Abstract
Background: The COVID-19 pandemic virus detections are dependent on complex diagnostic techniques, such as PCR molecular amplification, electrophoresis DNA separation, titrimetric fluorescence chromatography and other combined time losing skills.
Methods: However, in this study, a simplified virtual rapid sensing voltammetric micro working probe was developed, the detection time used only 60 seconds. which can be analyzed anywhere by anyone with simple instantaneous detection. Electrochemical antibody antigen redox titration current was amplified using platinum linked graphene nano powder working electrode.
Results: As the binding strength enhancer, an aqueous dispersion synthesized resin was used. Detection potential windows of -2.0V~2.0V cyclic, anodic and cathodic stripping accumulation time was just 30 seconds. The diagnostic circuit of the coin size voltammetric workstation used.
Conclusions: Titration current can be performed to 1.0×10-3A~1.0×10-9A amplification, coin size electrochemical workstation analyzer and tattoo mimic skin in vitro electrode sensing was used. Results are useable for anyone who can use it on their own telemetric wearable diagnosis.
Keywords: Rapid Sensing; Covid-19 Virus; Antibody; Blood; Platinum; Antigen; Graphene Probe
Abbreviations: COVID 19: Coronavirus disease 2019; WHO: World Health Organization; RT-PCR: Real time polymerase chain reaction.
Background
Introduction
In December 2019, a cluster of atypical pneumonia cases was detected in Wuhan, China, which soon spread throughout China before Chinese Lunar New Year in January 25, 2020. Outdoor movement restrictions were also adopted in a number of wide cities. However it was far too late. Here the novel coronavirus was named COVID-19 by the World Health Organization (WHO) on February 11, 2020. Later the COVID-19 epidemic was declared a public health emergency of international concern by WHO on January 30, 2020. Also later, on March 11, 2020 it was declared a pandemic by the WHO. On March 29, 2021, more than 126.89 million people have been confirmed to be infected (across over 223 countries, areas or territories), of which more than 2.78 million people died (WHO, 2020b) [1]. This is now pushing the whole of humanity into a biological warfare against viruses. Recent virus detection methods depend on the titrimetric PCR amplifications such as hemagglutination inhibition assay of the microneutralization [2], fluorescence spectrometric absorption method [3], photometric UV-Vis spectroscopy and electron microscopy [4] and others. Here conventional testing methods require approximately 1~8 days by virus culture, DNA amplification, electrophoresis separation and neutralization titration spectroscopic analysis. However, electrochemical in vivo or in vitro trace ion [5] and [6] cell [7,8] detection is fast, and only a few minutes are required. Diagnostic circuit fabrications are simple, so for this reason, we have developed modified probe sensing skills such as micro implementation [9] Diagnostic carcinoembryonic antigen tumor marker [10] detection, Pico molar ranges [11] working probe infrared photo diode electrodes [12,13] for in vivo cell [14] or human fluid [15] non treated tissue direct assay [16] and macro [17] synthesis [18] Human Blood Plasma [19] sensing ect, also which sensing circuit can be interfaced to telemetric WiFi wearable diagnostics for covid virus detection. Here developed results can replace the existing complex WHO method, such as PCR DNA amplification, Fluorescence Absorption Analysis, or chromatographic separation.
WHO Standard Diagnostic Protocol Comparison
Conventional WHO diagnostic protocols of PCR DNA amplication titrimetric methods is time consuming and replicated sophisticated skills are required. Also highly trained master’s and doctoral technicians are required: for example, Real-time RT-PCR assays for Eurasian H7HA standard protocols shown as follows
WHO Protocol 2
Real-time RT-PCR assays for Eurasian H7HA shown here.
Materials Required
1. QIAamp Viral RNA Mini Kit (QIAGEN®, Cat. No.52906) or
equivalent extraction kit
2. ThermoFisher Fast Virus 1-step Master Mix (Applied
Biosystems®, Cat. No. 44-2)
3. Ethanol (96–100%)
4. Microcentrifuge (adjustable, up to 13 000rpm)
5. Benchtop centrifuge with 96-well plate adaptor
6. Adjustable pipettes (10, 20, 100, 200μl).
7. Sterile, RNase-free pipette tips with aerosol barrier.
8. Vortexer
9. MicroAmp Fast Optical96-well reaction plate(Life Technologies,
Part No.4346906)
10. MicroAmp optical adhesive film (Life Technologies, Part No.
4311971)
11. Real-time PCR system (ViiA™ 7 Real-Time PCR System)
12. Positive control (Available from HKU, e-mail: llmpoon@hkucc.
hku.hk)
13. Primer and probe set.
Type/subtype
Influenza A (H7Nx). Gene:HA.
Name and Sequence
HA H7-1603F: TTTAGCTTCGGGGCATCATGTTT
H7-1674R: CAAATAGTGCACCGCATGTTTCCA
H7-1646P: FAM-TGGGCCTTGTCTTCATATGTGTAAA-MGB
Procedure from reference [20]
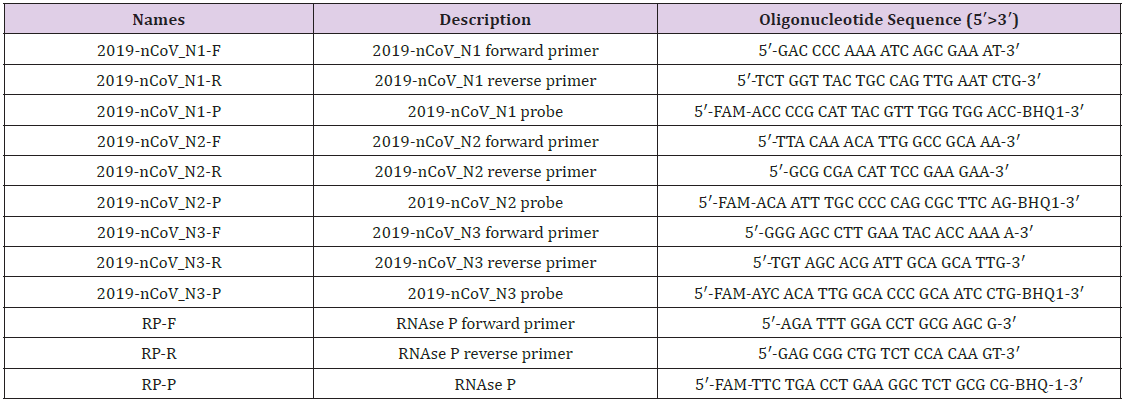
For more detailed examples of Covid 19 primers and probes
sequences shown here. also which sequence was used to antigen
N1~N3 mixing immobilized working probe fabrication. At this
work. Here Table 1 shows the coronavirus disease 2019.molecular
DNA sequence [21]. This DNA was used for the working probe
mixed antigen immobiliz. The following images showing synthetic
polyma in vitro film and sensing probe schimetic fabrication.
Materials and Methods Working Probe
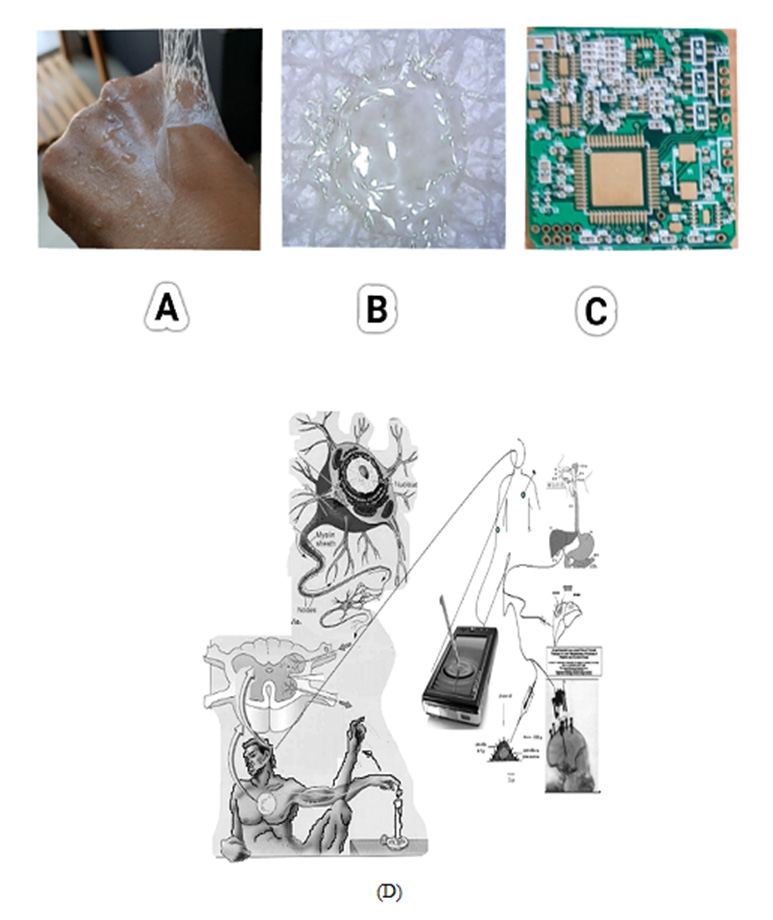
(Figure 1A) A image is shown for skin tattoo coated conduction film made from synthesizing water-dispersion resin with the following characteristics: Softness, Wear-resistance, life extension, Skin protection, Conductivity and Transparency. Image B is a tattoo working electrode at the in vitro skin muscle electrode. Image C is voltammetric bio-electroanalyzer workstation with Wi-Fi telecommunications. It can be used to wearable diagnostic circuit for virus detection. Here in three electrode systems of counter and reference electrode was made like image B. However, working electrode fabrication is shown here, and the probe binder was used by synthesizing an aqueous dispersion resin. The resin paste was made with graphene nanopowder, and covid antigen, and platinum catalys were mixed with conductivity resin. Here the antibody antigen redox titration potential was obtained to reductions of -0.14V, 0.26V anodic scanning and - 0.9V oxidation peak currents was obtained during cathodic scanning. Analytical in vitro WiFi application is shown at (D).
Figure 1: (A) Skin tattoo micro rasin coating film.
(B) In vitro muscle skin tattoo working probe.
(C) Voltammetric Wi-Fi wearable coin size workstation.
(D) Virtual diagnostic in vitro schematic diagram of the working probe redox voltammetric ionic reaction for antigen
immobilized on a graphene nanotube synthesize antibody antigen titration image.
Electrochemical Analyzer
Diagnostic circuit was used for the bioelectrochemical analyzer-2. voltammetric and our system, which can be designed to 5Cm×3Cm×60mm like physical coin size with telemetric WiFi control wearable workstation, potential windows of 2.0V~-2.0V redox potential windows, scanning. The current amplification of 1.0×10-3A~1.0×10-9A pico range and cyclic voltammetry redox, stripping voltammetry and chronoamperometry 2D visual control programs were used, power of USB interfaced ±2.5V strength was used.
Working Electrode Preparation
The diagnostic working probe was made by maxing a ratio of the graphene nano paste 2.0 g, mineral oil 0.5 g, Water-dispersible synthetic resin 0.5 g, platinum standard solution 1000 ppm 0.5 g. Paste was mixed completely during 20 minutes. The paste was coated on the graphite crystal load of 0.5 mm diameter 30 mm length, then connected to voltammetric working electrode terminal by using 0.35 mm pure copper enamel insulation coating wire, counter and reference electrode was made with the same method using graphite crystal load. The circuits are usable for wearable tattoo assay such as In-vivo or in vitro and feeling sensing analysis.
Results
Results and Discussion
Voltammetric analytical optimum para conditions were searched in PBS 5 mL electrolyte microtube conical rack, with 0.6 mL covid virus antigen spiking, at room temperature. The cyclic voltammetric antigen and antibody redox ionic voltammograms were searched.
Cyclic Voltammetric Optimization
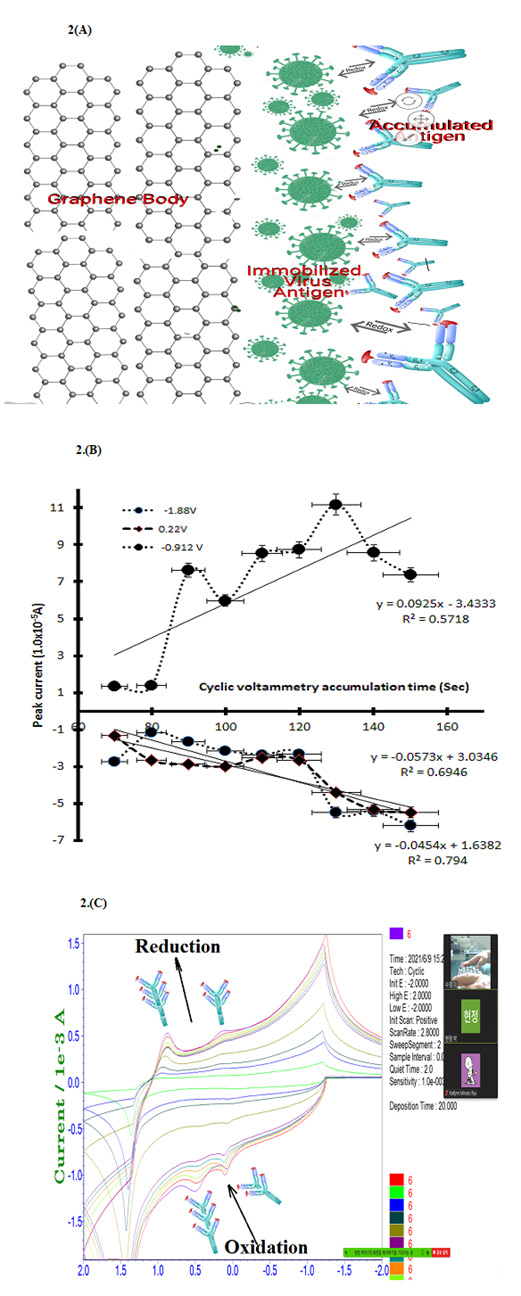
Cyclic potential scanning is related to covid 19 virus ionic redox activation. For this purpose, the cyclic scan rate was searched from 0.2mv/sec~0.9mv/sec eight points in 0.9 mL antibody contained PBS buffer, 30 sec stirring convection with magnetic bar, only changed for scan rate variations. The real voltammogrms shown at Figure 2(A) is antigen antibody titration working image. (B) is, 0.2mv/sec scan rate oxidation peak obtained two site at -0.14V peak high of (4.322×10-5A) and 0.14V peak high of (3.46×10-5A) anodic current obtained then reduction peak of -0.9V peak high of (10.4×10-5A) cathodic obtained, which anodic and cathodic linear statistic 3rd equations shown there slope gradient, intercept and relative standard deviations, here in -0.14V potentials current linear increased, dx/dy=-31.782, y intersect is 3.4125, R2=0.8351 obtained. Other statistic equations not shown here. Figure (C) is reduction potentials of two peaks and oxidation of one linear equation obtained, least square linear equation and relative statistic results not shown in this figure, and the accumulation time increase. The sensitivity increase obtain, here qualitative potentials windows was used to square wave stripping voltammetry.
Discussion
Stripping Voltammetric Optimization
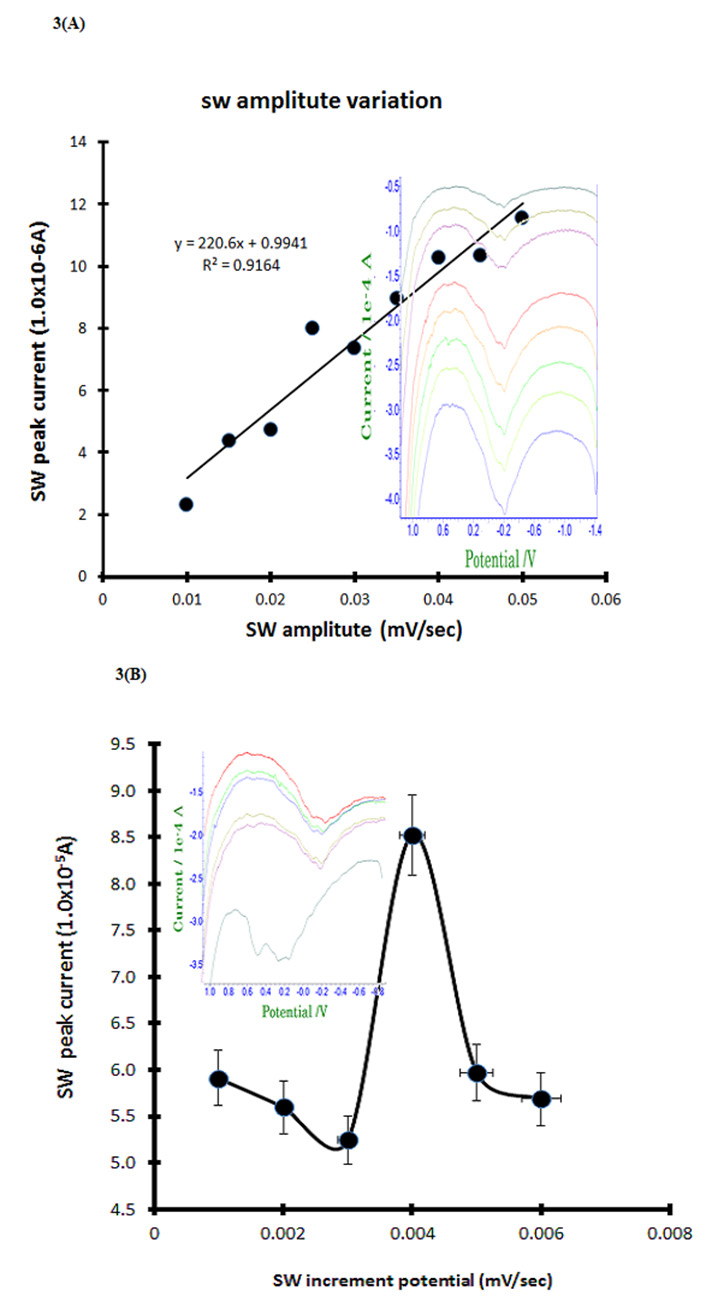
Cyclic voltammetric antigen and antibody anodic or cathodic ionization potentials were applied to sensitive stripping voltammetry. Here stripping accumulated amplifications are increasing to the detection limit, so anodic and cathodic scanning accumulation time variations were examined in 0.9 mL covid 19 antibody containing the PBS electrolyte. Stripping optimum amplified variations shown in Figure 3A, at the first point of 0.01 mV/Sec is 2.321×10-5A obtained in -0.2V potential windows anodic scanning, then continue variations performed during 10 steps by same methods, here final step is 0.05 mV/Sec 11.54×10-5A obtained on the linear curve. The least square linear equation is shown inserted in picture of Y=220.06x+0.9941, slop sensitivity is dx/ dy=220.06, intercept is 0.9941 and the relative standard deviation is R2=0.9164 obtained, at this para conditions square wave increments variation was examined from 0.001 mV/Sec to 0.006 mV/Sec with six steps, results shown in Figure 3B voltammograns and linear statistic equation, here in 0.004 mV/Sec is big current of 8.524×10-5A obtained, thus 0.05 V/Sec amplitude and 0.006 V/Sec increments are fixed. Under these conditions, diagnostic concentration effects were examined.
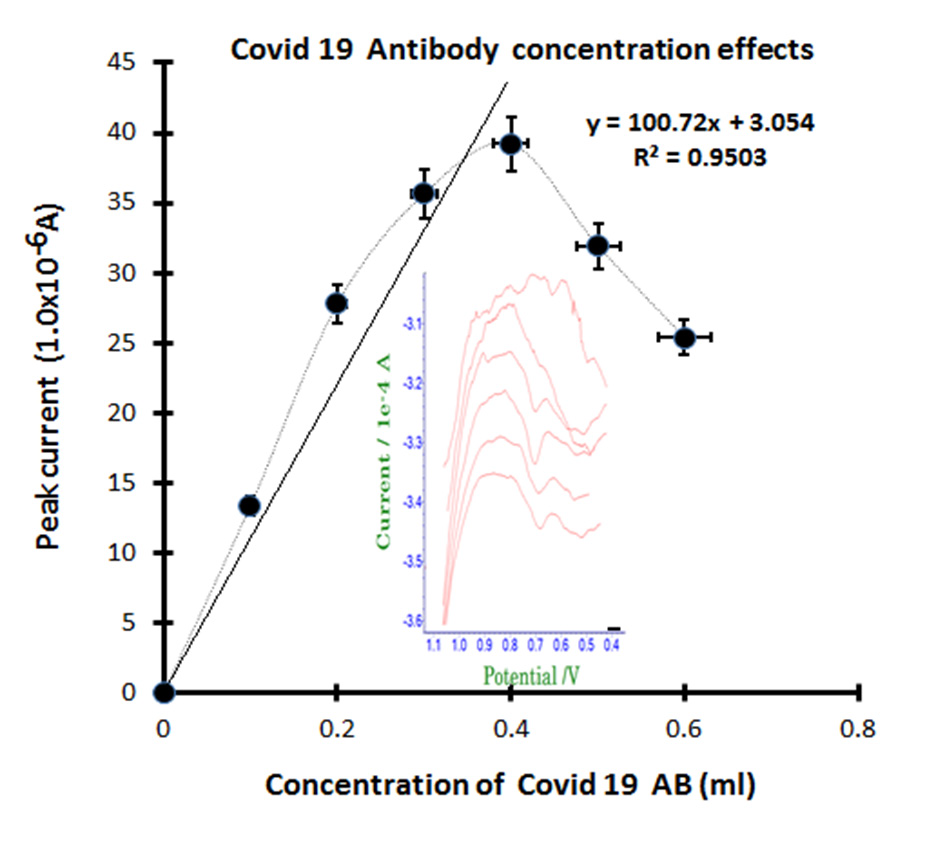
Figure 4 is analytical application effects for patients’ blood extracted antibody standard spike. The optimized final para conditions, diagnostic real antibody was examined in the PBS buffer electrolyte 8 mL volume. The first peak of electrolyte blank stripping is non signal appeared, then the 0.1 mL antibody spike was obtained to anodic of 0.7V oxidation current for 13.37×10-6A. In this figure, the inserted voltammograms shown slowly increased the sharp width, then continued spiking of 0.2, 0.3, 0.4, 0.5 and 0.6 mL the same conditions were added. Here the peak current 27.78×10-6A, 35.65×10-6A, 39.21×10-6A, linearly increased, then 31.92×10-6A, 25.37×10-6A decreased, which linear equations of dx/ dy=100.72, Y intercept is 3.054, the relative standard is R2=0.9504, which statistic results can be usable for real patients’ blood assay, anytime and anywhere immediately with self-detection. Thus, a more simplified tattoo wearable sensing circuit was fabricated, which can be used to telemetric remote control.
Figure 2: (A) Cyclic voltammetric scan rate variations for 0.2, 0.3, 0.4, 0.5, 0.6, 0.7, 0.8, 0.9 mV/Sec, insert voltammograms shown
reductions of -0.14V, 0.26V and -0.9V oxidation peak currents with 0.9 mL antigen spike in 3mL PBS electrolyte.
(B) Cyclic voltammetric accumulation time variations for 70, 80, 90, 100, 110, 120, 130, 140, 150 sec at (A) condition.
(C) Virtual real time voltammograms ionic imager.
Figure 3: (A) Square wave stripping amplitude variations for 0.01, 0.015, 0.02, 0.025, 0.03, 0.935, 0.04, 0.045, 0.05 V/Sec in PBS electrolyte
3mL, with 0.9mL Covid 19 antigen spike.
(B) Square wave increments potential variations for 0.001, 0.092, 0.03, 0.094, 0.005, 0.06 V/Sec of (A) electrolyte conditions.
Figure 4: Concentration effect of Covid-19 antibody for 0.1mL, 0.2mL, 0.3mL, 0 4mL, 0.5mL, 0.6mL standard spike, during stripping optimum para conditions, accumulation time was 60 sec string anodic -2.0V~-2.0V in PBS 8 mL buffer electrolyte.
Conclusion
Diagnostic optimum results be attained in 60 seconds accumulation time only. During the anodic scanning, stripping peak potentials of 0.7V oxidation current was obtained of antibody antigen ionic transfer redox strength. Electrolyte buffer solution needed 5 mL PBS only and didn’t require any titrate or separation solutions. Detection systems used to Wi-Fi telemetric voltamnetric workstations of wearable patch type simple circuit of -2.0V ~ -2.0V potential scanning cyclic amplifier, working electrode is tattoo synthetic art painting film sensors of three electrode. The current range was 1.0×10-4A ~ 1.0×10-5A. The developed diagnostic results be used for virus detection and in any other field requiring human computer interfaced wearable tattoo bioassay and in vivo remote telemetric treatments.
Acknowledgement
Declared none.
Authors’ Contributions
This idea from Dong Ho Kim, Jeeyu Choi, Hyeon Jeong Park,
Jeannette Sahnghee Kim, Kyung Lee and SuwYoung Ly, experiment
perform by all authors.
All authors read and approved the final manuscript.
Funding
This research was not supported by the any fund.
Availability of data and materials
All materials are available by the corresponding author.
Declarations
Ethics Approval and Consent to Participate
All experiments were performed according to established guidelines for the ethical use.
Consent for Publication
Not applicable.
Competing Interests
Declare No Conflict of Interest.
References
- Lili Ronga, Anming Zhang (2021) Assessing regional risk of COVID-19 infection from Wuhan via high-speed rail, Transport Policy 106: 226-238.
- Federica Siccaa, Donata Martinuzzi, Emanuele Montomolib, Anke Huckriedea (2020) comparison of influenza-specific neutralizing antibody titers determined using different assay readouts and hemagglutination inhibition titers: good correlation but poor agreement. Vaccine 38(11): 2527-2541.
- Janne O Koskinen, Raija Vainionpää, Niko J Meltola, Jori Soukka, Aleksi E Soini, et al. (2007) Rapid Method for Detection of Influenza A and B Virus Antigens by Use of a Two-Photon Excitation Assay Technique and Dry-Chemistry Reagents. journal of clinical microbiology 45(11): 3581-3588.
- Lior Sepunaru, Blake J Plowman, Stanislav V Sokolov, Neil P Young, Richard G Compton (2016) Rapid electrochemical detection of single influenza viruses tagged with silver nanoparticles. Chem Sci 7: 3892-3899.
- Suw Young Ly, Hai Soo Yoo, SeungKyu Chun (2013) Detection of trace metal in distilled alcoholic drinks. Food Chemistry 138: 168-171.
- Suw Young Ly, So Dam Yoo, Seung KyuChun (2012) Detection of Helicobacterpylori DNA in preliminary stage gastric cancer cells. Pathology 44(3): 251-2540.
- Suw Young Ly, Jin Hui Lee, Dong Ho Jung (2010) Radioactive uranium measurement in vivo using a handheld interfaced analyzer. Environmental Toxicology and Chemistry 29(5): 1025-1030.
- Suw Young Ly, Hwa Jin Heo, Min Jung Kim (2010) Real Time Analysis of Neurotransmitters in the Brain Using a Micro-Electrode System. Current Neurovascular Research 7(1): 32-38.
- Suw Young Ly, Dal woong Choi (2013) Implementation of a Biocircuit Implants for Neurotransmitter Release During Neuro Stimulation. Current Neurovascular Research 10(3): 238-246.
- Suw Young Ly, Jung Eun Kim, Woo Yeon Moon (2011) Diagnostic assay of carcinoembryonic antigen tumor markers using afluorine immobilized biosensor with handmade volt ammetric circuit. European Journal of Cancer Prevention 20(1): 58-62.
- Suw Young Ly, Hai Soo Yoo, Joon Young Ahn, kwang hee Nam (2011) Pico molar assay of riboflavin in human urine using voltammetry. Food Chemistry 127(1): 270-274.
- Suw Young Ly, Sung Hoon Choa, MyoungHo Shin, Seong Ryul Kim, Hai Soo Yoo, et al. (2010) Trace uranium assay using infrared photo diode electrodes. Analytical Letters 43(9): 1471-1480.
- Suw Young Ly, Yun Kym Kim (2006) Assay of glucose in urine anddrinking water with voltammetric working sensors of infrared photo diodeelectrode. Sensors and Actuators A: Physical 127(1): 41-48.
- Suw Young Ly, Myoung Ho Shin, Chang Hyun Lee, Jin Hui Lee, Mi Sook Kim, et al. (2013) Detection of trace cobalt ions in in vivo plant cells using a voltammetricinterlocking system. Water Science & Technology 67(6): 1339-1344.
- Suw Young Ly, Dal Woong Choi, DaeHong Kim (2013) Diagnostic Assay of Glucose in Diabetes Patients’ Urine. SENSOR LETTERS 11(10): 1996-1999.
- Suw Young Ly, Hai Soo Yoo, Nam Jeong Kim, Jin Hui Lee, Sung Soo Woo, et al. (2011) Diagnosticassay of trace lead in an ex vivo tissue using a combination electrode. Environmental Toxicology and Pharmacology 31(1): 51-56.
- Suw Young Ly, Lee Kyung, YuJung Choi, Brandon Sangwon Ra (2016) Septicemia Poisoning Detection at Vascular by Copper Immobilized Macro Probe. Sensor Lett 14(1): 93-96.
- Huck Jun Hong, Suw YoungLy (2017) Modified Synthesis of Tetrodo toxinand Novel Toxicity Diagnostic Sensors. Sensor Lett 15(7): 557-564.
- Suw Young Ly, Yeoung Chan Kim, Huck Jun Hong, Jeongha Kim, Kyung Lee (2018) Rapid Voltammetric Diagnosis of Escherichia Coli Contamination in Non-Treated Human Blood Plasma of Healthy and Sepsis-Infected Patients. IEEE Sensors Journal 18(6): 2200-2205.
- (2021) WHO information for the molecular detection of influenza viruses, Annex 1: Conventional RT-PCR protocols. February, p. 7-8.
- Claudia C dos Santos, Barbara A Zehnbauer, Uriel Trahtemberg, John Marshall (2020) Molecular Diagnosis of Coronavirus Disease 2019. Critical Care Explorations 2(9): 1-10.

 Research Article
Research Article