ABSTRACT
Abbreviations: TOF: Tetralogy of Fallot; VSD: Ventricular Septal Defect; RV: Right Ventricular; MAPCAs: Major Aortopulmonary Collateral Arteries; PA: Pulmonary Atresia; CMR: Cardiac Magnetic Resonance; CTA: Computer Tomography Angiography; TTE: Transthoracic Echocardiography; AO: Ascending Aorta
Description
Tetralogy of Fallot (TOF) is one of the most commonly encountered congenital heart malformations, comprising of a ventricular septal defect (VSD), right ventricular (RV) outflow tract obstruction, RV hypertrophy, and overriding aorta [1-3]. Major aortopulmonary collateral arteries (MAPCAs) are frequently found in association with pulmonary atresia with ventricular septal defect (PA/VSD) [4-6] However, some patients with MAPCAs do not have PA/VSD but have a variety of other “atypical” anatomic diagnoses [4]. Our case is a rare mild symptomatic adult noncorrected TOF, with preserved RV function, right aortic arch, and MAPCAs (“classic” thoracic MAPCAs but also abdominal MAPCAs). The anatomy of a complex congenital defect is well illustrated by cardiac magnetic resonance (CMR) and computer tomography angiography (CTA), in order to understand the relative clinical “silence” and to decide the best management.
Learning Outcomes
CMR provides vital details allowing a stepwise approach to correctly and completely define cardiac anomalies of the TOF. Moreover, CTA had already proved to be useful in the assessment of the aortopulmonary collateral vessels, which develop to supply under-perfused areas of the pulmonary bed [7] and pose a unique and challenging problem at the time of surgical repair. Both imaging techniques gives us complementary information about the anatomy and function of the TOF, helping in planning surgical management [8,9].
Patient Presentation
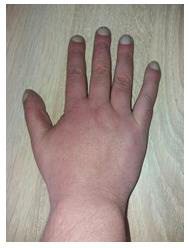
A 44 year-old Caucasian male underwent a routine cardiological examination. His past medical history revealed a minimally symptomatic childhood, describing dyspnea and cyanosis only during intense effort. He had mild kyphosis, asymmetry of the rib cage (with larger intercostal spaces on the left side), a fusion defect in the lower part of the sternum corresponding to the xiphoid process and finger clubbing (Figure 1). An anterior chest systolic murmur suggestive for VSD was detected. He also had important polycythemia. At the time of initial cardiological evaluation the patient was under beta-blocker treatment for tachycardia. We suspected a congenital disease and a transthoracic echocardiography (TTE) was performed [10].
Initial Work Up
ECG revealed sinus tachycardia and right axis deviation. TTE revealed a perimembranous VSD, mild RV hypertrophy and enlargement with preserved LV and RV function. Due to the extremely difficult acoustic window, the patient was referred to another imaging evaluation, in order to assess and characterize the complex anatomy.
Diagnosis and Management
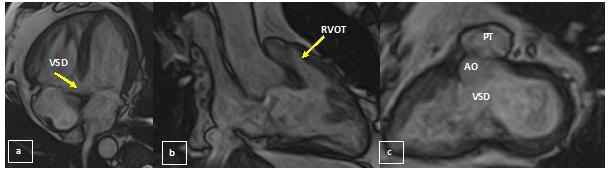
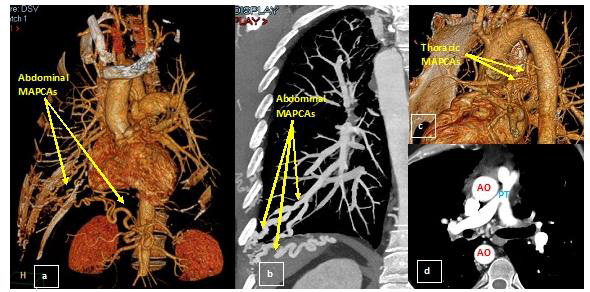
At CMR, steady-state free precession cine sequences (Figure 2) demonstrated normal left ventricular function and revealed a large perimembranous VSD of 15 mm, with the ascending aorta (AO) overriding the interventricular septum and the descending AO to the right of the spine. There was an infundibular stenosis and RV hypertrophy with preserved RV systolic function. A mild tricuspid regurgitation was present. Delayed contrast enhanced images revealed the presence of a nodular contrast enhancement at the level of the posterior septal insertion of the RV free wall. In order to assess major vascular anatomy and collateral vessels for pulmonary supply we performed a CTA examination (Figure 3). Thus, the presence of a right aortic arch was highlighted, AO having normal dimensions. An important periesophageal, peribronchial, intercostal collateral circulation (especially on the right side) was visualized and the right mammary artery was prominent. The main pulmonary artery (PT) had a small caliber (with an ascending aortic/pulmonary trunk ratio of 3.6), associating a small diameter of the origin of both pulmonary arteries.
Figure 2: CMR
(a) Horizontal long axis: Subaortic ventricular septal defect
(b) RVOT: infundibular stenosis (with an RV outflow tract diameter of 13 mm)
(c) Short axis: Subaortic ventricular septal defect and ascending aorta overriding the interventricular septum.
Figure 3: Pulmonary CTA (a) 3D reconstruction (b) MIP reconstruction: abdominal aortopulmonary collateral vessels (c) 3D reconstruction: thoracic aortopulmonary collateral vessels (d) Axial section: the main pulmonary artery (PT) had a small caliber (10 mm, with an ascending aortic / pulmonary trunk ratio of 3.6), associating a small diameter of the origin of both pulmonary arteries (10 mm on the right and 8 mm on the left).
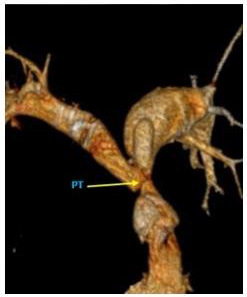
A sacciform ectasia was highlighted on the anterior contour of the proximal portion of the left pulmonary artery (Figure 4). The right subclavian artery presented proximal occlusion, distally loading through fine periesophageal collaterals originating at the level of the descending thoracic aorta and probably also through subclavian steal. We also noticed an aberrant arterial trajectory originating from the abdominal AO (caudal from the emergence of the superior mesenteric artery and cranial to the origin of the right renal artery) with an ascending perihepatic trajectory, then transdiaphragmatic, reaching the right costodiaphragmatic pulmonary angle, from where it continued with multiple smaller tortuous paths; more prominent branches of the right lower pulmonary artery also reached the level of this sinus, these changes being suggestive for MAPCAs.
Figure 4: Pulmonary CTA, 3D reconstructions the main pulmonary artery had a small caliber, associating a small diameter of the origin of both pulmonary arteries. A sacciform ectasia was highlighted on the anterior contour of the proximal portion of the left pulmonary artery.
Follow Up
We decided to refer the patient to a surgeon for step-by-step surgical procedures, in order to prevent sudden cardiac death. Although he is still in good clinical condition, without heart failure and with normal RV function, he has hypoxemia and polycythemia. Sudden death from ventricular arrhythmias has been reported in 1-5% of patients at a later stage of life [5]. However, we considered our case very challenging for the surgical team: a large VSD, small RVOT and PA, also associated with important dilation of left pulmonary artery. Above all, with multiple atypical MAPCAs. Importantly, when the MAPCAs are significant, patients tolerate symptoms well. Over time, they develop signs of heart failure secondary to MAPCAs or pulmonary hypertension secondary to a systemic arterial flow in the pulmonary vascular bed. The surgical approach in these situations is more complex and the repair is usually performed in several stages including: reconstructing the PA centrally through a unifocalization procedure, i.e. isolating the MAPCAs from their systemic origin and connecting them centrally to the pulmonary arteries; releasing the infundibular obstruction by a right ventricle-pulmonary artery (RV-PA) conduit and making them continuous with the unifocalized pulmonary arteries and closing the VSD. Knowledge of the vascular bed and of its supply is therefore essential before any surgery.
Conclusion
Our case is a very unusual combination of an adult noncorrected TOF with a large VSD but non-obstructive stenosis at the RVOT level, small PT and both left and right PA, mild RV hypertrophy and preserved RV function, right aortic arch and distinctive aortopulmonary collaterals (“classic” thoracic aortopulmonary collateral vessels but also abdominal aortopulmonary collateral vessels). These findings explain the clinical silence and long-term survival without major events. This seems to be, to the best of our knowledge, the first case of an adult noncorrected TOF with development of collaterals from the abdominal aorta. Pulmonary CTA, as well as CMR, each provided vital details allowing a gradual approach to describing the patient’s unique anatomy. Both imaging techniques were essential in understanding the clinical silence of the TOF and discovering the adaptive collateral vessels, in order to plan for surgery.
References
- Tomas Dobrocky, Thorsten Klink, Christian Weisstanner, Johannes Heverhagen, Andreas Christe (2014) Imaging findings in uncorrected tetralogy of Fallot and pulmonary atresia with major aortopulmonary collateral arteries and septic embolism. Acta Radiol Short Rep 3(1): 2047981613515211.
- Robin Boyer, Hyung Jin Kim, Rajagopal Krishnan (2020) Management of Unoperated Tetralogy of Fallot in a 59-Year-Old Patient. J Investig Med High Impact Case Rep 8: 2324709620926908.
- Sadashivappa Surhonne Prakash, Rajni Sharma, Navin Agrawal, Somanna Shankar (2014) An extraordinary survival at the age of 67 years in an unoperated case tetralogy of Fallot presenting as systemic hypertension despite RV dysfunction. BMJ Case Rep bcr2013202647.
- William L Patrick, Richard D Mainwaring, Olaf Reinhartz, Rajesh Punn, Theresa Tacy, et al. (2017) Major Aortopulmonary Collateral Arteries With Anatomy Other Than Pulmonary Atresia/Ventricular Septal Defect. Ann Thorac Surg 104(3): 907-916.
- Yvesd'Udekem, NelsonAlphonso, Martin A Nørgaard, Andrew D Cochrane, Leeanne E Grigg, et al. (2005) Pulmonary atresia with ventricular septal defects and major aortopulmonary collateral arteries: Unifocalization brings no long-term benefits. Surgery for congenital heart disease 130(6): 1496-1502.
- Gaia Spaziani, Silvia Favilli, Claudio Fonda, Enrico Chiappa (2013) Giant aorto pulmonary collaterals in pulmonary atresia and ventricular septal defect: long-term survival in unoperated adults. J Cardiovasc Med (Hagerstown) 14(8): 613-615.
- Illena Antonetti, Daniel Lorch, Brian Coe, Thomas S Maxey, Leelakrishna Nallamshetty, et al. (2013) Unrepaired tetralogy of fallot with major aortopulmonary collateral arteries in an adult patient. Congenit Heart Dis 8(1): E24-30.
- Sotiria C Apostolopoulou, Athanassios Manginas, Nikolaos L Kelekis, Michel Noutsias (2019) Cardiovascular imaging approach in pre and postoperative tetralogy of Fallot. BMC Cardiovasc Disord 19(1): 7.
- Tal Geva (2011) Repaired tetralogy of Fallot: the roles of cardiovascular magnetic resonance in evaluating pathophysiology and for pulmonary valve replacement decision support. Journal of Cardiovascular Magnetic Resonance 13(9).
- Shabir Bhimji(2021) Tetralogy of Fallot (TOF) in Adults.

 Mini Review
Mini Review