lobectomy was performed by a four-arm technique as outlined by Cerfolio [12]. All procedures were performed with the DaVinci X robot (Intuitive Surgical, Sunnyvale, CA). All cases were conducted by a single surgeon who had surpassed the initial learning curve for robotic lobectomy and achieved proficiency in both the three- and four-arm techniques prior to this study.
Lobectomy Approach
We placed the ports in line in the eighth intercostal space. In addition, a 12 mm port was placed in the ninth intercostal space to remove the dissected lymph node. We used the robotic-controlled endo-wrist stapler to staple the vessels and bronchus in all of the patients. After docking the robots, the lymphadenectomy was performed first, by mobilizing the pulmonal ligament, whereby we removed lymph node station 9. Thereafter, we continued upward and removed the LN stations 8, 7, 4R and 2R on the right side and 5 and 6 on the left side. This was followed by dissection of the anterior hilum. By removing the LN stations 10 and 11, we usually have the vessels exposed such that they could be easily surrounded. After dissection and exposing the vessels, we surrounded the vessels and elevated them before dividing with the stapler. Based on the indication and the lobe to be removed, we then divided the vein, the artery and the bronchus. We usually used the DaVinci stapler, which can be inserted through the 12 mm trocar. The specimens were removed in a plastic bag through the assistant’s port. We injected 4mg (2ml) ropivacaine each in the third to tenth intercostal space for pain control and local anesthesia.
Perioperative Goals
Patients were routinely extubated after closing the chest in the operative room and transferred to the intermediate care unit (ICU) for 2 to 5 hours. When stable, a chest X-ray is performed, and the patient is sent to the surgical ward and start mobilization. On day one post-operatively, we intensified the mobilization of patients. The drains were removed when the daily fluid production was under 300 ml and no air leaks were observed.
Analgesia Management
On the day of operation, our patients all received metamizole intravenously at 200 mg/hour. If necessary, piritramide could be additionally administered. On post-operative day one, we gave tapentadol at 100 mg twice a day until the drain was removed. Subsequently, we reduced the tapentadol to 50 mg twice a day. If needed, patients could receive rescue medication (ibuprofen or acetaminophen on on an as needed basis).
Outcomes
Outcomes data were captured in a prospectively maintained institutional quality database. Operative time was defined as the time from skin incision to skin closure. The number of lymph nodes harvested was derived from surgical pathology reports. Postoperative events and 90-day mortality were uniformly captured.
Statistical Analysis
Continuous data are expressed as means with standard deviation, or medians and quartiles based on normal distribution. For categorical measures, absolute and relative frequencies are reported. The normality of continuous measures was checked via quantile-quantile plots. For normally distributed data, a t-test for independent groups was used to compare two samples. If the data was not normally distributed, the Mann-Whitney U-test was applied instead (with exact p-values when n < 10). For comparison of more than two groups, the non-parametric Kruskal-Walli’s test was employed, as group sizes were insufficient to prove normality. Significant Kruskal-Walli’s test results were followed up by pairwise post-hoc analyses with Bonferroni correction. For comparison of categorical data between two samples, the chi-square test for cross tabulation was applied when the cell size was sufficiently high; otherwise, we made use of Fisher’s exact test for 2x2 cross tables. As an effect size measure, Cohen’s d was reported, and the data is visually shown by boxplots. The software SPSS 25 and a 5% level of significance was used.
Results
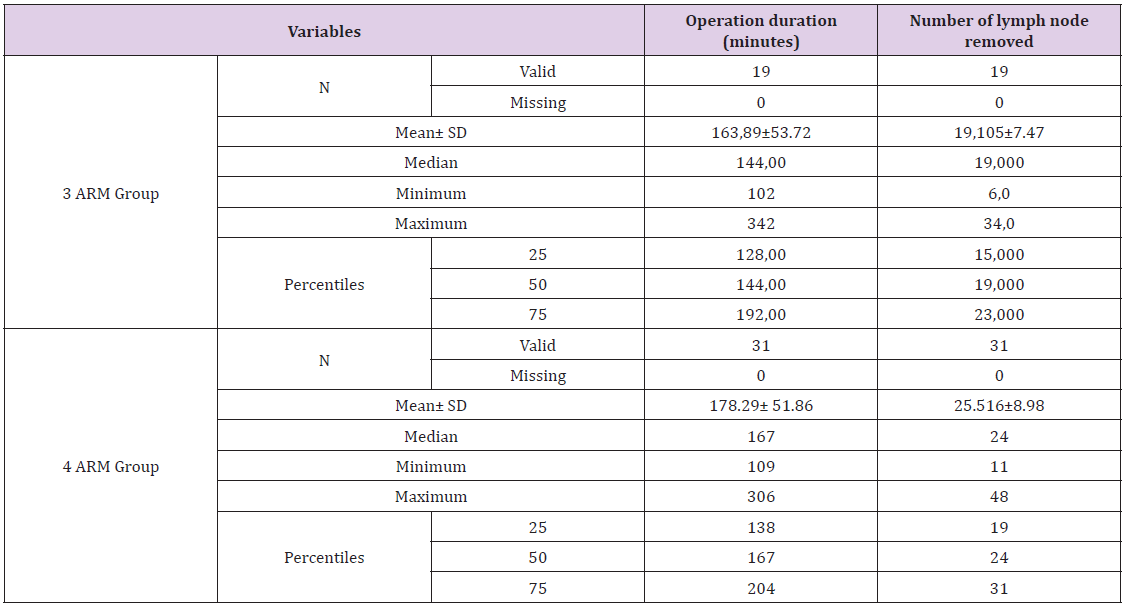
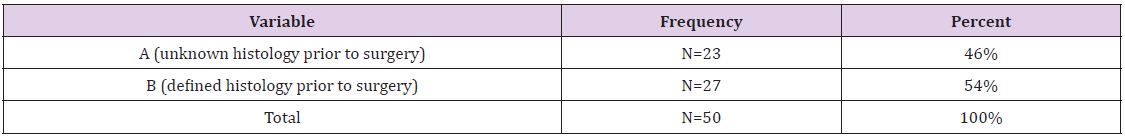
A total of 50 patients underwent lobectomy by the robotic three-arm (n=19) and four-arm (n=31) approach for non-small cell lung cancer. Patients’ demographic information’s are summarized in Table 1. There were 32 (62%) men, and the most common histological pattern was adenocarcinoma in 31 cases (62%). An intraoperative frozen examination was performed in 23 patients (46%) to define the histology prior to lobectomy. There was no difference in lung cancer stage between the three- and four-arm group (Tables 2-4).
Table 2: Number of Robotic Arms used, Age of patients, operation time and numbers of lymph nodes removed.
Table 4: Histology known prior to surgery by another modality viz. CT guided biopsy or bronchoscopic biopsy etc.
Note: A (unknown histology prior to surgery), B (defined histology prior to surgery).
Outcomes of Three- vs. Four Arm Lobectomy
The mean operation duration was 172 minutes and the mean number of lymph nodes removed was 23 (range 6-48). Two patients – one in the three-arm group and other in the four-arm group – were in clinical stage N0 but had pathological N2 disease. Comparing the two groups, the operation duration was slightly longer in the fourarm group (178.29 versus 164.29 minutes), although it was not statistically significant (p= 0. 26). However, we removed significantly more lymph nodes with the four-arm technique than with the three-arm technique (25.5 versus 19.1), which was significantly different (p=0.012). The subgroup analysis revealed that the fourarm technique required significantly more time for the left upper lobectomy than for the right lower lobectomy (p=0.040) when both were performed using the four-arm technique. In one patient (2%), we had to convert RATS into thoracotomy due to bleeding owing to injury to the apical trunk of the pulmonary artery during a left upper lobectomy. Few postoperative events were recorded. Cardiac fibrillation occurred in two (4%) of patients. There was no wound infection or chylothorax in any of the operated cases, and no mortality was observed within 90 days of surgery.
Discussion
In the present study, we report on our experience with three-arm and four-arm robotic-assisted lobectomy. Our data showed that the four-arm robotic technique was associated with significantly more lymph nodes harvest as compared with the three-arm technique. The operative time and perioperative outcomes were otherwise similar. This study is important, as it suggests that the evolution of the robotic technique can further improve outcomes for the individual surgeon. The improved lymph node yield is encouraging and advocates for further applications of robotic lobectomy with a four-arm technique in lung cancer patients. Lymphadenectomy was the focus of this study. Surgical lymph node staging is an integral part of the oncological operation which is crucial for accurate staging of patients and offers the best post-operative treatment. The number of dissected lymph nodes can be considered as a surrogate for surgical quality. The robotic platform can facilitate a radical lymph node dissection by several factors, including the improved visualization and the increased range of motion due to the wristed instruments. We used 30° optic instruments so that we could more easily view different parts of important structures and therefore enable an easier dissection of the intrathoracic structures.
Other authors mention the use of 0° optic instruments to avoid nerve damage in the intercostal space. To our knowledge and based on extensive literature search, there is no study comparing the usefulness of 0° or 30° optic instruments for lobectomy. There are publications about robotic lobectomy without using CO2 [13,14]. We use CO2 routinely to de-inflate the lung and push the diaphragm downwards to gain more space in the chest for intrathoracic maneuverability, in line with published experiences by Cerfolio and Dylewski [5,11]. Gharagozloo, et al. described a hybrid technique with three robotic arms positioned at the eighth, sixth and fifth intercostal spaces for the dissection of hilar structures [15,16]. In the current study, we selected patients in UICC Stage I and II without previous oncological treatment. This may lead to better results in term of number of lymph node removed. Since the tumor himself was not challenging, we focused more on the lymph node dissection. We dissected more lymph nodes with the four-arm technique. In our experience with the four-arm technique, the exposition is better and therefore a thorough lymphadenectomy could be possible. We begin all our procedures for lobectomy with a lymphadenectomy, as previously described [3], and subsequently removed lymph nodes with surrounding fat tissue. Cerfolio, et al. used the four-arm technique and could remove a median of thirteen lymph nodes for lobectomy [17], which is less than our yield. Egberts found advantages for using the four-arm technique during the exposition of structures and lymphadenectomy [18]. We achieved a upstaging rate of cN0 to pN2 in two cases (4%). This is lower in comparison with other authors [19,20], although this finding could be explained by the reduced number of patients in our case series.
The robotic approach and number of port placement may also affect the length of the operation, which is an important metric given the already high expense of the robotic platform and precious resource operating room time. Ninan et al. reported the efficiency of a completely portal robotic lobectomy with three arms in 74 patients [21]. We assumed that the use of a four-arm robotic system for lobectomy could further reduce the operation duration since the surgeon is more independent for retracting the lung and has a better exposition by him-/herself. Cerfolio, et al. previously demonstrated a reduction in the operation time by the number of trocars used from 3 to 4 [12], which is in contrast to our experience, as the duration of the robotic lobectomy with four arms was slightly longer than with the three-arm technique in our institution. The reason for this finding could be the additional time for changing the instruments, while another reason could be the collision of the four arms, which required repositioning of the robots’ arms. With the four-arm technique, the left upper lobectomy required significantly more time than the right lower lobectomy. This can be explained by the number of arterial branches supplying the left upper lobe, with five segments. The right lower lobe had one main artery branch that had to be divided. In some cases, the artery to segment number 6 had to be divided separately. Furthermore, in our experience the left upper lobectomy is more challenging since the arteries are short in length, making the insertion of the vascular stapler sometimes difficult.
Limitations and Conclusion
One of the limitations of the present study is that it was performed at a single center. In addition, it was a retrospective study with a limited number of cases. However, the single surgeon experience is a significant strength of this study due to the uniform patient selection, baseline operative experience level prior to this study, and conduct of the operation. It is therefore sensitive to detect differences in outcomes based on modifications of the operative technique even with a small number of patients, which enabled a comparison of the three-arm and the four-arm technique. Further investigations in prospective randomized control trials could bring lighter and confirm our observations and findings, which would benefit patients with more yield of the specimen and less operative time, with a consequent short hospital stay, less morbidity and reduced mortality.
Conflict of Interest
The author is proctor for Intuitive surgical.
References
- Melfi FM, Olivia Fanucchi, Federico Davini, Alfredo Mussi (2014) VATS-based approach for robotic lobectomy. Thorac Surg Clin 24(2): 143-149.
- Dylewski MR, RS Lazzaro (2012) Robotics The answer to the Achilles' heel of VATS pulmonary resection. Chin J Cancer Res 24(4): 259-260.
- Cheufou DH, Khaled Mardanzai, Till Ploenes, Dirk Theegarten, Georgios Stamatis, et al. (2019) Effectiveness of Robotic Lobectomy-Outcome and Learning Curve in a High-Volume Center. Thorac Cardiovasc Surg 67(7): 573-577.
- Li C, Jiantao Li, Long Jiang, Hao Lin, Peiji Lu, et al. (2019) Comparison of robotic-assisted lobectomy with video-assisted thoracic surgery for stage IIB-IIIA non-small cell lung cancer. Transl Lung Cancer Res 8(6): 820-828.
- Cerfolio RJ (2014) Total port approach for robotic lobectomy. Thorac Surg Clin 24(2): 151-156.
- Melfi FM, Marcello C Ambrogi, Marco Lucchi, Alfredo Mussi (2005) Video robotic lobectomy. Multimed Man Cardiothorac Surg 2005(628): p. mmcts 2004 000448.
- Whitson BA, Shawn S Groth, Susan J Duval, Scott J Swanson, Michael A Maddaus, et al. (2008) Surgery for early-stage non-small cell lung cancer: a systematic review of the video-assisted thoracoscopic surgery versus thoracotomy approaches to lobectomy. Ann Thorac Surg 86(6): 2008-2016.
- Kneuertz PJ, Mahmoud Abdel Rasoul, Dirk Theegarten, Susan D Moffatt Bruce, Clemens Aigner, et al. (2019) Propensity-score adjusted comparison of pathologic nodal upstaging by robotic, video-assisted thoracoscopic, and open lobectomy for non-small cell lung cancer. J Thorac Cardiovasc Surg 158(5): 1457-1466 e2.
- Jang HJ, Hyun Sung Lee, Seong Yong Park, Jae Ill Zo (2011) Comparison of the early robot-assisted lobectomy experience to video-assisted thoracic surgery lobectomy for lung cancer: a single-institution case series matching study. Innovations (Phila) 6(5): 305-10.
- Louie BE, Alexander S Farivar, Ralph W Aye, Eric Vallières (2012) Early experience with robotic lung resection results in similar operative outcomes and morbidity when compared with matched video-assisted thoracoscopic surgery cases. Ann Thorac Surg 93(5): 1598-604.
- Dylewski MR, AC Ohaeto, Pereira JF (2011) Pulmonary resection using a total endoscopic robotic video-assisted approach. Semin Thorac Cardiovasc Surg 23(1): 36-42.
- Cerfolio RJ, Ayesha S Bryant, Loki Skylizard, Douglas James Minnich (2011) Initial consecutive experience of completely portal robotic pulmonary resection with 4 arms. J Thorac Cardiovasc Surg 142(4): 740-746.
- Veronesi G, Bernardo Rocco, Alessandro Borri, Roberto Gasparri, Lorenzo Spaggiari, et al. (2011) Experience with robotic lobectomy for lung cancer. Innovations (Phila) 6: 355-360.
- Pardolesi A, Luca Bertolaccini, Jury Brandolini, Piergiorgio Solli (2017) Four arm robotic-assisted pulmonary resection-right upper lobectomy: how to do it. J Thorac Dis 9(9): 3302-3306.
- Gharagozloo F, Marc Margolis, Barbara Tempesta, Eric Strother, Farzad Najam (2009) Robot-assisted lobectomy for early-stage lung cancer: report of 100 consecutive cases. Ann Thorac Surg 88(2): 380-384.
- Meyer M, Farid Gharagozloo, Barbara Tempesta, Marc Margolis, Eric Strother, et al. (2012) The learning curve of robotic lobectomy. Int J Med Robot 8(4): 448-452.
- Cerfolio RJ, Asem F Ghanim, Mark Dylewski, Giulia Veronesi, Lorenzo Spaggiari, et al. (2018) The long-term survival of robotic lobectomy for non-small cell lung cancer: A multi-institutional study. J Thorac Cardiovasc Surg 155(2): 778-786.
- Egberts JH, Domenico Galetta, Lorenzo Spaggiari (2015) Robot-assisted minimally invasive lobectomy with systematic lymphadenectomy for lung cancer. Zentralbl Chir 140(1): 15-16.
- Darr C, Gerhard Weinreich, Thomas Hachenberg, Clemens Aigner, Sandra Kampe, et al. (2017) Robotic thoracic surgery results in shorter hospital stay and lower postoperative pain compared to open thoracotomy: a matched pairs analysis. Surg Endosc 31(10): 4126-4130.
- Cheufou DH, Dirk Theegarten, Georgios Stamatis, Sandra Kampe, Clemens Aigner, et al. (2018) Effectiveness of Robotic Lobectomy-Outcome and Learning Curve in a High Volume Center. Thorac Cardiovasc Surg 67(7): 573-577.
- Ninan M, MR Dylewski (2010) Total port-access robot-assisted pulmonary lobectomy without utility thoracotomy. Eur J Cardiothorac Surg 38(2): 231-232.

 Research Article
Research Article