ABSTRACT
Atrial fibrillation is a common supraventricular arrhythmia which affects approximately 1% to 4% of the global population. Atrial fibrillation is more common in older individuals; the prevalence of AF increases sharply in those over 80 years old. Atrial fibrillation is the most common cardiac arrythmia and is associated with costly cardiovascular morbidity such as stroke and heart failure. Inflammation has been implicated in the pathophysiology of various cardiac, as well as non-cardiac, diseases that are comorbid with atrial fibrillation. Heart failure and cardiomyopathy are associated with a 4–6-fold increase in atrial fibrillation prevalence. Inflammation has been implicated in the pathophysiology of heart failure and modulates the cell signaling activation patterns associated with fibrosis, apoptosis and hypertrophy; these are all forms of cardiac remodeling that, when they occur at the atrial level, predispose to atrial fibrillation. Rhythm control strategy should be preferred in symptomatic patients due to paroxysmal pattern of atrial fibrillation and the presence of a hyperdynamic circulation. β-blockers is effective in reducing symptoms when rhythm control is not possible Indicated also for HF; Calcium channel blockers (verapamil, diltiazem): Effective in reducing symptoms when rhythm control is not possible and Digoxin: Second line therapy when β-blockers or calcium channel blockers are not tolerated.
Keywords: Atrial Fibrillation; Pathophysiology; Management
Abbreviations: ADAMTS13: A Disintegrin and Metalloprotease with ThromboSpondin Motif Repeats 13; ADP: Action Potential Duration; AF: Atrial Fibrillation; CAD: Coronary Artery Disease; DADs: Delayed After Depolarization’s; IL: Interleukin; LA: Left Atrial; LVH: Left Ventricular Hypertrophy; MR: Mineralocorticoid Receptor; NETs: Neutrophil Extracellular Traps; NF-KB: Nuclear Factor-kB; iNOS: Inducible Nitric Oxide Synthase; POAF: Postoperative Atrial Fibrillation; ROS: Reactive Oxygen Species; RYR2: Ryanodine Receptor; SGLT2-Inhibitors: Sodium-Glucose Co-Transporter 2 (SGLT-2) Inhibitors; TGFβ1: Transforming Growth Factor β1; TNFa: Tumour Necrosis Factor a; vWF: von Willebrand Factor
Introduction
Atrial fibrillation is characterized by irregular and often very fast contractions of the atrial cardiomyocytes, resulting in an irregular heart rate, palpitations, dizziness, shortness of breath, and tiredness in the patient. AF can occur when abnormal electrical impulses suddenly start firing in the atria and override the heart’s natural pacemaker, which can no longer control the rhythm of the heart. Importantly, AF is associated with severe complications, such as thromboembolic events, heart failure, cognitive impairment, and increased mortality [1,2]. The progressive stages of AF are associated with structural changes that promote contractile dysfunction and the impairment of electrical conduction in the atrial myocardium [3]. Atrial Fibrillation (AF) is the most common sustained cardiac arrhythmia and is associated with considerable morbidity and mortality [4]. Biomarkers of inflammation (C-reactive protein and fibrinogen), myocardial injury (high sensitivity troponin I), renal dysfunction (fibroblast growth factor-23), and hemodynamic stress (NT-proBNP [N-terminal pro-B-type natriuretic peptide]) have been reported to be independently associated with prevalent and incident AF [5]. The prevalence of AF ranges from 2% in the general population to 10–12% in those aged 80 and older. Patients with AF are five times more likely to have a stroke and three times more likely to experience heart failure compared to healthy individuals. AF may also increase an individual’s socioeconomic burden through increased healthcare costs [6-8].
Etiological Contributors
A variety of etiological factors contribute to AF occurrence. In most patients, AF results from interactions among multiple factors operating simultaneously. Over 70% of AF cases have associated heart disease. Aging is a major risk factor, largely via structural remodeling. Congestive Heart Failure (CHF), hypertension, valvular heart disease, and Coronary Artery Disease (CAD) are common contributors. Less common predisposing conditions include pericarditis or myocarditis, atrial myxomas, and hypertrophic cardiomyopathy. Extracardiac conditions also promote AF occurrence. Sufficiently powered studies suggest that heavy alcohol consumption promotes AF. Hyperthyroidism is a wellrecognized contributor, and the roles of sleep apnea and obesity are increasingly recognized [9,10]. Modifiable risk factors associated with atrial fibrillation are hypertension; diabetes mellitus; coronary artery disease; heart failure; valvular heart disease; chronic kidney disease; obesity; obstructive sleep apnoea; hyperthyroidism; smoking; chronic obstructive pulmonary disease; excessive alcohol intake and excessive exercise [11].
Pathophysiology
Immunothrombosis refers to the complex participation of the innate immune system in the formation of intravascular thrombus through distinct cellular and molecular interaction. This local coagulation can promote more inflammatory processes, initiating atrial remodeling through direct and indirect tissue damage. Fibrinogen, Von Willebrand Factor (vWF), and A Disintegrin and Metalloprotease with ThromboSpondin motif repeats 13 (ADAMTS13), a vWF-cleaving protease, are biomarkers that play key roles in coagulation and inflammatory pathways, and perhaps therefore be associated with AF. However, prospective research on this is scarce. Activation of the innate immunity can cause neutrophils to release neutrophil extracellular traps (NETs). Besides their important role in actively killing pathogens by releasing chromatin and DNA, NETs also stimulate coagulation processes by recruiting and activating platelets, binding to tissue factor, and stimulating fibrinogen and vWF. This way, NETs are at the intersection between inflammation and thrombosis, both potentially major players in AF pathophysiology [12,13]. AF is initiated by focal ectopic firing and is maintained by re-entry mechanisms in a vulnerable atrial substrate. The ectopic firing seems to arise from myocyte sleeves within the pulmonary veins and is triggered by a diastolic leak of Ca2+ from the sarcoplasmic reticulum that in turn determines myocyte depolarization due to an inward Na+ current via Na+ -Ca2+ exchanger. The re-entry mechanism is promoted by slow conduction velocity of the depolarizing wave front and a shortened refractory period of cardiac myocytes. The presence of structural and electrophysiological atrial abnormalities favors the selfperpetuation of AF by promoting re-entry [14].
Focal Ectopic Activity
Delayed after depolarization’s (DADs) constitute the most important mechanism of focal atrial arrhythmias. They result from abnormal diastolic leak of Ca2+from the main cardiomyocyte Ca2+ storage organelle, the sarcoplasmic reticulum (SR Ca2+ enters cardiomyocytes through voltage dependent Ca2+ channels during the action potential plateau, triggering Ca2+ release from the SR via Ca2+ release channels known as ryanodine receptors (RyRs; RyR2 is the cardiac form). This systolic Ca2+ release is responsible for cardiac contraction. Following action potential repolarization, diastolic cardiac relaxation occurs when Ca2+ is removed from the cytosol back into the SR by a Ca2+ uptake pump, the SR Ca2+ ATPase (SERCA). DADs result from abnormal diastolic Ca2+ leak through RyR2 from the SR to the cytoplasm. Excess diastolic Ca2+ is handled by the cell membrane Na+, Ca2+-exchanger (NCX), which transports three Na+ ions into the cell per single Ca2+ ion extruded, creating a net depolarizing current (called transient inward current, or Iti) that produces DADs. DADs that are large enough to reach threshold cause ectopic firing. Repetitive DADs cause focal atrial tachycardias (tachycardia is a heart rhythm >100 bpm). RyR2s are Ca2+ sensitive, and RyR2 leak results from SR Ca2+ overload or intrinsic RyR2 dysfunction. RyR2 function is modulated by channel phosphorylation: hyperphosphorylation makes RyRs leaky and arrhythmogenic [15-17].
Reentry
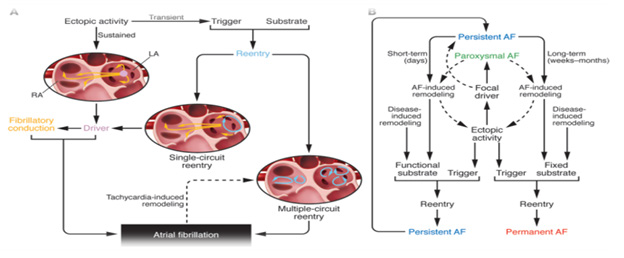
Reentry requires appropriate tissue properties, a vulnerable “substrate.” Reentry substrates can be caused by altered electrical properties or by fixed structural changes. Cardiac tissue exhibits a discrete refractory period (inexcitable interval following the last firing, governed by APD). Reentry initiation usually requires a premature ectopic beat that acts as a trigger. The resulting impulse conducts through the pathway leading to recording point ii, which is no longer refractory, but blocks in the pathway leading to recording point iii because of its longer refractory period. The premature impulse arrives at the distal end of previously refractory site iii and attempts to reenter [18] (Figure 1).
Figure 1: Tissue mechanisms leading to AF and clinical forms.
(A) Ectopic activity can act as a driver maintaining AF or as a trigger on a vulnerable substrate resulting in reentry (singleor multiple-circuit). Local driver mechanisms (ectopic or single-circuit reentrant) produce irregular fibrillatory activity via fibrillatory conduction. Rapid atrial activity (tachycardia) causes atrial remodeling, promoting multiple-circuit reentry.
(B) Clinical AF can manifest as paroxysmal AF (self-terminating), persistent AF (requires drug therapy or electrical cardioversion to terminate), and permanent AF (non-terminating). Focal ectopic drivers are principally associated with paroxysmal forms, functional reentrant substrates with persistent AF, and increasingly fixed substrates with permanent forms [51].
Aldosterone
Aldosterone itself has been shown to up regulate MR expression in cultured HL-1 cardiomyocytes, thus reinforcing its effects on the heart. Since the MR not only binds aldosterone, but also cortisol, it can be argued that some effects mediated by MR activation may be attributable to cortisol. 11â-hydroxysteroid dehydrogenase type 2, an enzyme which converts cortisol to inactive metabolites allowing aldosterone binding to the MR, is up regulated in the left atrial myocardium of patients with AF, thereby suggesting that MR activation in this setting is mainly due to aldosterone [19,20]. As regards to the potential physiopathological mechanism linking aldosterone excess to FA, aldosterone is thought to be involved in the genesis and perpetuation of AF not only by causing AH and electrolyte imbalance, but also by inducing inflammation, oxidative stress, fibrosis and electrophysiological changes; all these mechanisms contribute to structural and electrical atrial remodeling that are known to predispose to AF [21] (Figure 2). Oxidative Stress Over recent years, oxidative stress has been investigated as a potential essential mechanism in the development of AF. Reactive Oxygen Species (ROS) constitute the normal byproducts generated through the metabolism of oxygen. These molecules have been proven to have a multifaceted effect on the cells present in the heart tissue. The prevalence and incidence of AF were related to the redox potentials of glutathione (EhGSH) and cysteine, markers of oxidative stress. The study concluded that the prevalence of AF was 30% higher for each 10% increase in EhGSH, while the same alteration resulted in a 40% increase in the risk of incident AF [22]. Iron accumulation causes AF is increased oxidative stress [23].
Inflammation
Inflammation has been linked to the onset and maintenance of atrial fibrillation, according to accumulating evidence. Inflammation contributes to the atrial remodelling involving both structural and electrophysiological alterations that form the basis for the disease. A large-scale prospective study involving 24,734 women participants investigated the association of inflammatory markers such as CRP, fibrinogen, and intercellular adhesion molecule 1 (sICAM-1) with the incidence of AF. The results suggested that inflammation is a strong indicator for the incidence of AF with the median plasma levels of the biomarkers being independently correlated with the development of the disease in patients [24]. Inflammation has been implicated in the pathophysiology of various cardiac, as well as non-cardiac, diseases that are comorbid with AF. HF and cardiomyopathy are associated with a 4–6-fold increase in AF prevalence. Inflammation has been implicated in the pathophysiology of HF and modulates the cell signaling activation patterns associated with fibrosis, apoptosis and hypertrophy; these are all forms of cardiac remodeling that, when they occur at the atrial level, predispose to AF. CAD is a systemic, lipid-driven condition with immune inflammatory components. AF frequently occurs in acute myocardial infarction, especially in the early stages when the myocardial inflammatory reaction is at its maximal. Serum levels of interleukin-6 (IL-6), an inflammatory biomarker, were independently associated with AF risk in a cohort of subjects with known CAD [25,26].
Tumour Necrosis Factor A
TNF-α stimulates an acute immune cell reaction and induces inflammation. TNF-α is synthesized by various immune cells, including macrophages and lymphocytes [27].
C-Reactive Protein
C-Reactive Protein (CRP) is a highly reproducible but nonspecific inflammatory biomarker, synthesized primarily in the liver in response to inflammatory cytokines. The circulating level of CRP is increased in patients with AF compared with those without an AF history, and persistent-AF patients have higher CRP levels than do those with paroxysmal-AF [28].
Interleukin-6
IL-6 is produced by lymphocytes and stimulates inflammatory responses. IL-6 also has anti-inflammatory effects, via the inhibition of TNF-α signaling and the activation of IL-10, an anti-inflammatory cytokine [29].
Treatment
Lifestyle and dietary modifications including weight loss, alcohol reduction, and cardiometabolic risk factor management would be a cornerstone for AF prevention. In patients with short paroxysms of AF, therapeutic strategies should generally concentrate on providing control of the arrhythmia itself. Regardless of the arrhythmia pattern or the therapeutic strategy chosen, and in the absence of contraindications, patients should be considered for anticoagulation if they have one or more risk factors for thromboembolism. Patients at low or intermediate risk, and higher risk patients in whom warfarin is contraindicated, may benefit from antiplatelet treatment [30,31]. The medical prescription of medications other than anti-inflammatory agents, such as angiotensin-converting enzyme inhibitors, angiotensin receptor blockers, and aldosterone antagonists, can all help to reduce LA enlargement, atrial fibrosis, and TGF-β indicators, as well as atrial dysfunction. These are the most widely used drugs for AF and have to be considered for patients with a history of heart failure. The novel SGLT-2 inhibitors reveal beneficial effects in systolic heart failure included improved cardiac energy metabolism, the prevention of inflammation, oxidative stress, adverse cardiac remodelling, less LA enlargement, fibrosis, atrial mitochondrial dysfunction, inflammation, and AF inducibility [32-34].
Rhythm Control Strategy
Chelation Therapy: Effective in preventing both arrhythmic recurrences and iron overload;
Deferoxamine: this was the first iron chelator introduced in clinical practice. It has a short plasma life and is not absorbed in the gastrointestinal tract, so it must be administered parenterally. Deferoxamine can also be administered in continuous intravenous infusion when intensive chelation is needed [35].
Amiodarone: Effective safe in the short term; Amiodarone is often both effective in the control of arrhythmic recurrences and safe in the short term. However, its multiple adverse effects when taken chronically, associated with the frequent coexistence of organ damage from iron accumulation, suggest avoiding prolonged use. Long-term therapy, in fact, is often complicated by thyroid and hepatic dysfunction, as these organs are also targets of iron-mediated damage. Other antiarrhythmic drugs (flecainide, propafenone, sotalol).
Fewer Side Effects in the Long Term and Catheter Ablation: Avoiding side effects of antiarrhythmic drugs [36,37]. Catheter ablation has been shown to be a safe and effective option for rhythm control in patients with AF. Current international guidelines recommend ablation for patients with symptomatic AF, both paroxysmal and persistent, who have failed an antiarrhythmic drug therapy (class I of evidence). However, they also add that ablation may be used in selected patients even before a trial of antiarrhythmic drugs (class IIa) [38,39]. Flecainide and propafenone have been shown to be similarly effective at suppressing symptomatic paroxysms of AF and, in the absence of structural heart disease, neither drug appears to cause significant proarrhythmia. In general, these class Ic agents tend to be better tolerated and more effective than class Ia agents, such as quinidine and disopyramide [40].
Rate Control Strategy
Rhythm control strategy should be preferred in symptomatic patients due to paroxysmal pattern of AF and the presence of a hyperdynamic circulation [41]. β-blockers is effective in reducing symptoms when rhythm control is not possible Indicated also for HF; Calcium channel blockers (verapamil, diltiazem): Effective in reducing symptoms when rhythm control is not possible and Digoxin: Second line therapy when β-blockers or calcium channel blockers are not tolerated [42].
Anticoagulation
Warfarin: Frequent monitoring of coagulation state (INR) and DOACs (apixaban, dabigatran, edoxaban, rivaroxaban): More manageable and safer than warfarin [43].
Anti-Inflammatory Drugs
Statins: Statins have pleiotropic actions beyond cholesterol reduction, including improved endothelial function, reduced thrombogenesis, and suppression of oxidative stress and inflammation. In animal AF models, statins decreased AF vulnerability [44].
Colchicine: Colchicine suppresses leukocyte activation, endothelial cell adhesion and migration. Postoperatively initiated adjusted dose colchicine prevented POAF and shortened hospital stay without significant adverse effects in a double-blind randomized trial [45-47].
Corticosteroids: In an experimental study, tachycardiainduced atrial remodeling and AF vulnerability were attenuated by prednisone treatment; circulating CRP levels were also decrease [48-51].
Conclusion
AF is characterized by irregular and often very fast contractions of the atrial cardiomyocytes, resulting in an irregular heart rate, palpitations, dizziness, shortness of breath, and tiredness in the patient. Immunothrombosis refers to the complex participation of the innate immune system in the formation of intravascular thrombus through distinct cellular and molecular interaction. Inflammation contributes to the atrial remodelling involving both structural and electrophysiological alterations that form the basis for the disease. The medical prescription of medications other than anti-inflammatory agents, such as angiotensin-converting enzyme inhibitors, angiotensin receptor blockers, and aldosterone antagonists, can all help to reduce LA enlargement, atrial fibrosis, and TGF-β indicators, as well as atrial dysfunction.
Acknowledgment
The author would be grateful to anonymous reviewers by the comments that increase the quality of this manuscript.
Data Sources
Sources searched include Google Scholar, Research Gate, PubMed, NCBI, NDSS, PMID, PMCID, Scopus database, Scielo and Cochrane database. Search terms included: pathophysiology and management of arterial fibrillation.
Funding
None.
Availability of Data and Materials
The datasets generated during the current study are available with correspondent author.
Competing Interests
The author has no financial or proprietary interest in any of material discussed in this article.
References
- Sánchez FJ, Pueyo, E, Diez ER (2022) Strain Echocardiography to Predict Postoperative Atrial Fibrillation. Int J Mol Sci 23(3): 1355.
- Sagris M, Vardas EP Theofilis P, Antonopoulos AS, Oikonomou, et al. (2022) Atrial Fibrillation Pathogenesis Predisposing Factors and Genetics. Int J Mol Sci 23(1): 6.
- Krisai P Takamitsu Takagi, Tsukasa Kamakura, Elodie Surget, Clémentine, André, et al. (2022) Sex differences in ventricular arrhythmia epidemiology pathophysiology and catheter ablation. Rev Cardiovasc Med 23(1): 14.
- Tilly MJ, Sven Geurts , Samantha J Donkel , M Arfan Ikram , Natasja MS de Groot, et al. (2022) Immunothrombosis and new onset atrial fbrillation in the general population: the Rotterdam Study. Clinical Research in Cardiology 111(1): 96-104.
- Van Wijk SW, Su W, Wijdeveld LFJM, Ramos KS (2022) Brundel BJJM Cytoskeletal Protein Variants Driving Atrial Fibrillation: Potential Mechanisms of Action. Cells 11(3): 416.
- Meyre PB, Stefanie Aeschbacher, Steffen Blum, Gian Voellmin, Peter M Kastner, et al. (2022) Biomarkers associated with rhythm status after cardioversion in patients with atrial fibrillation. Scientifc Reports 12: 1680.
- Malagù M, Marchini F, Fiorio A, Sirugo P, Clò S, et al. (2022) Atrial Fibrillation in β-Thalassemia: Overview of Mechanism, Significance and Clinical Management. Biology 11(1): 148.
- Akintoye E, Sellke F, Marchioli R, Tavazzi L, Mozaffarian D (2018) Factors associated with postoperative atrial fibrillation and other adverse events after cardiac surgery. J Thorac Cardiovasc Surg 155(1): 242-251.
- Laredo M, Waldmann V, Khairy P, Nattel S (2018) Age as a Critical Determinant of Atrial Fibrillation A Two-sided Relationship. Can J Cardiol 34(11): 1396-1406.
- Liberale L, Montecucco F, Tardif JC, Libby P, Camici GG (2020) Inflamm ageing the role of inflammation in age dependent cardiovascular disease. Eur Heart J 41(31): 2974-2982.
- Stojanovic SD, Fiedler J, Bauersachs J, Thum T, Shedding DG (2020) Senescence induced inflammation: An important player and key therapeutic target in atherosclerosis. Eur Heart J 41(31): 2983-2996.
- Sanchez FJ, Gonzalez VA, Farrando M, Baigorria Jayat AO, Segovia Roldan M, et al. (2020) Atrial Dyssynchrony Measured by Strain Echocardiography as a Marker of Proarrhythmic Remodeling and Oxidative Stress in Cardiac Surgery Patients. Oxid Med Cell. Longev 2020: 8895078.
- Corradi D, Saffitz, JE, Novelli D, Asimaki A, Simon C, et al. (2020) Prospective Evaluation of Clinico-pathological Predictors of Post-operative Atrial Fibrillation an Ancillary Study from the OPERA Trial. Circ Arrhythmia Electrophysiol 13(8): 008382.
- Kawczynski MJ, Gilbers M, Van De Walle S, Schalla S, Crijns HJ, et al. (2021) Role of pre-operative transthoracic echocardiography in predicting post-operative atrial fibrillation after cardiac surgery A systematic review of the literature and meta-analysis. EP Eur 23(11): 1731-1743.
- Mandoli GE, Pastore MC, Benfari G, Bisleri G, Maccherini M, et al. (2020) Left atrial strain as a pre-operative prognostic marker for patients with severe mitral regurgitation. Int J Cardiol 324: 134-145.
- Diavati S, Sagris M, Terentes Printzios D, Vlachopoulos C (2021) Anticoagulation Treatment in Venous Thromboembolism: Options and Optimal Duration. Curr Pharm Des 28(4): 296-305.
- Siasos G, Skotsimara G, Oikonomou E, Sagris M, Vasiliki Chara M, et al. (2020) Antithrombotic Treatment in Diabetes Mellitus: A Review of the Literature about Antiplatelet and Anticoagulation Strategies Used for Diabetic Patients in Primary and Secondary Prevention. Curr Pharm Des 26(23): 2780-2788.
- Theofilis P, Sagris M, Antonopoulos AS, Oikonomou E, Tsioufis C, et al. (2021) Inflammatory Mediators of Platelet Activation Focus on Atherosclerosis and COVID-19. Int J Mol Sci 22(20): 1170.
- Liang F, Wang Y (2021) coronary heart disease and atrial fibrillation: A vicious cycle. Am J Physiol Heart Circ Physiol 320: 1-12.
- Sagris M, Theofilis P, Antonopoulos AS, Tsioufis C, Oikonomou E, et al. (2021) Inflammatory Mechanisms in COVID-19 and Atherosclerosis: Current Pharmaceutical Perspectives. Int J Mol Sci 22(12): 6607.
- Sagris M, Kokkinidis DG, Lempesis IG, Giannopoulos S, Rallidis, L, et al. (2020) Nutrition dietary habits and weight management to prevent and treat patients with peripheral artery disease. Rev Cardiovasc Med 21(4): 565-575.
- Levin MG, Judy R, Gill D, Vujkovic M, Verma SS, et al. (2020) Genetics of height and risk of atrial fibrillation: A Mendelian randomization study. PLoS Med 17(10): 1003288.
- Bonora, BM Raschi E, Avogaro A, Fadini GP (2021) SGLT 2 inhibitors and atrial fibrillation in the Food and Drug Administration adverse event reporting system. Cardiovasc Diabetol 20: 39.
- Dobrev D, Potpara TS (2020) Smart device-based detection of atrial fibrillation: Opportunities and challenges in the emerging world of digital health. Int J Cardiol 30: 108-109.
- Seshadri DR, Bittel B, Browsky D, Houghtaling P, Drummond CK Desai, et al. (2020) Accuracy of Apple Watch for Detection of Atrial Fibrillation. Circulation 141(8): 702-703.
- Roth GA, Mensah GA, Johnson CO, Addolorato G, Ammirati E, et al. (2020) Global burden of cardiovascular diseases and risk factors, 1990–2019: update from the GBD 2019 Study. Journal of the American College of Cardiology 76(25): 2982- 3021.
- Virani SS, Alonso A, Aparicio HJ, Benjamin EJ, Bittencourt MS, et al. (2021) heart disease and stroke statistics-2021 update: a report from the American Heart Association. Circulation 143(8): 254-743.
- Reynolds HR, Shaw LJ, Min JK, Spertus JA, Chaitman BR, et al. (2020) Association of sex with severity of coronary artery disease ischemia and symptom burden in patients with moderate or severe ischemia secondary analysis of the ISCHEMIA randomized clinical trial. JAMA Cardiology 5(7): 773-786.
- Li M, Parker BL, Pearson E, Hunter B, Cao J, et al. (2020) Core functional nodes and sex-specific pathways in human ischaemic and dilated cardiomyopathy. Nature Communications 11(1): 2843.
- Coppini R, Santini L, Olivotto I, Ackerman MJ, Cerbai E (2020) Abnormalities in sodium current and calcium homoeostasis as drivers of arrhythmogenesis in hypertrophic cardiomyopathy. Cardiovasc Res 116(9): 1585-1599.
- Pool L, Wijdeveld L, de Groot N, Brundel B (2021) The Role of Mitochondrial Dysfunction in Atrial Fibrillation Translation to Druggable Target and Biomarker Discovery. Int J Mol Sci 22(16): 8463.
- Jia Z, Zhang Y, Deng J, Guo Y, Du Y, et al. (2021) Novel LMNA indel mutation identified in a family with atrioventricular block and atrial fibrillation. Medicine 100(19): 25910.
- Gerbino A, Forleo C, Milano S, Piccapane F, Procino G, et al. (2021) Pro-inflammatory cytokines as emerging molecular determinants in cardiolaminopathies. J Cell Mol Med 25(23): 10902-10915.
- Ziki MDA, Bhat N, Neogi A, Driscoll TP, Ugwu N, et al. (2021) Epistatic interaction of PDE4DIP and DES mutations in familial atrial fibrillation with slow conduction. Hum Mutat 42(10): 1279-1293.
- Chalazan B, Mol D, Darbar FA, Ornelas Loredo A, Al Azzam B, et al. (2021) Association of Rare Genetic Variants and Early-Onset Atrial Fibrillation in Ethnic Minority Individuals. JAMA Cardiol 6(7): 811-819.
- Mavroidis M, Athanasiadis NC, Rigas PS, Kostavasili I, Kloukina I, et al. (2020) Desmin is essential for the structure and function of the sinoatrial node: Implications for increased arrhythmogenesis. Am J Physiol Circ Physiol 319(3): 557-570.
- Smolina N, Khudiakov A, Knyazeva A, Zlotina A, Sukhareva K, et al. (2020) Desmin mutations result in mitochondrial dysfunction regardless of their aggregation properties. Biochim. Biophys Acta BBA Mol Basis Dis 1866(6): 165745.
- Heffler J, Shah PP, Robison P, Phyo S, Veliz K, et al. (2020) Balance Between Intermediate Filaments and Microtubules Maintains Nuclear Architecture in the Cardiomyocyte. Circ Res 126(3).
- Rodríguez Sinovas A, Sánchez J, Valls Lacalle L, Consegal M, Ferreira González I (2021) Connexins in the Heart Regulation Function and Involvement in Cardiac Disease. Int J Mol Sci 22(9): 4413.
- Herrmann H, Cabet E, Chevalier NR, Moosmann J, Schultheis D, et al. (2020) Dual Functional States of R406W-Desmin Assembly Complexes Cause Cardiomyopathy with Severe Intercalated Disc Derangement in Humans and in Knock-In Mice. Circulation 142(22): 2155-2171.
- Song S, Shi A, Lian H, Hu S, Nie Y (2021) Filamin C in cardiomyopathy From physiological roles to DNA variants. Hear Fail Rev, p. 1-13.
- Hindricks G, Potpara T, Dagres N, Arbelo E, Bax JJ, et al. (2020) ESC Guidelines for the diagnosis and management of atrial fibrillation developed in collaboration with the European Association for Cardio-Thoracic Surgery (EACTS) The Task Force for the diagnosis and management of atrial fibrillation of the European Society of Cardiology (ESC) Developed with the special contribution of the European Heart Rhythm Association (EHRA) of the ESC. Eur Heart J 42(5): 373-498.
- Bollati M, Lopez C, Bioletto F, Ponzetto F, Ghigo E, et al. (2022) Atrial Fibrillation and Aortic Ectasia as Complications of Primary Aldosteronism: Focus on Pathophysiological Aspects. Int J Mol Sci 23(11): 2111.
- Parasiliti Caprino M, Lopez C, Prencipe N, Lucatello B, Settanni F, et al. (2020) Prevalence of primary aldosteronism and association with cardiovascular complications in patients with resistant and refractory hypertension. J Hypertens 38(9): 1841-1848.
- Boczar KE, Boodhwani M, Beauchesne L, Dennie C, Chan KL (2020) Aortic Stiffness Central Blood Pressure and Pulsatile Arterial Load Predict Future Thoracic Aortic Aneurysm Expansion. Hypertension 77(1): 126-134.
- Pandey AK, Okaj I, Kaur H, Belley Cote EP, Wang J, et al. (2021) Sodium-Glucose Co-Transporter Inhibitors and Atrial Fibrillation A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Journal of the American Heart Association 10(17): 02222.
- Virani SS, Alonso A, Benjamin EJ, Bittencourt MS, Callaway CW, et al. (2020) heart disease and Stroke Statistics-2020 Update: A Report from the American Heart Association. Circulation 141(9): 139-596.
- Packer M, Anker SD, Butler J, Filippatos G, Pocock SJ, et al. (2020) Cardiovascular and Renal Outcomes with Empagliflozin in Heart Failure. New England Journal of Medicine 383(15): 1413-1424.
- Bhatt DL, Szarek M, Steg PG, Cannon CP, Leiter LA, et al. (2021) Sotagliflozin in Patients with Diabetes and Recent Worsening Heart Failure. New England Journal of Medicine 384(2): 117-128.
- Seferović PM, Fragasso G, Petrie M, Mullens W, Ferrari R, et al. (2020) Heart Failure Association of the European Society of Cardiology update on sodium–glucose co-transporter 2 inhibitors in heart failure. European Journal of Heart Failure 22(11): 1984-1986.
- Zaidi Y, Aguilar EG, Troncoso M, Ilatovskaya DV, DeLeon Pennell KY (2021) Immune regulation of cardiac fibrosis post myocardial infarction. Cell Signal 77: 109837.

 Research Article
Research Article