ABSTRACT
Background: Cholelithiasis is associated with increased cholesterol and iron deficiency anaemia. Evaluation of levels of biliary cholesterol and ferritin and expression of Apo B-100 gene in Cholelithiasis was done.
Methods: 40 patients of cholelithiasis were included for measurement of serum and biliary cholesterol and ferritin by ELISA. Expression of Apo B-100 gene was also evaluated in their blood along with 40 healthy volunteers as controls for gene expression.
Results: 82.5% patients had hypercholesterolemia (Mean = 302.46mg/dL) and 80% had low HDL (Mean = 40.70mg/dL). Mean biliary cholesterol was 121.29mg/dL. Weak negative correlation seen between serum and biliary cholesterol(p=0.592) and moderate positive correlation between HDL and biliary cholesterol(p=0.001). 29/40 patients were anaemic with 25/40 having normal serum ferritin (Mean = 12.85ng/ mL). Mean biliary ferritin was 13.65ng/mL. Weak negative correlation seen between serum and biliary ferritin(p=0.992) and moderate positive correlation between biliary cholesterol and ferritin (p<0.001). 1.001 fold change upregulation in Apo B-100 gene was observed in patients compared to the controls.
Conclusion: HDL may be a better indicator than serum cholesterol for cholelithiasis. Ferritin plays an important role in the pathogenesis of cholelithiasis. Relationship between iron metabolism, lipid metabolism and cholelithiasis exists even at the genetic level.
Introduction
Gall stone disease is a common disorder around the world. Choleliths are bile precipitates resulting from abnormalities in the metabolism of cholesterol, bilirubin and bile acids. [1]. The prevalence of cholelithiasis in India ranges from 2-29%, being higher in North Indians and females [1-3]. Gall Stones are classified according to their chemical composition into Cholesterol stones, Mixed stones and Pigment stones [4]. In western populations, stones that are rich in cholesterol are the predominant type (80%) but lower frequencies of cholesterol stones and higher frequencies of pigment stones are seen in many parts of Asia [5]. The pathogenesis of cholesterol stones primarily involves supersaturation of bile with cholesterol accentuated by accelerated nucleation and impaired gallbladder emptying being other factors. Relative concentration of cholesterol, bile salts and phospholipids in GB bile determines precipitation [5]. Anaemia due to iron deficiency is associated with gall stone disease. Serum ferritin, a marker of iron stores, exhibits low levels in iron deficient patients. In a study done on patients with cholelithiasis by Aggarwal et al, in 2019, it was seen that serum ferritin levels were lower in cases of cholelithiasis as compared to those in the normal population [6]. Ferritin is also released in bile in hemochromatosis.
In this study biliary iron concentration (per millimole of bile acid), was increased about twofold (p < 0.05) and biliary ferritin concentration about fivefold (p < 0.001) [7]. The levels of biliary ferritin in gallstone disease have not been studied so far. Genetic variation in cholesterol metabolism can be brought about by different isoforms of lipid transport and receptor molecules in the general population. Apo E and apo B-100 are prominent transporters that exist in polymorphic states in humans whose certain isoforms help in better uptake and delivery of cholesterol [8]. Apo B-100 serves as a ligand for receptor-mediated endocytosis of LDL. The gene is located on chromosome 2. High frequency of X+ allele in apo B-100 genes has been reported in gallstone patients from China [9]. Heterozygosity (X+/X-) of apo B-100 may be associated with gallstone disease and gallbladder cancer in India [10,11]. In a study in transgenic rabbits, it was found that Apo B-100 expression is associated with higher serum cholesterol and triglyceride levels [12]. In another study, rats fed on iron rich diet had a higher expression of Apo B-100 gene and had higher serum cholesterol and triglyceride levels [13]. Hence this study was undertaken to determine the levels of biliary ferritin and biliary cholesterol in patients of cholelithiasis and to see the association of expression of Apo-B gene in them.
Methodology
This observational cross-sectional study was conducted in the Department of Surgery and the Department of Biochemistry at UCMS & GTB Hospital, Delhi from November 2018 to March 2020 with a sample size of 40 cases and equal number of controls for gene purposes. The research committee of the institute approved the study protocol. All consenting patients, except those with coexisting systemic and inflammatory illness and immunocompromise, diagnosed with symptomatic cholelithiasis willing to undergo laparoscopic/open cholecystectomy were included. For Apo B-100 expression, an equal number of healthy volunteers were included after ultrasound to exclude cholelithiasis in them. All patients of cholelithiasis and controls underwent history taking, radiological and biochemical investigations. A fasting blood sample of patients was taken for hematological evaluation, lipid profile, serum ferritin, and Apo B-100 gene expression. In the controls, only Apo B-100 gene expression was measured. Bile was obtained from the gall bladder of patients immediately after cholecystectomy for evaluation of biliary cholesterol and ferritin levels. Cholesterol and ferritin levels in both serum and bile were measured by ELISA. The reference range for serum ferritin was taken to be 29-248 ng/ml for males and 10-150 ng/ml for females [14].
Serum cholesterol levels less than 200 mg/dl were considered
desirable, 200-239 mg/dl as borderline high and >240 mg/dl as
high. Gene Expression comprised of 3 steps:
a) Extraction of RNA,
b) Synthesis of complementary DNA (cDNA) and
c) Quantification of Apo B-100 gene expression by
quantitative Real Time PCR.
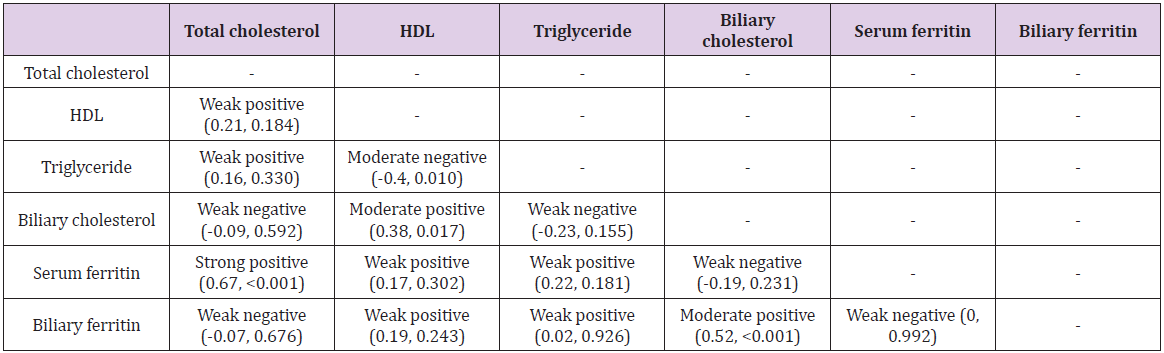
Statistical analysis was done using SPSS version 20.0. p-values were calculated using student t-test for normally distributed data. For non-normal distribution, non-parametric tests were used. Correlation was calculated by using Pearson test. Pearson’s coefficient (rho) values between 0 and 0.3 (0 and -0.3) indicate a weak positive (negative) linear relationship via a shaky linear rule. Values between 0.3 and 0.7 (-0.3 and -0.7) indicate a moderate positive (negative) linear relationship.
Results
Patient Profile
The mean age of the 40 patients included in the study was 34.65 with most patients belonging to the age group of 21-30 years (16/40). The mean age of controls was 34.20 ± 10.38 years and did not vary significantly. (p = 0.897). Females formed the majority of participants in both the groups, 37(92.5%) in cases and 33(82.5%) in controls. The mean BMI (Kg/m2) was 22.28 ± 2.81. with the majority (65%) falling in the normal range. The duration of illness ranged from 3 months to 60 months with a mean of 17.2 months.
Haemoglobin
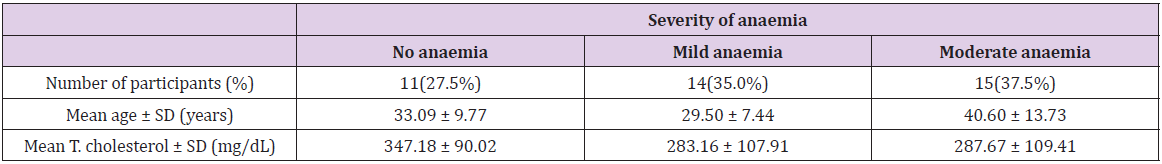
The mean haemoglobin level of the patients was 11.46 g/dl. On the basis of WHO classification, patients were divided into non anaemic, mildly anaemic and moderately anaemic. Most (72.5%) patients were anaemic (Table 1). The mean age of patients with moderate anaemia was significantly higher than those with no or mild anaemia (p=0.045) (Table 1).
Lipid Profile
Out of the 40 patients, 33 (82.5%) had raised serum cholesterol levels. The mean of T. Cholesterol (mg/dL) was 302.46. The mean HDL level of the patients was 40.70 mg/dl. Based on the American Heart Association criteria, 32 patients had low HDL levels (i.e. <40 for males and <50 for females) [15]. The mean triglyceride level of the patients was 122.53 mg/dl with 11/40 having raised triglyceride levels (>150 mg/dl) [16]. There was a significant moderate negative correlation between Triglycerides (mg/dL) and HDL (mg/dL) (rho = -0.4, p = 0.010).
T. Cholesterol and Anaemia
The mean total cholesterol levels in the patients grouped on basis of hemoglobin levels did not vary significantly (p=0.222), though patients without anaemia had a higher mean serum cholesterol level than those with anaemia (Table 1).
Biliary Cholesterol
The Biliary cholesterol (mg/dL) levels ranged from 17.01 to 311.68 with a mean of 121.29 mg/dL.
1. There was a significant moderate positive correlation between
HDL (mg/dL) and biliary cholesterol (mg/dL) (rho = 0.38, p =
0.017). For every 1 unit increase in HDL (mg/dL), the biliary
cholesterol (mg/dL) was seen to increase by 3.18 units.
2. There was a significant moderate positive correlation between
duration of illness (months) and Biliary cholesterol (mg/dL)
(rho = 0.49, p = 0.001).For every 1 unit increase in duration of
illness (months), the biliary cholesterol (mg/dL) was seen to
increase by 2.22 units.
Serum Ferritin
The mean serum ferritin (ng/mL) of the patients was 12.85. Out of the 40 patients enrolled, 25 (62.5%) patients had serum ferritin within normal range and 15(37.5%) patients had low serum ferritin levels [14].
1. Serum ferritin and anemia
There was a weak positive correlation between hemoglobin
(g/dL) and serum ferritin (ng/mL), and this correlation was not
statistically significant (rho = 0.07, p = 0.673). There was a visible
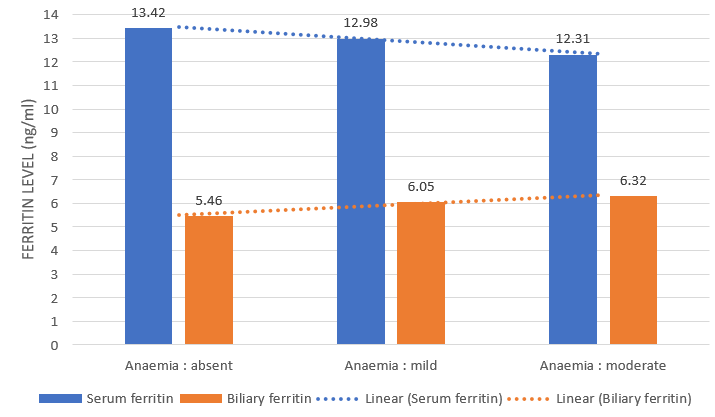
decrease in numerical value of serum ferritin as severity of anaemia
increased but this was not statistically significant (p = 0.967)
(Figure 1).
2. There was a significant strong positive correlation between T.
Cholesterol (mg/dL) and serum ferritin (ng/mL) (rho = 0.67, p
= <0.001). For every 1 unit increase in T. Cholesterol (mg/dL),
the serum ferritin (ng/mL) increases by 0.04 units.
3. There was a weak negative correlation between biliary
cholesterol (mg/dL) and serum ferritin (ng/mL), and this
correlation was not statistically significant (rho = -0.19,
p = 0.231). Though insignificant (p=0.783), higher biliary
cholesterol level was noted with low serum ferritin level
(mean biliary cholesterol = 123.16 mg/dL) in comparison to
patients with normal serum ferritin (120.16 mg.dL).
Biliary Ferritin
The mean biliary ferritin (ng/mL) of the 40 patients was 5.99 with a minimum value of 1.41 ng/mL and a maximum value of 13.65 ng/mL.
1. There was a weak negative correlation between haemoglobin (g/dL) and biliary ferritin (ng/mL), and this correlation was not statistically significant (rho = -0.19, p = 0.249).
Biliary ferritin levels were higher in patients with moderate anaemia as compared to mildly and non-anaemic patients but this trend was not statistically significant (p=0.456). While the mean values of serum ferritin were seen to decrease with increasing severity of anaemia, the mean values of biliary ferritin were seen to increase (Figure 1).
2. There was a weak negative correlation between serum ferritin (ng/mL) and biliary ferritin (ng/mL), and this was not statistically significant (rho = 0, p = 0.992). Mean serum ferritin (12.85 ng/mL) was found to be twice the mean biliary ferritin (5.99 mg/dL).
3. There was a moderate positive correlation between biliary cholesterol (mg/dL) and biliary ferritin (ng/mL), and this correlation was statistically significant (rho = 0.52, p = <0.001). For every 1 unit increase in biliary cholesterol (mg/dL), the biliary ferritin (ng/mL) increases by 0.03 units.
Apo B-100 Gene
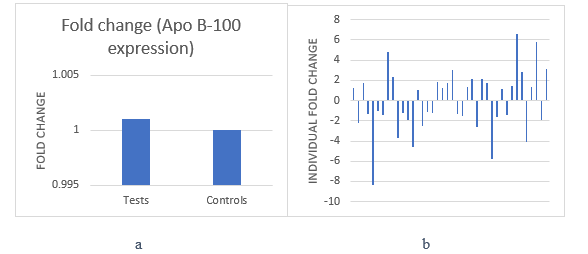
1. On taking all 40 patients as cases and all 40 healthy volunteers
as a control group, a 1.001 fold change upregulation of Apo
B-100 gene was observed. This difference was not significant
(p=0.740) (Figure 2a).
2. On taking the mean delta ct value of controls and evaluating
the expression of Apo B-100 gene in each patient against it,
the result showed that the gene was upregulated in 20 patients
and downregulated in 20 patients exhibiting equal distribution
on the individual level (Figure 2b).
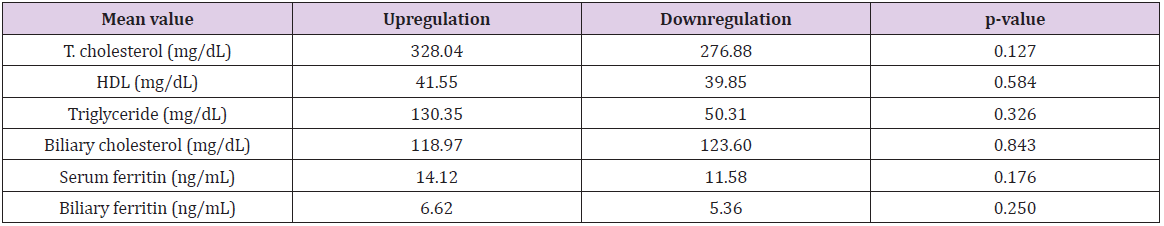
3. The mean T. Cholesterol (mg/dL) in patients with upregulation
of Apo B-100 gene was 328.04 while that in patients with
downregulation was 276.88 (Table 2). 18/20 (90%) patients
with upregulation of Apo B-100 gene had high cholesterol
levels and 12/20 (60%) with downregulation had high
cholesterol levels.
Figure 2: a. Fold change (Apo B-100 gene expression).
b. Individual fold change (Apo B-100 gene expression) of all patients.
Discussion
Cholelithiasis is a multifactorial disease having a complex interplay of metabolic, genetic and environmental factors that affect the disease process. In this study, the mean age of the 40 patients included was 34.65 years, with most patients in their 3rd and 4th decades of life. This age distribution was similar to the study conducted by Prakash, et al. [3] and Kanth, et al. [2]. Most other studies which aimed at studying the epidemiology of cholelithiasis found the disease to be more common in a relatively higher age group [1,17,18]. We also found the mean age to be significantly higher in the moderately anaemic group suggesting that anaemia increases with increasing age. Since anaemia and increasing age have both been described as independent risk factors for cholelithiasis [1,6], an additive effect of both seems to be at work in causation of the disease. A majority of the patients in this study were females (37/40, 92.5%). It is a well-established fact in literature that cholelithiasis occurs more commonly in the female population. Most of the patients in our study (26/40, 65%) had a BMI in the normal range suggesting that cholelithiasis is not just a disease of the obese; also seen by Kharga, et al. [19].
Lipid Profile
As found by Miquel, et al. [20] and Loria, et al. [21], in this study also, a majority (75%) of patients had raised total cholesterol and 7.5% patients had borderline raised cholesterol. Additionally, majority (32/40, 80%) of the patients had low HDL levels while only 11 out of the 40 patients (27.5%) had raised triglyceride levels. We found a significant negative correlation (rho = -0.4, p = 0.010) between HDL and triglyceride levels indicating an increase in triglyceride levels as HDL levels fall. Thijs, et al. [22] and Hayat, et al. [23] described an association of low HDL and raised triglyceride levels with gallstone disease but did not find any significant association of serum cholesterol levels with cholelithiasis. Alexander, et al. [24] conducted a study on 80 patients of cholelithiasis and found that only 12 (15%) patients had hypercholesterolemia while 79 (98.75%) patients had low HDL levels, and 23 (28.7%) patients had raised triglyceride levels. In our study, we found raised cholesterol levels (82.5%) and low HDL levels (80%) in a majority of the patients.
These findings lead us to believe that while serum cholesterol plays a role in pathogenesis of gallstone disease, low HDL levels may be a more relevant indicator for the disease. Da Luz, et al. observed in patients of coronary artery disease that the ratio of TG/ HDL and T. cholesterol/HDL are better indicators of disease than individual lipid values in serum [25]. Jiang, et al. found a strong positive association of CAD with cholelithiasis [26]. In the present study, we also found that as serum HDL levels decreased, there was an increase in triglyceride levels. Based on these findings we would like to postulate that it is the ratio of lipid parameters and not the individual levels of each parameter that may serve as better indicators for cholelithiasis. The alterations in ratios can identify individuals prone to cholelithiasis and an attempt can be made at disease prevention through lifestyle modification and management of dyslipidemia.
Biliary Cholesterol
In our study, there was a weakly negative correlation between serum and biliary cholesterol (rho = -0.09) and this was not statistically significant (p=0.592, (Table 3)); in contrast to Kanwar, et al. [27] who found a positive correlation between serum cholesterol and biliary cholesterol (rho = 0.61, p=0.001). We expected higher biliary cholesterol levels based on the prevalent concept of cholesterol supersaturation leading to gallstone formation, but our findings were contrary to this. It has been shown by Nagi and Arora [28] that the cholesterol stones (13.5%) are much less common in North Indian population than mixed stones (80.7%). In a study conducted by Chandran, et al. [29], amongst 200 patients of cholelithiasis, 52 (26%) had cholesterol stones, 76 (38%) had mixed stones and 72 (36%) had pigment stones. Such high proportion of pigment and mixed stones may be able to explain the lower levels of biliary cholesterol in our population as pigment stones are known to mainly form due to unconjugated bilirubin [30]. Since these studies had a small sample size, further evaluation with larger sample size along with stone analysis is warranted. We also found a significant moderate positive correlation between HDL and biliary cholesterol (rho = 0.38, p = 0.017, (Table 3)). HDL carries cholesterol in circulation and transports excess peripheral cholesterol to the liver for secretion into bile.
This process is termed reverse cholesterol transport. Biliary cholesterol can be derived from plasma lipoproteins like HDL and LDL or it can be synthesized in the liver. The more relevant source of cholesterol secreted into the bile is cholesterol derived from plasma lipoproteins. Of these, high density lipoprotein (HDL) appears to be the preferential contributor for cholesterol secreted into bile [31,32]. We found a significant moderate positive correlation between duration of illness and biliary cholesterol (rho = 0.49, p = 0.001). Supersaturation of bile with cholesterol is an ongoing process and does not stop after stone formation with progressively increasing biliary cholesterol levels over the course of time [33,34]. This also corresponds to the fact that increased chances of gallstone development occur as time progresses in predisposed individuals.
Serum Ferritin
In the present study, serum ferritin showed a numerical decrease with increase in severity of anaemia (p = 0.967 (Figure 1)). A significant, strong positive correlation was seen between T. cholesterol and serum ferritin (rho = 0.67, p = <0.001, (Table 3)). A similar significant positive correlation was also found by Kim, et al. [35]. While we found no significant association between serum cholesterol and anemia, the mean serum cholesterol level in non anaemics (347.18 mg/dL) was higher than that found in anaemics (285.41 mg/dL). This was similar to the findings of Chowta et al. [36] and Choi et al. [37] who studied the relationship of anaemia with lipid profile and Kumar, et al. [38] who also found lower serum cholesterol levels in anaemic patients of cholelithiasis. This was explainable through multiple factors like increased erythropoiesis requiring moe cholesterol, plasma dilution and increased uptake of cholesterol by the reticuloendothelial system in anaemics [36]. Thus, anaemics tend to have lower ferritin levels and lower cholesterol levels, supporting the findings of the present study. Iron deficiency anaemia, low serum ferritin levels and raised serum cholesterol levels were expected to be associated with cholelithiasis [6,39,40] We also found that as serum cholesterol levels increased, there was an increase in serum ferritin levels.
This contradiction suggests that there is a complex interplay of ferritin, cholesterol and anaemia in cholelithiasis and cant be simply explained by the individual levels of these factors. There was a higher biliary cholesterol level in patients with low serum ferritin than in patients with normal serum ferritin, though not significant (p=0.783), similar to a study conducted by Sahu, et al. [41]. Iron deficiency causes altered motility of gall bladder leading to biliary stasis leading to an increase in cholesterol saturation of bile [42,43].
Biliary Ferritin
There was a weak negative correlation between serum ferritin and biliary ferritin (rho = 0, p = 0.992, (Table 3)). While the mean values of serum ferritin were seen to decrease with increasing severity of anaemia, the mean values of biliary ferritin were seen to increase. There is a lack of adequate literature available on excretion of ferritin in bile. We could not find any studies which studied the role of biliary ferritin in cholelithiasis. However, Hultcrantz, et al. [7], in 1989, conducted a study in 10 patients of idiopathic hemochromatosis and 2 groups of controls (14 patients of gallstone disease and 16 healthy individuals). They found that hepatic iron concentration was increased 8 fold, biliary iron concentration was increased twofold and biliary ferritin concentration was increased about fivefold in patients of hemochromatosis as compared to the controls. They found higher levels of biliary ferritin in gallstone patients than in healthy individuals. Hultcrantz, et al. also concluded that hepatocytes secrete ferritin into bile since very little ferritin was detected in bile duct cells on electron microscopy making them an unlikely source of biliary ferritin. Hepatocyte excretion of ferritin into bile has also been established in animal models earlier [44]. Verma, et al. [45] found that the levels of biliary iron were higher than those of serum iron in cases of cholelithiasis as well as controls.
A significantly higher amount of iron in pigment stones than in cholesterol stones was also found. Chandran, et al. [29] found a significantly higher amount of iron in mixed stones than in pigment and cholesterol stones. While the amount of iron was higher in pigment stones than cholesterol stones, this difference was not significant. An increase in biliary ferritin as anaemia increases and relatively lower levels of biliary cholesterol suggest a more frequent occurrence of pigment and mixed stones in our population. Since we found an increased biliary ferritin with increase in anaemia (which is an iron deficient state) in patients of cholelithiasis, it suggests that the relationship between cholelithiasis and iron deficiency anaemia is more complex than thought previously. Ferritin may have a larger role to play in the pathogenesis of cholelithiasis than to just be a marker of iron deficiency anaemia and it is not just a decrease in gallbladder motility and resulting biliary stasis due to the deficiency of iron which cholelithiasis [46]. It is possible that the biliary route of excretion of ferritin is an incidental finding with no serious implications on gallstone formation. The concentration of ferritin in bile may also be due to fluid absorption from the gall bladder leading to concentration of its contents along with biliary stasis secondary to iron deficiency.
Based on our findings, we would like to hypothesize that ferritin gets concentrated in bile in the gall bladder after excretion from hepatocytes and delivers iron into the bile which will play a role in the formation of gallstones. In our study, there was a significant moderate positive correlation between biliary cholesterol and biliary ferritin (rho = 0.52, p = <0.001, (Table 3)). It is known that cations in bile such as iron, calcium, copper and zinc promote cholesterol crystallization [45]. We would like to postulate that in patients with cholelithiasis, with the biliary cholesterol increase, there is a tendency for these cations to increase as well and lead to crystallization of cholesterol and formation of gallstones. An increase in biliary ferritin levels suggests its role in delivering iron which serves as a cation in the biliary cholesterol rich bile leading to cholelithiasis.
Apo B-100 Gene
On taking all 40 patients as cases and all 40 healthy volunteers as a control group, a 1.001 fold change upregulation of Apo B-100 gene was observed (p=0.740). On assessing individual cases, equal distribution of up and downregulation in patients of cholelithiasis was seen (Figure 2b). In 2005, Wang, et al. assessed intestinal cells of mice and found that absence of expression of intestinal Apo B-48, but not Apo B-100, reduces biliary cholesterol secretion and cholelithogenesis, possibly by decreasing intestinal absorption and hepatic bioavailability. While the findings of Apo B-100 expression were similar to the present study, they studied the gene expression in intestinal cells of mice while this was studied in blood from humans in our study. Since we found a relatively higher expression of Apo B-100 in the patient group, though not statistically significant, it suggests Apo B-100 can have a role in etiology of cholelithiasis.
Apo B-100 and Lipid Profile: In our study we found that the levels of total cholesterol and serum triglyceride levels were much higher in patients with upregulation of Apo B-100 gene than patients who showed downregulation (Table 2). Even though our findings were not significant, they were in concordance with the findings of Fan, et al. [12], who found a threefold increase in levels of serum triglyceride and serum cholesterol levels in the transgenic rabbits with human Apo B-100 gene as compared to the non-transgenic controls. 90% patients with upregulation of the gene had raised serum cholesterol levels, while only 60% in the downregulation group had raised serum cholesterol levels. While this difference is not significant (p value = 0.081), it points to a higher frequency of raised cholesterol levels in the upregulation group than in the downregulation group and justifies the association of raised serum cholesterol levels with increased incidence of cholelithiasis. Since Apo B-100 upregulation is associated with higher cholesterol and triglyceride levels, assessment of gene expression may predict patients prone to develop hypercholesterolemia and hypertriglyceridemia and further, an increased risk of gallstone formation.
Apo B-100 and Ferritin: In our study we found higher levels of serum ferritin and biliary ferritin in patients with upregulation of Apo B-100 gene than in patients with downregulation (Table 2). Silva, et al. [13] studied the effects of dietary iron on lipid metabolism in rats. They found that rats on iron dextran diet had a higher expression of Apo B-100 and higher levels of serum cholesterol and triglycerides, than rats who did not have iron in their diet. Since ferritin stores iron, and higher levels of serum and biliary ferritin were found in patients with upregulation of Apo B-100 gene, it is possible that the higher levels of ferritin led to the upregulation of Apo B-100 gene which further led to an increase in serum cholesterol and triglyceride levels leading to an enhanced risk of cholelilthiasis.
Conclusion
Findings in the present study indicate that elevated serum cholesterol and anaemia play a role in the pathogenesis of cholelithiasis. HDL is a major source of cholesterol in bile. HDL levels and ratios of lipid parameters may be better indicators of cholelithiasis. We found a positive correlation between biliary cholesterol and biliary ferritin which seemed to correspond with a positive correlation between serum cholesterol and serum ferritin. We also found an increase in levels of biliary ferritin with increased severity of anaemia in patients of cholelithiasis. These findings suggest a role of ferritin in delivering iron to bile leading to an increased risk of gallstone formation. The levels of biliary cholesterol were relatively less and biliary ferritin levels increased with increase in anaemia. Iron levels are known to be higher in pigment stones. Thus, we can conclude this to be the reason for pigment stones being more common in our study population. We found a net upregulation of Apo B-100 gene in patients of cholelithiasis; also, serum cholesterol and triglyceride levels in Apo B-100 upregulated patients were raised, suggesting a possible role of Apo B-100 in cholelithiasis. The higher levels of serum and biliary ferritin could have led to the upregulation of Apo B-100 gene. This relationship between iron metabolism, lipid metabolism and cholelithiasis, which exists even at the genetic level requires further studies for evaluation.
References
- Bansal A, Akhtar M, Bansal A K (2016) A clinical study: prevalence and management of cholelithiasis. Int Surg J 1(3): 134-139.
- Kanth S, Verma A, Madan A (2017) Cholelithiasis in rural population of Haryana. PANACEA J Med Sci 7(2): 92-94.
- Prakash C P, Toppo S, Pratap V (2016) Prevalence and Management of Cholelithiasis in East India. IOSR J Dent Med Sci 15(12): 34-37.
- Gaharwar A (2013) Factors Favouring Cholelithiasis in North Indian Population.
- Roberts Thomson I C, Butler W J (2005) Polymorphism and gallstones. J Gastroenterol Hepatol 20(2): 319-320.
- Aggarwal D D, Agrawal D V, B D Banerjee (2020) EVALUATION OF SERUM FERRITIN LEVEL AND IRON REGULATORY PROTEIN-1 GENE EXPRESSION IN PATIENTS WITH CHOLELITHIASIS. Glob J Res Anal 8(12).
- Hultcrantz R, Angelin B, Björn Rasmussen E, Ewerth S, Einarsson K, et al. (1989) Biliary excretion of iron and ferritin in idiopathic hemochromatosis. Gastroenterology 96(6): 1539-1545.
- Ko C W, Beresford S A A, Alderman B, Jarvik G P, Schulte S J, et al. (2000) Apolipoprotein E genotype and the risk of gallbladder disease in pregnancy. Hepatology 31(1): 18-23.
- Han T, Jiang Z, Suo G, Zhang S (2000) Apolipoprotein B-100 gene Xba I polymorphism and cholesterol gallstone disease. Clin Genet 57(4): 304-308.
- Mittal B, Mittal R (2002) Genetics of gallstone disease. J Postgrad Med 48(2): 149.
- Mahendra Kumar S, Udai Bhan P, Uday Chand G, Itha Srivenu, Vinay Kumar K, et al. (2003) Apolipoprotein B-100 XbaI gene polymorphism in gallbladder cancer. Hum Genet 114(3): 280-283.
- Fan Jianglin, McCormick Sally P A, Krauss Ronald M, Taylor Stacy, Quan Ricky, et al. (1995) Overexpression of Human Apolipoprotein B-100 in Transgenic Rabbits Results in Increased Levels of LDL and Decreased Levels of HDL. Arterioscler Thromb Vasc Biol 15(11): 1889-1899.
- Silva M, Guerra J F da C, Sampaio A F S, Lima W G de, Silva M E, et al. (2015) Iron Dextran Increases Hepatic Oxidative Stress and Alters Expression of Genes Related to Lipid Metabolism Contributing to Hyperlipidaemia in Murine Model. BioMed Res Int 2015: 272617.
- Murray R, Bender D, Botham K, Kennelly P, Rodwell V, et al. (2012) Harper’s illustrated biochemistry (29th)., USA: Lange Medical Books/McGraw-Hill.
- Toth Peter P (2005) The “Good Cholesterol”. Circulation 111(5): e89-e91.
- Miller Michael, Stone Neil J, Ballantyne Christie, Bittner Vera, Criqui Michael H, et al. (2011) Triglycerides and Cardiovascular Disease. Circulation 123(20): 2292-333.
- Karlatti S S, Kumar G R (2016) Incidence of Various Types of Gallstones in Patients of Cholelithiasis in Belagavi 4(7): 3.
- Weerakoon H, Navaratne A, Ranasinghe S, Sivakanesan R, Galketiya K B, et al. (2015) Chemical Characterization of Gallstones: An Approach to Explore the Aetiopathogenesis of Gallstone Disease in Sri Lanka. PLOS ONE 10(4): e0121537.
- Kharga B, Sharma B K, Singh V K, Nishant K, Bhutia P, et al. (2016) Obesity Not Necessary, Risk of Symptomatic Cholelithiasis Increases as a Function of BMI. J Clin Diagn Res JCDR 10(10): PC28-PC32.
- Miquel J F, Núñez L, Amigo L, González S, Raddatz A, Rigotti A, et al. (1998) Cholesterol saturation, not proteins or cholecystitis, is critical for crystal formation in human gallbladder bile. Gastroenterology 114(5): 1016-1023.
- Loria P, Lonardo A, Lombardini S, Carulli L, Verrone A, et al. (2005) Gallstone disease in non-alcoholic fatty liver: prevalence and associated factors. J Gastroenterol Hepatol 20(8): 1176-1184.
- Thijs C, Knipschild P, Brombacher P (1990) Serum lipids and gallstones: A case-control study. Gastroenterology 99(3): 843-849.
- Hayat S, Hassan Z, Changazi S H, Zahra A, Noman M, et al. (2019) Comparative analysis of serum lipid profiles in patients with and without gallstones: A prospective cross-sectional study. Ann Med Surg 42: 11-13.
- Alexander N, Edwin R, Purushothaman P, Sanniyasi S (2018) Relationship Between Cholesterol and Gallstones, is There Really a Link? A Review of 80 Cases 12.
- Da Luz P L, Favarato D, Junior J R F-N, Lemos P, Chagas A C P, et al. (2008) High Ratio of Triglycerides to HDL-Cholesterol Predicts Extensive Coronary Disease. Clin Sao Paulo Braz 63(4): 427-432.
- Jiang Z Y, Sheng X, Xu C Y, Li W W, Chang X-X, et al. (2013) Gallbladder Gallstone Disease Is Associated with Newly Diagnosed Coronary Artery Atherosclerotic Disease: A Cross-Sectional Study. PLoS ONE 8(9):
- Kanwar R S, Merwaha D C, Gupta R R, Shukla P S, Minhas S S, et al. (1996) Phospholipid and cholesterol levels in serum and bile in cholelithiasis. Indian J Clin Biochem 11(2): 173.
- Nagi G S, Arora R (2015) Incidence of various types of gallstones in patients of cholelithiasis in North India. J Evol Med Dent Sci 4(97): 16213-16215.
- Chandran P, Kuchhal N K, Garg P, Pundir C S (2007) An extended chemical analysis of gallstone. Indian J Clin Biochem 22(2): 145-150.
- Vítek L, Carey M C (2012) New pathophysiological concepts underlying pathogenesis of pigment gallstones. Clin Res Hepatol Gastroenterol 36(2): 122-129.
- Dikkers A, Tietge U J (2010) Biliary cholesterol secretion: More than a simple ABC. World J Gastroenterol WJG 16(47): 5936-5945.
- Botham K M, Bravo E (1995) The role of lipoprotein cholesterol in biliary steroid secretion. Studies with in vivo experimental models. Prog Lipid Res 34(1): 71-97.
- Reshetnyak V I (2012) Concept of the pathogenesis and treatment of cholelithiasis. World J Hepatol 4(2): 18-34.
- Donovan JM, Carey MC (1991) Physical-chemical basis of gallstone formation. Gastroenterol Clin North Am 20(1): 47-66.
- Kim Y E, Kim D H, Roh Y K, Ju S Y, Yoon Y J, et al. (2016) Relationship between Serum Ferritin Levels and Dyslipidemia in Korean Adolescents. PLoS ONE 11(4): e0153167.
- Chowta N K, Reddy S B, Chowta M N, Shet A, Achappa B, et al. (2017) Lipid profile in anemia: Is there any correlation? Ann Trop Med Public Health 10(4): 837.
- Choi J W, Kim S K, Pai S H (2001) Changes in Serum Lipid Concentrations during Iron Depletion and after Iron Supplementation. Ann Clin Lab Sci 31(2): 151-156.
- Kumar M, Goyal B, Mahajan M, Singh S (2006) Role of iron deficiency in the formation of gall stones. Indian J Surg 68(2).
- Pamuk G E, Ümit H, Harmandar F, Yeşil N (2009) Patients with iron deficiency anemia have an increased prevalence of gallstones. Ann Hematol 88(1): 17-20.
- Bhadre R B, Verma R, Halgoankar P (2016) Correlation of gallstone formation with serum iron levels. J Evid Based Med Health 3(60): 3243-3247.
- Sahu S, Jain R, Prakash A, Bahl D, Sachan P, et al. (2017) Correlation Of Gallstone Disease With Iron-Deficiency Anaemia: A Prospective Study, p. 4.
- Swartz Basile D A, Goldblatt M I, Blaser C, Decker P A, Ahrendt S A, et al. (2000) Iron deficiency diminishes gallbladder neuronal nitric oxide synthase. J Surg Res 90(1): 26-31.
- Salomons H, Keaveny A P, Henihan R, Offner G, Sengupta A, et al. (1997) Nitric oxide and gallbladder motility in prairie dogs. Am J Physiol-Gastrointest Liver Physiol 272(4): G770-G778.
- Bradford W D, Elchlepp J G, Arstila A U, Trump B F, Kinney T D, et al. (1969) Iron metabolism and cell membranes. I. Relation between ferritin and hemosiderin in bile and biliary excretion of lysosome contents. Am J Pathol 56(2): 201-228.
- Verma G R, Pandey A K, Bose S M, Prasad R (2002) Study of serum calcium and trace elements in chronic cholelithiasis. ANZ J Surg 72(8): 596-599.
- Johnston S M, Murray K P, Martin S A, Fox Talbot K, Lipsett P A, et al. (1997) Iron deficiency enhances cholesterol gallstone formation. Surgery 122(2): 354-361; discussion 361-362.

 Research Article
Research Article