ABSTRACT
Introduction: Breast cancer is a large health problem. One of the therapies is percutaneous thermal tumor ablation, e. g. cryotherapy. In this prospective study we tried to show the histologic changes and to analyse the effectiveness of cryotherapy for the treatment of breast cancer.
Material and Methods: 53 patients with age of 38 – 81 years and with biopsy ensured breast carcinoma were handled with cryotherapy. After sonographically guided placing of a cryoprobe a complete intervention has been carried out in 51 patients. The patients underwent 1 to 35 days after cryotherapy a breast saving (48) or mastectomy (5) operation and treated according to the directive.
Results: The histologic examination of all cases displayed at least a partial destruction of tumor and variable amount of cryotherapy related changes (bleeding, necrosis, fibrosis, xanthogranulomatous inflammation with giant cells, etc.). In 27 breast cancer cases who were handled between 1999 and 2001 by cryotherapy (group 1) only 6 patients showed no residual tumor. The second group of patients (26 cases) were handled between 2002 and 2007. In this group only 3 patients had residual tumor after cryotherapy and 2 other cases had lymph node metastasis. 10 patients (group 1) and 3 (group 2) died.
Conclusion: The present study showed that breast carcinoma can be handled by cryotherapy. Especially small tumors with diameter of 15 mm or less can be successfully managed. In our view invasive lobular carcinomas and ductal carcinoma in situ are less appropriate for cryotherapy. The percutaneous cryotherapy produces no significant local or systematic complication in postoperative phases.
Keywords: Breast Cancer, Cryotherapy, Cryoablation, Breast Biopsy, Breast Surgery, Breast pathology
Abbreviations: DCIS: Ductal Carcinoma in Situ; IDC: Infiltrating Ductal Carcinoma; ILC: Infiltrating Lobular Carcinoma; ACC: Adenoid Cystic Carcinoma; ITC: Infiltrating Tubular Carcinoma; bl: Bilateral; NI: Not Identified; CAU: Caudal; CRA: Cranial; med: Medial; lat: Lateral; NRT: No Residual Tumor; LN: Lymph Node; RT: Radiotherapy; CT: Chemotherapy; HT: Hormone Therapy; D: Dead; A: Alive; MTS: Metastasis; OSS: Osteal
Introduction
The breast carcinoma with annually and worldwide more than one million new cases is one of the most frequent malignancies of women [1]. Every year about 72.000 new cases of breast cancer are detected in Germany. That means about one tenth of women experience breast cancer in their life. Among them about 20.000 cases are younger than 60 years. Accordingly breast cancer is also one of the most frequent malignancies of women in Germany. The mortality rate of breast carcinoma is about 18.000 women und 100 men each year [1-4]. The improvement of diagnostic procedures has led to earlier diagnosis of breast cancer. The multimodal therapy of locally advanced breast carcinoma with initial (neoadjuvant) systemic chemotherapy and the following local and regional therapies are performed increasingly [5]. Now a day breast saving therapy is carried out as a preferred therapeutic method and the results are comparable with mastectomy.
During last few years there was a trend for treating breast cancer with less invasive or minimal invasive procedures. Some methods are based on percutaneous tumor ablation by inducing local heat. They include Interstitial Laser Therapy (ILT), Radiofrequency ablation (RF) or Highly Intensive Focused Ultrasound (HIFU). Another method for local tumor management is cryotherapy. The aim of this method is to apply a local extremely low temperature to induce tissue degeneration. The procedure is carried out by using -180°C fluid nitrogen. The coldness is conducted by pouring the fluid nitrogen to the tumor location through a so called Cryoprobe [6-11]. Primarily this method has been used for treatment of prostate and liver tumors and gradually has been applied for the management of other tumors e.g. breast and kidney tumors. Recently some studies were published about the cryotherapy of breast cancer and fibroadenoma [12-19]. In the present study we analysed the effects of cryotherapy on the histologic examination of post treatment tumor resection specimens in a series of patients with breast carcinoma. As far as possible we analyzed also the follow up of the patients.
Materials and Methods
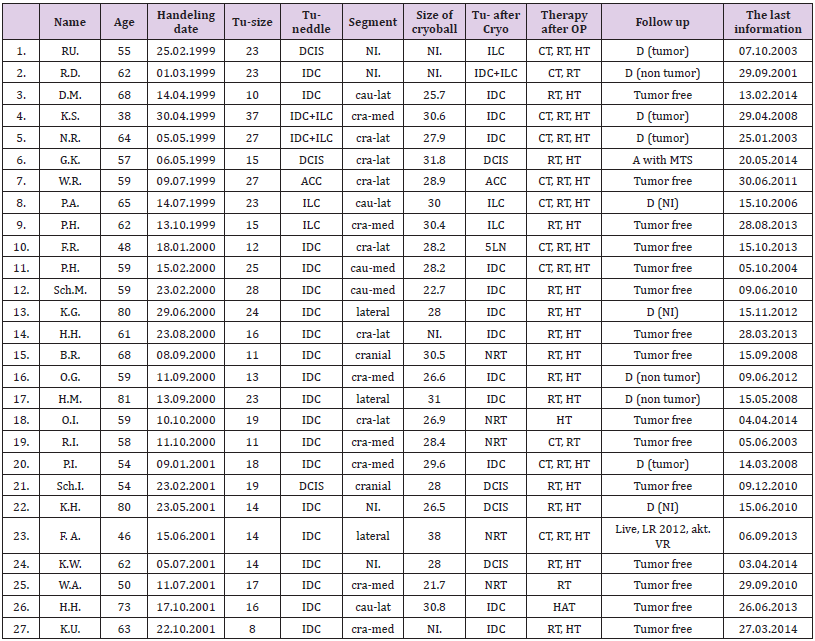
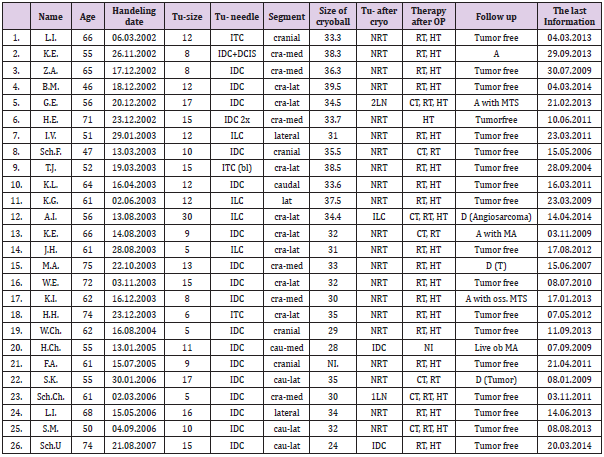
53 patients in the age range of 38 – 81 years (mean 61) underwent percutaneous ultrasound guided cryotherapy. The number of cases between 1999 and 2001 was 27 (tumor size 8 – 40 mm, mean 19.2 mm, group 1, Table 1). The number of patients between 2002 and 2007 was 26 (tumor size 5 – 30 mm, mean 11.7, group 2, Table 2). In all cases the diagnosis of breast cancer was confirmed in a needle biopsy sample. The cases included 39 cases of Invasive Ductal Carcinoma (IDC), 6 cases of Invasive Lobular Carcinoma (ILC), 3 cases of Invasive Tubular Carcinoma (ITC), 2 cases of IDC with components of ILC, one case of Adenoid Cystic Carcinoma (ACC) and 2 cases of Intraductal Carcinoma (DCIS). After local anesthesia and under the guidance of sonography a probe of Argon gas was placed at the tumor center. The procedure is performed in two cycles and each cycle included 7 minutes of freezing phase with following 5 minutes of thawing phase. The size of ice ball was measured in one minute intervals (21.7 – 39.5 mm, mean 31 mm). In five cases there was no available data. The continuing measurement of temperature at the top of probe showed a temperature of -146 ± 15.5°C. The patients were operated 1 to 35 days after cryotherapy and the specimens were analysed histologically. The type of operation was in 48 cases breast saving surgery and in 5 cases mastectomy.
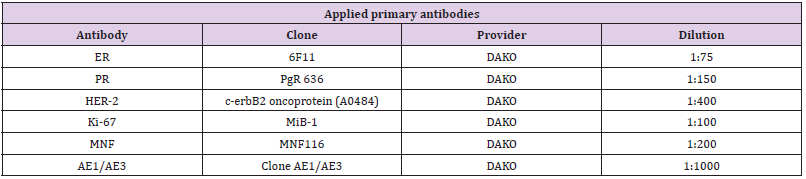
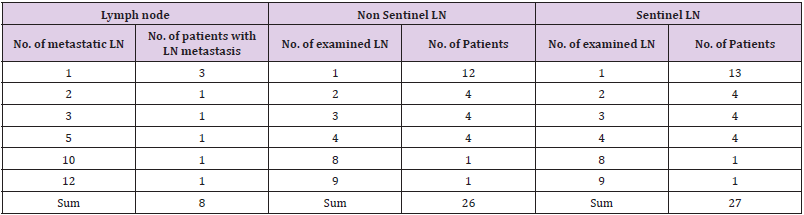
In two cases the tumor could not be excised completely in a breast saving surgery and accordingly this type of operation was followed by a mastectomy. The pathologic examination of needle biopsies and resection specimens performed in the Institute of Pathology, FSU, Jena, according to the international recommendations and S3-guidelines. The mastectomy and segmentectomy (or quadrantectomy) specimens were labelled on two or three facets, respectively. The specimens were fixed in 4% buffered formalin solution and then sectioned in about 5 mm thick intervals along the long axis of specimen. The segmentectomy or quadrantectomy specimens were embedded totally in paraffin blocks. The sections were stained by HE and examined microscopically. In mastectomy specimens the tumor area was sectioned and embedded completely alongside about 5 cm of adjacent macroscopic tumor free breast tissue. Immunohistochemical expression of pan cytokeratin (MNF), AE1/AE3 and Ki67 was examined in three, one and eight cases, respectively. In 26 cases the ipsilateral lymph nodes were also examined (in average 11 lymph nodes in each case). In 27 cases a sentinel lymph node resection was performed.
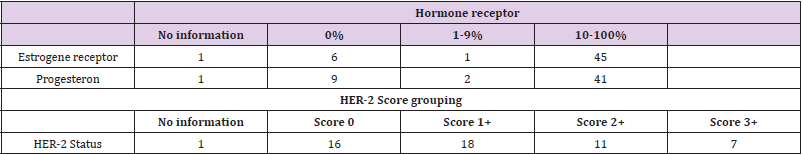
The status of hormone receptors as well as growth factor receptor c-erbB-2 (Her2/neu) was evaluated immunohistochemically in all needle biopsy specimens. For immunohistochemical examinations the sections were deparaffinised, rehydratet and stained after boiling for 20 minutes by microwave in a sodium citrate buffer pH 6.0. The primary antibodies, the level of dilutions and the providers are listed in Table 3. The analysis of estrogen and progesterone receptors was done semiquantitatively on the basis of staining intensity and the percent of positive cell nuclei. In the cases with an at least weak nuclear reactivity in more than 10% of tumor cells the reaction is considered positive. For the evaluation of Her2 the usual international score system of 0 – 3+ was used. In one case there was no tumor tissue in the sections for immunohistochemistry (Table 4). The collection of data and statistical analyses were carried out with Microsoft Excel 2007 and SPSS 15.0 (Chicago, USA). The difference between groups of data were analysed using Mann-Whitney U-Test and considered as significant if the p values is ≤ 0.05. According to the current guidelines the patients were handled by radiotherapy, chemotherapy and hormonotherapy.
Results
The intervention was done completely in 51 cases by placing the cryoprobe in the tumor location under the guidance of sonography. In one case the procedure has to be finished prematurely after 5 minutes from the beginning of freezing cycles because of technical problems. In another case the cryotherapy has to be stopped because of gas leakage. The size of tumor foci was between 0 and 55 mm (mean 27.4 mm) by macroscopic pathologic examination. In one case no tumor focus was detectable at macroscopic evaluation. In addition in two cases the tumor size was not recorded. The macroscopic appearance of tumor area was characteristic. The tumoral area showed variably dark red color appearance which can be related to the bleeding, necrosis and reparative changes after application of cryotherapy. In the cases which were operated within three days after cryotherapy the typical microscopic changes were haemorrhagic, necrosis and presence of tumoral shadow cells. Afterward xanthogranulomas inflammation as well as lymphocytic and plasmacytic infiltration became evident. After two weeks variable amount of tissue fibrosis and formation of granulation tissue constituted the dominant histologic picture.
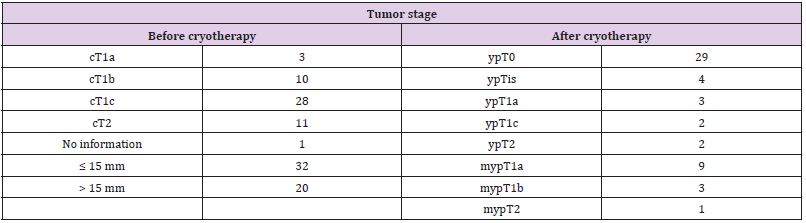
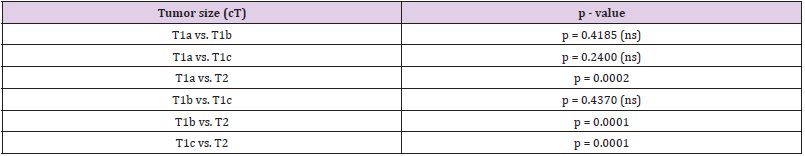
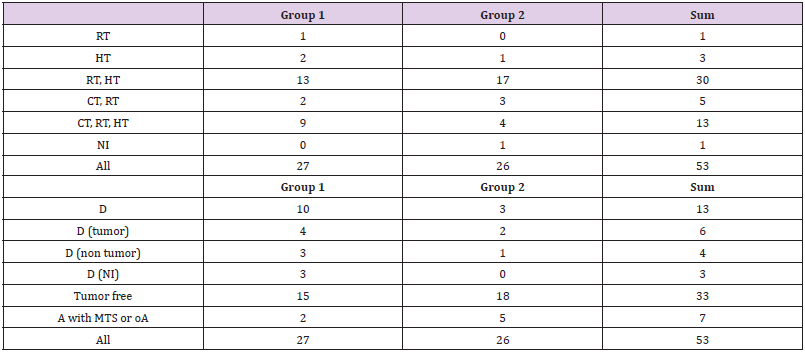
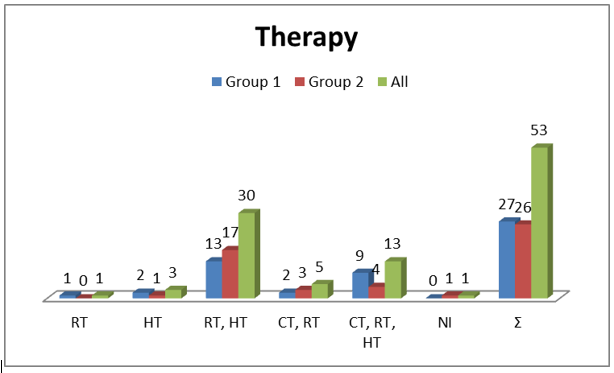
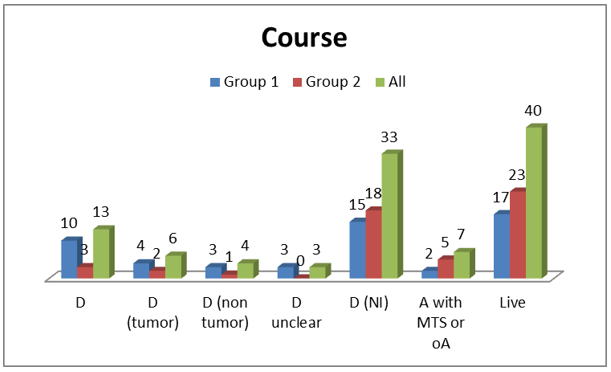
In all cases at least partial tumor destruction was detectable. The tumor staging before and after cryotherapy is depicted in Table 5. In 29 cases with the tumor size up to 19 mm a complete destruction of tumor was identified. In 24 cases the vital tumor rests were detected. They included one case with IDC with invasive lobular components, 4 cases with ILC, 4 cases with DCIS, 1 case of adenoid cystic carcinoma and 14 cases with IDC (Tables 1 & 2). Although in three cases no tumor rest has been detected in resection specimen, the examination of lymph nodes showed metastases. Five cases had the local tumor residue and lymph node metastases both (Table 6). Because of low number of these cases a statistical analysis was not possible. The tumor size between pT1 and pT2 has an essential influence on ten effectiveness of cryotherapy (Table 7). The Mann-Whitney-U-test showed no meaningful difference between histologic tumor type and the effectiveness of cryotherapy. The form of postoperative therapy is shown in the Table 8 and Diagram 1. The outcome of the patients was displayed in Table 8 and Diagram 2.
Discussion
Neoadjuvant chemotherapy is used preferably to eliminate the tumoral tissue and to help to preserve the breast tissue in cases with breast cancer. In most of these cases a complete regression of tumoral tissue is not accomplished [20]. Contemporary methods of breast saving treatment modalities consist of lumpectomy, chemotherapy, hormone therapy and radiation therapy, although not in all cases the cancer cells can be completely eradicated. That in turn can make a new course of radiation therapy mandatory. In this situation the side effects are not rare. The minimal invasive methods have been implicated increasingly during last few years for treatment of breast cancer. One of these methods is cryotherapy which has been approved for the treatment of liver and prostate tumors [13]. The preoperative application of cryotherapy can provide better results with induction of tumor regression and even complete disappearance of tumor without any significant side effect. Until now only few and mostly experimental reports with few number of patients (maximal 29) were published about the application of cryotherapy in breast tumor patients [13,18-22].
On the other hand the histopathologic evaluation of postoperative specimens was more and less descriptive. As far as we know other than a study by Pusztaszeri et al there is no real histopathologic study of such cases in human [23]. The present study with histologic verification of treatment effects after cryotherapy in 53 breast cancers is in our view the largest published case series. We have released some of the data before in 3 other publications [11,21,22]. The vital and avital tumor cells can be discriminated easily from each other. In questionable cases finding the tumor cells by the aid of immunohistochemistry for proliferation marker Ki67 and pancytokeratin MNF can be helpful. Roubidoux et al believe that it is necessary to have at least 7 days difference between cryoablation and lumpectomy for better histologic evaluation of residual tumoral tissue during routine light microscopic examination, because before this interval it is not possible to differentiate the vital and the avital or deadly injured tumor cells from each other [12]. If the tumor diagnosis is histologically confirmed on the needle biopsy, it is not really important to provide information about the tumor differentiation on post treatment samples.
These findings are compatible with other studies. Most studies reported the histopathologic effects of cryotherapy basically in first 4 - 5 weeks with no detailed description about the exact pathologic changes even in animal studies [12,13,16,18,19,24-26]. A successful cryotherapy is dependent on the size of tumor and the exact positioning the probe. The DCIS and ILC are usually multifocal and hence are less suitable for cryotherapy. The accurate size of tumors was in this series of cases difficult to measure. Many authors believe that the cryotherapy can be carried out successfully in handling of benign tumors such as fibroadenoma. The cryotherapy of fibroadenoma is a safe, effective, practical and pain free method of therapy with good cosmetic results. Particularly in cases who want to be treated without open surgery cryoablation of benign breast lesions is a preferred method [17]. Between year 1999 and 2001 in our institute 27 cases of breast cancer (tumor size 8 - 40 mm, mean 19.21 mm) were treated by cryotherapy (group 1) and in only 6 of them no residual tumor was found. The number of cases between 2002 and 2007 was 26 (tumor size 3 - 50 mm, mean 11.68). In this group of cases (group 2) in only 3 cases residual local tumor was detectable and two other cases had lymph node metastasis. In this recent group only one tumor was 30 mm in largest diameter. All other tumors were smaller with mean size of 17 mm. In all cases of this study we found after cryotherapy 15 cases of IDC 13 of them belonged to the group 1. The size of 10 tumors was more than 1.5 cm. In 4 cases the tumor was ILC and all of them were more than 2.3 cm in size.
One adenoid cystic carcinoma was among the cases of group 1 and had a diameter of 2.7 cm before the cryotherapy. The residual tumor after cryotherapy measured 1.5 cm in this case. In group 1 we had in addition 4 cases of DCIS. One of these 4 tumors had a diameter of more than 1.5 cm. Before the cryotherapy we had 6 patients with ILC. After cryotherapy in two cases the residual tumor in form of ILC and in one case residual tumor in form of intraductal carcinoma was detected. In all these three cases the size of primary tumor was more than 2.3 cm. The size of tumor in two cases with intraductal carcinoma was 1.9 and 2.3 cm. The examination after cryotherapy showed an intraductal and an invasive lobular carcinoma. A woman had two foci of invasive ductal carcinoma each one 1.5 cm in diameter. In this case we could not find any tumor residue after cryotherapy. The comparison of the results between these two groups showed the importance of experience of operator and selection criteria for a successful cryotherapy. In our view the most important cause for the presence of rest tumor in cases with small tumor size (≤ 1.5 cm) is the false positioning of probe. The most types of carcinomas can be easily diagnosed in needle biopsy specimens. In problematic cases it is possible to use immunohistochemistry. The evaluation of hormone and Her2 status is any way obligatory.
Our results are widely in concordance with the results of other studies. Apparently, the results showed that cryotherapy is more successful in handling of IDC in compare with ILC and DCIS. Roubidoux et al reported about their experience with cryotherapy of small breast tumors (< 2 cm) [12]. By ultrasonographic examination had 7 of 9 patients no residual tumor. Sabel et al reported about the results of cryotherapy in 21 patients with invasive tumors with diameter of ≤ 2 cm [13]. The surgical resection followed 1-4 weeks later and showed that all tumors with diameter of <1 cm could be completely eradicated. That means no residual tumor could be detected histologically. In cases with tumors with diameter of >1.5 cm they found residual invasive carcinoma (1 case) and residual DCIS (4 cases) after cryotherapy. In the study of Pusztaszeri et al only two patients had a complete remission without residual vital tumor cells. Seven patients had partial remission with residual areas of invasive carcinoma. Two patients had intraductal carcinoma. The authors believe that the tumor destruction has no relationship with tumor size (clinically <2 cm) or other histologic parameters. They had in their series only 1 case with ILC and 10 cases with IDC (23). A pilot study from Vlastos et al showed also encouraging results. Histologically proved invasive breast carcinomas (≤ 2 cm) were successfully managed by cryotherapy which was confirmed by a surgical examination after one month [14].
Our study sowed that the tumor size between cT1 and cT2 is a significant deciding factor for a successful cryotherapy. All 29 cases in our study with tumor free status after cryotherapy were in cT1 stage. The Mann-Whitney-U-Test showed that the histologic type of tumor has no significant effect on the results of cryotherapy. It has to be pointed out that DCIS and ILC are often accompanied by numerous foci of tumor and in our view are not good candidates for a successful cryotherapy. We think that cryotherapy is in general an appropriate method for handling of small primary breast tumors. Ultrasonography is a valuable adjunct tool for better visualisation and control of ablation area. Its application also ensures an adequate protection of skin in cases with superficial tumors near to the skin in the way that they can be handled without significant skin complication. Most importantly is the choosing of appropriate patients and exact positioning of probe. Patients with breast cancer of less than 1.5 cm in maximal diameter with no invasive lobular or DCIS component are appropriate cases for cryoablation [13].
The histologic evaluation of all specimens revealed a R0 situation, although in 5 cases this has been achieved after a mastectomy operation. According to the HER2-status as well as the recommendations of current guidelines all the patients received postoperative radio-, chemo- and hormonotherapy. The results of therapeutic measures were similar to the patients without cryotherapy. The most frequent treatment combination in both groups was radio- and chemotherapy or radio-, chemoand hormonotherapy (30 and 13 patients, respectively). Because of small number of patients in each group a statistical evaluation of the results was very difficult and sometimes impossible. A similar situation was seen in the survival data. Thirteen patients (24.5%) were died between 2 and 12 years after detection of carcinoma among them 10 patients (36.3%) belonged to the group 1 and 3 patients (11.5%) belonged to group 2. It shows an evident increased death rate in patients of group 1. In this group of patients 4 patients were died by carcinoma and in 3 cases the cause of death was not evident. In the whole cohort 40 patients (75.5%) were alive among them 17 patients (63.7%) were in group 1 and 23 patients (88.5%) were in group 2. The survival time in group 1 patients varied between 2 and 15 years (in 12 cases more than 10 years). The survival time in group 2 patients varied between 1 and 11 years (more than 10 years in 4 women). Not all the patients were available for follow up. In summary it is possible to conclude that patients of group 2 had a better survival rate and very low mortality rate. They are patients with a tumor size of 5 to 30 mm (mean 11.68 mm) and constitute the group of patients who predictably can benefit from cryotherapy.
Conclusion
The present study showed that breast carcinoma can be managed by cryotherapy. Particularly small tumors with the diameter of 15 mm or less can be handled successfully. Most important influencing factors for a successful treatment are the experience of operator and the selection of appropriate patients. The following inclusion criteria are in our view essential: sonographically detectable tumors, histologic confirmation of tumor in clinical stage of T1 (≤ 15 mm), more than 10 mm distance from the skin and tumor localisation out of nipple area. In addition it is necessary to have the informed consent of the patient. We believe that the invasive lobular carcinoma and DCIS are not suitable tumor types for a successful cryotherapy. According to the S3 guideline in all cases with histologically ensured breast cancer the examination of sentinel lymph nodes is necessary. Although further studies with larger number of cases and long follow up is necessary, the results of this study show that the percutaneous, ultrasonographically supported cryotherapy is a simple and safe method for handling of small breast carcinomas. This method causes no significant local or systematic complication in postoperative phases.
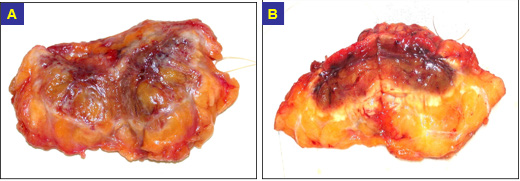
Figure 1: Resetion specimens after cryotherapy: Cut surface of specimens at 9 days
(A) and 27 days
(B) after treatment.
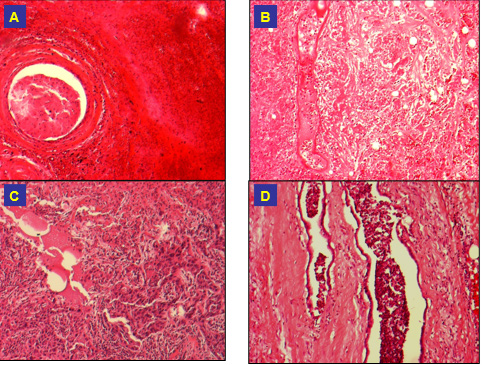
Figure 2: Microscopic changes at day 2 after cryotherapy:
A and B Hemorrhage and necrosis;
C and D infiltrations of an invasive ductal Carcinoma (H&E. 100x).
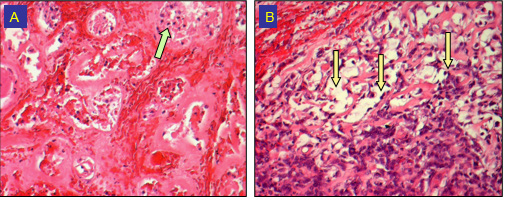
Figure 3: Microscopic changes at day 5 after cryotherapy:
(A) A Hemorrhage necrosis and avital tumor cells (arrow) (H&E. 100x) ),
(B) B Hemorrhage (left up) and vital tumoral cells of an adenoid cystic carcinoma (arrows) (H&E, 200x).
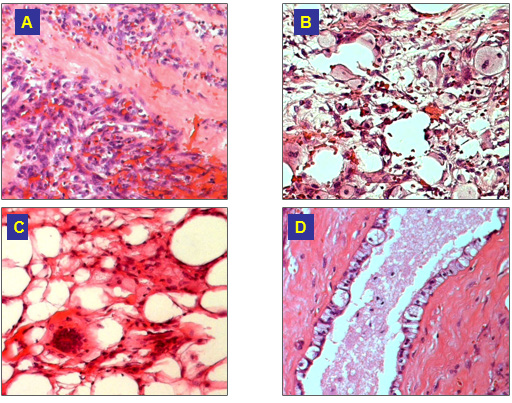
Figure 4: Microscopic features at day 9 after cryotherapy:
(A) A Necrosis and granulation tissue;
(B) B Foamy cells;
(C) C Gaint cells and macrophages;
(D) D Hydropic degeneration of epithelial cells (H&E, 100x).
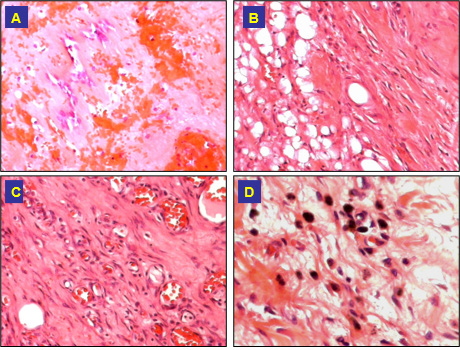
Figure 4: Microscopic changes at day 25 after cryotherapy:
(A) A Remained bledding and fibrosis;
(B) B Fibrosis and granulation tissue;
(C) C Granulation tissue with abundant small vessels (H&E, 100x);
(D) D Fibrosis and pigment laden macrophages (H&E, 200x).
Conflicts of Interest
Conflict of Interest relevant to this article was not reported.
References
- Boyle P, Ferlay J (2005) Cancer incidence and mortality in Europe, 2004. Ann Oncol 16(3): 481-488.
- Beckmann MW (2005) Women with primary breast cancer - the conflicting priorities between medicine and politics. Geburtshilfe und Frauenheilkunde 65: 137-138.
- Fisher B, Redmond C, Poisson R, Margolese R, Wolmark N, et al. (1989) Eight year results of a randomized clinical trial comparing total mastectomy and lumpectomy with or without irradiation in the treatment of breast cancer. N Engl J Med 320(13): 822-828.
- Baras N, Barnes B, Bertz J, et al. (2012) Breast in Cancer in Germany 2007/2008. RKI 8: 64-67.
- Piccart MJ, Kerger J, Tomiak E, Perrault DJ (1992) Systemic treatment for locally advanced breast cancer: what we still need to learn after a decade of multimodality clinical trials. Eur J Cancer 28(2-3): 667-672.
- Pfleiderer SO, Reichenbach JR, Wurdinger S, Marx C, Freesmeyer MG, et al. (2003) Interventional MR-mammography: manipulator-assisted large core biopsy and interstitial laser therapy of tumors of the female breast. Z Med Phys 13: 198-202.
- Hilger I, Hiergeist R, Hergt R, Winnefeld K, Schuber H, et al. (2002) Thermal ablation of tumors using magnetic nanoparticles: an in vivo feasibility study. Invest Radiol 37(10): 580-586.
- Gianfelice D, Khiat A, Amara M, Belblidia A, Boulanger Y (2003) MR imaging-guided focused US ablation of breast cancer: histopathologic assessment of effectiveness initial experience. Radiology 227(3): 849-855.
- Suzuki Y (1995) Cryosurgical treatment of advanced breast cancer and cryoimmunological responses. Skin Cancer 10: 19-26.
- Tanaka S (1995) Cryosurgical treatment of advanced breast cancer. Skin Cancer 10: 9-18.
- Gajda MR, Mireskandari M, Baltzer PA, Pfleiderer SO, Camara O, et al. (2014) Breast pathology after cryotherapy. Histological regression of breast cancer after cryotherapy. Pol J Pathol 65(1): 20-28.
- Roubidoux MA, Sabel MS, Bailey JE, Kleer CG, Klein KA, et al. (2004) Small (<0-cm) breast cancers: mammographic and US findings at US-guided cryoablation - initial experience. Radiology 233(3): 857-867.
- Sabel MS, Kaufman CS, Whitworth P, Chang H, Stocks LH, et al. (2004) Cryoablation of early-stage breast cancer: Work-in-progress report of a multi-institutional trial. Ann Surg Oncol 11(5): 542-549.
- Vlastos G, Verkooijen HM (2007) Minimally invasive approaches for diagnosis and tratment of early-stage breast cancer. The Oncologist 12(1): 1-10.
- Staren ED, Sabel MS, Gianakakis LM, Wiener GA, Hart VM, et al. (1997) Cryosurgery of breast cancer. Arch Surg 132(1): 28-33.
- Morin J, Traoré A, Dionne G, Dumont M, Fouquette B, et al. (2004) Magnetic resonance-guided percutaneous cryosurgery of breast carcinoma: technique and early clinical results. Can J Surg 47(5): 347-351.
- Kaufman CS, Littrup PJ, Freeman Gibb LA, Smith JS, Francescatti D, et al. (2005) Office-based cryoablation of breast fibroadenomas with long-term follow-up. Breast J 11(5): 344-350.
- Manenti G, Perretta T, Gaspari E, Pistolese CA, Scarano L, et al. (2011) Percutaneous local ablation of unifocal subclinical breast cancer: clinical experience and preliminary results of cryotherapy. Eur Radiol 21(11): 2344-2353.
- Littrup PJ, Jallad B, Chandiwala Mody P, D Agostini M, Adam BA, et al. (2009) Cryotherapy for breast cancer: a feasibility study without excision. J Vasc Interv Radiol 20(10): 1329-1341.
- Fisher B, Brown A, Mamounas E, Wieand S, Robidoux A, et al. (1997) Effect of preoperative chemotherapy on local-regional disease in women with operable breast cancer: findings from National Surgical Adjuvant Breast and Bowel Project B-18. J Clin Oncol 15(7): 2483-2493.
- Pfleiderer SOR, Freesmeyer MG, Marx C, Kühne Heid R, Schneider RA, et al. (2002) Cryotherapy of breast cancer under ultrasound-guidance: initial results and limitations. Eur Radiol 12(12): 3009-3014.
- Pfleiderer SO, Marx C, Camara O, Gajda M, Kaiser WA (2005) Ultrasound-guided, percutaneous cryotherapy of small (≤ 15 mm) breast cancers. Invest Radiol 40(7): 472-477.
- Pusztaszeri M, Vlastos G, Kinkel K, Pelte MF (2007) Histopathological study of breast cancer and normal breast tissue after magnetic resonance-guided cryotherapy ablation. Cryobiology 55(1): 44-51.
- Rand RP, Eggerding FA, Field M, Denbesten L, King W, et al. (1985) Cryolumpectomy for breast cancer: an experimental study. Cryobiology 22(4): 307-318.
- Rabin Y, Julian TB, Olson P, Taylor MJ, Wolmark N (1999) Long-term follow-up post-cryosurgery in a sheep breast model. Cryobiology 39(1): 29-46.
- Otterson MF, Redlich PN, McDonald A, Kristin Thorsen M, Klaas KK, et al. (2003) Sequelae of cryotherapy in breast tissue. Cryobiology 47(2): 174-178.

 Research Article
Research Article