ABSTRACT
Cynodon dactylon is a perennial herb found in all tropical and subtropical areas, which is a well-known traditional medicine and possessed various pharmacological activities. The present work was aimed to explore the analgesic and anti-inflammatory activities of ethanolic extract of Cynodon dactylon on stressed rodent models. In this study, the anti-inflammatory activity of C. dactylon was determined using the carrageenan induced rat paw edema method, in which the inflammation was generated by intraperitoneal administration of a 1% Carrageenan (0.1 ml/100g) suspension, where ibuprofen was used as standard. After assessing the paw volume, it has been observed that the ethanolic extract of C. dactylon could significantly decrease the formation of edema induced by carrageenan. For the evaluation of both peripheral and central analgesic activity, the acetic acid writhing test as well as tail-flick method were utilized and aspirin was chosen as the reference standard. In the acetic acid writhing test, C. dactylon extract showed a significant reduction in pain against acetic acidinduced abdominal contractions produced by 1% acetic acid (10ml/kg). On the other hand, in the tail-flick method, the central analgesic effect of the C. dactylon extract against heat stress on their tails; was found to be significant at high doses (P<0.01) than low and medium doses. It can, therefore, be inferred that Cynodon dactylon could be utilized as a good alternative therapy in the treatment of inflammation and pain.
Keywords: Cynodon Dactylon; Anti-Inflammatory; Analgesic; Rat; Carrageenan; Acetic Acid Writhing Test
Abbreviations: IASP: International Association for the Study of Pain; NSAIDs: Nonsteroidal Anti-Inflammatory Drugs; COX: Cyclooxygenase; PGs: Prostaglandins
Introduction
Pain is currently defined by the International Association for the Study of Pain (IASP) as “an unpleasant sensory and emotional experience associated with actual or potential tissue damage, or described in terms of such damage.” This definition was recommended by the Subcommittee of Taxonomy and adopted by the IASP Council in 1979 [1]. Pain creates an uncomfortable feeling and is part of human illness. Regardless of whether the pain is acute or chronic, peripheral or central, nociceptive or neuropathic, the underlying cause is inflammation and the inflammatory response. Inflammation is a bodily process that involves ones white blood cells and the substances they produce to protect from bacteria and viruses that have invaded the body. In some cases, such as arthritis, the body’s immune system causes inflammation even though there are no invaders to fight. In more recent times, the term inflammation has been defined as the sequential set of changes that happens in a living tissue when it is damaged, with the condition that the injury is not severe enough to destroy both the structure and the health of the tissue at the same time [2].
Certain drugs, such as paracetamol, acetaminophen, diclofenac, ketorolac, opioid etc., are used as analgesics or anti-inflammatories. A basic analgesic regimen consists of aspirin, codeine, and morphine [3]. Nonsteroidal anti-inflammatory drugs (NSAIDs) exert their therapeutic effects by inhibiting cyclooxygenase (COX), the enzyme responsible for the production of prostaglandins (PGs) [4]. Aspirin, ibuprofen, naproxen, and indomethacin are some of the most often used anti-inflammatory medications. However, these medications have a several serious side effect, including gastrointestinal, cardiovascular, renal, central nervous system, hypersensitivity etc. [5-7]. When opioid medicines are used as analgesics, patients develop an addiction and dependence on them. Opioid administration frequently results in the following side effects: sedation, dizziness, nausea, constipation, vomiting, constipation, tolerance and respiratory depression [8,9]. Adverse reactions to synthetic drugs account for about 8% of all hospital admissions in the United Stated. These toxins cause at least 100,000 individuals to die annually [10]. Apart from the severe side effects, these synthetic medicines are expensive, and the patient may encounter financial difficulties in order to finish the whole treatment regimen.
As a result, the development of powerful analgesic and antiinflammatory medicines with fewer side effects is required. According to medicinal plant researchers, there is a possibility that novel chemical compounds derived from medicinal plants may have therapeutic value. Because of this, scientists are actively searching for alternative or plant-based herbal remedies to cure many ailments, including pain, inflammation, and many other illnesses. For a long time, medicinal herbs have been widely utilized to prevent these side effects. Thus, it is pivotal to seek out therapeutic plants with a variety of chemically active components. The plant is the source of many chemically active compounds, Because of having many chemical constituents like phenols, alkaloids, terpenoids, saponins, glycosides, tannins, flavonoids, resins, polysaccharide, plant lipid, essential oil etc., these medicinal plants can give varieties of pharmacological and therapeutic effects [11,12]. More than only analgesic and anti-inflammatory characteristics, medicinal plants include a wide range of other therapeutic effects such as antiviral, anticancer, antimalarial, and anti-inflammatory capabilities [13]. Again, the concentration of the plant’s chemical components, whether rising or decreasing, may trigger the desired therapeutic effect that can be done through genetic modification of the plant. For example, we can increase the bio-synthesis of secondary metabolites like alkaloid through a reverse genetics process [14]. By increasing alkaloid to a plant, we get antimalarial, antipyretic, anti-smallpox, analgesic and anti-inflammatory effects anti-parasitic activity, antibacterial, antifungal, and antiarrhythmic activity, CNS stimulant activity from a certain plant [15].
Cynodon dactylon belonging to the family Poaceae, commonly called as Bermuda grass, Dhoob, dūrvā grass, ethana grass, dubo, dog’s tooth grass etc. This plant is found in Europe, Africa, Australia, many parts of Asia, New Zealand, Philippines, Indonesia, New Guinea etc. [16]. It contains 28.17% enzymes, 11.79% ash, 10.47% proteins, phenolic phytotoxins viz. ferulic, syringic, paracoumaric, vanillic, para hydroxyl benzoic and orthohydroxy phenyl acetic acid, glycosides and flavonoids vitamin C, β carotene, fats, palmitic acid carotenoids: beta-carotene, neoxanthin, etc [17,18]. This plant has anti-diabetic, anti-arrhythmic, cardiovascular, anti-microbial, anti-ulcer, anti-oxidant, dermatological, anti-nephrolithiasis, CNS, diuretic, analgesic anti-inflammatory, anti-viral, antipyretic, antidiarrrhoeal activity etc. [19-21]. Another study found that, Cynodon dactylon chloroform extract protected against Achinduced bronchospasm which is similar to atropine [22]. Our study aims to determine the analgesic and anti-inflammatory effects of Cynodon dactylon. Further research may assist in the isolation and purification of the active component for analgesic and antiinflammatory properties from this plant which may lead to the discovery of new medicines. In this way, we can offer medications with fewer side effects and at a more affordable price.
Method and Materials
Plant Collection
The fresh Cynodon Dactylon leaf was obtained from the garden of Curzon Hall at University of Dhaka. The specimen was then verified by Department of Pharmacy, University of Dhaka.
Extraction
The moist Cynodon Dactylon leaf was air dried and roughly pulverized. The powdered bark was then extracted for several days with 50% ethanol. Every 3 days, the extract was filtered. The extract was dried at low temperature and pressure in the rotary evaporator. The crude residue was finally employed for necessary pharmacological testing.
Botanical Authentication
According to the rule of national herbarium, the herbarium authorities completed the appropriate measures after we submitted a sample of each section of our plant species. But due to covid pandemic the authority restricted the access of outsiders for a very longtime. That’s why we don’t get botanical authentication (accession number) yet.
Drugs and Chemicals
Alloxan was bought from the sigma company, USA. Acetic acid was procured from Chemical.co.uk. Aspirin as analgesic and ibuprofen as anti-inflammatory are used as standard drug. Aspirin and ibuprofen were collected from Incepta Pharmaceutical Ltd. as a gift sample. We preferred aspirin over the other analgesics like paracetamol as the use of aspirin does not appear to be related with an increased risk of cardiovascular events and skin rash which may be found in case of paracetamol. At the same time ibuprofen was preferred rather than diclofenac or ketorolac as it is an OTC drug having fewer side effects like impaired kidney function, edema etc.
Experimental Animal Procurement, Nursing and Grouping
A total of 500 male rats (120-150 grams) were bought from the Jahangirnagar university, Savar. Each of them was housed in the Institute of Nutrition & Food Science in a climate-controlled environment (temperature 25±3°C, relative humidity 55±5%, and a 12hour light/dark cycle) at University of Dhaka. They have been treated with a normal food system and allowed to drink cleaned water. Prior to the trial, all of the animals were housed in this environment for at least one week. All experimental procedures were carried out in accordance with the Institutional Animals Ethics Committee (IEAC).Four hundred ninety rats were continually dispersed to 49 groups of 10 rats each. In all studies, rats were assigned to groups at random.To increase the validity of the investigatio, we took 10 rats in each group.Because our rats were confined in the animal house throughout thepandemic lockdown, the lab curator was the only one caring for the animals, and we, the scientists, visited the lab twice a week.We, on the other hand, used to keep an eye on the rat every day during the breeding season. There are positive control groups and negative control groups in our study.
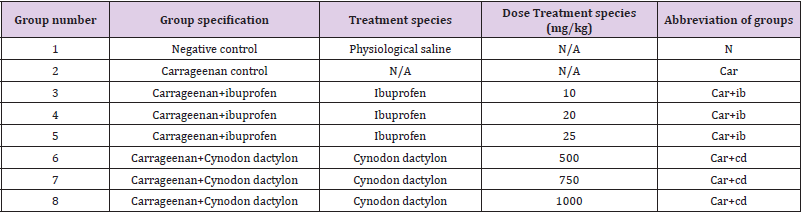
Evaluation of Anti-Inflammatory Activity
(Table 1).
Carrageenan-Induced Acute Inflammatory Model
Carrageenan induced rat hind paw edema was employed as the animal model of acute inflammation in this study. The animals are divided into four groups each having 10 rats. An antiinflammatory test was conducted using a specific type of tool called a plethysmometer. Initially, the volume of each rodent’s paw was measured.Acute inflammation was induced in rats by subplantar injection of 0.1 ml per 100g of 1% carrageenan suspension in the right hind paw. Extract, ibuprofen and distilled water were administered orally 30 minutes prior to the sub plantar injection of carrageenan. Using a plethysmometer, the paw volume was estimated at 0, 20, 40, 60, 80, 100, 120, 140, 160, and 180 minutes following Carrageenan infusion. The rate hindrance of edema was then calculated using the procedure below.

Here,
Vt= volume of animals’ paw after injection
V0=volume of animals’ paw before injection.
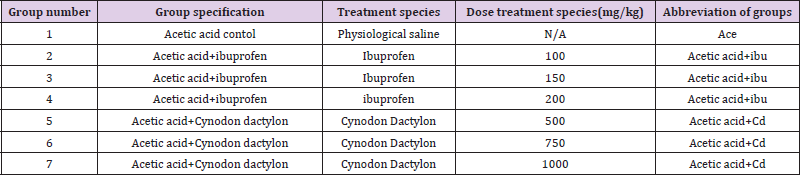
Evaluation of Analgesics Activity
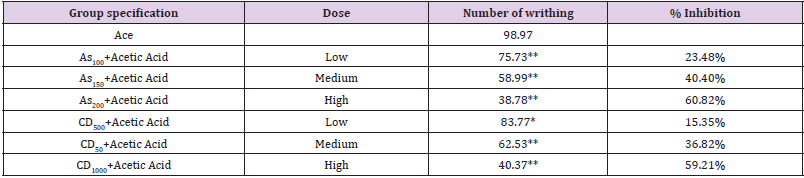
(Table 2) To determine peripheral analgesic activity, a writhing test with acetic acid was used [23,24]. The rats are split into three groups, each with ten rats., 30 minutes prior to the intraperitoneal delivery of acetic acid, plant extract and test medication ibuprofen were given. To induce writhes in rats, an intraperitoneal injection of 1% acidic acid (10ml/kg) was administered. The number of writhes (muscular contraction ions) was counted during a 20-minute period, commencing 5 minutes after the administration of acetic acid. When the number of writhes in each group was compared to the number of writhes in the control group, the percent decrease in writhes counts was determined as follows:

Where, T Control = the mean number of the writhing of each test group.
After a seven-day interval, the extract’s analgesic efficacy is evaluated using the “Tail Flick Method” on the same experiment rat model. By this time, the injected acetic had lost its efficacy.
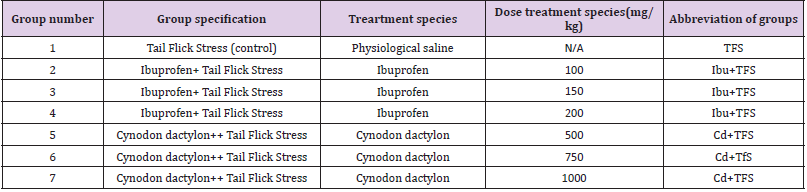
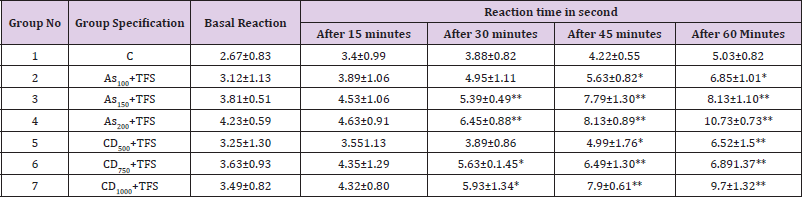
Tail Flick Method
(Table 3) D’Amour and Smith (1941) determined central analgesic activity by assessing drug-induced alterations in the sensitivity of prescreened (reaction time: 2-44 sec) mice to heat stress given to their tails. The animals were divided in the groups showed in the table. Rat reaction latencies are measured using a radiant heat programmed tail-flick analgesiometer (UGO BASILE®, Germany). The device’s nichrome wire was warmed to the proper temperature and maintained with the help of heat controls. The current intensity flowing through the bare nicrome wire was 4 amperes. Radiant heat is applied to the mice’s tails 5cm away from the tip of the tail to cause pain. Time of response for rats or animals treated with a test drug and plant extract was measured. The test was carried out at 0, 0.5, 1, 1.5, 2, 3, 4, 6, and 8 hours after the test drug was used. Following 24 hours, the rats were observed again contemplating remaining activity.
Results
Anti-Inflammatory Response
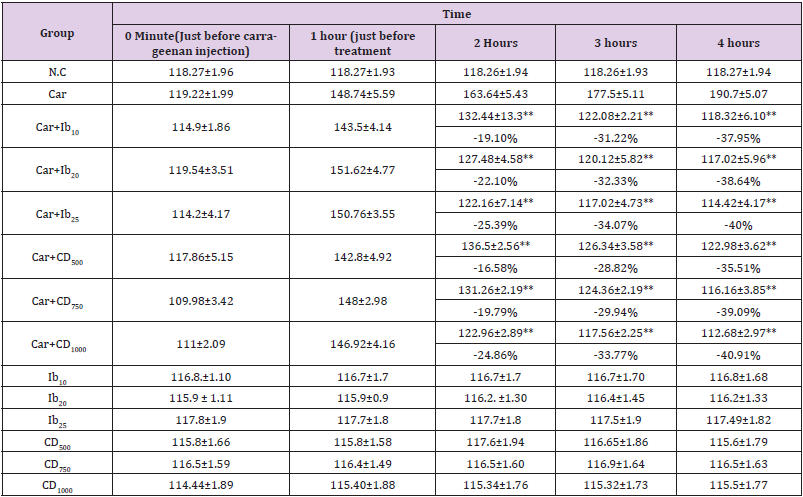
There was a statistically significant difference in edema conditions between the positive control group and the treatment groups. C. dactylon extract, like normal drugs, significantly inhibits the development of edema in low, medium or high dosages considerably (Table 4).
Table 4: Assesment of anti-inflammatory activity of cynodon dactylon extract and ibuprofen through paw oedema test in a rat model.
Note: (*presents the level of significance of result).
Analgesic Activity of Cynodon Dactylon
C. dactylon extract was shown to decrease acetic acid-induced abdominal contractions, but only in a dose-dependent way Table 5. Low dose of extract reduced the pain but medium and high doses cause a significant reduction in pain whereas Aspirin reduced the pain in low, medium and high doses. A high dose of C. dactylon produced statistically significant data(P<0.01) after 45 minutes than the low and medium dose of the extract(P<0.05). On the other hand, the standard drug produced high significant data in both medium and high doses (Table 6).
Table 5: An evalution of analgesic effect of different doses of Cynodon dactylon, Aspirin by acetic acid writhing test.
Note: (*presents the level of significance of result).
Table 6: An evaluation of the analgesic activity of Cynodon dactylon and Aspirin by the tail-flick test method.
Discussion
Anti-Inflammatory Effects
In this study analgesic and anti-inflammatory activity were evaluated to the animal model. There are several techniques available for inflammation measurement, but the most frequent approach is edema development in rat paws where the aqueous and ethanolic extracts of Cynodon Dactylon were evaluated in experimental rat models [25]. Paw edema in rats caused by carrageenan is a common experiment that occurs in a matter of hours and is directly connected to the release of kinins, prostaglandins, cyclooxygenase, histamine, and serotonin [26-28]. Table 4 indicates that low, medium, and high doses, the standard drug, C. dactylon extract, significantly reduces edema formation. This decrease shows that C. dactylon extracts are effective in treating inflammation in the rat model. The reason for giving significant anti-inflammatory effects is for high amount of flavonoids [29,30]. Similar results were found in Acacia catechu, Cassia fistula, Aerva lanata, Sarca asoca, Albizia lebbeck [31-35].
In our investigation, we found that C. dactylon began imparting its anti-inflammatory action one hour after injecting carrageenan. Carrageenan-induced edema, which is thought to be biphasic, has been widely utilized as an experimental animal model for acute inflammation. The carrageenan model’s early phase (1-2 h) is primarily mediated by histamine, serotonin, and enhanced prostaglandin production in the injured tissue surrounds. Prostaglandin release sustains the late phase, which is mediated by bradykinin, leukotrienes, polymorphonuclear cells and prostaglandins generated by tissue macrophages [36,37]. Furthermore, the mechanisms of anti-inflammatory action would be linked with the antiphlogistic activity imparted by tannins. Thus, our extract may exert its anti-inflammatory action through serotonin and histamine inhibition. As a result, the extract may produce anti-inflammatory action by reducing histamine’s pharmacological activity. Furthermore, the extract includes flavonoids, which have the ability to suppress the synthesis of prostaglandins. So, Cynodon dactylon extract may have anti-inflammatory action by blocking histamine and reducing prostaglandin synthesis [38,39].
Analgesic Activity
In our study the analgesic efficacy of C. dactylon was assessed using a tail-flick test as well as an acetic acid writhing test since both procedures are regarded as classic pharmacological models that employ a natural product to evaluate analgesia [40,41]. The acetic acid-induced writhing test is used mostly for medicines that act peripherally [42]. In our investigation, we found differences between the effects of Aspirin and test extract. The low amount of active chemicals contained in the test extract is thought to be the source of the discrepancy between the effects of Aspirin and the test extract. A significant reduction in pain in the treatment groups compared to the positive control group shows that C. dactylon extract has analgesic efficacy. The reasons for giving analgesic activity is for having high amount of alkaloids and flavonoids which are established in previous studies [43,44]. Similar results were found in aqueous fraction of MeOH extract of Carthamus lanatus, Azadiracta indica, Centella asiatica, Hibiscus rosa sinensis Linn, Imperata cylindrica (L.)[45-49].
The central analgesic efficacy of the planting material was investigated using the Medicraft Analgesiometer Mask N (D’Amour and Smith, 1941) by assessing drug-induced changes in pre-screen sensitivity (response time: 2-4 sec) of mice against heat stress on their tails. C.dactylon demonstrated significant antinociceptive activity in the tail-flick test at high doses (1000mg). There is a significant difference between the treatment groups with different dosages and time durations. One possible explanation for this behavior is the extract having lower concentration of active chemicals. The overall improvement in illness state shows that C. dactylon is effective in the tail-flick test. In quantitative research on the screening of phytochemical components, a comprehensive literature research has verified the presence of tannin, flavonoids, gums, alkaloids and carbohydrates. In a prior study, flavonoids were shown to be able to carry out analgesic action through prostaglandins inhibition [50]. Furthermore, alkaloids are well known for their ability to reduce pain perception.
Conclusion
In our study, it has been evidenced that the whole extract of our plant can significantly improve the disturbed pathological state induced by different types of chemical and physical stress. Further meticulous studies regarding the isolation and modification of antiinflammatory and analgesic compounds from whole extracts of C. dactylon may bring about more precise therapeutic constituents in the disease management system.
References
- Raja SN, Carr DB, Cohen M, Finnerup NB, Flor H, et al. (2020) The revised International Association for the Study of Pain definition of pain: concepts, challenges, and compromises. Pain 161(9): 1976-1982.
- Punchard NA, Whelan CJ, Adcock I (2004) The journal of inflammation. J inflamm 1(1): 1.
- Twycross RG (1984) Analgesics. Postgraduate medical journal 60(710): 876-880.
- Munir MA, Enany N, Zhang JM (2007) Nonopioid analgesics. Anesthesiology clinics 25(4): 761-774.
- Ricardo Buenaventura M, Rajive Adlaka M, Nalini Sehgal M (2008) Opioid complications and side effects. Pain physician 11(2): S105-S120.
- Bjarnason I, Hayllar J (1993) Side effects of nonsteroidal anti-inflammatory drugs on the small and large intestine in humans. Gastroenterology 104(6): 1832-1847.
- Lazzaroni M, Bianchi Porro G (2004) Gastrointestinal side‐effects of traditional non‐steroidal anti‐inflammatory drugs and new formulations. Alimentary pharmacology & therapeutics 20: 48-58.
- Vane JR, Botting RM (1998) Anti-inflammatory drugs and their mechanism of action. Inflammation Research 47(2): 78-87.
- Raghavan S, Harvey AD, Humble SR (2011) New opioid side effects and implications for long-term therapy. Trends in Anaesthesia and Critical Care 1(1): 18-21.
- Aye MM, Aung HT, Sein MM, Armijos C (2019) A review on the phytochemistry, medicinal properties and pharmacological activities of 15 selected Myanmar medicinal plants. Molecules 24(2): 293.
- Yang L, Stöckigt J (2010) Trends for diverse production strategies of plant medicinal alkaloids. Natural product reports 27(10): 1469-1479.
- Saxena M, Saxena J, Nema R, Singh D, Gupta A (2013) Phytochemistry of medicinal plants. Journal of pharmacognosy and phytochemistry 1(6): 168-182.
- Harlan JR, De Wet JMJ (1969) Sources of variation in Cynodon dactylon (L). Pers. 1. Crop Science 9(6): 774-778.
- Shendye NV, Gurav SS (2014) Cynodon dactylon: A systemic review of pharmacognosy, phytochemistry and pharmacology. Int J Pharm Pharm Sci 6(8): 7-12.
- Debnath B, Singh WS, Das M, Goswami S, Singh MK, et al. (2018) Role of plant alkaloids on human health: A review of biological activities. Materials today chemistry 9: 56-72.
- Nasri H (2013) Toxicity and safety of medicinal plants. Journal of HerbMed Pharmacology 2(2): 21-22.
- Pengelly A (2021) The constituents of medicinal plants. Cabi.
- Khajavi Rad A, Hajzadeh MAR, Rajaei Z, Sadeghian MH, Hashemi N, et al. (2011) Preventive effect of Cynodon dactylon against ethylene glycol-induced nephrolithiasis in male rats. Avicenna Journal of Phytomedicine 1(1): 14-23.
- Das S, Morya S, Neumann A, Chattu VK (2021) A Review of the Pharmacological and Nutraceutical Properties of Cynodon dactylon. Pharmacognosy Research 13(3): 104-112.
- Kumar S, Shachi K (2020) Hypoglycemic Medicinal Effects of Cynodon Dactylon in Alloxan Induced Diabetic Albino Rats. Adv Endoc and Daib: AEAD, pp. 110.
- Ashokkumar K, Selvaraj K, Muthukrishnan SD (2013) Cynodon dactylon (L.) Pers.: An updated review of its phytochemistry and pharmacology. Journal of Medicinal Plants Research 7(48): 3477-3483.
- Patel MR, Bhalodia YS, Pathak NL, Patel MS, Suthar K, et al. (2013) Study on the mechanism of the bronchodilatory effects of Cynodon dactylon (Linn.) and identification of the active ingredient. Journal of ethnopharmacology 150(3): 946-952.
- RA Devi, SK Tandan, D Kumar, SP Dudhgaonkar, J Lal (2008) Analgesic Activity of Caesalpinia bonducella Flower Extract. Pharm Biol 46(10-11): 668-672.
- S Ahmed, A Naved, RA Khan, S Siddiqui (2015) Analgesic Activities of Methanol Extract of Terminalia chebula Fruit. Pharmacol Amp Pharm 6(6): 547-553.
- Fereidoni M, Ahmadiani A, Semnanian S, Javan M (2000) An accurate and simple method for measurement of paw edema. Journal of Pharmacological and Toxicological Methods 43(1): 11-14.
- Morris CJ (2003) Carrageenan-induced paw edema in the rat and mouse. Inflammation protocols 225: 115-121.
- Guay J, Bateman K, Gordon R, Mancini J, Riendeau D (2004) Carrageenan-induced paw edema in rat elicits a predominant prostaglandin E2 (PGE2) response in the central nervous system associated with the induction of microsomal PGE2 synthase-1. Journal of Biological Chemistry 279(23): 24866-24872.
- Posadas I, Bucci M, Roviezzo F, Rossi A, Parente L, et al. (2004) Carrageenan‐induced mouse paw oedema is biphasic, age‐weight dependent and displays differential nitric oxide cyclooxygenase‐2 expression. British journal of pharmacology 142(2): 331-338.
- Falodun A, Okunrobo LO, Uzoamaka N (2006) Phytochemical screening and anti-inflammatory evaluation of methanolic and aqueous extracts of Euphorbia heterophylla Linn (Euphorbiaceae). African Journal of biotechnology 5(6): 529-531.
- Rathee P, Chaudhary H, Rathee S, Rathee D, Kumar V, et al. (2009) Mechanism of action of flavonoids as anti-inflammatory agents: a review. Inflammation & Allergy-Drug Targets 8(3): 229-235.
- Stohs SJ, Bagchi D (2015) Antioxidant, anti‐inflammatory, and chemoprotective properties of Acacia catechu heartwood extracts. Phytotherapy Research 29(6): 818-824.
- Ilavarasan R, Malika M, Venkataraman S (2005) Anti-inflammatory and antioxidant activities of Cassia fistula Linn bark extracts. African journal of traditional, complementary and alternative medicines 2(1): 70-85.
- Pieczykolan A, Pietrzak W, Gawlik-Dziki U, Nowak R (2021) Antioxidant, Anti-Inflammatory, and Anti-Diabetic Activity of Phenolic Acids Fractions Obtained from Aerva lanata (L.) Juss. Molecules 26(12): 3486.
- Ahmad F, Misra L, Tewari R, Gupta P, Mishra P, et al. (2016) Anti-inflammatory flavanol glycosides from Saraca asoca bark. Natural product research 30(4): 489-492.
- Saha A, Ahmed M (2009) The analgesic and anti-inflammatory activities of the extract of Albizia lebbeck in animal model. Pakistan journal of pharmaceutical sciences 22(1): 74-77.
- Gupta M, Mazumder UK, Gomathi P, Selvan VT (2006) Antiinflammatory evaluation of leaves of Plumeria acuminata. BMC Complementary and alternative medicine 6(1): 1-6.
- Antonio MA, Brito AS (1998) Oral anti-inflammatory and anti-ulcerogenic activities of a hydroalcoholic extract and partitioned fractions of Turnera ulmifolia (Turneraceae). Journal of ethnopharmacology 61(3): 215-228.
- Yogesh HS, Kichadi SC, Muchandi IS, Gopalakrishna B (2013) Evaluation of Anti-Inflammatory activity of Cynodon dactylon On carrageenan induced paw edema in rats 4(2): 151-154.
- Sindhu G, Ratheesh M, Shyni GL, Helen A (2009) Inhibitory effects of Cynodon dactylon on inflammation and oxidative stress in adjuvant treated rats. Immunopharmacology and Immunotoxicology 31(4): 647-653.
- Hayes RL, Bennett GJ, Newlon PG, Mayer DJ (1978) Behavioral and physiological studies of non-narcotic analgesia in the rat elicited by certain environmental stimuli. Brain research 155(1): 69-90.
- Saha A, Masud MA, Bachar SC, Kundu JK, Datta BK, et al. (2007) The analgesic and anti-inflammatory activities of the extracts of Phyllanthus reticulatus. in mice model. Pharmaceutical Biology 45(5): 355-359.
- Lu TC, Ko YZ, Huang HW, Hung YC, Lin YC, et al. (2007) Analgesic and anti-inflammatory activities of aqueous extract from Glycine tomentella root in mice. Journal of ethnopharmacology 113(1): 142-148.
- Abdullahi MH, Anuka JA, Yaro AH, Musa A (2014) Analgesic and anti-inflammatory effects of aqueous leafextract of Combretum micranthumg. Don (Combretaceae). Bayero Journal of Pure and Applied Sciences 7(2): 78-82.
- Uche FI, Aprioku JS (2008) The Phytochemical Constituents, Analgesic and Anti-inflammatory effects of methanol extract of Jatropha curcas leaves in Mice and Wister albino rats. Journal of Applied Sciences and Environmental Management 12(4).
- Bocheva A, Mikhova B, Taskova R, Mitova M, Duddeck H (2003) Antiinflammatory and analgesic effects of Carthamus lanatus aerial parts. Fitoterapia 74(6): 559-563.
- Emran TB, Nasir Uddin MM, Rahman A, Uddin Z, Islam M (2015) Phytochemical, antimicrobial, cytotoxic, analgesic and anti-inflammatory properties of Azadirachta indica: A therapeutic study. J Bioanal Biomed.
- Qureshi M, Jahan N, Muhammad S, Mohani N, Wazir A, et al. (2015) Evaluation of neuropharmacological, analgesic and anti-inflammatory effects of the extract of Centella asiatica (Gotu kola) in mice. African Journal of Pharmacy and Pharmacology 9(41): 995-1001.
- Sawarkar A, Jangde CR, Thakre PD, Kadoo R, Shelu S (2009) Analgesic Activity of Hibiscus rosa sinensis Linn in Rat. Veterinary world 2(9): 353-354.
- Razafindrakoto ZR, Tombozara N, Donno D, Gamba G, Nalimanana NR, et al. (2021) Antioxidant, analgesic, anti-inflammatory and antipyretic properties, and toxicity studies of the aerial parts of Imperata cylindrica (L.) Beauv. South African Journal of Botany 142(40): 222-229.
- Ganguly A, Al Mahmud Z, Kumar Saha S, Abdur Rahman SM (2016) Evaluation of antinociceptive and antidiarrhoeal properties of Manilkara zapota leaves in Swiss albino mice. Pharmaceutical biology 54(8): 1413-1419.

 Research Article
Research Article